Abstract
Hypothesis/introduction:
The association of ACE I/D polymorphism and hemodynamic response to exercise have been limited to primarily aerobic exercises. We hypothesized that D allele carriers would show greater hemodynamic response to resistance exercise, as has been observed with aerobic. This study aimed to investigate the association of ACE I/D polymorphism and hemodynamic (blood pressure (BP), heart rate (HR) and rate-pressure product (RPP)) response to resistance exercise in young healthy subjects.
Materials and methods:
ACE I/D polymorphisms were studied by PCR analysis from 75 healthy men. Subjects completed a resistance exercise session of three sets of 10 knee extension repetitions with loads of 50, 75 and 100% of 10RM and two-minute rest intervals. Hemodynamic measures were recorded before and immediately after each set. Analysis of variance was used to identify significant differences among ACE genotypes.
Results:
ACE I/D polymorphism is associated with hemodynamic response to resistance exercise, as healthy subjects with ACE D allele were prone to higher responses. In addition, this phenotypic difference seems to be a load-dependent trend.
Conclusion:
ACE DD carriers exhibit greater heart work during resistance exercise. Future studies should focus on the influence of resistance training period with different workloads on the hemodynamic response in healthy individuals with different ACE genotypes.
Introduction
Blood pressure (BP) rises in response to physical exercise and it is known that the magnitude of this response is dependent on select aspects of the exercise (e.g. type (aerobic or resistance exercises; static or dynamic), duration, intensity). 1 However, factors not related to these exercise aspects, such as genetic factors, may also be associated with the BP response to exercise.2,3 Genetic factors are estimated to account for ~30% of the variance in resting BP of an adult population 4 and may account to some extent for the variability in BP response to exercise in a load-dependent way. 2
The study of “candidate genes” to certain phenotypic responses, such as the hemodynamics response to exercise, is one of the strategies used to verify possible associations among genetic factors and physiological response to exercise. 5 Among many candidate genes that may be associated with the BP response to exercise, the genes from elements in the renin-angiotensin system (RAS) have attracted considerable attention.
Among the elements of the RAS, the angiotensin-converting enzyme (ACE) plays a key role in regulating BP owing to the influence on vascular tone through the activation of the vasoconstrictor angiotensin II 6 and inactivation of the vasodilatory peptide bradykinin. 7 Inter-individual differences in blood ACE levels are, at least, in part explained by the presence of an insertion/deletion (I/D) polymorphism in intron 16 of the ACE gene,3,8 with greater ACE levels observed in D allele carriers. Notwithstanding, it has been postulated that the D allele carriers have a higher level of angiotensin I conversion to angiotensin II than I allele carriers. 9
The relationship between candidate genes (i.e. ACE gene) and hemodynamics response to exercise have been proposed by many researchers.3,10 –12 Previous findings indicated that D allele carriers have a greater hemodynamic response to exercise when compared to I allele carriers, but these studies have been restricted to aerobic exercises.11,12 Thus, it is unclear if these findings are applicable to resistance exercise, since the BP response to exercise is influenced by specific components, such as type, intensity, or mode.
Furthermore, the inclusion of resistance exercises in physical activity and rehabilitation routines has been strongly recommended by the American College of Sports Medicine13,14 and American Heart Association 15 owing to its beneficial effects for skeletal muscle and cardiovascular systems.
In light of the fact resistance exercise has not been studied concerning this issue and the importance of resistance exercise as a component of being physically active, the current study was undertaken. The purpose of this study was to investigate the association of ACE I/D gene polymorphism and BP response to resistance exercise in young healthy subjects. We hypothesized that D allele carriers would show greater BP response to resistance exercise, similar to what has been seen with aerobic exercises.
Methods
Subjects
Seventy-five healthy men, originally from the state of Bahia in the northeast part of Brazil (eight white Brazilians (ACE genotypes: one DD, six ID and one II), 37 pardo Brazilians(ACE genotypes: 15 DD, 15 ID and seven II) and 30 black Brazilians (ACE genotypes: 16 DD, eight ID and six II), volunteered to participate in the current study. All subjects underwent a clinical examination before being admitted into the Brazilian military service and were considered healthy (i.e. no muscle, joint or cardiovascular disease). Subjects were starting military service and had no recent history of an exercise training routine. The purpose and procedures of the experiment were explained and consent was obtained prior to the commencement of the experiment. All procedures were approved by the local ethics committee according to the Declaration of Helsinki.
Genotype analysis
Blood samples were collected from an antecubital vein in a seated position for detection of the ACE I/D polymorphism using the polymerase chain reaction (PCR) amplification of the region of intron 16 of the gene for ACE to determine if the Alu repeat sequences were present. In addition, individual DNA sequences were amplified using primers flanking the polymorphic region (sense 5’CTG-GAG-ACC-ACT-CCC-ATC-CTT-TCT3’ and antisense 5’GAT-GTG-GCC-ATC-ACA-TTC-GTC-AGA-T3’). PCR profiling and genotyping of the I/D polymorphisms were conducted as described previously. 8 To avoid mistyping I/D heterozygotes as DD homozygotes because of the preferential amplification of the D allele in heterozygous samples, all DD homozygous samples were subjected to independent PCR amplification using a primer set that identifies an insertion-specific sequence that has been previously reported (sense 5’TGG-GACCAC-AGC-GCC-CGC-CAC-TAC3’ and antisense 5’TCG-CCA-GCCCTC-CCA-TGC-CCA-TAA3’). 8 The reproducibility was greater than 99%, with most instances of error being due either to transcription or data entry. When discrepancies were found, the genotyping of the sample was repeated.
All genotyping was performed by the same researcher who was blinded to subject data. The study design was a double-blind approach with respect to the participants’ genotype.
Body composition measures
Body weight was assessed using a digital scale to the nearest 0.1 kg, and height was recorded with a stadiometer to the nearest 0.1 cm. Measurements were made with the subjects in light clothes and shoes removed. Body mass index (BMI) was calculated as body mass (in kilograms) divided by the square of height (in meters). Percentage of body fat and lean body mass was approximated using a three-site skin-fold method as proposed by the American College of Sports Medicine. 16 A well-trained technician measured skin-folds three times at each site and the mean of the values was used.
Ten repetition maximum (10RM) tests and exercise protocol
In the week before the experiment, the load for 10RM was determined for each subject in a knee extension machine by using the maximum weight that could be lifted for 10 consecutive repetitions. If the subject did not accomplish 10RM in the first attempt, the weight was adjusted by 4–10 kg and a minimum of five minutes of rest was given before the next attempt. Only three trials were allowed per testing session. To increase the reliability of 10RM testing, the following strategies were employed: (a) the 10RM was measured on two nonconsecutive days that were separated by 72 hours, (b) exercise technique was monitored and corrected as needed and (c) all subjects received verbal encouragement. The reliability of the 10RM loads was assessed with the intraclass correlation coefficient (ICC) and showed high reliability (ICC, r = 0.95). A week after the last 10RM test, subjects performed three sets of 10 repetitions of knee extension at 50% (first set), 75% (second set) and 100% (third set) of 10 RM, with two-minute rest intervals between sets. Subjects were instructed on proper breathing technique to avoid the occurrence of a Valsalva maneuver. To ensure that all subjects were moving at approximately the same velocity for each repetition, each set was timed using a metronome, which called out a cadence for the eccentric and concentric phases of each repetition. The repetition cadence consisted of a two-second eccentric phase followed by a two-second concentric phase.
Hemodynamics measures
Using standardized techniques, 17 a well-trained technician measured systolic and diastolic blood pressure by auscultation using a calibrated aneroid sphygmomanometer (BD®, Germany) and a stethoscope (BD®, Germany). Systolic blood pressure (SBP) was recorded as the moment of hearing the first Korotkoff sound and diastolic blood pressure (DBP) as the moment of disappearance of the last Korotkoff sound. SBP and DBP were measured once before (i.e. at rest) and immediately after the end of each set of the exercise. BP values were also measured immediately after each exercise set, because our aim was to study the acute response to resistance. BP increases immediately after resistance exercise execution, and then lowers to the pre-exercise values in just a few seconds (i.e. BPs can reduce very quickly following resistance exercise). Thus, to measure the acute hemodynamic response, BP was measured only immediately after each resistance exercise execution.
The mean blood pressure (MBP) was calculated using the formula: MBP = DBP + (SBP–DBP/3), for each BP measure (i.e. before and after each exercise set). To avoid the intersubject variability, the same well-trained technician performed all BP measures.
Intra-arterial pressure measurement is considered the gold-standard method for assessing BP, and the auscultation method tends to underestimate this parameter. However, the intra-arterial measurement is an invasive procedure that might put participants at excessive risk, which leads to a recommendation to avoid its use in healthy subjects.18,19
Heart rate (HR) was monitored with a heart rate monitor (HRM) (Polar, Kempele, Finland) and recorded before (i.e. at rest) and immediately after the end of each set of the exercise, simultaneously to BP measures. The rate-pressure product (RPP) was calculated as the product of HR (beats per minutes) and systolic arterial pressure (mmHg) multiplied by 10−3, following recommendations.20 –22
RPP was included to evaluate the association between the ACE I/D gene polymorphism and heart work during the resistance exercise protocol. RPP is directly proportional to the work performed by the heart and considered to be a useful index of myocardial oxygen consumption during exercise,23 –25 which is the most important indicator of the load on the heart. 22 Myocardial oxygen consumption is difficult to measure directly, which makes RPP a non-cumbersome measure of myocardial oxygen consumption during ambulatory conditions
Statistical analysis
After genotype analyses, subjects were placed into three groups according to allele frequency: II, ID or DD. A χ2 test was used to determine whether the observed genotype frequency was in Hardy-Weinberg equilibrium (expected frequency of alleles: (p2+2pq+q2=1), where p is the more common allele and q is the less common allele). 26 The reliability of the 10RM load assessments was determined with the ICC and the reliability was categorized as “excellent” for ICC values in the range of 0.8–1.0 and “good” for 0.6–0.8, whereas values <0.6 were “poor.” 27
A one-way analysis of variance (ANOVA) was used to test the differences in age, body composition, exercise load (10RM) and hemodynamic variables before the first exercise set among the ACE I/D gene polymorphisms (i.e. II, ID and DD). A three-way ANOVA (three genotypes × two time (PRE- and POST-exercise) × 3 sets) was used to compare SBP, DBP, MBP, HR and RPP during exercise. Significant main effects were analyzed using pairwise comparisons and followed by Tukey post hoc tests. Statistical significance for all analyses was accepted at p ≤ 0.05. Statistical analysis was completed using PASW 18 statistical package (SPSS Inc, Chicago, IL, USA.).
Results
A χ2 analysis indicated that the genotype frequencies of the subjects were in Hardy-Weinberg equilibrium (p = 0.61). Table 1 shows the body composition, exercise load (10RM) and resting blood pressure of the subjects after separation according to ACE genotype group. There were no significant differences between groups in age and any of the body composition measures (height, body weight, BMI, percentage of body fat and lean body mass) (p > 0.05). None of the subjects enrolled in the study displayed hypertension (i.e., blood pressure ≥140/90 mmHg); 16 although subjects with ACE DD genotype had clinically similar but significantly higher resting SBP when compared to II and ID genotypes (p < 0.05; see Table 1).
Mean±SE of body composition, exercise load (10 RM) and rest blood pressure of volunteers by ACE genotypes (II, ID and DD).

ACE: angiotensin-converting enzyme; BMI: body mass index; RM: repetition maximum; bpm: beats per minute. aSignificant difference among genotype DD and other genotypes (p < 0.05).
SBP showed a significant main effect for time (F1,432 = 738.38; p < 0.001; observed power = 1.00), sets (F2,432 = 55.70; p < 0.001; observed power = 1.00) and genotypes (F2,432 = 15.70; p < 0.001; observed power = 0.99) (Figure 1). The post hoc analysis indicated that subjects carrying DD alleles had greater SBP responses with each exercise set when compared to other genotypes (i.e. II or ID) (p < 0.05). Additionally, we observed a progressive increase in SBP with the sets (p < 0.05); that is, with the increasing exercise load.
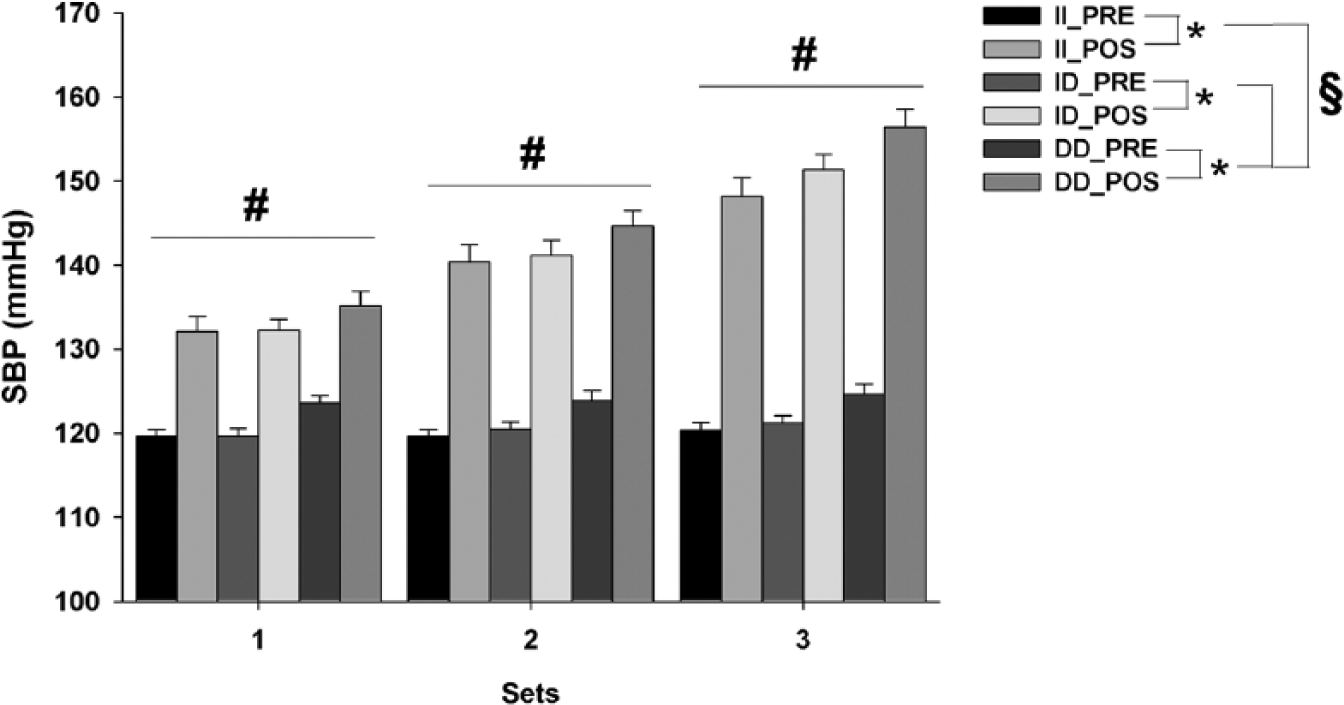
Mean±SE of SBP, before and after each exercise set, from ACE gene genotypes II, ID and DD. Note that the scale starts at 100 mmHg. *Significant difference between PRE- and POST-exercise measures (p < 0.05); #Significantly different from other exercise sets (p < 0.05);
DBP showed a significant main effect for time (F1,432 = 58.83; p < 0.001; observed power = 1.00), sets (F2,432 = 3.20; p < 0.05; observed power = 0.61) and genotypes (F2,432 = 13.75; p < 0.001; observed power = 0.99) (Figure 2). The post hoc analysis indicated that subjects carrying DD alleles have greater DBP response with each exercise set when compared to other genotypes (i.e. II or ID) (p < 0.05). Between the sets, significant differences existed only between the first and third sets (p < 0.05).
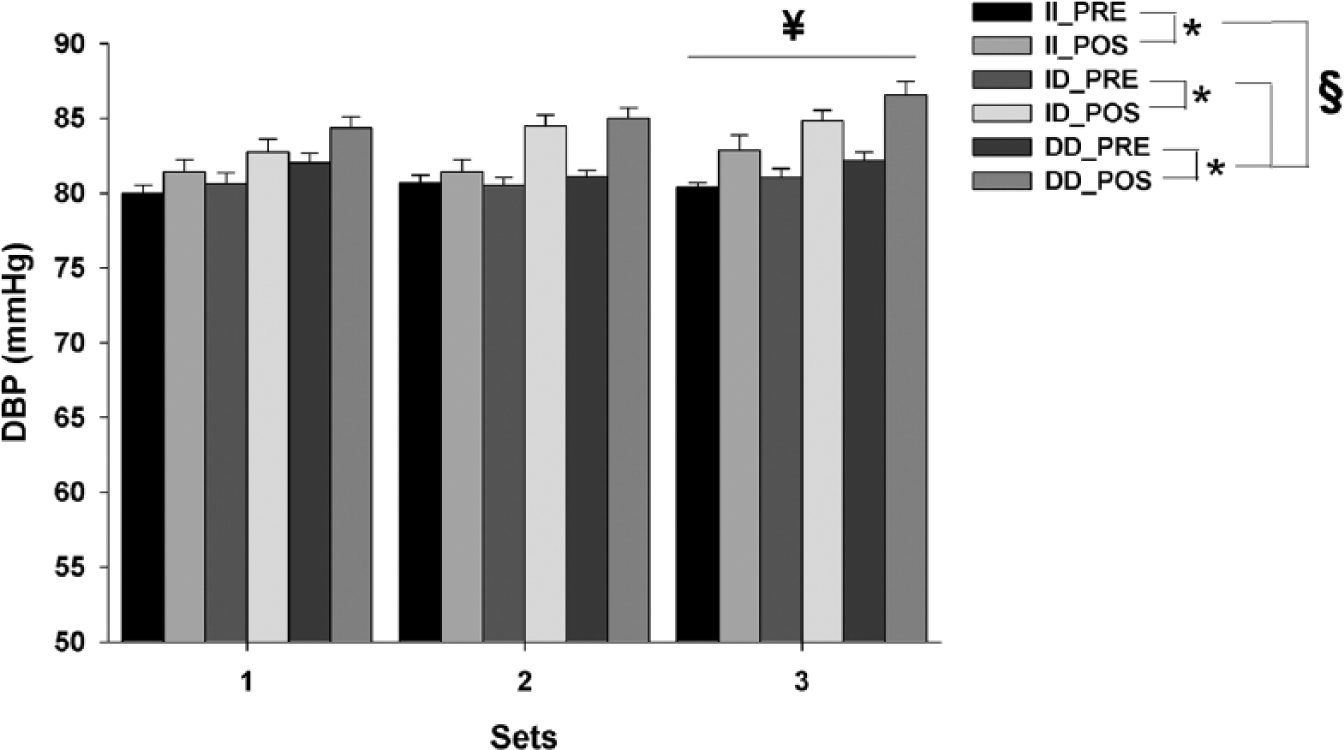
Mean±SE of DBP, before and after each exercise set, from ACE gene genotypes II, ID and DD. Note that the scale starts at 50 mmHg. *Significant difference between PRE- and POST-exercise measures (p < 0.05); ¥significantly different from first exercise set (p < 0.05); §significant difference among genotype DD and all other genotypes (p < 0.05).
MBP also showed a significant main effect for time (F1,432 = 485.75; p < 0.001; observed power = 1.00), sets (F2,432 = 33.17; p < 0.001; observed power = 1.00) and genotypes (F2,432 = 21.91; p < 0.001; observed power = 1.00) (Figure 3). The post hoc analysis indicated that subjects carrying DD alleles have greater MBP response with each exercise set when compared to other genotypes (i.e. II or ID) (p < 0.05). Genotype differences in MBP were observed both before and immediately after exercise, with particularly marked differences after the last sets (p < 0.05). Additionally, we observed a progressive increase in MBP with the sets (p < 0.05); that is, with the increasing exercise load.

Mean±SE of MBP, before and after each exercise set, from ACE gene genotypes II, ID and DD. Note that the scale starts at 60 mmHg. *Significant difference between PRE- and POST-exercise measures (p < 0.05); #significantly different from other exercise sets (p < 0.05);
HR showed a significant main effect for time (F1,432 = 448.96; p < 0.001; observed power = 1.00), sets (F2,432 = 23.680; p < 0.001; observed power = 0.61) and genotypes (F2,432 = 4.49; p = 0.01; observed power = 0.77) (Figure 4). Post hoc analysis indicated that subjects carrying DD alleles have greater HR response with each exercise set when compared to ID carriers (p < 0.05), while the difference between DD and II carriers was borderline significant (p = 0.06). Between the sets, significant differences were observed only between first and third sets (p < 0.05).

Mean±SE of HR, before and after each exercise set, from ACE gene genotypes II, ID and DD. Note that the scale starts at 40 bpm. *Significant difference between PRE- and POST-exercise measures (p < 0.05); #significantly different from other exercise sets (p < 0.05);
RPP also showed a significant main effect for time (F1,432 = 706.97; p < 0.001; observed power = 1.00), sets (F2,432 = 49.13; p < 0.001; observed power = 1.00) and genotypes (F2,432 = 9.46; p < 0.001; observed power = 0.98) (Figure 5). The post hoc analysis indicated that subjects carrying DD alleles have greater RPP response with each exercise set when compared to other genotypes (i.e. II or ID) (p < 0.05). Additionally, a progressive increase in RPP was observed with the sets (p < 0.05); that is, with the increasing exercise load.

Mean±SE of RPP, before and after each exercise set, from ACE gene genotypes II, ID and DD. Note that the scale starts at 4 mmHg/beats.10−3. *Significant difference between PRE- and POST-exercise measures (p < 0.05); #significantly different from other exercise sets (p < 0.05);
Discussion
Our main study finding was that healthy subjects with the ACE D allele had higher hemodynamic responses to resistance exercise. In addition, young healthy DD carriers showed a significantly greater resting SBP and MBP before the exercise.
Previous studies reported that the magnitude of increase in BP during exercise is an independent predictor of left ventricular (LV) hypertrophy, and the risk of stroke and death.28,29 Interestingly, the ACE D allele has been associated with exercise-induced LV hypertrophy30 –33 after exercise training (military training involving resistance and endurance exercises); specifically, training-induced LV mass increases could be 2.7 times greater in the DD homozygotes than in the II homozygotes. 31
The genotypic difference in the hemodynamic and LV mass responses to exercise may be related to the greater ACE levels observed in D allele carriers, compared to I allele carriers. The greater ACE levels observed in D allele carriers may lead to higher levels of angiotensin I conversion to angiotensin II than I allele carriers. 9 The susceptibility for LV hypertrophy from ACE D allele carriers may therefore result from direct action of angiotensin II over the ventricular muscle, 34 and/or an indirect way, through vasoconstrictor action over arterioles, 6 generating additional blood flow resistance and potentially a myocardial overload.
Angiotensin II is a known vasoconstrictor mediator acting on type 1 (AT1) receptors, increasing the resistance to blood flow, 6 which leads to LV hypertrophy 35 owing to an increased pressure load (“after load”). In addition, circulating angiotensin II has peripheral and central effects on the autonomic nervous system (ANS), increasing sympathetic outflow.6,36,37 This can lead to additional blood flow resistance and the potential for myocardial overload during exercise.
DD carriers had a greater HR during the resistance exercise protocol, which suggests a higher sympathetic outflow. Yet, the relationship between the ANS and angiotensin II could be more complex, since the activation of β1-receptors in juxtaglomerular (JG) cells increases renin secretion, the rate-limiting enzyme in the formation of angiotensin II. 38 Despite our hypothesis about the interplay between the ACE gene polymorphism and ANS, further studies are required to investigate this issue, since HR is influenced by the ANS among other factors, and the sympathetic outflow was not measured directly.
The BP and RPP response to resistance exercise was associated with the ACE I/D polymorphism, with the greatest responses occurring in the D allele carriers, especially DD homozygous, which partially explains the previous findings about the association between exercise-induced LV hypertrophy and ACE I/D polymorphism.30 –33 The greater RPP of DD carriers during exercise reinforces this hypothesis since this hemodynamic parameter is a useful index of heart work and myocardial oxygen consumption during exercise.23,24
The greater ACE activity at rest of D allele carriers, as reported by a previous study, 9 may also have existed in our subjects and thus explains our observed greater SBP and MBP before the exercise protocol (i.e. at rest) when compared to I allele carriers. Although our sample was healthy young subjects (~19 years) without signs of cardiovascular disease after a clinical examination, and with no recent history of an exercise training routine, heart structural remodeling could already be present and influence the hemodynamics. Yet, our study investigated only the hemodynamic response to exercise, and the absence of a specific heart structural exam is a limitation of our study. Despite this, it is important to note that all subjects enrolled in this study were normotensive and the mean resting SBP from the ACE DD genotype was only 124 mmHg (i.e. only slightly above that of the other groups).
There was also a trend for load-dependent differences in the BP response to resistance exercise for subjects with the different ACE gene polymorphism. Physical effort can stimulate the RAS,10,39 –41 dependent on the load 42 increasing the ACE activity and circulating angiotensin II. Therefore, physical exertion and subsequent sympathetic activity stimulates renin release from the kidney, increasing RAS activity. 41 This may explain the differences among genotypes for HR and RPP only during exercise. Notwithstanding, another study 43 found that men with the ACE DD genotype exhibit a greater pressor response to angiotensin I infusion than did men with the other ACE genotypes. Therefore, a greater sympathetic activity during intense exercise could lead to a greater renin secretion and angiotensin I synthesis, inducing a greater pressor response to exercise in subjects with the ACE DD genotype. Of course, this is only speculation, as we did not conduct hormonal assessment, but nonetheless it is highly plausible. 44
Based on our findings, we hypothesized that greater exercise loads may induce greater ACE activity and circulating angiotensin II, but this idea should be tested in further studies. Our results point to the need for future investigations about the relationship between the ACE I/D polymorphism and BP response to resistance exercise, and suggest this is a promising field for longitudinal investigations where the LV mass outcome may be observed after resistance exercise training with different loads (i.e. 50, 75 or 100% of 10RM).
In summary, the ACE I/D polymorphism is associated with the hemodynamic response to resistance exercise, with healthy subjects having the ACE D allele being prone to higher hemodynamic (i.e. BP, HR and RPP) responses to resistance exercise. In addition, this phenotypic difference seems to induce a load-dependent trend in exercise responses. Based on our findings, we recommend that future studies should focus on two issues: 1) the long-term influence of a resistance training program with different workloads on the BP response and LV mass from healthy subjects with different ACE genotypes; 2) the influence of other polymorphisms of the RAS on the BP response and LV mass from healthy subjects submitted to a resistance training program (since we included only the ACE I/D polymorphism, because it is the most reported polymorphism of the RAS related to the hemodynamic and LV mass response to exercise).3,11,12,30 –33
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
None declared.
