Abstract
Hypothesis:
Polymorphisms in angiotensin II type-1/2 receptor genes (AGTR1/AGTR2) may be involved in the pathogenesis of primary aldosteronism. The present study aims to reveal some loci susceptible to the disease on the genes in a group of Chinese Han nationality.
Materials and methods:
A case-control study was conducted in 202 patients and 188 controls. Ten tagging SNPs on AGTR1/AGTR2 were genotyped for all subjects via the method of multiplex PCR-ligase detection reaction. Statistical analysis was performed with chi-square test and logistic regression analysis.
Results:
rs3772616 on the AGTR1 gene was a factor for susceptibility to primary aldosteronism (p<0.001), and the TT genotype significantly decreased the risk of primary aldosteronism compared with the CC homozygote (p=0.008, adjusted OR=0.13; 95%CI: 0.03–0.59). The rs3772616 polymorphism was associated with primary aldosteronism under the additive and dominant models. The female carriers of the G allele in rs5193 showed a significant difference compared with the T allele.
Conclusions:
The AGTR1 rs3772616 polymorphism can be considered as a hereditary marker for primary aldosteronism, and in the Chinese Han population the rs5193 G allele seems to predispose to it only in women.
Keywords
Introduction
Primary aldosteronism (PA) is one of the most common forms of secondary hypertension. The reported prevalence of PA varies widely, ranging from 1.4–32%, due to varying study design and screening criteria. However, autonomous aldosterone secretion may be present in more than 30% of hypertensive patients, according to recent findings. 1 The recently investigated prevalence of PA in the Chinese population is 7.1% within refractory hypertension, and within this the proportion of hypokalemia is up to 52.54%. 2
PA comprises a wide spectrum of conditions ranging from a solitary unilateral nodule producing aldosterone to bilateral diffuse or nodular hyperplasia, with several intermediate phenotypes. Two major causes of PA are recognized: idiopathic hyperaldosteronism (IHA), accounting for 60–70% of patients with PA, and aldosterone-producing adenoma (APA), accounting for 30–35%. 3
The renin–angiotensin system (RAS) mediates several classical physiological processes, including body water and electrolyte homeostasis, blood pressure (BP) regulation and aldosterone generation, based on the angiotensin II type 1and type 2 receptors (AGTR1, AGTR2) subtype system. Genetic or environmental factors may affect the expression and activity of AGTR1 and AGTR2, which may impair their regulatory functions and eventually lead to the development of related diseases.
The AGTR1 gene is located on chromosome 3q21-25 with a length of about 45 kb, containing five exons and four introns. Its expression product – AGTR1 – plays an integral role in BP, and is implicated in the pathogenesis of hypertension. The association between AGTR1 gene polymorphisms and essential hypertension has been widely studied, but the results remain controversial. 4 Otherwise, AGTR1 expression was reported repeatedly to be correlated with the functional status of adrenocortical tumors, including APA. 5
The gene encoding AGTR2 is located on chromosome Xq22-q23, which contains a limited number of 15 known single nucleotide polymorphism (SNP) loci. For its part, AGTR2 mainly plays a role in cardiovascular function and programmed cell death. It has also been demonstrated that expression of AGTR2 in APA tissue was lower than that in normal adrenal gland tissues. Our previous research showed that the rs5194 SNP in the AGTR2 gene was significantly associated with the risk of APA. 6 On the basis of our previous study and other relevant literature, we decided to further investigate the associations of multi-polymorphisms in two AGTR genes with the risk of PA.
Materials and methods
Subjects
Through screening thousands of suspicious patients from the Han population from Fujian Province of China with the aldosterone plasma renin activity ratio, our study enrolled 202 sporadic patients who were hospitalized and diagnosed with PA after endocrinal confirmatory tests, mostly the saline infusion test and Captopril Challenge test. Computed tomography scanning of the adrenal glands and adrenal vein sampling were undertaken for subtype diagnosis according to the latest guideline for PA. Another 188 healthy Chinese volunteers without hypertension or any adrenal disease were recruited as the control group from the health examination center of the First Affiliated Hospital of Xiamen University. The study was approved by the Institutional Ethical Committee (EC-ZD-002•02) of the hospital, and written informed consent forms were obtained from each participant before any examination or data collection.
Selection of SNPs loci
Based on public SNP databases (http://www.ncbi.nlm.nih.gov/SNP/ and http://www.hapmap.org/) of CHB populations and published literature, data for SNPs in the AGTR1/AGTR2 genes were selected for our study. We then applied Haploview software version 4.2 to choose the tagSNPs, which enabled us to capture all common SNPs within the entire AGTR1/AGTR2 gene sequences according to r2 linkage disequilibrium (LD) (threshold ≥0.8) and a minor allele frequency (MAF) ≥0.05. The 10 tagSNPs were rs12695877, rs2638360, rs1492100, rs37726216 and rs2675511 in intron 2, rs385338, rs275649, rs6801836 in intron 3 of the AGTR1 gene and rs5193 in 3’-UTR exon 3, rs1403543 in intron 1of the AGTR2 gene.
DNA extraction
Venous blood of each individual was sampled into sterile anticoagulation tubes and cryopreserved. Genomic DNA was isolated from peripheral blood leukocytes using the TIANamp Blood DNA Kit (TIANGEN Biotech, Beijing, China) according to the manufacturer’s instructions.
Genotyping
The SNP genotyping work was performed using an improved multiplex ligation detection reaction (iMLDR) technique developed by Genesky Biotechnologies Inc. (Shanghai, China). A multiplex PCR-ligase detection reaction method was used in the iMLDR. For each SNP, the alleles were distinguished by different fluorescent labels of allele-specific oligonucleotide probe pairs. Different SNPs were further distinguished by different extended lengths at the 3’end. A random sample accounting for ~5% (n=20) of the total DNA samples was directly sequenced using Big Dye-terminator version 3.1 and an ABI3730XL automated sequencer (Applied Biosystems) to confirm the results of iMLDR.
Statistical analysis
Demographic and clinical data between groups were compared using the χ2test or Student’s t-test. Quantitative variables were expressed as the mean ±SD. Multiple comparisons were made using one-way analysis of variance (ANOVA), and Bonferroni correction was applied to avoid false positives.
The allele and genotype frequencies of both groups were compared using χ2 statistics or Fisher’s exact test. Compliance with Hardy–Weinberg equilibrium in each group was subsequently assessed using the Haploview. An expectation maximization algorithm was used for all haplotype analysis on the SHEsis, a web-based program which also helped to manage the calculation of LD and genetic association.7,8 Odds ratios (ORs) and their adjustment for relevant variables (gender, age, body mass index (BMI), smoking and alcohol consumption) were calculated as estimates of risk relative to disease, and 95% confidence intervals (CI) were obtained by logistic regression analysis. Statistical analysis was performed using SPSS version 13.0 (Chicago, Illinois, USA). A p-value less than 0.05 or OR equal to 1.00 not in the range of 95% CI indicated statistical significance.
Results
Basic characteristics of the study population
Demographic and clinical characteristics of the two groups are listed in Table 1. There was no significant difference in gender, age and BMI; controls were matched for age and gender. Significant differences in PA-related parameters and some blood lipid levels were observed between groups.
Clinical characteristics of the patients and controls.

M: male; F: female; BMI: body mass index; SBP: systolic blood pressure; DBP: diastolic blood pressure; K+: serum potassium; PAC: plasma aldosterone concentration; PRA: plasma renin activity; ARR: aldosterone plasma renin activity ratio; HDL-C: high-density lipoprotein cholesterol; LDL-C: low-density lipoprotein cholesterol; CHO: cholesterol; TG: triglyceride.
Hardy–Weinberg equilibrium
There was no deviation of genotype frequencies from the Hardy–Weinberg expectation in all SNPs, which indicated that the subjects were representative in the field (all p>0.05, data not shown).
Analysis of AGTR1/AGTR2 gene polymorphisms
The C allele of rs3772616 in AGTR1 was more prevalent in the patients with PA compared with control subjects (p<0.001). Other allelic distributions of SNPs between groups were statistically insignificant (Table 2). Table 3 shows the genotypic distributions and their association analyses under the assumed co-dominant model. The TT genotype of rs3772616 showed a significant difference (p=0.008, adjusted OR=0.13; 95%CI: 0.03–0.59) compared with the CC homozygote, whether considering the confounding factors or not. A small difference was observed in the GA genotype of rs1403543 in AGTR2 between groups, which without including the influence of other covariates would be a false positive (p=0.04). Meanwhile, in rs5193, a significant difference was seemingly revealed in GT versus GG after adjustment for the mixed effects of gender, age, BMI, smoking and alcohol consumption (p=0.03, adjusted OR=0.45; 95%CI: 0.22–0.93).
Allele distribution of 10 SNPs between patients and controls.

Genotype distributions and the ORs for the AGTR1 and AGTR2 genes between patients and controls.

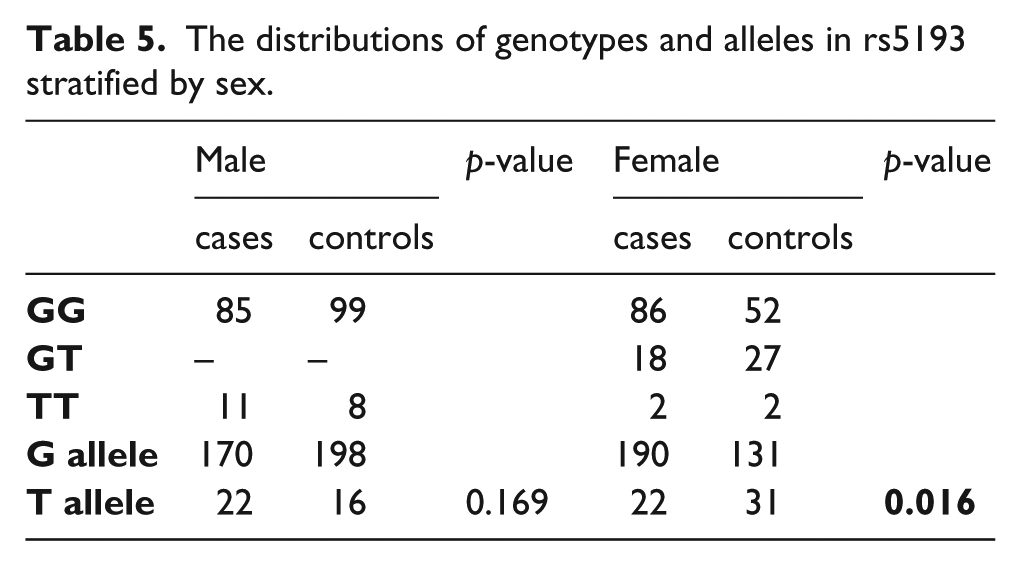
Furthermore, four common genetic models were assumed in order to verify their possible hereditary characteristics (Table 4). The rs3772616 polymorphism in the AGTR1 gene was significantly associated with PA in additive (OR=0.52, 95%CI: 0.35–0.78, p=0.001) and dominant (OR=0.54, 95%CI: 0.35–0.86, p=0.008) patterns. Regarding the rs5193 polymorphism under a heterozygous advantage model, individuals with the GT genotype had a 0.37-fold decreased risk of developing PA after adjusting for the known risk factors (p=0.009; 95%CI: 0.179–0.79). Given that rs5193 is located on the X chromosome, to which sex-linked inheritance belongs, stratification analysis based on different gender was subsequently adopted. As a result (Table 5), the T allele showed a significant difference compared with the G allele in female subjects, meaning that the G allele in rs5193 may reflect a genetic predisposition to PA in women (p=0.016).
Relationship between polymorphisms (rs5193, rs3772616) and PA in different genetic models.
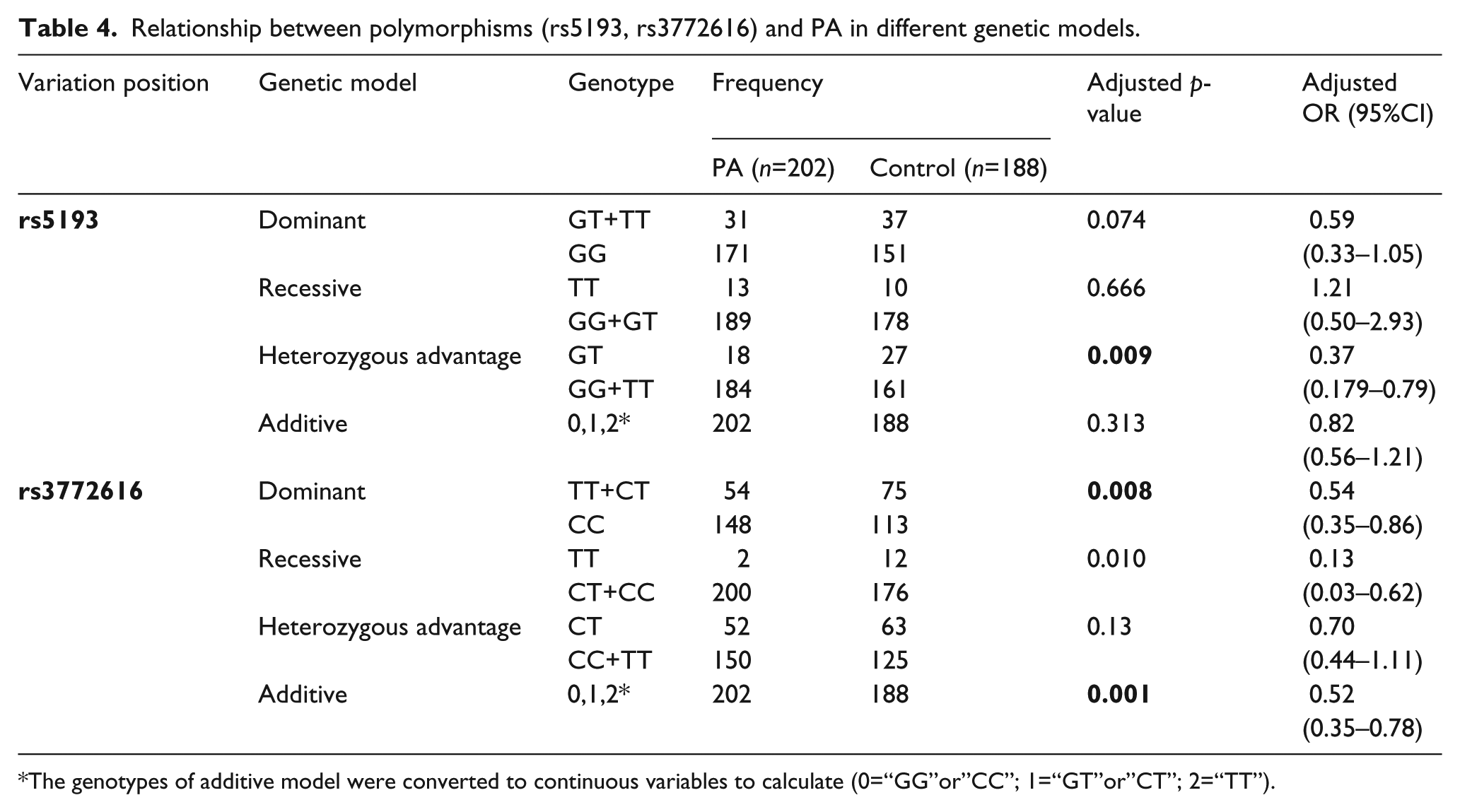
The genotypes of additive model were converted to continuous variables to calculate (0=“GG”or”CC”; 1=“GT”or”CT”; 2=“TT”).
The distributions of genotypes and alleles in rs5193 stratified by sex.
Haplotype analysis
As the selected eight tagSNPs in the AGTR1 gene are sufficient to construct most of the haplotype structures in this genomic region, we explored further to identify the most significant one to explain the hereditary susceptibility of PA. Seven haplotypes of the AGTR1 gene with frequencies above 3% either in cases or controls are shown in Table 6. Hap4 TATCACGC had a lower incidence in the cases group (0.009) than in the controls group (0.032), but the p-value did not reach the significant level after Bonferroni correction (p=0.027 > 0.05/7). No significant difference in frequencies of AGTR2 haplotypes was observed between two groups (data not shown).
Haplotype frequencies in AGTR1 between patients with PA and controls.

The haplotype structure of the AGTR1 gene was rs12695877, rs2638360, rs1492100, rs3772616, rs2675511, rs385338, rs275649 and rs6801836. Haplotypes with frequencies <0.03 are not included in the table.
Discussion
As a pathogenetically heterogeneous disease, the etiology of PA is still elusive and vague. Based on several basic investigations the intra-adrenal RAS, which is a major regulator of aldosterone generation, was extensively focused on in investigating the mechanism of excessive aldosterone secretion.9,10 To date, the pathophysiological role of the RAS in PA has been elucidated and components of the RAS such as AGTR1, AGTR2, CYP11B1, CYP11B2 and mineralocorticoid receptor have been reported to be widely involved in the regulation of autonomous aldosterone. It is known that the activation of AGTR1 can trigger the synthesis and secretion of aldosterone. The increased expression of AGTR1 mRNA in APA has long been confirmed. 11 Previous research has suggested that AGTR2 may partially contribute to the overproduction of aldosterone in angiotensin II (AT II)-responsive APA, 12 which may derive from its antagonistic effect towards the AGTR1-mediated function of AT II including cell growth, vasoconstriction and cardiovascular fibrosis. Otherwise, a pathological continuum between APA and IHA featuring a transition from hyperplasia to a nodular phase has been proposed, suggesting that pathogenic mechanisms common to these conditions exist. 13 Our earlier study preliminarily demonstrated that down-regulation of AGTR2, leading to a deficiency in counterbalancing the effects of angiotensin II mediated by AGTR1, could finally promote adrenal zona glomerulosa cell proliferation and aldosterone hypersecretion. 14 According to a series of investigations concerning the connections between AGTR1/2 and the pathology of the adrenal cortex, we speculate that the overexpression of AGTR1 and reduced expression of AGTR2 may be associated with the pathophysiological processes of PA and the relative gene risk variants inevitably involved in this disease, by altering the activity of genes and affecting their transcription and post-transcription. Although such an assumption needs more studies for clarification, AGTR1 and AGTR2 could be presumed to be factors in association with the susceptibility of PA.
The aim of the present pilot study was to explore whether genetic variants of AGTR constitute associated factors for the hereditary susceptibility of PA in the Chinese population. To date, very few epidemiological studies have been published related to the predisposition to PA, mostly focused on APA, and these have analyzed few SNPs. In this sense, our study is novel and takes us one step further in finding the genetic risk of developing this disease.
Based on the LD patterns and empirical genome-wide data from the international HapMap project, we obtained a set of informative SNPs (tagSNPs) which are representative of the remaining SNPs, so that the genotyping burden can be reduced without much loss of power for disease association studies.15,16 In this context, eight tagSNPs within the intron of AGTR1 were selected to cover all the genetic information of this region. Another two tagSNPs located in intron 1 (rs1403543) and 3’UTR exon3 (rs5193) identically shared information in their vicinity. However, such a method inevitably misses some rare SNPs associated with this disease since MAF≥0.05 was used to select the tagging SNPs based on the limited sample size.
In relation to our results, the rs3772616 polymorphism has been investigated elsewhere to study the relationship between genetic variants in RAS and assumed salt-related BP. This study reported that rs3772616 was associated with diastolic BP (DBP) and mean arterial pressure (MAP). In particular, the number of T alleles was associated with an increased mean DBP response to low-sodium intervention. 17 Considering that the mineralocorticoid aldosterone can regulate the level of sodium in the body and may interact with sodium loading to induce hypertension as well as cardiac injury, 18 PA and sodium intake probably share some genetic pathways or markers in multiple environmental–genetic interactions, which may play an important role in salt-sensitive hypertension and aldosterone modulation. 19 Our findings that carriers of the C allele in rs3772616 were found more frequently among patients with PA raised an issue that the major allele corresponded to the risk allele. Such a phenomenon is not rare in genome-wide association studies, especially in complex disease. Carriers of the major allele have a higher risk of developing some less prevalent disease, indicating that the mutation makes the locus protective in terms of its own susceptibility. However, it is more likely that this locus represents only a small increment in risk together with some main causative variants near or far from this region which have the main genetic effect on the association. The underlying rationale of this inconsistency derives from the complexity of the genetic architecture, and must be explained in terms of heredity, variation and evolution. In addition, Gu et al. 20 found that a potential interaction of CYP11B2-AGTR1 is involved in the etiology of hypertension. Against the background that the pathogenesis of essential hypertension is undefined, the question of whether secondary hypertension caused by PA follows a similar gene–gene interactive model is worthy of further research. If this is the case, AGTR1 interacting with other genes may constitute a disease locus of secondary hypertension caused by hyperaldosteronism.
With regard to rs5193, the GT genotype seemed to protect against PA, with a p-value of 0.009 (adjusted OR=0.37; 95%CI: 0.18–0.79) under the heterozygote advantage or overdominance model. However, such a tiny difference in statistics and the insignificant difference in the distribution of corresponding alleles drove us to analyze in more depth, since only a small proportion of loci had polymorphisms maintained by heterozygote advantage. 21 After stratification by sex, we found that a significant difference only occurred in the female subgroup (p=0.016), and not in males (p=0.169). Knowing that females originally showed a higher prevalence of PA than males, this result could be interpreted that polymorphisms of rs5193 may rooted in this sex-linked disease. Otherwise, Min Zhang et al. 22 reported that the T allele of rs5193 linking with the A allele of rs5194 had cardioprotective effects on essential hypertension. As we know, an excess of autonomous aldosterone secretion causes great cardiovascular damage independent of secondary hypertension. Interestingly, our finding that the T allele of rs5193 protected against PA was consistent with its cardioprotective role to some degree. Thus, we wonder whether the rs5193 gene polymorphism is associated with cardiovascular events caused by hypertension secondary to PA, although more targeted research is required to prove this in the future.
In our study, no haplotype was found to be significantly different between cases and controls. The one with most potential – Hap4 TATCACGC – failed to show significance after Bonferroni correction. However, there is considerable controversy about adjusting the p-value after multiple tests in clinically orientated studies. 23 With this in mind, whether a truly important difference was hidden by the increased likelihood of type II errors remains unknown. In addition, our effort to explore any gene marker to distinguish IHA and APA found no significant difference, which may be mainly due to the small sample size of APA patients (n=57), despite their intrinsic similarity.
To the best of our knowledge, this is the first attempt to explore AGTR-associated loci in PA pathogenesis. Therefore, some limitations of the study must be taken into account for correct interpretation of our data. First, as a retrospective case-control study, the number of patients is relatively small because of the very low prevalence of PA, and the statistical power is accordingly reduced. Larger case-control studies, as well as independent cohorts of patients, are suggested to validate our findings. Second, more data in Chinese, especially in other ethnic groups, are necessary in future studies in order to make results more representative, since all our subjects are confined to the Han people of Fujian Province, southeast of China. Third, the confounding factors between groups are not limited to those mentioned above. It is difficult to adjust for other environmental factors, such as diet, physical activity and metabolic level. Last but not the least, it should be emphasized that the genetic pathways underlying this complex disease remain largely elusive due to unpredictable factors such as polygenic inheritance, genetic heterogeneity, incomplete penetrance, gene–environment interaction and others. 24 Thus, the variations of AGTR may be related just to risk factors, inducing factors or impact factors, rather than directly to the disease itself.
Conclusions
The current study suggests that the rs3772616 polymorphism in AGTR1 may play a role in susceptibility to PA in the Han Chinese population, with the C allele predisposing to it. Meanwhile, females carrying the G allele on rs5193 of AGTR2 may be also susceptible to PA. Due to the complexity of definite diagnosis of PA and our limited number of patients, the present results require confirmation in larger studies.
Footnotes
Acknowledgements
We wish to express our gratitude to Dr. Xin Zeng for her help in revising this paper.
Funding
This project was supported by the National Natural Science Foundation of China (Grant No. 81200578) and Natural Science Foundation of Fujian Province of China (Grant No. 2011D011).
Conflict of interest
The authors declare that there is no conflict of interest.
