Abstract
Introduction:
Tacrolimus is frequently used as immunosuppressive agent in organ transplantation but its clinical use is limited due to its marked nephrotoxicity.
Materials and methods:
Male Wistar albino rats weighing 150–200 g (10–12 weeks old) were used. Animals were divided into four groups. Group 1 served as control group and received normal saline, group 2 served as toxic group and received 2 mg/kg tacrolimus i.p., group 3 served as treatment group and received 2 mg/kg tacrolimus i.p. followed by 2 mg/kg aliskiren orally and group 4 served as drug per se group and received 2 mg/kg aliskiren orally. Tacrolimus-induced nephrotoxicity was assessed biochemically and histopathologically.
Results:
Treatment with aliskiren decreased the tacrolimus-induced changes in biochemical markers of nephrotoxicity such as blood urea nitrogen and creatinine. Aliskiren also attenuated the effects of tacrolimus on oxidative stress parameters such as malondialdehyde, reduced glutathione and catalase. Histopathological and ultrastructural studies showed that aliskiren attenuated tacrolimus-induced renal damage.
Conclusion:
These results suggest that aliskiren has protective effects against tacrolimus-induced nephrotoxicity; implying that renin inhibitor may counteract nephrotic syndrome associated with immunosuppressant use.
Introduction
Tacrolimus, a calcineurin inhibitor, is used clinically as an immunosuppressive agent to reduce the rejection rate in organ transplantation.1–4 As compared with cyclosporine, tacrolimus treatment seems to induce fewer hemodynamic and fibrogenic effects but the resultant nephrotoxicity remains a serious problem.5–8 In renal transplant biopsies, almost similar histopathological injuries have been reported with cyclosporine and tacrolimus-treated patients, suggesting that tacrolimus in the long term results in the same structural damage as cyclosporine. 9
The mechanism of tacrolimus-induced acute nephrotoxicity is still unclear. Previous literature suggests that the ability of tacrolimus to produce reactive oxygen species (ROS) via activation of NADPH oxidase pathway and cause disturbance in antioxidant defense may be responsible for renal toxicity. 10 Several studies suggest that the renin–angiotensin system (RAS) plays a key role in the regulation of blood pressure (BP), volume homeostasis and development of cardiac hypertrophy, failure and reperfusion injury.11,12 Subsequently, its importance in renal failure has also been recognized and it has been established that inhibition of RAS is an effective way to intervene with the pathogenesis of these disorders.13–15 It is reported that treatment with tacrolimus leads to impairment in RAS. It has been shown previously that tacrolimus treatment significantly increases blood urea nitrogen (BUN) and serum creatinine level, while it decreases endogenous creatinine clearance and is inversely proportional to plasma renin activity. The rise in plasma renin activity significantly correlates with the increase in fractional excretion of Na.16,17 Similarly, renin mRNA levels are also increased in the renal cortex in tacrolimus-treated rats. 18 All these studies suggest that tacrolimus disturbs the RAS and is also associated with increased oxidative stress.
Aliskiren (ALK) is the first clinically used orally active renin inhibitor, which selectively inhibits renin enzymatic activity without affecting other systems.19,20 ALK attenuates gentamicin-induced nephropathy by suppression of inflammatory, profibrotic and apoptotic factors through inhibition of the nuclear factor and mitogen-activated protein kinase signaling pathways. 20 Direct renin inhibition with ALK prevents intra-renal RAS activation, leading to mitigation in the development of glomerulonephritis. 21 Moreover, transgenic mice overexpressing renin show disappearance of podocyte foot processes as well as perivascular and peri-glomerular inflammation in the kidney and these changes are reversed by treatment with AT1 receptor antagonist. 22 Based on these studies we hypothesized that ALK may reduce/attenuate tacrolimus-induced renal damage and oxidative stress through renin inhibition. Therefore, the present study was designed to investigate the effect of ALK on tacrolimus-induced nephrotoxicity in rats using biochemical markers of oxidative stress and renal function, and histopathological measures of cellular damage.
Materials and methods
Animal model
In this study male Wistar albino rats weighing 150–200 g (10–12 weeks old) were used. The animals were obtained from Experimental Animal Care Center, College of Pharmacy at King Saud University. They were housed under ideal laboratory conditions (12 h light/12 h darkness cycle, 45–55% relative humidity and temperature 23–25°C), maintained on standard pellet diet and water ad libitum throughout the experimental period. All experiments were carried out according to the guidelines of the Animal Care and Use Committee at King Saud University.
Drugs and chemicals
Tacrolimus from Sigma Aldrich, USA and aliskiren from Novartis Ltd were used in the study. Biochemical parameters were done using kits (Dimension, Siemens, USA). All of the other chemicals used were of analytical grade.
Experimental protocol
Rats were randomly divided into four groups: Group 1, control group, received normal saline for 14 days. Group 2, toxic group, received tacrolimus (2 mg/kg, intraperitoneally (i.p.)) for 14 days. 23 Group 3, treatment group, received tacrolimus (2 mg/kg, i.p.) for 14 days similarly to group 2, and also ALK [(50 mg/kg, dissolved in distilled water administered per os (p.o.)] for 28 days. 24 Group 4, drug per se group, received ALK (50 mg/kg, dissolved in distilled water administered p.o.) for 28 days. 24
Tacrolimus administration (in group 2 and 3) started on day 15 and was continued till the end of the study. All the rats were sacrificed at the end of the study by decapitation under ether anesthesia, as per the protocol. Blood samples were collected followed by serum separation at 3000 g for 10 min. Samples were then kept at −20 ºC until analysis of renal function parameters.
The rats’ kidneys were isolated and washed in ice cold physiological saline and used for assessment of oxidative stress, histopathology and ultrastructural changes.
Biochemical estimation
Biochemical estimations were done by autoanalyzer (Dimension RXL MAX, Siemens, USA).
Determination of lipid peroxides, measured as malondialdehyde (MDA)
The level of MDA, a product of membrane lipid peroxidation, was estimated by reacting it with thiobarbituric acid (TBA), by the method of Okhawa et al., 25 using the standard calibration curve prepared from tetraethoxy propane. MDA was expressed as nmoles of MDA per mg of protein. Protein was estimated by the method of Lowry et al. 26
Determination of reduced glutathione (GSH)
GSH content was estimated by the method of Sedlack and Lindsay. 27 The absorbance of reaction mixture as read within 5 min of addition of dithibis-2-nitrobenzoic acid at 412 nm using a UV-spectrophotometer, against a reagent blank.
Determination of catalase (CAT)
Kidney tissue was homogenized and post-mitochondrial supernatant (PMS) was used to assay CAT activity. CAT activity was estimated using the method of Claiborne. 28 The reaction mixture consisted of 1.95 ml of phosphate buffer (0.1 M, pH 7.4), 1.0 ml of hydrogen peroxide (0.019 M) and 0.05 ml of PMS in a final volume of 3 ml. Changes in absorbance were recorded at 240 nm every minute for 5 min. The enzyme activity was calculated as nmoles of H2O2 consumed/min/mg protein.
Histopathological studies
Kidneys were harvested from the rats and fixed in 10% buffer formosaline. Paraffin sections of thickness 3–4 μm were prepared and stained with hematoxylin and eosin (H&E) for histopathological examination under light microscopy. 29
Ultrastructural studies
Immediately after removal of kidney from the dissected rats, tissues were sliced into small size (1 mm3) and fixed in 3% buffered glutaraldehyde. Tissue specimens were then post fixed in 1% osmium tetaroxide (OsO4) for 90 min. Dehydration of the fixed tissue was performed using ascending grades of ethanol followed by transfer of tissue to epoxy resin via propylene oxide. After impregnation with the pure resin (SPI Resin), tissue specimens were embedded in the same resin mixture. 31 Ultra-thin sections of silver shades (60–70 nm) were cut using an ultra-microtome (Leica, UCT, Tokyo, Japan) with a diamond knife; sections were then placed on copper grids and stained with uranyl acetate (20 min) and lead citrate (5 min). Stained sections were observed under Transmission Electron Microscopy (JEOL JEM-1011Tokyo, Japan) operating at 80 kV.30–32
Statistical analysis
All results are expressed as mean±standard error of the mean (SEM). Comparisons among different groups were analyzed by analysis of variance (ANOVA), followed by Tukey-Kramer multiple comparisons test to identify significance among groups. Values were considered statistically significant when p<0.05. Statistical analysis was carried out using GraphPad Prism 3.0.
Results
Effects of ALK on tacrolimus-induced parameters of renal function in serum
In this study, two weeks of treatment of rats resulted in tacrolimus-induced renal damage as evidenced by a significant (p<0.05) increase in serum creatinine and BUN levels as compared to the control group. Treatment with ALK significantly (p<0.05) decreased both serum creatinine and BUN levels compared to the toxic group. A significant (p<0.05) decrease in serum total protein and albumin was seen in the toxic group which was reversed by treatment with ALK. the drug group per se had no significant changes in any of parameters compared to control group (Table 1).
Effect of aliskiren on tacrolimus induced changes in renal function.

BUN: blood urea nitrogen.
The data are expressed as mean±standard error of the mean (SEM) (n=6).
p<0.05 vs control group; bp<0.05 vs toxic group. Analysis of variance (ANOVA) followed by Tukey-Kramer multiple comparison test.
Effects of ALK on tacrolimus-induced parameters of oxidative stress in kidney
The results are summarized in Figure 1–3. Two weeks treatments of rats with tacrolimus (2 mg/kg) resulted in a significant (p<0.05) increase in kidney MDA contents compared to the control group. Treatment with ALK showed a significant (p<0.05) decrease in tacrolimus-induced kidney MDA level (Figure 1). Consequently, a significant (p<0.05) decrease in kidney GSH level (Figure 2) and catalase activity (Figure 3) was found in tacrolimus-treated rats as compared to the control group. Treatment with ALK showed a significant increase in tacrolimus-induced kidney GSH level (p<0.05) and catalase activity (p<0.05). The drug per se group had no significant changes in any of parameters compared to control group.

Effect of aliskiren on tacrolimus-induced changes in lipid peroxidation in kidney of different experimental groups. The data are expressed as mean±standard error of the mean (SEM) (n=6). *p<0.05 vs control group; **p<0.05 vs toxic group. Analysis of variance (ANOVA) followed by Tukey-Kramer multiple comparison tests. MDA: malondialdehyde.

Effect of aliskiren on tacrolimus-induced changes in glutathione (GSH) levels in kidney of different experimental groups. The data are expressed as mean±standard error of the mean (SEM) (n=6). *p<0.05 vs control group; **p<0.05 vs toxic group. Analysis of variance (ANOVA) followed by Tukey-Kramer multiple comparison tests.

Effect of aliskiren on tacrolimus-induced changes in catalase activity in kidney of different experimental groups. The data are expressed as mean± standard error of the mean (SEM) (n=6). *p<0.05 vs control group; **p<0.05 vs toxic group. Analysis of variance (ANOVA) followed by Tukey-Kramer multiple comparison tests.
Effects of ALK on tacrolimus-induced parameters of histopathological changes in kidney
Normal morphological structures of renal tissue were seen in the control group (Figure 4a). However two weeks treatment with tacrolimus resulted in hypertrophy of epithelial cells and renal tubules epithelia degeneration. The dilatation of capillaries filled with erythrocytes was the most advanced change all over the kidney. Renal glomeruli and epithelial cell enlargement in the cortical part of the kidney were also observed. In a few renal tubules, single epithelial cells desquamated from the lumen were also noted (Figure 4b). Four weeks treatment with ALK ameliorated tacrolimus-induced damage in renal tissue to that observed in normal tissue (Figure 4c). No morphological changes were observed in the drug per se group (Figure 4d).
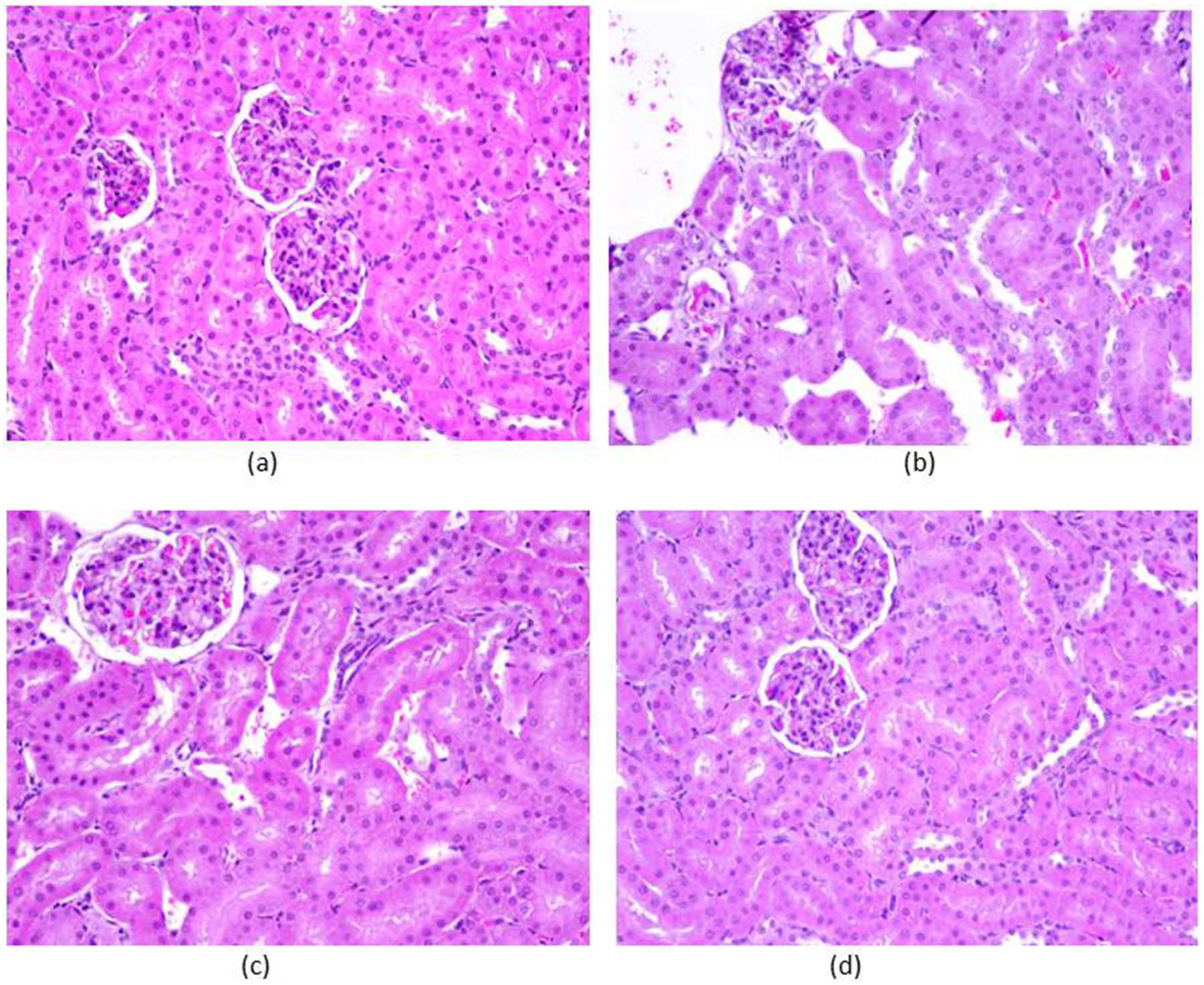
Effect of aliskiren on tacrolimus-induced changes histopathology in kidney of different experimental groups: (a) control group; (b) toxic group; (c) treatment group, and (d) drug per se group (n=6 per group; magnification ×20).
Effects of ALK on tacrolimus-induced parameters of ultrastructural changes in kidney
Ultrastructural changes in cellular structure were further visualized by transmission electron microscopy (TEM) studies. At ultra-structural level, normal structures of kidney were seen in the control group (Figure 5a). By contrast, nephropathic changes were observed in the cortico-medullary region of the toxic group which were characterized by glomerular damage, including dilatation of glomerular blood vessels, exfoliation and shedding of proximal tubular cells into the tubular lumen, thickened basement membrane, and interstitial inflammation (Figure 5b). Cellular damage was accompanied by ‘scarring’ of some glomeruli (focal glomerular sclerosis) and tubules (tubular interstitial fibrosis), as well as by tubular regeneration (Figure 5b). We also noticed degenerated mitochondria, numbers of multi-vesicle bodies, glomerular epithelial injury, and cell debris. Under normal conditions, tubular regeneration serves to restore the loss of damaged cells by a transient increase of cell proliferation. However, in ALK-treated rats, the kidneys demonstrated massive and sustained regenerative renal cell proliferation, resulting in simple tubular hyperplasia, indicated by a broad increase of cell number and multilayered tubules. ALK treatment was associated with normalization of the tacrolimus-induced renal cell damage and the treated rats showed normal cellular features (Figure 5c). Normal cellular structures were observed in the drug per se group (Figure 5d).

Effect of aliskiren on tacrolimus-induced ultrastructural changes in the kidney of different experimental groups: (a) control group; (b) toxic group; (c) treatment group, and (d) drug per se group (n=6 per group). Podocyte foot process and mitochondrial integrity were assessed using transmission electron microscopy (magnification= ×10,000).
Discussion
ALK is one of a new class of orally active agents which selectively inhibits renin enzymatic activity without affecting other systems involved in cardiovascular regulation. Recently, in some studies, the protective effects of ALK on nephrotoxicity were evaluated using animal models (acute murine model) 33 or drugs (such as gentamycin and doxorubicin).19,34 The concentration of creatinine and BUN depend on the glomerular filtration rate (GFR). Renal dysfunction reduces glomerular filtration of creatinine and BUN, and thus levels of creatinine and BUN rise. If the serum creatinine and BUN levels double, the GFR is considered to have been halved.34,35 Previous reports have confirmed that tacrolimus-induced nephropathy is manifested by elevation in the serum levels of creatinine and BUN.19,34,36,37 In the present study, tacrolimus-treated rats showed elevated levels of serum creatinine and BUN. However, treatment with ALK caused a significant reduction in creatinine and BUN levels which may be due to improvement in glomerular filtration damage induced by tacrolimus. These results are in agreement with an earlier report. 38 Hypo-albuminemia is a characteristic feature of the nephrotic syndrome. 39 In this study, serum albumin level was found to be low in tacrolimus-treated rats, similar to the findings of previous studies.40–42 Treatment with ALK reversed tacrolimus-induced hypo-albuminemia. 38 Therefore, it is suggested that ALK improves renal function by counteracting tacrolimus-induced nephropathy.
In our study, we observed a significant increase in the concentration of MDA level in renal tissues of tacrolimus-treated animals as compared to the control group. The elevated level of MDA may be due to enhanced production of ROS (superoxide radicals, hydrogen peroxide and hydroxyl radicals). Ibrahim et al., also reported a significant increase in kidney MDA content in rats with a cumulative dose of tacrolimus. 33 Similar studies were also reported by other authors.43,44 Zhou et al., in 2004, reported that tacrolimus enhanced production of ROS leading to cell death. 17 In this study, treatment with ALK significantly decreased renal MDA level which suggests its role in combating oxidative stress generated by tacrolimus. As MDA is a secondary product of lipid peroxidation of unsaturated fatty acids, its decrease by ALK could be taken as an index for decreased oxidative stress and subsequent reduction in cytotoxicity. 45
Tissue GSH depletion is one of the primary factors which permit lipid peroxidation. 46 The decreased CAT activity in renal tissue may be due to excess production of superoxide and hydrogen peroxide which promote lipid peroxide formation. In the present study, administration of tacrolimus decreased GSH level and CAT activity as compared to the control group, while treatment with ALK reversed the tacrolimus-induced decrease in renal GSH content and CAT activity. Increased intracellular GSH content and CAT activity might be due to up-regulation of enzymatic/nonenzymatic antioxidants or decreased ROS production in the ALK-treated group. ALK has been shown to decrease oxidative stress in earlier studies.47,48
In histopathological examination of the toxic group, the epithelia of the renal tubules show degeneration with infiltration of mononuclear cells, dilation of glomeruli as well as hyperemia of medullary and cortical parts. The dilatation of capillaries filled with erythrocytes was the most advanced change all over the kidney. Further, the present study showed tubular vacuolization in toxic group. Examination of the renal cellular ultrastructure showed round, oval mitochondria within the tubular epithelium in tacrolimus-treated rats. Further ultrastructural analysis showed disturbances in podocyte foot processes i.e. podocyte foot injury and podocyte foot process effacement. Similar histopathological and ultrastructural changes are associated with tacrolimus-related nephrotoxicity, including tubular vacuolisation, arteriolar hyalinosis, interstitial fibrosis and juxtaglomerular hyperplasia which have been reported earlier.38,49–52 Tacrolimus-induced histopathological and ultrastructural changes were reversed by treatment with ALK. There was restoration of podocyte architecture along with epithelial integrity after treatment with ALK. Tubular vacuolization induced by tacrolimus was also attenuated by treatment with ALK. Earlier studies have shown that RAS blockade leads to preservation of podocyte architecture, mitochondrial function and epithelial integrity.22,47,53
Some additional parameters like measurement of blood pressure and plasma renin activity would have further strengthened our findings, however these could not be measured and this is a limitation of the current study.
Treatment with ALK ameliorated tacrolimus-induced nephrotoxicity implying that renin inhibition leads to improvement in renal function. The protective effect of ALK was accompanied by a significant attenuation of oxidative stress parameters in kidney (MDA, GSH and CAT), improvement in renal function biochemistry in serum as well as restoration of renal structures as observed by histopathological and ultrastructural findings. Thus, the current study suggests that ALK treatment may protect against nephrotic syndrome associated with immunosuppressant use.
Footnotes
Acknowledgements
The authors acknowledge the Department of Pharmacology and Toxicology, College of Pharmacy, King Saud University for its facilities and funding from Deanship of Scientific Research, College of Pharmacy (Project No. RGP-VPP-305).
Conflicts of interest
The authors declare that there are no conflicts of interest.
Funding
The present work was funded by the Deanship of Scientific Research, King Saud University, Riyadh, Kingdom of Saudi Arabia (Project No. RGP-VPP-305).
