Abstract
Introduction:
Despite recognition of stress as a causation of severe neuropsychological dysfunctions, no casual and clinically effective anti-stress therapeutic strategy has yet been found. We have previously shown that blockade of initial stress response by angiotensin receptor blockers alleviates the negative effect of prolonged stress on cognitive non-spatial functions of rats. Here we aimed to find whether telmisartan reduces stress-related memory decline in spatial hippocampal-dependent learning tasks conditioned upon differences in level of stress induced by aversive nature of memory tests.
Methods:
Male Wistar rats were exposed to chronic restraint stress for three weeks and daily treated with either vehicle or telmisartan (1 mg/kg). Afterwards rats were tested in three spatial learning and memory paradigms: Morris water maze (MWM), radial arm maze (RAM), and Barnes maze (BM).
Results:
Stressed animals demonstrated significantly impaired performance in all the tests, which was normalized in the animals stressed and treated with telmisartan. Interestingly, despite the fact that MWM and RAM are more stressful, which affects animal behavior, therefore considered less sensitive than BM, more significant effect of telmisartan was found in MWM and RAM than BM.
Conclusions:
AT1 angiotensin receptor blockade attenuates negative effect of both acute and chronic stress on spatial memory.
Abbreviations
ACE-I Angiotensin-converting enzyme inhibitor
Ang II Angiotensin II
ANOVA I One-way analysis of variance
ARB Angiotensin II type 1 receptor blocker
AT1 Angiotensin II type 1 receptor
BM Barnes maze
CNS Central nervous system
HPA Hypothalamic-pituitary-adrenal axis
LTP Long-term potentiation
MWM Morris water maze
PPARγ Peroxisome proliferator-activated receptor γ
RAM Radial arm maze
SEM Standard error of mean
Introduction
Stressful experiences are generally well remembered, 1 especially if the context of perceived stress matches the context of the information being encoded. 2
However, this is an example of adaptive behavior in healthy individuals and deregulation of the underlying mechanism might result in cognitive decline. 3 Chronic and acute stress modulates learning and memory processes, acquisition, consolidation, retention, and retrieval. Prolonged experience of continuous physiological arousal negatively affects cognitive performance. 4 Acute stimulus might enhance memory performance, 5 but acute intense event in previously stressed subjects results in amplification of hypothalamic-pituitary-adrenal (HPA) axis deregulation, which might trigger memory loss. 6
Stress exceeding adaptation mechanisms is potentially harmful due to over activity of three major neuroendocrine systems – the sympatho-adrenergic system (SAS), the HPA axis, and the renin–angiotensin system (RAS). 7 However, this effect might be reduced by suppression of the brain RAS system, as we have previously shown using highly selective angiotensin type one (AT1) receptor blockade, which effectively prevented deleterious effects of chronic stress on retrieval of memory in rats.8,9
In this study, we attempted to evaluate the effect of AT1 receptor blockade using telmisartan in hippocampal-dependent memory tasks. Spatial and non-spatial cognition are two important aspects of cognitive function. Various types of behavioral tasks have been designed to evaluate specific type of memory in rodents.10–13 Most of the spatial memory tasks use positive or negative reinforces, such as food for radial arm maze (RAM) or water immersion for a Morris water maze (MWM) test, however those effects are minimized in Barnes maze (BM). Therefore, we decided to use all three paradigms to evaluate potential protective effect of telmisartan on memory loss due to chronic stress and learning abilities under stressful conditions in a variety of intensification. Moreover, using mazes with diverse reinforces seems useful to incorporate results of animal studies with the human population.
Materials and methods
Animals
The experiments were conducted on male Wistar rats, weighing approximately 150 g at the beginning. Animals were housed five to a cage in a temperature (22°C) and humidity (50–60%) controlled room, on a 12 h: 12 h light/dark cycle with light on from 6:00 a.m. Free access to standard laboratory food and tap water was provided. All animals were handled daily for 2 min each until the day of experiment. All the procedures were conducted between 1:00 p.m. and 6:00 p.m. A 30 min adaptation period in the experimental room preceded all the tests. To minimize the influence of hemodynamic changes, we did not study hypertensive animals. All procedures involving animals were approved by the local Ethics Commission for Animal Experimentation.
Drugs
The specific AT1 receptor antagonist – telmisartan – was suspended in 0.5% methylcellulose (vehicle) at concentration of 1 mg/ml. Telmisartan or its vehicle was dosed by oral gavage at 1 mg/kg body weight, considered to be a non-hypotensive dose in rats.14,15 One hundred and twenty male Wistar rats were randomly assigned to four groups: (1) 30 control rats – receiving 0.5% methylcellulose as a vehicle; (2) 30 rats receiving telmisartan suspended in 0.5% methylcellulose; (3) 30 rats receiving 0.5% methylcellulose subjected to repeated restraint stress procedure; (4) 30 rats receiving telmisartan suspended in 0.5% methylcellulose subjected to repeated restraint stress procedure. The subjects received either telmisartan or vehicle each day approximately 30 min before the beginning of stressing procedure. Animal weight was measured each day prior to drug administration.
Stress procedure
Two groups of animals (30 rats each) were subjected to chronic restraint stress, 2 h daily for 21 days.16,17 The restraint was imposed during the light phase from 1:00 p.m. to 3:00 p.m. The restrainer was made of transparent perforated plastic tube, 20 cm long, and 7 cm in diameter. A rat was eased into the restrainer, head first, and once in the tube it was closed with a Plexiglass lid. The animals fit tightly into the restrainers and it was not possible for them to move or turn around. At the same time not stressed control rats were briefly handled and returned to their home cages. Randomly chosen animals subjected to stress were checked for gastric ulceration on the next day after ending the behavioral tests. Rats were anaesthetized with the mixture of ketamine (50 mg/kg) and xylazine (7.5 mg/kg) injected intraperitoneally and sacrificed. Exposed gastric mucosa was visually examined under the 5× magnification lens for gastric ulceration. No visible signs of injury were found, that could potentially interfere with the results of behavioral tests.
Behavioral tests
Hippocampal-dependent cognitive performance of rats was estimated the next day after ending stress and drug administration procedures. Three different paradigms with three different levels of stress induced by the procedure were used to assess its influence on working memory. Moreover this work has benefited from evaluating spatial perception, spatial cognition, and spatially directed action in both allocentric (MWM, BM) and egocentric (RAM) representations. Each animal would participate in one behavioral paradigm only (40 animals assigned to MWM, 40 participating in BM and 40 tested in RAM, from total number of 120 rats).
Morris water maze
Forty rats (different from those used for RAM and BM) randomly assigned to four groups as described above were used. This task was adapted from the paradigm originally described by Morris. 13 The water maze was a black circular pool (180 cm in diameter, 60 cm high), filled with water (26±1°C) made opaque with a grain instant coffee, to the depth of 30 cm, placed in a room rich in consistently located spatial cues (door, shelves, illumination lights, posters). The pool was divided into four quadrants designated Northeast (NE), Northwest (NW), Southeast (SE), and Southwest (SW). An escape platform (9 cm in diameter) was placed in the middle of one quadrant (NW), 1.5 cm below the water surface, equidistant from the sidewall and middle of the pool. The platform provided the only escape from the water and was located in the same quadrant conducted through trials each day, but its location was changed daily. Four different starting points for rats were equally spaced around the perimeter of the pool (N, E, S, and W). On each of the three training days, three start points were used once each in a pseudo-random sequence. A trial began by placing the animal in the water facing the wall of the pool at one of the starting points. If the animal failed to escape on the platform within 60 s it was gently placed there by the experimenter and allowed to stay for 15 s.
The interval was 5 min. After each trial, the rats were towel and fan dried and returned to their home cages. All animals were trained one session of three trials daily for three consecutive days. The latency to reach the original position of the platform, the number of crossings over its previous location during 1 min, and time spent by rat in the target quadrant and in the opposite quadrant and the border zone were measured manually.
Eight-arm radial maze
Forty rats (different from those used for MWM and BM) randomly assigned to four groups as described above were used. The apparatus was an elevated eight arm radial-maze which was made of 0.5 cm thick gray wooden planks. Each arm (68 × 10 cm2) extended from an octagonal-shaped center hub (30 cm in diameter). A small cup (4.5 cm in diameter) was placed at the end of each arm. The maze was elevated 80 cm above the floor and placed in a sound attenuated room with a masking noise of 70 dB above the human thresholds. 18 The rats were submitted to three habituation sessions (one per day). They were put on the central platform and allowed to explore the maze entries for 10min.At the end of habituation stage, they were deprived of food for 24 h before the beginning of the training phase. Rats were given 12 sessions (one per day). The training session lasted until the rat made eight choices or 15 min elapsed. The cups at the end of each arm contained arachnid peanuts. The experimenter recorded order of the eight choices made by each rat. One hour after the completion of the session, rats were given access to food for 10 min. The following data was considered: mean number of errors – an error was an entrance in a previously visited arm; mean number of correct entries – correct choices made in sequence before the first error in each session; mean time spend by rats to visit eight maze arms within each session.
Barnes maze
Forty rats (different from those used for MWM and RAM) randomly assigned to four groups as described above were used. On the first day of testing, the animals (were brought in the testing suite) and allowed to enter the goal box through one of the holes in the circular surface (120 cm diameter) of the maze. Once the animal entered the hole, a black Plexiglass cover was positioned over the hole to prevent escape. A 4-min habituation in the goal box was given prior to the first training trial. Next, the animal was placed for 30 s in a 20 cm diameter by 30 cm high round nontransparent holding box that was positioned in the center of the maze. The holding box was then removed, a timer begun, and the experimenter moved behind the curtain. An escape was counted when all four paws of the animal were in the goal box. Following successful location of the goal box, the animals were allowed to stay there for 60 s. A maximum of 4 min was allowed for each trial and if an animal did not locate in the goal box during this 4 min period it was removed from the maze and placed in the goal box for 60 s. During the 60 s period in the goal box, the maze was wiped with a paper towel to remove any feces or urine prior to the beginning of the next trial. The test paradigm consisted of two trials per day for 3 days. Before the start of the second trial, the animal was returned to its home cage for 1 min and then placed again in the holding box in the center of the maze to start the second trial as described above. The maze was then rotated a random number of holes (between 1 and 8 holes using a random numbers table) to prevent use of odor trails in solving the task. The hole under which was the escape tunnel was changed to assure its constant position relative to extra-maze cues (shelves, posters, and laboratory bench) for 3 days. Following the rotation of the table, the procedure was repeated with the next animal. Two measures were recorded on each trial. The first was the latency to find the goal box. The second was the number of errors committed by each animal. An error was defined as a head poke or exploration of any hole other than the hole above the goal box and including preservative investigations of the same hole. At the end of each trial the maze was cleaned using a 70% ethanol solution. 11
Statistical analysis
Data were presented as means of ± standard error of mean (SEM). A two-way analysis of variance (ANOVA II) (treatment × days) with repeated measures followed by the post hoc Bonferroni test for multiple comparisons was used for latencies to reach platform in the MWM and mean number of errors, latencies, and number of correct entries in RAM. One-way analysis of variance (ANOVA I), followed by post hoc Bonferroni test for chosen group comparisons, was applied for calculating performance of the rats in MWM (savings ratios, border zone time, and swimming speed), Barnes maze (escape latencies and number of errors), and body weight gain. Levels were deemed significant at p < 0.05.
Results
Effects of chronic stress, chronic telmisartan treatment, or both in combination on rats’ performance in the Morris water maze
Figure 1 presents mean ± SEM escape latency in the MWM. The stressed subjects reached the platform significantly later than the control (p < 0.01), telmisartan (p < 0.001), and stress + telmisartan (p < 0.05) group of rats. Telmisartan administration not only abolished adverse effects of stress on spatial working memory, but also improved performance of not stressed subjects. ANOVA II of the latencies to reach platform in the water maze revealed significant treatment effect (F3,36 = 7.226; p < 0.001), but no significant treatments × days interaction (F6,72 = 1.367; p > 0.05). Nevertheless, the fact that most of the rats effectively learned the task meant the day effect was not significant (F2,6 = 1.818; p > 0.05).

Effects of chronic stress (daily restraint for 2 h, 21 days), chronic telmisartan (1 mg/kg p.o., daily, 21 days), or both in combination on mean ± SEM escape latency in the Morris water maze over three days (three trials per day), n = 10. *p < 0.05 stress + telmisartan vs. stress, **p < 0.01 control vs. stress, ***p < 0.001 telmisartan vs. stress.
Figure 2 presents mean ± SEM of the time spent in border zone in the MWM. ANOVA I of the time spent in the border zone from nine trials yielded F3,36 = 4.663; p < 0.05, indicating that stressed subjects spent significantly more time in the border zone than non-stressed rats. This effect was abolished by telmisartan treatment of stressed rats.

Effects of chronic stress (daily restraint for 2 h, 21 days), chronic telmisartan (1 mg/kg p.o., daily, 21 days), or both in combination on the task-solving strategy in the Morris water maze. Bars represent means + SEM of the time spend in the border zone from nine trials (three trials per day for three days), n = 10. *p < 0.05 vs. control and telmisartan.
Figure 3 presents performance of rats in MWM expressed as mean ± SEM savings ratio, a proportion of second trial latency to total trial time for the session. According to the interpretation suggested by Glenn and Mumby, 19 the smaller savings ratios the better retention of the platform position. ANOVA I of the calculated savings ratios from MWM test yielded F3,36 = 4.595; p < 0.05, showing statistically significant differences between stressed rats in comparison to control and telmisartan groups.

Effects of chronic stress (daily restraint for 2 h, 21 days), chronic telmisartan (1 mg/kg p.o., daily, 21 days), or both in combination on working memory in the Morris water maze expressed in mean savings ratios. Bars represent means + SEM, n = 10. *p < 0.05 vs. control and telmisartan.
Figure 4 presents mean ± SEM swimming speed, calculated as number of line crossings divided by escape latency. The results suggest that all rats were performing the task with equal swimming speed and this parameter did not influence the outcome of their performance. ANOVA I of the results obtained in MWM test revealed no significant (F3,36 = 3.265; p > 0.05) differences between the groups.

Effects of chronic stress (daily restraint for 2 h, 21 days), chronic telmisartan (1 mg/kg p.o., daily, 21 days), or both in combination on swimming speed (line crossed/s) in the Morris water maze. Bars represent means + SEM, n = 10. No statistically significant differences were found.
Effects of chronic stress, chronic telmisartan treatment, or both in combination on rats’ performance in the radial arm maze
Figure 5(a) presents the mean ± SEM numbers of errors, cumulated in three four-day blocks. ANOVA II of these results yielded F2,6 = 20.523; p < 0.001, showing significant decrease of the errors made by all rats across 12 days. No significant treatments × days interaction was found (F6,72 = 0.576; p > 0.05). ANOVA I of the total number of errors made by the rats over 12 days yielded F2,6 = 7.496, p < 0.01 showing significant treatment effect. These results demonstrate that impairment of working memory displayed by chronically stressed rats that is partially diminished by telmisartan.

Points represent means ± SEM from 4 days each of the values obtained from 10 animals. (a) Mean number of errors, **p < 0.01 control and telmisartan vs. stress; (b) mean number of correct sequential entries before the first error, **p < 0.01 control vs. stress, *p < 0.05 telmisartan vs. stress; (c) mean time of eight arms exploration, *p < 0.05 control and telmisartan vs. stress.
Figure 5(b) presents mean number ± SEM of correct and sequential entries to arms of the radial maze, prior to first error. ANOVA II of the daily numbers of the correct entries made by each rat over 12 days yielded F2,6 = 18.791; p < 0.001, showing significant time effect, i.e. increase of the correct entries made by all rats across 12 days. No significant treatments × days interaction was found (F6,72 = 0.664; p > 0.05). ANOVA I of the total number of the correct entries made by each rat over 12 days yielded F3,36 = 5.592; p < 0.01 control vs stress; p < 0.05 telmisartan vs stress.
Figure 5(c) presents the mean ± SEM of the time spent by rats visiting eight arms. ANOVA II of the times used by all rats yielded F6,72 = 4,38; p < 0.001, pointing to the significant increase of the rats performance in time of visiting maze arms. Significant treatments × days interaction was found (F6,72 = 4.380; p < 0.001). ANOVA I of the times spent by rats visiting all arms over 12 days yielded F3,36 = 3.726; p < 0.05, showing significant differences among stressed and not stressed subjects.
Effects of chronic stress, chronic telmisartan treatment, or both in combination on rats’ performance in the Barnes maze
Figure 6 presents mean ± SEM number of errors made by rats in the Barnes maze. ANOVA I of the results reviled significant difference between stressed and control subjects (F3,36 = 2.867; p < 0.05) in selected pair comparison. Telmisartan treatment of stressed rats reduced this effect but further post hoc comparisons made with Bonferroni revealed that the difference is not statically significant.

Effects of chronic stress (daily restraint for 2 h, 21 days), chronic telmisartan (1 mg/kg p.o., daily, 21 days), or both in combination on working memory in the Barnes maze. Bars represent mean + SEM number of errors from six trials (two trials per day for three days), n = 10. *p < 0.05 stress vs. control.
Figure 7 presents mean ± SEM of escape latency in the Barnes maze. Exploration time was extended in the stressed subjects and this effect was lowered in telmisartan treated group, but ANOVA I of the results from all the trials and post hoc comparisons made with Bonferroni revealed that the differences are not statically significant.
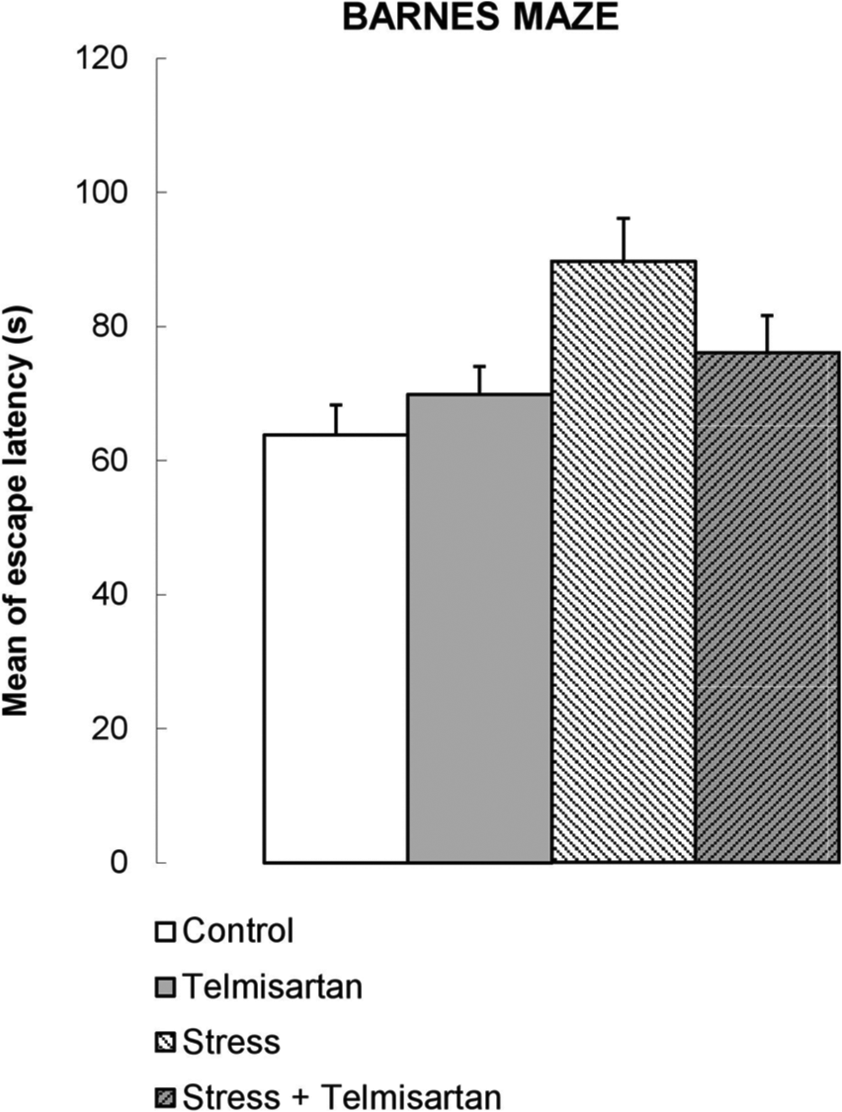
Effects of chronic stress (daily restraint for 2 h, 21 days), chronic telmisartan (1 mg/kg p.o., daily, 21 days), or both in combination on working memory in the Barnes maze. Bars represent mean + SEM time of escape latency from six trials (two trials per day for three days), n = 10. No statistically significant differences were found.
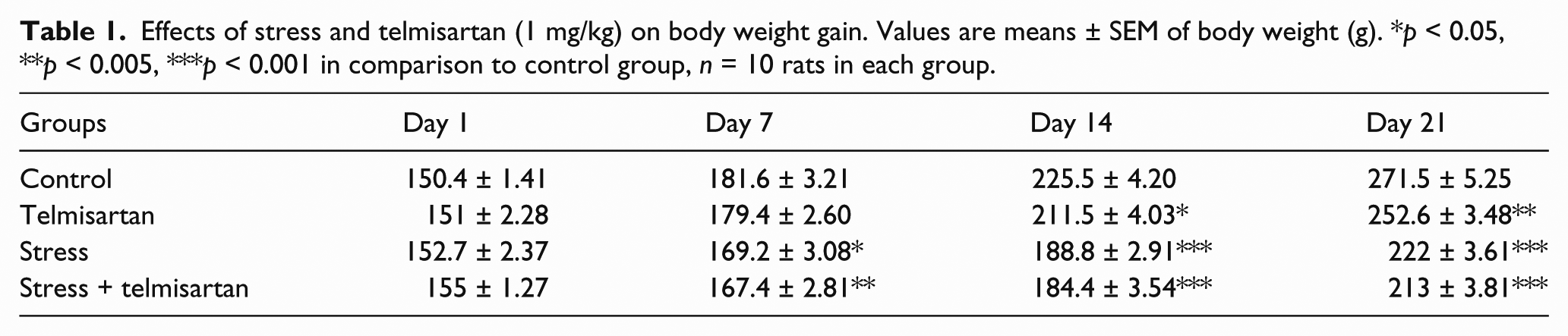
Effects of stress and telmisartan on body weight gain
As shown in Table 1, all of the rats gained weight during the course of the study, but growth rate differed among the groups. Restraint induced a significant weight loss (p < 0.001) that was even greater in the rats receiving telmisartan. The effect became apparent during the first week of restraint. Moreover, animals that were not subjected to stress procedure and received telmisartan also gained significantly (p < 0.005) less weight than control group; however, this effect appeared after two weeks of the study.
Effects of stress and telmisartan (1 mg/kg) on body weight gain. Values are means ± SEM of body weight (g). *p < 0.05, **p < 0.005, ***p < 0.001 in comparison to control group, n = 10 rats in each group.
Discussion
Despite rapid development of technology and biochemical methods, behavioral tests are still recognized as very sensitive tools in testing animal cognition, which might reveal minor hippocampal-induced impairment before, or even in the absence of, neuron death. 20 Although the Morris water maze, the radial arm maze and the Barnes maze are well established means of testing spatial memory,11,13,21,22 neither might be considered optimal for studying the behavioral effects of stress. The MWM is itself highly stressful to the animals. 23 The RAM relies on a food reward to motivate the animals, and glucocorticoids have profound effects on hunger and satiety. 24 While strong aversive stimuli (i.e. water or food deprivation) are likely to produce stress in the animal that influences the performance in the task, weak aversive stimulation might result in lack of motivation to escape from the circular platform in the Barnes maze. 25 Performance of rodents with hippocampal damage in all the mazes mentioned above is impaired, which strongly supports hypothesis of the spatial nature of all the tasks.26,27 All the paradigms are challenging tasks for rodents and employ a variety of sophisticated mnemonic processes. 28 These processes comprise the acquisition and spatial localization of relevant visual cues that are subsequently processed, consolidated, retained, and then retrieved in order to successfully navigate using allocentric (MWM, BM) or egocentric (RAM) representations. The general processes used for visuospatial navigation in rats also contribute considerably to human day-to-day cognitive processes. 29 Therefore, to broaden utility of this study all three paradigms were used (MWM, RAM, and BM) as it might become relevant to neurodegenerative and neuropsychiatric illnesses where cognition is impaired.30–32
As a continuation of our previous studies, 9 and screening for compounds with potential cognitive enhancing effects, as well as delineating deleterious effects of stress on cognition, we proceeded with development studies of telmisartan. The results of the present study clearly demonstrate that pre-treatment with telmisartan significantly reduces stress-related cognitive impairment in rats. Parameters of cognitive performance (latencies, errors and correct choices, savings ratios) in MWM, RAM, and BM were significantly deteriorated in chronically stressed subjects. This effect was reduced in stressed subjects that received telmisartan. Interestingly, the most profound effect was reviled in the MWM, which is the most stressful paradigm therefore, capitalizes upon aversive motivation. Moreover, the MWM allows for the assessment of spatial memory without potential experimental manipulation incorporating food reward. Rats exhibit a robust increase in corticosterone on the first day of water maze testing that is stable and only slightly diminished throughout days of testing. 33 No such data are available for the RAM and BM. According to inverted-U-shaped fashion, suggesting that an optimal level of corticosterone will enhance memory and higher levels may impair it, 26 telmisartan treatment presumably diminish excessive release of corticosterone in stressed subjects, suppressing over-activation of the HPA axis and enhanced corticotrophin-releasing hormone, as has been recently described by Braszko et al. 8 Noteworthy, time latencies in the MWM seems to efface among the stressed and non-stressed subjects on the second and the third day of testing. It is possible that control rats sooner than stressed rats switch from spatial to non-spatial strategies with overtraining. 4 The least profound effect of protective outcome of telmisartan treatment in stress induced cognitive decline was reviled in the Barnes maze, the least stressful of all three mazes. Number of errors made by stressed subjects in the BM was significantly higher and telmisartan treatment reduced this effect, but no statistically significant differences in the escape latency were found. However, unitary measures such as latency and path length are more susceptible to non-mnemonic influences and are not the best indicators of cognitive performance in learning tasks. 34 Moreover, the BM induces a modest increase in corticosterone, 35 which might be insufficient for motivation of animals to perform in the task. 36 In our study, adaptation procedure was not effective enough, to exert animals for more readily exploration of the holes on the surface of circular platform of the BM. Rats often hesitated to enter the escape hole and started to inspect other holes, what clearly had a direct effect on latency and distance measures. Control rats needed more than 60 s to find the escape hole in the BM, which is relatively long when compared to the MWM performance (<45 s) in which the diameter of the apparatus is much longer. Execution of the task in the RAM was impaired in the stressed subjects and expressed by mean number of errors. Interestingly mean number of correct entries was slightly lower in rats receiving telmisartan, as those rats chose more often not to enter arms sequentially, but without committing more errors than controls. Despite the fact that RAM is generally used to measure spatial memory, using this paradigm has notable limits. For example, even blind rats can learn to enter arms in one direction (i.e. constantly turn left), which might be a failure in long-term testing procedure, when hippocampal mediated processes become independent from the hippocampus over weeks. 37 Another limitation for using RAM in stressed subjects, is the fact that chronic stress may influence motivation with clearly interferes with the results of the test. The effects of stress and telmisartan treatment tested in the RAM were slightly less profound than in MWM, but more expressed in comparison to the BM. The data presented here, indicating the differences among three paradigms, are assumed to be caused by the negative correlation between learning and level of corticosterone release. However, certain causative direction cannot be drawn and needs further investigation.
Repeated restraint is used in this study as a model of moderate psychological stress, which is a complex response that also affects metabolism, therefore commonly considered as an animal model of anorexia nervosa. 38 Repeated stressors in rats generally seem to reduce food intake and body weight. 39 Stress-induced effects on metabolism and energy balance have been demonstrated as a result of repeated daily restraint and as an acute response in the hours immediately after a single restraint. 40 In our study weight loss induced by repeated restraint appeared within the first week of stress sessions and became more pronounced after each exposure to stress, which indirectly indicates the effectiveness of the restraint procedure. Significant attenuation of weight gain was also a result of telmisartan administration, which is not very surprising since reduced adipogenesis and weight gain have already been reported as the effects of this ARB in rodents. 41 In addition to being a blocker of AT1 receptor, telmisartan is also a partial agonist of the peroxisome proliferator–activated receptor-gamma (PPAR-γ), a well-known target of insulin sensitizing drugs used to treat type 2 diabetes. Only telmisartan, and to a lesser extent candesartan, are the only commercially available ARBs producing PPAR-γ activation, when tested at concentrations typically achieved in plasma with conventional oral dosing.42,43 Telmisartan influences the expression of PPAR target genes involved in carbohydrate and lipid metabolism and reduction of glucose, insulin, and triglyceride levels, 44 but the significance of PPAR-γ activation in this study goes beyond its metabolic effects. These nuclear receptors are considered to be attractive targets for the treatment of cognitive impairment due to their ability to suppress microglial-mediated inflammatory responses, 45 promote tissue repair, 46 and induce the degradation of the Aβ peptides in the brain by activating genes responsible for reverse cholesterol transport. 47 Nevertheless, PPAR-γ are expressed in the fetal brain as a factor involved in cellular development, its expression is rather limited in the healthy mature brain. 48 Moreover, the use of the PPAR-γ activators (i.e. rosiglitazone, pioglitazone) for CNS-targeted disease treatments is compromised due to their poor blood brain barrier (BBB) penetrance. 49 Telmisartan, however, effectively penetrates BBB in a dose- and time-dependent manner, to inhibit centrally mediated effects of Ang II for over 4 hours following peripheral administration of a dose as low as 1 mg/kg. 50 However, it is essential that a non-hypotensive dose of 1 mg/kg does not result in a significant reduction in blood pressure and does not affect cerebral blood flow. 51 Although blood pressure (BP), even in normotensive rats, as a continuous, not a static, variable might be altered due to restraint, which clearly is a factor for development and progression of cognitive decline, the negative effects of stress are simultaneously mediated through angiotensin II and glucocorticoids, affecting both circadian BP rhythm and cognitive performance. Therefore, stress induced alterations of BP cannot be considered as an independent trigger of memory loss. Stress induces an increase in BBB permeability, 52 and disruptions of BBB are casually associated with cognitive decline. 53 Interestingly, treatment with an ARB ameliorates cognitive impairment in type-2 diabetic subjects, 54 which is caused most likely due to decrease in BBB permeability. 55 Moreover, recent studies suggest that salt-dependent hypertension and activation of the brain RAS contributes to BBB malfunction and cognitive decline, which is reversed with an ARB treatment independent of blood pressure changes, by preventing BBB permeability. 56 These data indicates importance of the brain RAS in the pathogenesis of impaired cognitive performance. The role of the Ang II is well established in a complexity of sympathoadrenal response. 57 In rats, high density of AT1 receptors is present in brain regions that are involved in the regulation of sympathetic tone, such as the endothelial cells and neurons of the circumventricular organs outside the BBB (e.g. SFO, OVLT), cerebrovascular endothelial cells of the BBB, and neuronal AT1 receptors selectively localized in many brain areas located inside the BBB (e.g., PVN, SCh, and MnPO). 58 Significant numbers of AT1 receptors are located in higher stress-regulatory structures: hippocampus, septum and amygdala. AT1 receptors are also expressed in parvocellular corticotrophin-releasing factor (CRF) forming neurons controlling CRF release during stress. 59 These brain structures are important therapeutic targets for ARBs, especially those lipophilic like telmisartan that readily crosses BBB to inhibit the brain RAS, 50 as a treatment of stress related CNS disorders. It has been reported that sartans normalize cerebrovascular remodeling and autoregulation in hypertension, prevents and reverses cerebrovascular inflammation, reduces vulnerability to brain ischemia, and protects from stroke, 60 but explanation of many positive effects of ARBs would go beyond AT1 receptor blockade. Besides central functions of Ang II, other naturally occurring neuroactive peptides of the RAS are also involved in memory processing. 61 The effects of angiotensin IV (Ang IV) and des-Phe(6)Ang IV include improvement of several memory aspects (i.e. recall of appetitively and aversively motivated behaviors and learning of spatial tasks). Moreover, angiotensin metabolites modulate central functions by increasing levels of some of the other brain peptides, including oxytocin, which is associated with anxiolytic effects. 62
As a consequence of AT1 blockade, ARBs increase not only Ang II levels, but also levels of Ang metabolites. Interestingly, some data suggest that angiotensin neuropeptide release, occurring in mild stressed subjects, results in memory enhancement that could be attenuated by both ACE-inhibitors and ARBs. 63 ACE-inhibitors decreases the formation of Ang II, Ang III, and Ang IV from Ang I, while ARBs selectively block AT1 receptor, thus diminishing Ang II-mediated biological actions without affecting the actions of other angiotensin neuropeptides, suggesting the key role of Ang II not only in stress-induced memory impairment but also enhancement. This indicates flexibility and complexity of brain RAS system demanding further evaluation conditioned upon different levels of stress.
Nevertheless, accumulating preclinical studies and clinical reports call attention on the therapeutic and neuroprotective effects of ARBs,64,65 including stress attenuating properties of telmisartan,14,66–68 although at this point narrowing the group of ARBs, excluding those that are hydrophilic (i.e. losartan, eprosartan),69,70 to indicate the most potent of withdrawing negative effects of stress, seems unreasonable.
In conclusion, data presented in this study indicate that AT1 angiotensin receptor blockade with telmisartan attenuates negative effect of both, acute and chronic stress on memory, which is a novelty in the pursuit of effective therapeutic approach to stress induced cognitive decline.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Medical University of Bialystok.
