Abstract
Aim:
To investigate the effect of telmisartan on the lipometabolisms and the proinflammatory factors secreted from 3T3-L1 adipocytes and to explore the possible mechanisms.
Materials and methods:
Telmisartan was applied to interfere with mature 3T3-L1 adipocytes. The culture’s free fatty acids, interleukin 6 (IL-6) and tumor necrosis factor α (TNFα) were evaluated. Oil Red O staining was used to determine the adipogenesis of 3T3-L1 adipocytes. 18F-FDG uptake levels corrected for protein content were determined by cellular radioactivity. The total RNA was isolated for hybridization experimentation in the microarray.
Results:
Telmisartan reduced lipid storage and increased 18F-FDG uptake in a dose-dependent manner, reduced the levels of IL-6 and TNFα and increased those of free fatty acids. One hundred and fifty-seven differentially expressed genes were found by microarray. The mitogen-activated protein kinase (MAPK) signaling pathway involved in the secretion of proinflammatory factor and lipid metabolisms was affected by telmisartan. The expression of endothelial nitric oxide synthetase gene 3 (Nos3) and carnitine palmitoyl transferase 1α (CPT1α) was up-regulated by telmisartan.
Conclusions:
Telmisartan affected lipometabolisms and the proinflammatory factors secreted from adipocytes. Nos3, CPT1α and the MAPK pathway being affected by telmisartan may be the underlying cause of the improvement in lipid metabolisms and secretion of proinflammatory factors of differentiated 3T3-L1 adipocytes.
Introduction
The metabolic syndrome, as the cluster of hypertension, diabetes mellitus, dyslipidemia and body obesity, is a common cause of atherosclerotic cardiovascular diseases and one of the most serious threats to public health. It is understood that adipose tissue produces and secretes some proinflammatory factors. Dysregulated production of proinflammatory factors, such as tumor necrosis factor-α (TNFα), interleukin-1β (IL-1β) and interleukin-6 (IL-6), is associated with the pathophysiology of obesity-related disorders.
Patients with essential hypertension and obesity are at increased risk of type 2 diabetes. Several recent clinical trials have suggested that blockade of the renin–angiotensin system (RAS) may protect against the development of de novo diabetes in ‘at risk’ patients. 1 Evidence shows that the RAS may have a direct role in the pathogenesis of diabetes. Local RAS was activated in the adipocyte, which makes the adipose tissue the potential target of angiotensin II receptor blockers.
In this context, the present study was designed to elucidate the effect of telmisartan, an angiotensin II receptor blocker, on the proinflammatory factors’ secretion using 3T3-L1 adipocytes. We hereby show that telmisartan can decrease the proinflammatory factors’ secretion from 3T3-L1 adipocytes and decrease the adiposity, which may favor the adipocytes with insulin sensitization.
Materials and methods
Cell culture and adipocyte differentiation
3T3-L1 cells (Shanghai Institute of Life Science of CAS, Shanghai, China) were cultured and differentiated into adipocytes as described previously. 2 Briefly, 3T3-L1 cells were immersed in DMEM plus 10% heat-inactivated FCS (10% FCS/DMEM), housed in a humidified incubator set at 5% CO2 and 37°C. Cells were grown for two days post-confluence (referred to as day 0) in 10% FCS/DMEM. Differentiation was induced with 10% FCS/DMEM containing 0.5 mM 3-isobutyl-1-methylxanthine (IBMX) (Sigma-Aldrich, MO, USA), 1 µM dexamethasone (DX) and 10 µg/ml insulin for three days. The cells were then incubated in 10% FCS/DMEM with insulin for three days and maintained thereafter with 10% FCS/DMEM to day 12. The medium was changed every other day. After differentiation, more than 90% of the adipocytes displayed a round shape with a cytoplasm completely filled with multiple fat droplets. Only cultures with a differentiation density ≥90% were used in experiments.
Treatments of differentiated 3T3-L1 adipocytes with various concentrations of telmisartan
The experiment was designed for five groups according to the concentrations of telmisartan (Sigma-Aldrich, MO, USA): control group (DMSO), T1 group (0.01 µg/ml), T2 group (0.1ug/ml), T3 group (1ug/ml) and T4 group (10ug/ml). Telmisartan was dissolved in 100% DMSO at 1 mg/ml, diluted in RPMI 1640 medium to a concentration of 10 µg/ml and stored at –20°C until use. The final concentration of DMSO was less than 0.1% and considered to be non-toxic to the differentiated 3T3-L1 adipocytes.
Oil Red O staining
The cells were washed with phosphate-buffered saline (PBS) twice, fixed in 3.7% formaldehyde for 1 h and then stained with 0.6% (w/v) Oil Red O solution (60% isopropanol, 40% water) for 2 h at room temperature. Cells were then washed with water to remove unbound dye. Stained Oil Red O was eluted with isopropanol and quantified by measuring the optical absorbance at 550 nm.
Proinflammatory factors and free fatty acids assay of cultural medium
The concentrations of proinflammatory factors TNF-α and IL-6 were measured using a commercially available enzyme-linked immunosorbent assay (ELISA) kit for rat (Rapidbio, CA, USA) according to the manufacturer’s protocol. Each sample was measured in duplicate with a microplate reader and expressed as means ± SEM. The values of free fatty acid (FFA) concentrations were obtained by enzymatic assay on an automatic analyzer. 3
Cellular 2-[18F]-2-deoxy-D-glucose uptake measurements
Uptake measurements were performed in 3T3-L1 mature adipocytes treated with 0.1% DMSO (control) and various concentrations of telmisartan for three days by adding 370 kBq (10 μCi) of 2-[18F]-2-deoxy-D-glucose (18F-FDG; Nuclear Medicine Department of Chinese PLA General Hospital, Beijing, China) to the culture of cells and incubating in a humidified atmosphere of 5% CO2 at 37°C. Time course experiments showed that 18F-FDG uptake in 3T3-L1 adipocytes reached a relative plateau after 25 min incubation (data not shown). Hence, the remaining 18F-FDG uptake experiments were performed with 25-min incubation, when the adherent cells were washed twice with PBS and then lysed in 1 ml distilled water before the radioactive counts were measured on a high-energy γ-counter (Wallac). 18F-FDG uptake levels of each sample were corrected for protein content as determined by the Bradford method and expressed as percentage of uptake relative to that of the control cells.
High throughput gene expression profiling by microarray
In order to detect the possible genes involved in the effects of telmisartan on the 3T3-L1 adipocytes, a cDNA microarray analysis was performed for the chosen two groups: control group and T2 group (telmisartan 0.1µg/ml). The control and telmisartan-treated groups were treated with 1 ml Trizol reagent (Invitrogen, MD, USA). RNA isolation and quality were checked by spectrophotometry. All procedures of the hybridization experiments of oligonucleotide microarray analysis (CapitalBio 36k Mouse Genome Array) were conducted by CapitalBio Corp. (Beijing, China). 36k Mouse Genome Oligo Array comprises 35,852 70-mer oligo probes from Mouse Genome Version 4.0 of Operon (http://www.operon.com), representing approximately 25,000 genes and 38,000 transcripts of Mouse Genome. The housekeeping genes ACTB, GAPD, LDHA and RPL5 were printed as internal positive controls of hybridization. The results were analyzed with LuxScan 3.0 and the KEGG and BioCarta genome databases retrieved with CapitalBio Molecule Annotation System 4.0 software for pathway analysis. 4
Quantitative real time polymerase chain reaction
Parts of the differential expressed genes screened by the microarray were verified by quantitative real time polymerase chain reaction (Q-PCR). The control and telmisartan treated group (0.1 µg/ml) were treated with 1 ml Trizol reagent (Invitrogen, MD, USA). RNA isolation and quality were checked by spectrophotometry. Q-PCR was performed by SunBio Corp. (Beijing, China). The primer sequences for data presented in relation to this study are as follows: GAPDH: forward primer 5′ TGC TGA GTA TGT CGT GGA G 3′, reverse primer 5′ GTC TTC TGA GTG GCA GTG AT 3′; Nos3: forward primer 5′ GGG CTC CCT CCT TCC GGC TGC CAC 3′, reverse primer 5′ GGA TCC CTG GAA AAG GCG GTG AGG 3′; CPT1A: forward primer 5′ CTC AGT GGG AGC GAC TCT TCA 3′, reverse primer 5′ GGC CTC TGT GGT ACA CGA CAA 3′; Fos: forward primer 5′ GAG GAG GGA GCT GAC AGA TAC ACT 3′, reverse primer 5′ GAT TGG CAA TCT CAG TCT GCA A 3′; Fgf22: forward primer 5′ GAG ATC CGT TCT GTC CGT GT 3′, reverse primer 5′ TTG TAG CCG TTC TCC TCG AT 3′; Prkcc: forward primer 5′ AAA TTG CAC CTC CTT TCA GAC CA 3′, reverse primer 5′ GAC TAG AGA GGG TAG ATG GGT GTT 3′.
Statistical analyses
The data are presented as means ± SEM. Statistical significance was assessed by one way analysis of variance (ANOVA) and the ANOVA post-hoc Bonferroni test with SPSS 12.0. A p<0.05 was considered to be statistically significant. All experiments (except microarray) were performed more than three times and similar results were observed every time.
Results
Telmisartan reduced fat drop and lipid storage of the 3T3-L1 adipocytes
To explore the potential effect of telmisartan on the lipid storage of the differentiated 3T3-L1 cells, 3T3-L1 preadipocytes were differentiated with standard differentiation inducers. During day 12 to 16 of differetiation, telmisartan was added to the media every day. As shown in Figure 1, higher doses of telmisartan (T2, T3 and T4 groups) reduced fat drop and lipid storage of the differentiated 3T3-L1 cells in a dose-dependent manner, while a lower dose (T1 group) did not (Figure 1).

Effect of telmisartan on the fat drop and lipid storage of the 3T3-L1 adipocytes. Telmisartan was dissolved in DMSO (vehicle) and added to the culture from day 12. The differentiated 3T3-L1 adipocytes were incubated with various concentrations of telmisartan for five days. (a) After incubations with telmisartan for five days, the 3T3-L1 adipocytes were fixed and stained with Oil Red O. Macroscopic and microscopic pictures (magnitude 200×) of cells are shown. (b) Lipid storage was assessed by quantification of OD550 in destained Oil Red O with isopropanol. Telmisartan significantly reduced fat drop and lipid storage of the differentiated 3T3-L1 cells in a dose-dependent manner in T2 (0.1 µg/ml, 15.2 ± 5.3% decrease), T3 (1 µg/ml, 26.8 ± 5.7% decrease) and T4 (10 µg/ml, 29.0% ± 6.9% decrease) groups, whereas a lower dose (T1 groups, 0.01 µg/ml) did not.
Effect of telmisartan on secretions of the mature 3T3-L1 adipocytes
To examine a potential impact of telmisartan on the FFAs and proinflammatory factors secreted from the differentiated 3T3-L1 adipocytes, an assay of the culture levels of IL-6 and TNFα was performed. After differentiation of 3T3-L1 cells (day 12), the cells were incubated with vehicle (DMSO) and 0.1 µg/ml (T2) and 1 µg/ml (T3) of telmisartan for 12 h. Then the levels of the proinflammatory factors and FFAs in the cultural solution were measured. Compared with the control, the higher dose (T3) of telmisartan decreased the levels of IL-6 and TNFα and increased those of FFAs. The lower dose (T2) of telmisartan reduced the levels of IL-6 and had no significant effects on the levels of TNFα and FFAs (Figure 2).

Telmisartan reduced the levels of proinflammatory factors (a) and increased the level of free fatty acids (FFAs) (b) from the differentiated 3T3-L1 adipocytes. The levels of tumor necrosis factor-α (TNFα) and interleukin-6 (IL-6) were measured after 12-h incubation of vehicle and 0.1 and 1 µg/ml telmisartan. Compared with control, higher doses (T3) of telmisartan decreased the levels of IL-6 and TNFα and increased the level of FFAs. Lower dose (T2) of telmisartan reduced the levels of IL-6 and had no significant effects on the levels of TNFα and FFAs.
Effect of telmisartan on 18F-FDG transport in the mature 3T3-L1 adipocytes
To examine the potential impact of telmisartan on glucose transport in differentiated 3T3-L1 adipocytes, an 18F-FDG (2-DG) uptake assay was performed. Exposure of differentiated 3T3-L1 adipocytes to telmisartan resulted in a significant enhancement in 18F-FDG uptake in a dose-dependent manner (Figure 3). In the absence of insulin, treatment with 1 and 10 µg/ml telmisartan for 72 h increased 18F-FDG uptake to 174.5% ± 31.2% (p<0.05) and 246.1 ± 38.6% (p<0.01), respectively. In the presence of 1 µM insulin, 0.1, 1 and 10 µg/ml telmisartan significantly augmented glucose uptake to 149.3% ± 21.5% (p<0.05), 166.8% ± 28.7% (p<0.05) and 273.4% ± 47.2% (p<0.05), respectively.

Relative 18F-FDG uptake levels in differentiated 3T3-L1 adipocytes. 18F-FDG uptake was measured in the absence (a) or presence (b) of 1 µM insulin. Exposure of differentiated 3T3-L1 adipocytes to telmisartan resulted in a dose dependence of 18F-FDG uptake levels after 72-h incubation with various concentrations of telmisartan. Data are mean ± SD of percentage uptake relative to controls from a single experiment representative of three separate experiments.
Result from microarray study and Q-PCR
To determine which gene might be involved in the adiposity, we used 3T3-L1 adipocytes from the control and telmisartan (0.1 µg/ml) groups for microarray analysis. One hundred and fifty-seven genes differentially expressed between the groups were found from the microarray. These genes were involved in transport, cell adhesion and signal transduction of the adipocytes; preliminary analyses of these gene functions and their relationship were performed (unpublished). In the present study, we focused on analyzing the gene expression profile related to the processes of lipometabolism and the secretion of proinflammatory factors from the differentiated 3T3-L1 adipocytes. By retrieving the KEGG and BioCarta database, we propose that some cell signaling pathways involved in the secretion process of adipocytokines and proinflammatory factors were affected by telmisartan. The significant pathways affected by telmisartan are listed in ascending order of p-values (and multiple-comparison corrected Q-value) 5 in Table 1. According to the pathway analysis, the mitogen-activated protein kinase (MAPK) signaling pathway was significantly different. The analysis revealed nine differentially expressed genes involved in the MAPK signaling pathway (Table 2). Moreover, the result also showed that Nos3 and CPT1α (Table 3), which encode the endothelial nitric oxide synthase and carnitine palmitoyl transferase 1A respectively, were up-regulated. Consistent results were obtained by the Q-PCR with Nos3, CPT1α and the three select genes, which are Fos, Prkcc and Fgf22, from the MAPK signaling pathway (Table 3).
The pathways affected most by telmisartan analyzed by use of the KEGG and BioCarta databases.
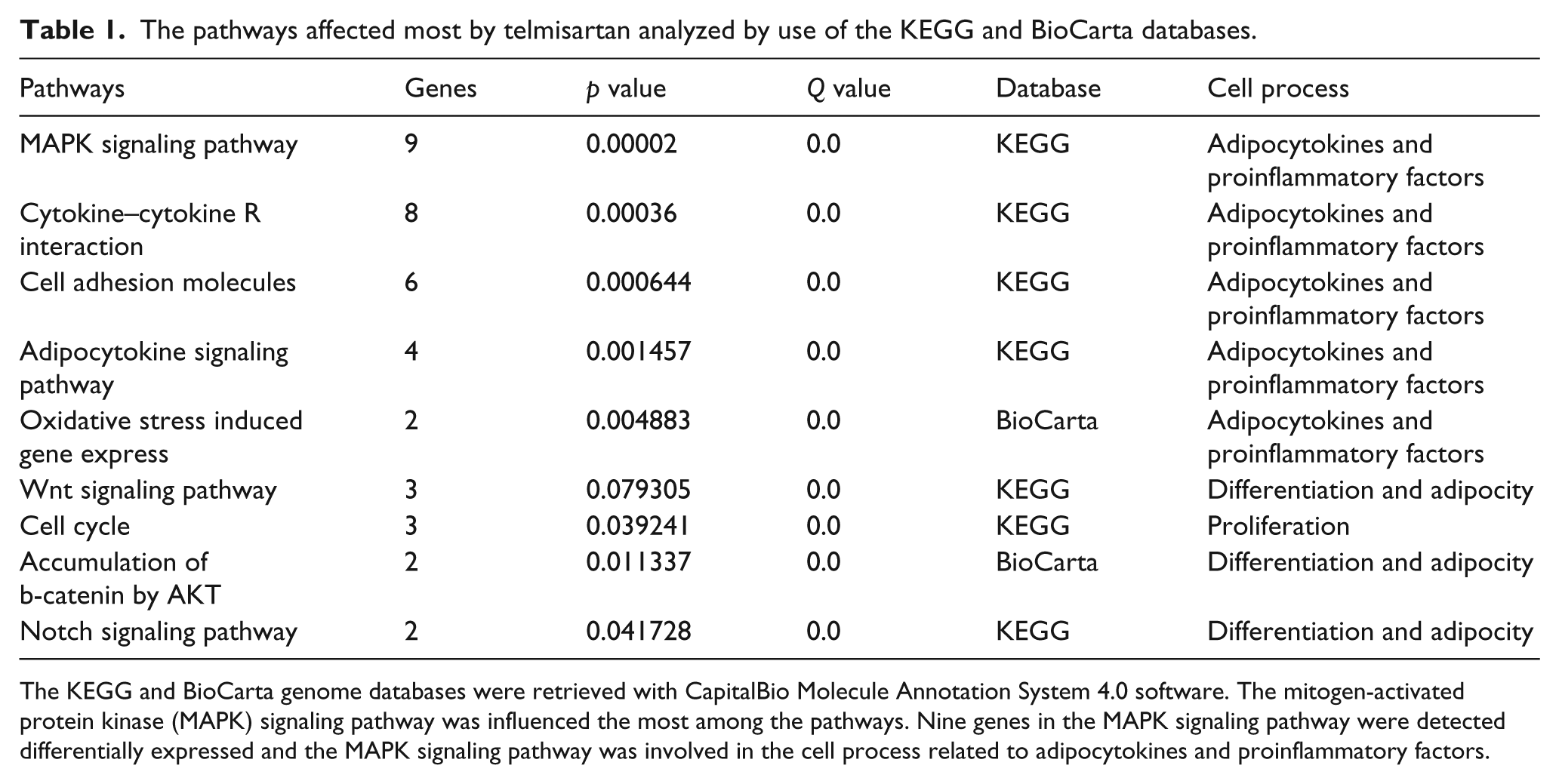
The KEGG and BioCarta genome databases were retrieved with CapitalBio Molecule Annotation System 4.0 software. The mitogen-activated protein kinase (MAPK) signaling pathway was influenced the most among the pathways. Nine genes in the MAPK signaling pathway were detected differentially expressed and the MAPK signaling pathway was involved in the cell process related to adipocytokines and proinflammatory factors.
The nine genes in the mitogen-activated protein kinase signaling pathway detected differentially expressed by microarray assay.

R1: compared with control by microarray, R1 = Cy3/Cy5.
The value of Cy3/Cy5 >1.5 or <0.6 indicated significant up-regulation or down-regulation of the gene expression.
Comparison of microarray and real-time PCR result.

R1: compared with control by microarray, R1 = Cy3/Cy5; R2: compared with control by real-time PCR.
p<0.01.
p<0.05.
Discussion
We have demonstrated that telmisartan reduced the lipid storage and increased the 18F-FDG uptake in differentiated 3T3-L1 adipocytes in a dose-dependent manner. At the same time, telmisartan reduced the levels of IL-6 and TNFα and increased that of FFAs in the culture medium. Microarray analysis revealed that the MAPK signaling pathway and oxidative stress were implicated in the secretion of proinflammatory factor and lipometabolisms and were affected by telmisartan. Regulation of Nos3 and CPT1α expression by telmisartan, which was detected by the microarray, may also be the underlying cause of the improvement in the lipometabolisms.
Currently, more than 50 different adipokines are recognized. The adipokines are implicated in a wide range of physiological processes, including appetite and energy balance, glucose homeostasis, lipid metabolism, blood pressure regulation, hemostasis and angiogenesis. IL-6 and TNFα are two of the inflammation-related proteins whose circulating levels are increased during obesity and are directly linked to inflammation and inflammatory response. 6 These proteins also include C-reactive protein, interleukin-18, plasminogen activator inhibitor-1 and haptoglobin, which are the basis for the view that obesity is characterized by chronic low grade inflammation. Adipose tissue is one of the main origins of IL-6 and TNFα. 7 In addition, RAS components, including angiotensin II receptor, exist in the adipocyte, which makes the adipose tissue a potential target of angiotensin II receptor blockers. Numerous clinical studies in subjects with a variety of cardiovascular conditions have shown that blockade of the renin–angiotensin system demonstrates suppression of proinflammatory cytokines including IL-6 and TNFα.8–10 Our study showed that telmisartan reduced the levels of IL-6 and TNFα, which suggests that the direct effect of telmisartan on adipocyte secretion is one of the causes of the suppression of IL-6 and TNFα in vivo. In addition, the microarray analysis indicates that the MAPK pathway and oxidative stress implicate this effect of telmisartan.
We then tested the hypothesis that telmisartan exerted a direct impact on adipocyte metabolism using the 3T3-L1 adipocytes model. The results showed that telmisartan reduced fat drop and lipid storage of the adipocytes. Previous research found that Ang II can significantly increase triglyceride content in 3T3-L1 adipocytes and in human adipose cells. These effects were mediated through the Ang II AT2 receptor, but not Ang II type 1 receptor. 11 As one of the Ang II AT1 receptor blockers, the action of telmisartan on adipocyte may be independent of the Ang II AT1 or AT2 receptor. We also demonstrate that telmisartan dose-dependently augments 18F-FDG uptake both in the basal and in the insulin-stimulated state of 3T3-L1 adipocytes. These data are consistent with a previous report, which speculated that telmisartan induces glucose transporter type 4 (GLUT4) expression and subsequent glucose uptake via peroxisome proliferator activated receptor γ (PPARγ) activation in adipocytes. 12 Enhancement of GLUT4 expression and PPARγ activation can facilitate lipid accumulation in the process of 3T3-L1 cell differentiation from preadipocytes to adipocytes. 12 During the in vitro studies, evidence has suggested that PPARγ activation increases body weight and causes a shift in the fat distribution from visceral to subcutaneous adipose tissue. 13 Telmisartan is effective at reducing body weight and adiposity 14 and can prevent thiazolidine (PPARγ agonist)-induced weight gain without interfering with its insulin-sensitizing properties. 15 In human studies, telmisartan has also been proven to reduce fat content and affect fat distribution in individuals with the metabolic syndrome. 16 These facts raise a question regarding the differential effects of telmisartan and thiazolidine, both PPARγ agonists, on body weight and adiposity. Some investigators hold the view that reduction of body weight is associated with reduction of food intake and a down-regulation of orexigenic peptides after AT1-blockage.17,18 Others presume that it is a direct effect of angiotensin receptor blockers (ARBs) on the lipid metabolisms in the liver and/or the adipose tissue. 19 In the present study, we observed the effect of telmisartan on the lipid metabolisms of differentiated 3T3-L1 adipocytes and provided evidence that the telmisartan has direct impact on the adipocytes, reduces lipid storage and increases FFA secretion from the cells. Thereafter the possible mechanisms were explored by the microarray, which showed that telmisartan affected the MAPK signaling pathway and gene expressions of Nos3 and CPT1α during the reduction of lipid storage. Local endothelial nitric oxide synthetase can increase the nitric oxide synthesis of adipocytes and also regulates energy metabolism via influencing mitochondrial biogenesis 20 and lipolysis. 21 Carnitine palmitoyl transferase 1A is the key regulatory enzyme of the long-chain fatty acid enzyme of β-oxidation. Recent studies suggest that a moderate increase in carnitine palmitoyltransferase 1A activity is sufficient to substantially reduce hepatic triglyceride levels. Overexpression of CPT1α increased the rate of β-oxidation and was accompanied by a significant reduction in triglyceride accumulation by hepatocytes which have potential benefits in the treatment of fatty liver. 22 In adipocytes, it is known that increasing the expression of CPT1α can also enhance the fatty acid oxidation. 23
Abuissa et al., 1 in a meta-analysis of 12 recent randomized controlled clinical trials that enrolled patients with hypertension, chronic heart failure or coronary heart disease, showed that angiotensin-converting enzyme (ACE) inhibitors and ARBs produced a highly significant 25% reduction in the incidence of new-onset diabetes when compared with placebo, diuretics, beta-blockers or calcium channel antagonists. However, these mechanisms are not fully understood. Our experiment suggests that the adipocyte is one of the major targets for telmisartan, and the actions of reducing proinflammatory factors from adipocyte, increasing glucose uptake and decreasing the lipid content of adipocytes may enhance the beneficial metabolic effects of telmisartan. Collectively, the present findings of our experiments indicate that telmisartan may be involved in regulation of adipose mass and secretion of adipocytes. Local effects of telmisartan on the adipocytes could thus be expected to change the factors known to regulate adipocyte metabolism. Additional studies will be necessary to determine whether there are similar effects of telmisartan on regulation of lipid metabolisms and proinflammatory factors secretion in vivo.
Conclusions
Telmisartan, an angiotensin II receptor blocker, affected lipometabolisms and the proinflammatory factors secreted from adipocytes. Our research revealed that Nos3, CPT1α and the MAPK pathway affected by telmisartan may be the underlying cause of the improvement in the lipid metabolisms and proinflammatory factors affecting the secretion of differentiated 3T3-L1 adipocytes.
Footnotes
Conflict of interest
The authors declare that there is no conflict of interest.
Funding
This work was supported by the National Natural Science Foundation of China (grant number 30671095) and the National Basic Research Program of China (grant number 2006CB503903).
