Abstract
Introduction:
Asymmetric dimethylarginine (ADMA), an endogenous inhibitor of nitric oxide synthase, is involved in hypertension. We tested whether aliskiren treatment in early postnatal life can reduce ADMA and regulate the renin-angiotensin system to prevent hypertension in rat offspring exposed to maternal caloric restriction (CR).
Materials and methods:
Four groups of 12-week-old male offspring were sacrificed: control, CR, CR+aliskiren, and CR+losartan group. The CR group included offspring from 50% food-restricted maternal rats. The CR+aliskiren and CR+losartan groups were produced by treating CR offspring with oral aliskiren 10 mg/kg/day or losartan 20 mg/kg/day between 2–4 weeks of age, respectively.
Results:
Blood pressure increased in CR rats, which was prevented by aliskiren or losartan. CR increased plasma ADMA levels, which aliskiren prevented. Renal renin and prorenin receptor (PRR) expression increased in CR rats treated with aliskiren, whereas both were reduced by losartan. Both aliskiren and losartan decreased renal mRNA expression of angiotensinogen, angiotensin II type 2 receptor, and Mas in CR rats. However, aliskiren increased angiotensin II type 2 receptor and Mas protein levels in CR kidneys.
Conclusions:
Early aliskiren therapy prevents CR-induced hypertension via ADMA reduction, decreases angiotensinogen expression, and increases renal angiotensin II type 2 receptor and Mas protein.
Introduction
Hypertension is a highly prevalent disorder, which might originate during early life. The nitric oxide (NO)/reactive oxygen species (ROS) imbalance has been documented in experimental and human hypertension.1,2 Maternal undernutrition can impair NO/ROS balance and elicit epigenetic alterations in genes for nephrogenesis in the kidney, ultimately leading to developmental programming of hypertension in later life.
Studies have reported that increased asymmetric dimethylarginine (ADMA), an endogenous inhibitor of NO synthase (NOS), is involved in hypertension and programmed hypertension.3–5 ADMA can reduce the synthesis of NO while inducing superoxide production. Thus, ADMA levels tightly regulate NO/ROS balance. Another cause of NO/ROS imbalance is activation of the renin-angiotensin system (RAS), which is critically linked to the development of hypertension. 6
The RAS plays a fundamental role in the regulation of blood pressure (BP) and kidney development.7,8 Studies showing that blockade of RAS by angiotensin-converting enzyme (ACE) inhibitor captopril or angiotensin II type 1 receptor blocker (ARB) losartan between 2–4 weeks of age offsets the effects of nutritional programming on BP were supportive of a role for the RAS, linking maternal undernutrition to adulthood hypertension.9,10 We recently found that three-month-old offspring of dams exposed to 50% caloric restriction (CR) during late pregnancy and lactation had NO/ROS imbalance, increased ADMA, and increased BP. 11 We also found that treatment with the direct renin inhibitor, aliskiren, reduces ADMA and prevents the development of hypertension in young spontaneously hypertensive rats. 12 Thus, we intended to study whether aliskiren in early postnatal life reduces ADMA and prevents hypertension in offspring exposed to maternal CR.
The RAS has been reported to be critical in nephrogenesis. Nephrogenesis-related and RAS genes can be epigenetically controlled via histone deacetylases (HDACs).13,14 As another important member of the RAS, angiotensin-converting enzyme 2 (ACE2) appears to adjust angiotensin II type 2 receptor (AT2R) and angiotensin (1-7) receptor Mas in a way that opposes the development of hypertension. 15 We also intended to elucidate whether type I HDAC isoforms and ACE2/AT2R/Mas are differentially regulated by CR and aliskiren in this study.
In this study, because ADMA and the RAS are involved in programmed hypertension and aliskiren can reduce BP and ADMA concurrently, we intended to elucidate whether early aliskiren therapy can reduce ADMA and regulate RAS to prevent the development of hypertension in CR offspring.
Materials and methods
Animals and pharmacological treatment
This study was approved and performed according to the guidelines for animal experiments of Chang Gung Memorial Hospital and Chang Gung University. The treatment of animals conformed to the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health. 16 Virgin Sprague-Dawley rats (aged 12–16 weeks, n=8) were obtained (BioLASCO Taiwan Co., Ltd., Taipei, Taiwan) and given free access to tap water and standard rat chow. Male Sprague-Dawley rats were caged with individual female rats until mating was confirmed. After birth, each litter was left with the mother until weaning. Food-restricted maternal rats received 11 g/day of a standard chow from day 11 of pregnancy until the day of delivery (day 23) and 20 g/day of the same chow during the entire lactation period as we described previously (CR offspring group). 11 Another group of CR offspring was produced by treating young male rats with oral aliskiren 10 mg/kg/day via gastric gavage (Novartis Pharmaceutical, New York, USA) between 2–4 weeks of age (CR+A group, n=6). A third group of CR offspring was produced by treating young male rats with oral losartan 20 mg/kg/day via gastric gavage between 2–4 weeks of age (CR+L, n=7). This group was used as the antihypertensive control. Last, a normal control group consisted of male offspring of maternal rats with free access to standard rat chow (C group, n=7). The dose of aliskiren or losartan used here was based on previous studies conducted in rats.9,12 The male offspring were weighed and their BP measured on postnatal days 21, 28, 56, and 84. These offspring were sacrificed at 12 weeks of age. Heparinized blood samples were collected at the time of sacrifice. Kidneys were harvested after perfusion with phosphate buffered saline (PBS). Plasma levels of nitrite (NO2–) and NOx (NO2− + NO3−) were measured by the Griess reaction as previously described. 11
Detection of l -arginine, ADMA, and symmetric dimethylarginine by high-performance liquid chromatography
Plasma and tissue
Western blot analysis
Western blot analysis was performed as described. 4 We used the following antibodies: for ACE2, rabbit anti-rat ACE2 (1:1000, overnight incubation; Santa Cruz Biotechnology, Santa Cruz, California, USA); for AT2R, rabbit anti-rat AT2R (1:250, overnight incubation; Santa Cruz Biotechnology); for Mas, a rabbit antibody (1:1000, overnight incubation; Santa Cruz Biotechnology). We used a rabbit anti-rat β-actin antibody (1:20,000 dilution, 30 min incubation; Chemicon, Temecula, California, USA), followed by a secondary goat anti-rabbit antibody. The bands of interest were visualized using ECL reagents (PerkinElmer, Waltham, Massachusetts, USA) and quantified by densitometry (Quantity One Analysis software; Bio-Rad, Hercules, California, USA), calculated as the integrated optical density (IOD) minus the background value. The IOD was factored for β-actin to correct for any variations in total protein loading. Protein abundance was represented as IOD/β-actin.
Quantitative real-time polymerase chain reaction
RNA was extracted using TRI Reagent (Sigma, St. Louis, Missouri, USA) and treated with deoxyribonuclease I (Ambion, Austin, Texas, USA) to remove DNA contamination, and 2 µg were reverse transcribed (SuperScript II RNase H Reverse Transcriptase, Invitrogen, Bethesda, Maryland, USA) with random primers (Invitrogen) in a total volume of 40 µl. Control reverse transcription reactions were performed by omitting the reverse transcription enzyme, and polymerase chain reaction (PCR) was amplified to ensure that DNA did not contaminate the RNA. Two-step quantitative real-time PCR was conducted using Quantitect SYBR Green PCR reagents (QIAGEN, Valencia, California, USA) on an iCycler iQ Multi-color Real-Time PCR Detection System (Bio-Rad). Primers were designed using GeneTool Software (Biotools, Edmonton, Alberta, Canada) with an annealing temperature of 57–60°C and PCR products between 80–150 bp (Table 1). R18S was used as a reference. All samples were run in duplicate (2.5 µl of cDNA per well in a 96-well format). For the relative quantification of gene expression, the comparative threshold cycle (Ct) method was employed. The averaged Ct was subtracted from the corresponding averaged r18S value for each sample, resulting in ΔCt. ΔΔCt was achieved by subtracting the average control ΔCt value from the average experimental ΔCt. The fold-increase was established by calculating 2−ΔΔCt for experimental vs reference samples.
Polymerase chain reaction (PCR) primer sequences.

ACE: angiotensin-converting enzyme; AGT: angiotensinogen; AT1R: angiotensin II type 1 receptor; AT2R: angiotensin II type 2 receptor; HDAC: histone deacetylase; PRR: prorenin receptor.
Detection of superoxide by electron paramagnetic resonance
Superoxide production was measured by electron paramagnetic resonance (EPR) spectroscopy with hydroxylamine spin probe 1-hydroxy-3-carboxypyrrolidine (CPH), as we previously described. 5 Kidney homogenate was prepared, and 10 μg of protein was added to 1 mM CPH and 0.1 mM diethylenetriaminepentaacetic acid in a total volume of 100 μl of Chelex-treated PBS. Samples were placed in a 50μl glass capillary (Wilmad Glass, Buena, New Jersey, USA). The EPR spectra were recorded using an EMX Plus EPR spectrometer (Bruker Biospin, Rheinstetten, Germany) equipped with a EMX-m40X microwave bridge operating at 9.87 GHz.
Statistics
First, the Shapiro-Wilk normality test was used to determine which data were normally distributed. Normally distributed data are given as mean±standard error of the mean (SEM). For most parameters, statistical analysis was done using one-way analysis of variance (ANOVA) with Tukey’s post-hoc test for multiple comparisons. BP was analyzed by two-way repeated-measures ANOVA and Tukey’s post-hoc test. A p-value<0.05 was considered statistically significant. All analyses were performed using the Statistical Package for the Social Sciences (SPSS) software (Chicago, Illinois, USA).
Results
As shown in Table 2, body weight decreased in all three CR offspring groups compared with controls. The heart weight did not differ among the four groups, but the ratio of heart weight to body weight in CR offspring increased compared with controls, which was not prevented by treatment with aliskiren or losartan. The kidney weight was lower in the CR offspring group but higher in the CR+A and CR+L groups than in the control group. The ratio of kidney weight to body weight in the CR offspring group was unaltered compared with controls, whereas this ratio was increased by treatment with aliskiren or losartan.
Weights and functional parameters in caloric restriction (CR) offspring and control rats at 12 weeks of age.

Male offspring of mother exposed to 50% caloric restriction (CR) during late pregnancy and lactation; CR+A indicates CR offspring that received aliskiren 10 mg/kg between 2–4 weeks of age; CR+L indicates CR offspring that received losartan 20 mg/kg between 2–4 weeks of age.
p<0.05 vs control.
p<0.05 vs CR.
The mean arterial pressure increased in the untreated CR offspring group over eight weeks compared with controls (Figure 1). There was a similar reduction in mean arterial pressure in the CR+A group versus the CR+L group at eight and 12 weeks of age. As shown in Table 2, systolic and diastolic BP decreased in both CR offspring groups treated with aliskiren or losartan.

Effects of aliskiren and losartan on mean arterial pressure in caloric restriction (CR) offspring and controls. Male offspring of mother exposed to 50% caloric restriction during late pregnancy and lactation; CR+Aliskiren indicates CR offspring that received aliskiren 10 mg/kg between 2–4 weeks of age; CR+Losartan indicates CR offspring that received losartan 20 mg/kg between 2–4 weeks of age; *p<0.05 vs control; #p<0.05 vs CR.
As shown in Table 3, plasma
Plasma levels of

CR indicates male offspring of mother exposed to 50% caloric restriction (CR) during late pregnancy and lactation; CR+A indicates CR offspring that received aliskiren 10 mg/kg between 2–4 weeks of age; CR+L indicates CR offspring that received losartan 20 mg/kg between 2–4 weeks of age; ap<0.05 vs control; bp<0.05 vs CR; cp<0.05 vs CR+A.
Next, we evaluated the renal expression of RAS components. The expression of renin, PRR, angiotensinogen (AGT), ACE, ACE2, angiotensin II type 1 receptor (AT1R), AT2R, and Mas in the kidney did not differ between the CR offspring and control groups (Figure 2(a)). However, renal renin and PRR expression significantly increased in CR offspring rats treated with aliskiren, while it decreased by losartan therapy (Figure 2(b) and (c)). Both aliskiren and losartan treatment similarly decreased renal AGT, AT2R, and Mas expression in CR offspring rats.

Effects of (a) caloric restriction (CR) vs control (b) CR+aliskiren (A) vs CR, and (C) CR+losartan (L) vs CR on gene expression of renin-angiotensin system (RAS) components in the kidney. Male offspring of mother exposed to 50% CR during late pregnancy and lactation; CR+A = CR offspring that received aliskiren 10 mg/kg between 2–4 weeks of age; CR+L = CR offspring that received losartan 20 mg/kg between 2–4 weeks of age; *p<0.05 vs control. ACE: angiotensin-converting enzyme; AGT: angiotensinogen; AT1R: angiotensin II type 1 receptor; AT2R: angiotensin II type 2 receptor; PRR: prorenin receptor.
We further analyzed protein levels of ACE2, AT2R, and Mas in the kidney. We observed that renal ACE2 expression was not different among the four groups (Figure 4(b)). AT2R protein abundance in the kidney was higher in the CR+A and CR+L groups than in CR offspring rats (Figure 3(c)). Aliskiren therapy significantly increased renal Mas protein level compared with the other groups (Figure 3(d)).
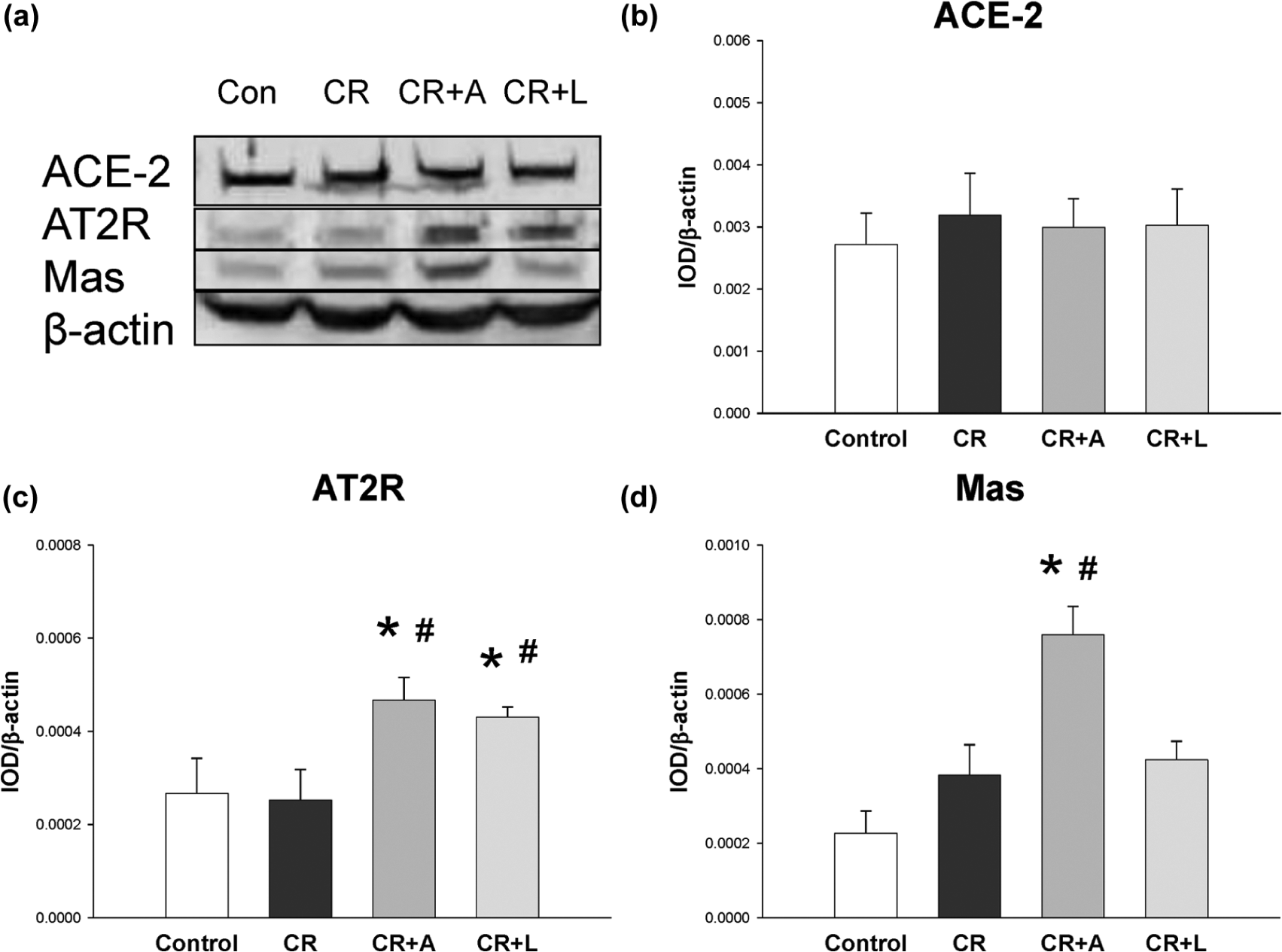
Representative western blots (a) showed angiotensin-converting enzyme 2 (ACE2) (90 kDa), angiotensin II type 2 receptor (AT2R) (50 kDa), and Mas (37 kDa) bands in control and caloric restriction (CR) offspring rats at 12 weeks of age. Relative abundance of renal cortical (b) ACE2, (c) AT2R, and (d) Mas as quantified. Male offspring of mother exposed to 50% CR during late pregnancy and lactation; CR+A indicates CR offspring that received aliskiren 10 mg/kg between 2–4 weeks of age; CR+L indicates CR offspring that received losartan 20 mg/kg between 2–4 weeks of age; *p<0.05 vs control; #p<0.05 vs CR.
We next examined whether type 1 HDACs were involved in the regulation of RAS expression in response to CR, aliskiren, and losartan. As shown in Figure 4, CR upregulated HDAC1 expression in the kidney, which losartan prevented. However, aliskiren therapy increased renal expression of HDAC2, HDAC3, and HDAC8 in CR offspring rats.

Effects of aliskiren and losartan on gene expression of (a) histone deacetylase (HDAC)1, (b) HDAC2, (c) HDAC3, and (d) HDAC8 in the kidney. Caloric restriction (CR) male offspring of mother exposed to 50% CR during late pregnancy and lactation; CR+A indicates CR offspring that received aliskiren 10 mg/kg between 2–4 weeks of age; CR+L indicates CR offspring that received losartan 20 mg/kg between 2–4 weeks of age; *p<0.05 vs control; #p<0.05 vs CR.
Discussion
This study demonstrates that treatment of male CR offspring with aliskiren or losartan between the ages of 2–4 weeks postnatally prevents the development of hypertension at 12 weeks of age. Our data are in agreement with previous studies showing that blockade of RAS by ACE inhibitor captopril or ARB losartan in early postnatal life offsets the effects of nutritional programming in the low-protein diet programming model.9,10 To our knowledge, our study is the first to show that the renin inhibitor, aliskiren, can prevent the development of hypertension in offspring exposed to maternal CR. Consistent with growing evidence that prenatal nutritional programming can lead to alterations in the RAS in the offspring, 17 our data support the view that early blockade of RAS affords protection against the development of hypertension. Like ACE inhibitors and ARBs, early treatment with aliskiren in the prehypertensive stage represents a novel way to prevent the development of hypertension later in life.
BP was not decreased with either aliskiren or losartan treatment in the first two weeks of treatment. The antihypertensive effect was attenuated starting from eight weeks of age and over time. These findings indicate that the reduction of BP might be due to the restoration of NO/ROS balance or regulation of the RAS instead of an acute effect.
The direct renin inhibitor, aliskiren, inhibits the enzyme renin by binding to its catalytic site, thus blocking the RAS. In this respect, angiotensin I, angiotensin II, and aldosterone levels are decreased. In addition, aliskiren blocks renin and prorenin activity while renin and prorenin levels remain high. 18 In this study, we found that aliskiren therapy significantly increased renin and PRR, whereas losartan treatment decreased renin and PRR expression. Activation of the renin-PRR pathway has been reported to induce hypertrophy, 19 which might explain the renal hypertrophy in the CR+A group. Although both aliskiren and losartan can prevent hypertension in CR offspring, our findings indicate that differentially protective mechanisms are involved, including RAS components and the ADMA/NO pathway.
First, prevention of elevation of BP with aliskiren therapy in CR offspring might be, at least in part, due to restoration of ADMA/NO balance. We found that aliskiren therapy decreased ADMA levels but increased the ratio of
Another protective mechanism of aliskiren on programmed hypertension is related to reduction of renal AGT expression and increased protein levels of AT2R and Mas in the kidney. Unlike aliskiren, losartan therapy decreased renin and PRR in the CR offspring kidney. In addition, losartan therapy decreased the AGT mRNA and increased AT2R protein levels in the kidney. It is now well established that the signals formed by AT2R or Mas appear to represent an endogenous counter-regulatory pathway within the RAS, the actions of which are in opposition to the vasoconstrictor arm of the RAS. Thus, one might expect early losartan therapy to adjust the RAS in a way that opposes the development of hypertension in CR offspring rats.
Nevertheless, we also found discrepancies in mRNA and protein levels as a result of programming at the transcriptional and posttranscriptional levels. Aliskiren and losartan both reduce the renal expression of AT2R and Mas mRNA. Despite the decreases in mRNA, the protein levels of AT2R and Mas were increased by aliskiren. Thus, the downregulation of mRNA by aliskiren or losartan might be a negative feedback compensatory response to their BP-lowering effect. The mechanism through which aliskiren and losartan differentially regulate Mas protein expression remains unclear. Our previous observations showed that aliskiren enhances renal cortical neuronal NOS (nNOS) and prevents the development of hypertension in young spontaneously hypertensive rats. 12 Control of nNOS activity can be exerted by the binding protein postsynaptic density-95 (PSD95) through interaction with the NOS PDZ domain. 21 Other investigators recently reported that PSD95 enhances Mas protein expression by increasing the stabilization of the receptor. 22 Thus, it is possible that aliskiren may enhance nNOS/PSD95 dimer formation to increase Mas protein expression.
Type I HDACs are critical for normal embryonic kidney homeostasis, 14 which is supported by our results showing that CR increases HDAC1 expression in the kidney related to hypertension, which losartan prevented. Nevertheless, aliskiren therapy increased expression of HDAC2, HDAC3, and HDAC8 in the CR offspring kidney. Several players in the RAS system such as AGT, renin, ACE, and AT1R have been reported to be epigenetically controlled via HDACs. 13 Further studies are warranted to elucidate whether nutritional programming regulates specific genes in the RAS via HDACs, which can be reprogrammed by blockade of RAS in early life.
Conclusions
We conclude that early blockade of RAS by aliskiren or losartan offsets the effects of maternal CR-induced programmed hypertension, primarily through restoration of the balance between ADMA/NO and RAS in favor of vasodilatation in the kidney. Our data highlight the differential effects of early blockade of RAS with two anti-hypertensive agents, aliskiren and losartan, on ADMA and RAS components despite both having a similar BP-lowering effect. The possible implications of the underlying epigenetic changes for the regulation of hypertension in later life remain to be explored. Our data demonstrate that there is a critical window in the early postnatal period during which the adult blood pressure can be modified. The present study supports the assertion that early aliskiren treatment in childhood might prevent the transition from prehypertension to hypertension in adult life.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Chang Gung Memorial Hospital, Kaousiung, Taiwan (grant number CMRPG8B0351), and the Taiwan Society of Nephrology, Taiwan (grant number SMRPG8B0011).
