Abstract
Introduction:
C-type natriuretic peptide (CNP) selectively binds to the guanylyl cyclase coupled natriuretic peptide receptor (NPR)-B and exerts more potent antihypertrophic and antifibrotic properties. Elimination of CNP occurs mainly by neutral endopeptidase (NEP) and NPR-C.
Methods:
We established a rat model of unilateral ureteral obstruction (UUO) to examine the continuous change of the CNP expression and to assess the correlations of NPR-B, NPR-C, NEP with CNP in the obstructed kidneys.
Results:
The expressions of CNP mRNA and protein in the obstructed kidneys tended to be higher immediately after ligation and declined at later time points compared to sham-operated rats, measured by real-time polymerase chain reaction (PCR) and western blot analysis. Subsequent correlation analysis indicated that CNP mRNA was positively correlated with NPR-B mRNA (
Conclusions:
A high level of CNP may contribute to the elevated expression of NPR-B in the early phase of UUO. More interestingly, paradoxical expressions of NPR-C and NEP may account for the decline of CNP in the obstructed kidneys.
Keywords
Introduction
In 1990, Sudoh and his colleagues primarily isolated C-type natriuretic peptide (CNP) from porcine brain. 1 It is now known that endothelium has been identified as the most major site of CNP synthesis. CNP comprises a ring structure of 17 amino acids linked by a cysteine disulphide bridge, which is essential for receptor binding and bioactivity (Figure 1). Distinct from the other members of natriuretic peptide family, CNP selectively binds to guanylyl cyclase coupled natriuretic peptide receptor (NPR)-B and results subsequently in the highest elevation in intracellular cyclic guanosine monophosphate (cGMP). 2 In addition, CNP exerts more potent antihypertrophic and antifibrotic properties, but has little effect on water and salt excretion, partly due to the deficiency of carboxy-terminal extension. 3 On the other hand, there are two pathways involved in the catabolism of CNP: removal by the clearance receptor (NPR-C) and hydrolysis by neutral endopeptidase (NEP). 4

Amino acid sequence of the human C-type natriuretic peptide (CNP). CNP comprises a ring structure of 17 amino acids linked by a cysteine (Cys) disulphide bridge, and conserved sequences of natriuretic peptide family are labeled in black.
Currently, CNP is believed to be produced locally in normal kidneys. Cataliotti et al.,
5
by immunohistochemical method, showed that CNP was predominantly present in tubular epithelial cells, including proximal, distal, and medullary collecting duct cells. CNP immunoreactivity was also observed in the glomeruli. Renal tissue expression of CNP is often up-regulated in several pathological conditions, such as nephrotic syndrome, function renal failure in cirrhosis and diabetic nephropathy.5–7 Our latest study published in
Materials and methods
Animals and treatment
Male Wistar rats weighing 190–250 g were housed at an ambient temperature of 23±1°C and exposed to a daily 12-h light-dark cycle (light on 07:00–19:00) with free access to tap water and a pellet diet. Animals were treated humanely using approved procedures in accordance with the guidelines of the Institutional Animal Use and Care Committee of Anhui Medical University. A total of 96 rats were separated into 16 experimental groups: eight groups undergoing left proximal unilateral ureteral obstruction (UUO) (
Real-time PCR
Total RNA was extracted from renal tissue using TRIzol reagent (Invitrogen Co. Ltd, USA). Use of an ultraviolet spectrophotometer measuring absorbance, agarose gel electrophoresis confirmed that there had been no degradation of RNA. Specific primers were synthesized according to the published sequences. The sequences of oligonucleotides and sizes of PCR products were, for CNP, sense: 5’-AACATCCCAGACCGCTCATG-3’, antisense: 5’-CAA GAA GGG CTT GTC CAA AGG-3’(73 bp) 9 for NPR-B, sense: 5’-AAC GGG CGC ATT GTG TAT ATC TGC GGC-3’, antisense: 5’-TTA TCA CAG GAT GGG TCG TCC AAG TCA-3’ (692 bp) 10 for NPR-C, sense: 5’-ATA GTG CGC TAC ATC CAG GGC AGT-3’, antisense: 5’-TCC AAA GTA ATC ACC AAT GAC CTC CTG GGT ACC TGC-3’ (573 bp) 10 for NEP, sense: 5’-CCC CGC CGG CAT TT-3’, antisense: 5’-GCC CCC ATA GTT CAA TGA GTT G-3’ (66 bp) 11 for glyceraldehyde-3-phosphate dehydrogenase (GAPDH), sense: 5’-TGT GAG GGA GAT GCT CAG TG-3’, antisense: 5’-GGC ATT GCT CTC AAT GAC AA-3’ (229 bp). 12 One microgram total RNA from the renal tissue of each rat was reverse transcribed into cDNA with an ExScript RT reagent kit (Takara Biotechnology Co. Ltd, Dalian, China). CNP, NPR-B, NPR-C and NEP were amplified with SYBR Premix Ex Taq (Takara Biotechnology Co. Ltd). Gene expression of GAPDH was also measured in each sample and used as an internal control for loading and reverse transcription efficiency. Each sample analysis was repeated three times. The average threshold cycle (Ct, the cycles of template amplification to the threshold) was worked out as the value of each sample. Relatively quantitative 2−ΔΔCt was used to compare the mRNA expression. 13
Protein extraction and western blot analysis
Renal tissues were homogenized in ice-cold buffer (40 mM KCl, 10 mM Hepes, pH 7.9, 3 mM MgCl2, 5% glycerol, 0.5 µg/ml leupeptin, 0.1 µg/ml aprotinin, 1.5 µg/ml pepstatin and 100 µg/ml phenylmethylsulphonyl fluoride) with a Polytron homogenizer for 15–20 s. The homogenates were centrifuged at 500 g for 10 min at 4°C and the supernatants were recentrifuged at 12000 g for 60 min at 4°C. The pellets were resuspended in 0.5 ml homogenizing buffer containing 0.5% Nonidet P-40 (Boster, Wuhan, China) and total protein concentration of the supernatant was determined by the dye-binding method using
Statistical analyses
All values are expressed as mean±standard error of the mean (SEM). Comparison of mean values between groups was made using one way analysis of variance (ANOVA), and post-hoc analysis was calculated using the Student-Newman-Keuls test. The correlations of NPR-B, NPR-C, NEP with CNP were assessed by linear regression. A value of
Results
Renal expression of CNP, NPR-B, NPR-C and NEP mRNA at the time of sacrifice are shown in Figure 2. The relative quantities of CNP transcript were significantly higher throughout the obstructed kidneys compared to the corresponding SORs at each time point (

Real-time polymerase chain reaction (PCR) analysis of (a) C-type natriuretic peptide (CNP), (b) natriuretic peptide receptor (NPR)-B, (c) NPR-C, (d) neutral endopeptidase (NEP), and collagen IV (Col-IV) mRNA. *
Renal expression of CNP, NPR-B, NPR-C and NEP protein at time of sacrifice are shown in Figure 3. By western blot analysis, an elevated protein expression of CNP was also observed in UUO rats as early as 24 h post-ligation (a 47% increase,

Western blot identification of C-type natriuretic peptide (CNP), natriuretic peptide receptor (NPR)-B, NPR-C, neutral endopeptidase (NEP), and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) protein. SOR: sham-operated rat; UUO: unilateral ureteral obstruction.
The correlations of NPR-B, NPR-C, NEP with CNP in the obstructed kidneys are shown in Figure 4. CNP mRNA was positively correlated with NPR-B mRNA (
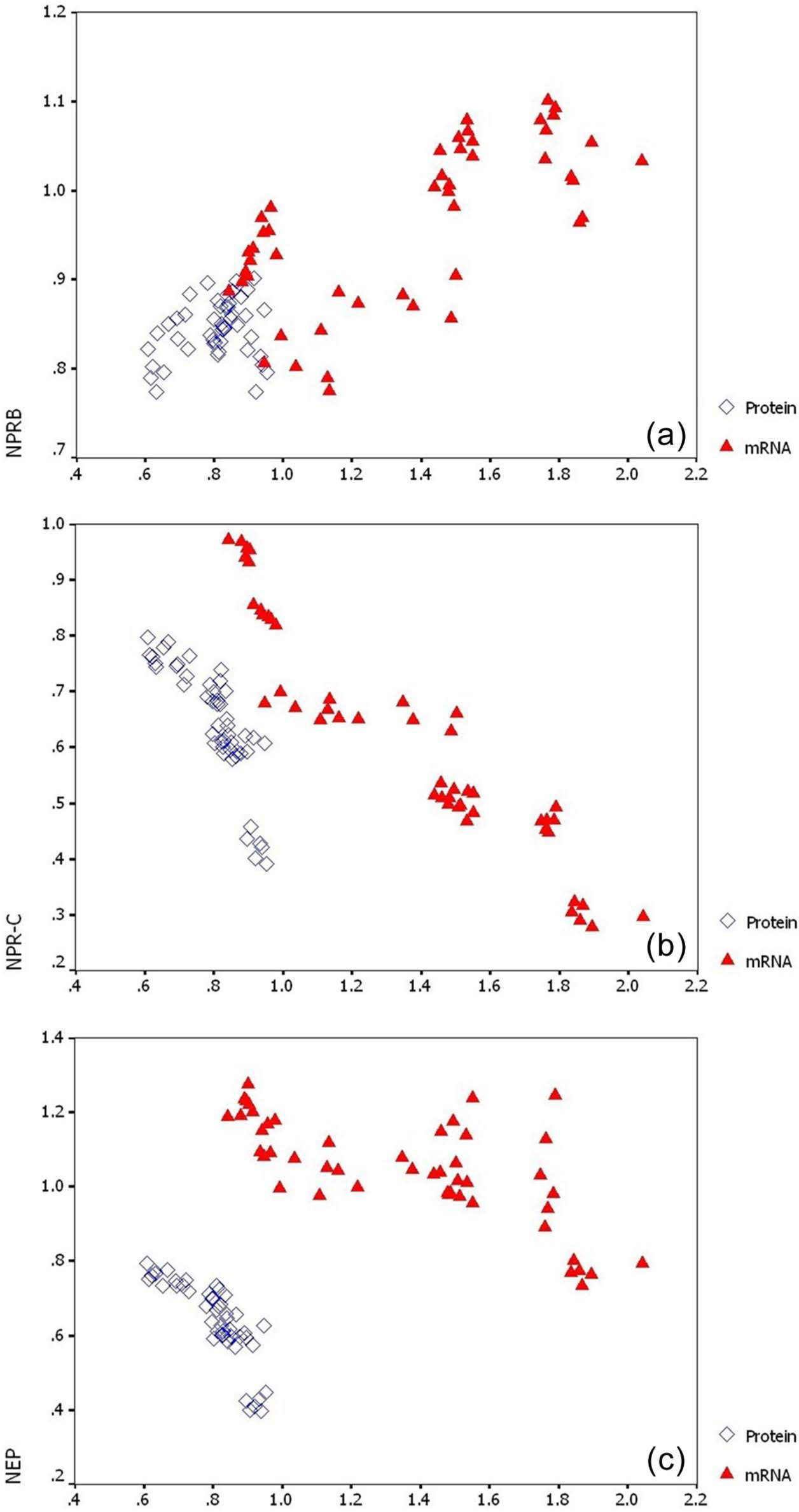
The correlations of (a) natriuretic peptide receptor (NPR)-B, (b) NPR-C, (c) neutral endopeptidase (NEP),with C-type natriuretic peptide (CNP) in the obstructed kidneys.
Discussion
During the last two decades, there has been a substantial increase in our knowledge of CNP, especially in the area of renal pathophysiology. It is clear that CNP expression is elevated in the obstructed kidneys. Our latest study published in
NPR-B, a single-span transmembrane protein, possesses a particulate guanylyl cyclase domain and is thought to specifically mediate the biological actions of CNP through the intracellular accumulation of cGMP.
16
Several prior studies have attempted to determined the location of NPR-B in the kidney. According to the report of Lohe et al.,
17
using real-time PCR of mRNA for NPR-B from microdissected sections of nephron, NPR-B mRNA was noted in the rat distal convoluted tubule, and cortical, outer medullary, and inner medullary tubule. Millul and coworkers revealed that NPR-B was expressed in a human cortical collecting duct cell line.
18
Similarly, CNP mRNA was also predominantly expressed in tubular epithelial cells. The presence of NPR-B and CNP in rat kidney confirms a role for CNP as an autocrine or paracrine factor. In the present study, our results demonstrated that NPR-B expression was significantly higher in the obstructed kidneys, compared to the left kidneys of SORs; subsequent correlation analysis indicated that CNP mRNA was positively correlated with NPR-B mRNA (
NPR-C does not possess the guanylyl cyclase domain and is thought to be engaged in the receptor-mediated degradation of CNP.
22
An earlier study conducted by Zhao et al. has demonstrated that NPR-C mRNA was mainly detected in glomerular mesangial and epithelial cells.
23
In the current study, our results indicated that UUO could increase NPR-C expression, which was accompanied by a significant decline in CNP (
NEP is a 90–110 kD zinc-dependent metallopeptidase, identical to the common acute lymphoblastic leukemia antigen.
25
It is expressed in brain tissue, on polymorphonuclear leukocytes and lymphoid progenitor cells, and on epithelial cells within nonlymphoid organs, such as the kidneys, the liver, the breasts and the lungs.
26
As the first human podocytic antigen, NEP has been documented to induce the onset of proteinuria in membranous nephropathy.
27
In our previous report, NEP was not only involved in the disarrangement of CNP catabolism, but also produced the enhanced glomerular permeability to protein in membranous nephropathy.
28
The current study focused on the expressive features of CNP and NEP in the obstructed kidneys for the first time, and found that NEP expression was markedly augmented in UUO rats during the period from 72 h to three months post-ligation, negatively correlated with CNP expression (
In summary, we demonstrated that CNP expression tends to be higher immediately after ligation and declined at later time points. A high level of CNP may contribute to the elevated expression of NPR-B in the early phase of UUO. More interestingly, the sustained upregulation of NPR-C and NEP observed throughout the study period (that is up to three months) helps to, at least partly, explain the subsequent decline of CNP.
Footnotes
Conflicts of interest
None declared.
Funding
This study was supported by the National Natural Science Foundation of China (No. 81000306) and the Post-Doctoral Foundation of Anhui Medical University (No. 2009KJ02).
