Abstract
Introduction:
adenosine monophosphate-activated protein kinase (AMPK) plays a prominent role as a metabolic stress sensor, and it has recently been suggested that the renin-angiotensin system, in addition to its role in stress regulation, may play a significant role in regulating the AMPK system. This study aimed to evaluate the effects of candesartan, an angiotensin II receptor blocker, on cardiac and hepatic AMPK activity basally as well as after surgical stress under general anesthesia.
Materials and methods:
Male Wistar rats were treated with 5 mg/kg/day candesartan in their drinking water for two weeks. Levels of cardiac and hepatic AMPK activity were determined, using a kinase activity assay, basally and after surgical stress under general anesthesia.
Results:
Chronic administration of candesartan increased hepatic AMPK activity approximately 4 times (p<0.05) while no significant change was demonstrated in cardiac AMPK. Cardiac and hepatic AMPK activities were not significantly increased by surgical stress alone performed under anesthesia. However, chronic treatment with candesartan decreased AMPK activity in both liver and heart after surgical stress under anesthesia (p<0.01 for both comparisons).
Conclusions:
While chronic candesartan treatment may stimulate AMPK activity in certain organs such as the liver, when combined with surgical stress under anesthesia it inhibits pathways regulating AMPK activity.
Introduction
Adenosine monophosphate (AMP)-activated protein kinase (AMPK), an enzyme functioning as a cellular sensor of low energy, promotes adaptive changes in metabolism.1,2 It works mainly in terms of activation in response to an increasing AMP/adenosine triphosphate (ATP) ratio, improving survival under metabolic stress. 3 The activation of AMPK in vivo is complex and varies according to the magnitude and type of the stress as well as the organ studied. 4 The AMPK system plays an important role in both heart and liver, 5 which are known as important organs involved in the stress response. However, it is necessary to clarify the tissue-specific and stress-specific activation mechanism of AMPK activation. 6
It has been suggested that the renin-angiotensin system may play a significant role in regulating the AMPK system. 7 Angiotensin II receptor blockers (ARBs) are considered to play a protective cardiovascular role through blood pressure lowering and beyond.8,9 Indeed, the use of ARBs in perioperative medicine has increased, 10 although the role of these agents in surgical practice has not been fully elucidated. While AMPK has been extensively studied in terms of its response to metabolic fuel deprivation, 11 very few studies are available on the in vivo modifications of AMPK responses to such a common stress scenario as laparotomy. We have recently shown that basal hypothalamic AMPK is upregulated by the ARB candesartan, although it was diminished in candesartan-treated animals submitted to surgical stress under anesthesia. 12
We have therefore sought to evaluate cardiac and hepatic AMPK activities under basal conditions and in response to the surgical stress of laparotomy in anesthetized rats chronically treated with candesartan in order to assess the pattern of AMPK activity response in the heart and the liver.
Material and methods
Animals
Male Wistar rats of 7–9 weeks of age (provided by the Federal University of Minas Gerais Medical School Animal Facility) were maintained under temperature-controlled conditions (25±2 °C) with an artificial 12-hour light-dark cycle, with lights on from 07:00–19:00, and allowed standard chow and water ad libitum. Rats were housed in individual cages and were handled daily.
A total of 28 animals were treated either with candesartan cilexetil (AstraZeneca, Cotia, Brazil) 5 mg/kg/day in the drinking water for two weeks (n=14) or no additions (vehicle, n=14) for the same period in their drinking water for two weeks utilizing metered drinking bottles with air-holes according to a well-established protocol13,14 for glucose and AMPK measurements. The University Ethics Committee for Animal Experimentation approved all procedures (Protocol No. 110/2007).
Experimental design
Following the two-week treatment with candesartan, rats were transferred in the early morning on the day of the experiment to the temperature-controlled experimental room and were left to rest for 60 min in their home cages. Noise was kept at a minimum. Experiments were performed from 08:00–12:00, and the animals had no access to water or food during the experiments.
Experiment animals were then anesthetized with 0.075 mg/g intraperitoneal ketamine (Cristália, Itapira, Brazil) and 0.01 mg/g xylazine (Schering-Plough Coopers, Cotia, Brazil) according to the protocol of the local Ethics Committee for Animal Research. This drug combination was suggested by the local Ethics Committee to induce a non-inhaled general anesthesia. After an average of 5 min, when rats were deeply anesthetized, a median laparotomy was performed, followed by evisceration and exposure of the bowels for 20 min 15 with exposed tissues kept warm and moist.
Non-stressed animals and those after 20 min of stress were moved to an adjacent room where they were killed by decapitation without anesthesia, followed by immediate removal of liver and heart. The organs were then placed in liquid nitrogen and stored at −80°C. Blood from the trunk was collected in sodium citrate 6% syringes and kept in ice until centrifugation (900 g, 20 min, 4°C). Plasma was then stored for subsequent glucose measurements.
Assays
The AMPK activity of the heart and liver tissues were evaluated at both baseline and after 20 min of surgical stress. We have chosen to evaluate AMPK activity after 20 min of stress based on our and others’ previous experience showing a stress peak, as evaluated by the hyperglycemic response, at around 20 min.15–18 Heart and liver of rats were weighed and homogenized with Precellys 24 using CK14 tubes containing ceramic beads (Stretton Scientific, Stretton, UK) at 3600 g for one cycle of 20 s in lysis buffer containing 50 mM Tris-HCl, 50 mM NaF, 5 mM Na pyrophosphate, 1 mM Ethylenediaminetetraacetic acid (EDTA), 250 mM sucrose, 1% Triton X-100, 1 mM Dithiothreitol (DTT), 1 mM benzamidine, 0.1 mM phenylmethane sulfonyl fluoride, 5 µg/ml soybean trypsin inhibitor. Tissue protein content was determined using bicinchoninic acid (BCA) assay (Pierce, Rockford, USA). AMPK activity was assayed as previously described: 19 briefly, AMPK was immunoprecipitated with an equal mixture of α1AMPK and α2AMPK antibodies, 19 and AMPK activity was determined by the entity of phosphorylation of SAMS (amino acid sequence: HMRSAMSGLHLVKRR, Pepceuticals Ltd., Nottingham, UK), a synthetic peptide substrate of AMPK. 19 AMPK activity was calculated using the difference of the counts between SAMS containing and SAMS negative samples and expressed as nanomoles of ATP incorporated per minute per milligram of sample of peptide. The sensitivity of the AMPK assay was 5 pmol/ATP/min/mg protein with an intra- and inter- assay coefficients of variation of 10% and 15%, respectively.
Statistical analysis
Non-parametric tests (Kruskal-Wallis test followed by Conover-Inman comparison) were performed for AMPK measurements, as these data were not normally distributed (Kolmogorov–Smirnov test). StatsDirect (Addison Wesley Longman, Cambridge, UK) and GraphPad Prism4 (San Diego, California, USA) were utilized for statistical analysis. All data are expressed as means ± standard error of the mean (SEM), and significance was taken at p<0.05.
Results
There was no difference between the two-week fluid intake of animals who on the day of the stress tests were assigned to stress or to control groups, suggesting that they had equal exposure to candesartan. There were no significant differences in plasma glucose levels as related to candesartan treatment when compared to respective non-stressed or stressed controls, as appropriate. Marked hyperglycemia as an index of sympathetic discharge was shown in surgically stressed animals utilized in this study. 12
Cardiac AMPK
After two weeks of candesartan treatment, there was no significant change in cardiac AMPK activity in candesartan-treated non-stressed rats when compared to the vehicle-treated non-stressed controls (Figure 1).

Cardiac adenosine monophosphate-activated protein kinase (AMPK) activity at baseline and after surgical stress under anesthesia in rats treated with candesartan for two weeks (n=6–8 for each group, Kruskal-Wallis test followed by the Conover-Inman test).
After 20 min of surgical stress under general anesthesia, no significant change could be observed in cardiac AMPK in the vehicle-treated stressed animals when compared to the non-stressed control group (0.018±0.001 vs 0.018±0.003 nmol/min/mg). However, there was a significant decrease in cardiac AMPK levels in candesartan-treated anesthesia-stressed animals when compared to the other groups (0.010±0.002 vs 0.023±0.003 nmol/min/mg, p<0.01 for comparison with candesartan-treated non-stressed animals; 0.010±0.002 vs 0.018±0.003 and 0.010±0.002 vs 0.018±0.001 nmol/min/mg, p<0.05 for comparisons with vehicle-treated non-stressed animals and with no-candesartan stressed animals, respectively; Figure 1). These data suggest that the surgical stress of laparotomy under general anesthesia is not associated with a stress-induced increase in cardiac AMPK activity. However, the combination of candesartan and laparotomy with anesthesia inhibits AMPK activity levels.
Hepatic AMPK
In the liver, two weeks candesartan treatment increased hepatic AMPK activity levels (0.045±0.011 vs 0.013±0.003 nmol/min/mg, p<0.05, Figure 2). After 20 min surgical stress under general anesthesia, there was a non-significant increase in hepatic AMPK in vehicle-treated stressed animals when compared to the non-stressed control group (0.029±0.007 vs 0.013±0.003 nmol/min/mg, p>0.05, Figure 2).
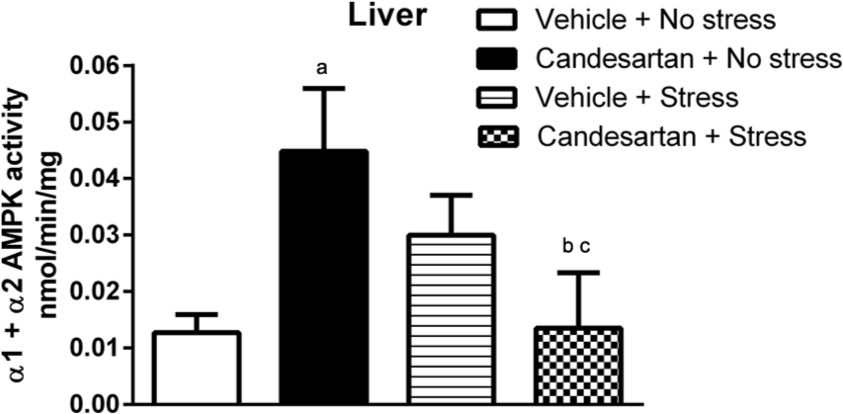
Hepatic adenosine monophosphate-activated protein kinase (AMPK) activity at baseline and after surgical stress under anesthesia in rats treated with candesartan for two weeks (n=6–7 for each group, Kruskal-Wallis test followed by the Conover-Inman test).
However, there was a significant decrease in hepatic AMPK activity levels in the candesartan-treated anesthesia-stressed group when compared to both the candesartan-treated non-stressed group (0.014±0.010 vs 0.045±0.011 nmol/min/mg, p<0.01, Figure 2) and the vehicle-treated stressed animals (0.014±0.010 vs 0.029±0.007 nmol/min/mg, p<0.05, Figure 2). These data show that the stress of laparotomy under general anesthesia (surgical stress) does not significantly change hepatic AMPK activity. However, when the same surgical stress is combined with candesartan it restores hepatic AMPK activity to values which are similar to non-stressed controls, suggesting that inhibitory AMPK pathways are switched on by this stress which can overcome the effects of candesartan upon hepatic AMPK activity observed on basal values.
Discussion
In this study, we have demonstrated that chronic candesartan treatment stimulates hepatic AMPK activity in non-stressed animals. Cardiac AMPK levels are not changed by chronic candesartan treatment. We have also shown that cardiac and hepatic AMPK are not changed significantly by surgical stress under general anesthesia, suggesting that the stress-anesthesia procedure inhibits pathways which we might anticipate should lead to a stress-induced increase in AMPK activity. However, when candesartan is combined with anesthesia and surgical stress, other mechanisms seems to be switched on: the ARB blocks the antiotensin receptor type 1 (AT1)-mediated effects of stress-induced angiotensin II release, 20 thus preventing stress-increases of both cardiac and hepatic AMPK, as demonstrated in vascular smooth muscle cells. 21 Consequently, chronic treatment with candesartan in combination with the inhibitory actions on AMPK of both anesthetics and stress-induced hyperglycemia inhibits AMPK activity levels after surgical stress in both heart and liver (Figure 3).

Proposed scheme for AMP-activated protein kinase (AMPK) regulation in an in vivo stress model under anesthesia and an angiotensin II receptor blocker (ARB). Thickness of the arrows is related to intensity of effects.
AMPK is a stress-activated protein kinase that works as metabolic sensor of cellular ATP levels. 1 AMPK not only senses energy status, but it also functions at the tissue and whole organism level to promote context-specific responses to physiological signals of metabolic status in response to ATP depletion and related stimuli. Several stress stimuli, such as exercise, anoxia and myocardial ischemia, have been described as activators of AMPK. 22
This is the first study, to the best of our knowledge, to identify the changes of cardiac and hepatic AMPK in the surgical stress of laparotomy. ARBs are widely used in clinical medicine, but very little is known regarding their effects on the AMPK system. 23 We have shown here that candesartan significantly increases hepatic AMPK activity in non-stressed animals, while surgical stress under anesthesia does not significantly change hepatic or cardiac AMPK activity. We have further addressed the in vivo complex interaction of candesartan, anesthetics and the hyperglycemic response in surgical stress, demonstrating that this combination ultimately shuts down cardiac and hepatic AMPK activity levels.
The AMPK system plays a major role in the regulation of hepatic and lipid metabolism through its acute effects on energy metabolism and long-term effects on gene expression pattern in the liver. 24 Candesartan has been otherwise shown to increase the expression of peroxisome proliferator-activated receptor-gamma (PPAR-γ) and adiponectin, 25 which are both known to increase hepatic AMPK activity.26–28 Therefore, both PPAR-γ agonism and the effect on adiponectin might have contributed to the observed increase on basal hepatic AMPK activity.25,29,30 In addition, as candesartan not only reduces the actions of angiotensin II, but also increases angiotensin-(1–7), we might also speculate that the alternative renin angiotensin system axis formed by angiotensin-converting-enzyme 2 (ACE2)-angiotensin-(1–7)/Mas receptor could play a role in hepatic AMPK activation. Likewise, we have recently shown that candesartan stimulates the hypothalamic AMPK system, and others have shown that another ARB, telmisartan, acts on skeletal muscle through activation of the PPAR/AMPK pathway. 23
Activation of hepatic AMPK causes a switch from an anabolic to a catabolic state and entails positive metabolic consequences such as inhibition of hepatic glucose production with restoration of blood glucose levels and regulation of cellular lipid metabolism through stimulation of fatty acid oxidation. Indeed, our findings of elevated basal hepatic AMPK levels in candesartan-treated animals are in agreement with possible beneficial effects of ARB on metabolism, pointing to an anti-steatotic effect in the liver. 31 However, despite the well-known protective effects of candesartan in the heart, 32 we did not detect in this study a significant elevation in cardiac AMPK activity of rats chronically treated with candesartan. Similarly, a recent study showed that activation of intrinsic cardiac ACE2 protects the heart against diabetes-induced dysfunction without significant changes in AMPK expression. 33
Interestingly, surgical stress under general anesthesia did not significantly change cardiac and hepatic AMPK activity. Indeed, general anesthesia reduces overall metabolism sparing AMPK, thus precluding stress-increases of AMPK in cardiac and hepatic cells as it turns off regulatory AMPK pathways. Likewise, we recently demonstrated the absence of significant changes in AMPK after surgical stress under this same anesthetic protocol for hypothalamic cells. 12 The role of anesthesia is further corroborated by a significant increase in hypothalamic AMPK activity after restraint, when anesthesia is not utilised. 12
The α2-adrenoceptor agonist xylazine utilized in these experiments acts centrally to decrease efferent sympathetic activity and circulating noradrenaline concentrations, thus preserving energy consumption in the periphery. 34 Ketamine, an N-methyl-D-aspartic acid receptor antagonist, has been recently demonstrated to lower brain AMPK activity when administered to rats before ischemia. 35 Thus, the anesthetic combination of ketamine and xylazine may have prevented a significant increase in AMPK due to surgical stress. However, these data may also be related to the marked hyperglycemia evoked by surgical stress under general anesthesia, thus reducing AMPK activity. 36
The role of AT1 receptors in the regulation of stress responses have been previously investigated.20,37,38 The production of circulating and local angiotensin II, as well as the expression of AT1 receptors increase during stress, 20 and the peripheral administration of an AT1 receptor antagonist otherwise blocks peripheral and brain AT1 receptors. 39 Furthermore, AT1 receptor mediates nicotinamide adenine dinucleotide phosphate hydrogen (NADPH) oxidase activation and increases reactive oxygene species (ROS) production, 40 which are known to upregulate AMPK. 41 Importantly, it has been demonstrated that the angiotensin II type I receptor antagonist valsartan significantly inhibits angiotensin II-induced AMPK activation in rat vascular smooth muscle cells, strongly suggesting that angiotensin II-induced AMPK activation is specifically AT1 receptor-dependent. 21 Furthermore, these data pointed to an AMPK inhibition effect on the proliferation signal induced by angiotensin II.
It has previously been demonstrated that the renin-angiotensin system is activated in anesthetized, surgically stressed rats. 42 In this study, candesartan blocked the AT1 receptor, thus inhibiting angiotensin-II-induced AMPK activation. This effect, in combination with the inhibitory effects of anesthetics and stress hyperglycemia, suppresses AMPK activity in the heart and liver (Figure 3). Further studies should address the molecular pathways as well as the significance of these changes. Interestingly, a possible role for angiotensin-(1–7) may also be considered to explain the observed changes in the candesartan-stressed group of animals. Our preliminary data, using the same stress protocol in transgenic rats over-expressing angiotensin-(1–7), suggest a reduction in hepatic and cardiac AMPK in these stressed rats when compared to the wild type (data not shown).
We have not included a group of rats with anesthesia alone (without surgery), as we wished to demonstrate the effects of combined anesthesia and surgery mirroring a ‘real life’ situation. However, as AMPK has been implicated in a wide range of metabolic pathways, apparent paradoxes and conflicts may be brought about, especially when utilizing an in vivo model.
In summary, this in vivo study shows that candesartan stimulates basal hepatic AMPK activity but has no significant effect on heart AMPK activity. It also shows that surgical stress under anesthesia is not associated with a significant increase in cardiac and hepatic AMPK activity. Furthermore, and in line with the fairly complex position of AMPK as a regulatory system, two weeks’ treatment with candesartan down-regulated cardiac and hepatic AMPK activity after surgical stress under anesthesia. These changes may need to be considered in the clinical situation where ARBs are in widespread use.
Conclusions
We show for the first time that chronic treatment with candesartan increases hepatic AMPK, while the combination of candesartan with surgical stress under anesthesia inhibits pathways regulating AMPK activity, suggesting a role of candesartan in modulating cardiac and hepatic AMPK activity in surgical stress under anesthesia.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Council of Technological and Scientific Development (CNPq), by the Fapemig (Fundação para o Desenvolvimento da Pesquisa do Estado de Minas Gerais), and by the Wellcome Trust.
