Abstract
Introduction:
This study was designed to investigate the underlying mechanisms of synergistic antihypertensive effect produced by combination therapy of losartan and pioglitazone in metabolic syndrome (MS) rats.
Materials and methods:
An MS model was induced by feeding rats a high-fat, high-sodium diet and 20% sucrose solution. Losartan (20 mg/kg/day), pioglitazone (10 mg/kg/day), and their combination were orally administered for eight consecutive weeks. Systolic blood pressure (SBP) and mean arterial pressure (MAP) were measured using the tail-cuff method and carotid arterial catheterization, respectively. The aortas were isolated and in vitro vascular reactivity studies were performed. The protein expression of angiotensin type 1 receptor (AT1), endothelial nitric oxide synthase (eNOS), phosphorylated eNOS and nicotinamide adenine dinucleotide phosphate (NADPH) oxidase subunit p47phox, level of nitrotyrosine as well as activity of eNOS and NADPH oxidase in aortas of MS rats were detected.
Results:
After eight weeks of treatment, the SBP and MAP in the losartan (115 ± 5 and 106 ± 6 mmHg), pioglitazone (130 ± 6 and 118 ± 6 mmHg), and combination therapy (105 ± 6 and 98 ± 5 mmHg) groups were lower than those in the model group (150 ± 8 and 136 ± 9 mmHg). Combination therapy of losartan and pioglitazone reduced BP more than either monotherapy, and showed additive effects on improving endothelial dysfunction and abolishing the increased vascular responsiveness to angiotensin II. These synergistic effects were associated with further reductions in protein expression of p47phox and AT1, NADPH oxidase activity, and nitrotyrosine level.
Conclusions:
Our data indicate that combined treatment exerts more beneficial effects on lowering BP and improving vascular lesions.
Introduction
Losartan (angiotensin II receptor blocker, ARB) is a commonly used drug indicated for the treatment of hypertension. Pioglitazone (peroxisome proliferator-activated receptor-γ, PPARγ agonist) is a useful prescription drug to improve insulin resistance and to treat type 2 diabetes. Several experimental studies have shown that pioglitazone has an antihypertensive effect in insulin-resistant and/or hypertensive animal models.1–10Clinical studies have reported that pioglitazone is either neutral 11–13 or associated with a mild decrease14–17 in blood pressure (BP). The BP-lowering effect of pioglitazone has been described in patients with type 2 diabetes, 14 type 2 diabetics with difficult-to-control hypertension, 15 nondipping diabetic patients, 16 and patients with congenital adrenal hyperplasia. 17 Interestingly, pioglitazone treatment abolishes the increased response of BP to high salt intake in salt-sensitive patients. 12
Our recent experimental findings suggested that consumption of a high-fat, high-sodium (HFS) diet and 20% sucrose solution for 16 consecutive weeks induces a metabolic syndrome (MS) rat model that mimics major characteristics of human MS, such as insulin resistance, dyslipidemia and hypertension.18,19 Furthermore, our 18,19 and other 20 reports revealed that the combination of pioglitazone and an ARB results in a synergistic effect on lowering BP in MS rats. Peredo et al. reported that combination therapy of losartan and pioglitazone exerts a marked trend toward more reduction of BP in fructose-overloaded Sprague-Dawley (SD) rats. 3 However, to the best of our knowledge, the underlying mechanisms of synergistic antihypertensive effect engendered by combination therapy are still unknown.
It is of note that endothelial function and the angiotensin II (Ang II) signaling pathway play essential roles in the modulation of BP. Aortic endothelial dysfunction and enhanced Ang II-induced contraction have been induced in SD rats fed a high-fat, high-sucrose or high-salt diet.21–25 Therefore, we hypothesized that vascular dysfunctions could also be induced in our abnormal diet-induced MS rats. In addition to its insulin-sensitizing action, the hypotensive effect of pioglitazone is attributed to the amelioration of endothelial function and abolishment of increased responsiveness to Ang II.2,4,5 As a result, this study was designed to evaluate whether pioglitazone enhanced the blood pressure-lowering effect of losartan in MS rats through these pathways.
Materials and methods
Reagents
A nitrotyrosine enzyme-linked immunosorbent assay (ELISA) kit was purchased from Cell Biolabs (San Diego, CA, USA). A total protein assay kit and superoxide dismutase (SOD) were purchased from Beyotime Institute of Biotechnology (Shanghai, China). A nitric oxide synthase (NOS) activity assay kit was purchased from Jiancheng Institute of Biotechnology (Nanjing, China). Polyclonal rabbit anti-endothelial nitric oxide synthase (eNOS), nicotinamide adenine dinucleotide phosphate (NADPH) oxidase subunit p47phox and angiotensin type 1 receptor (AT1) antibodies were purchased from Santa Cruz Biotechnology Inc (Santa Cruz, CA, USA). Polyclonal rabbit anti-phosphorylated eNOS (Ser1177) was purchased from Cell Signaling Technology Inc (Danvers, MA, USA). Monoclonal mouse anti-β-actin antibody was purchased from Boster Biotechnology Inc (Wuhan, China). Norepinephrine, phenylephrine (Phe), acetylcholine (Ach), sodium nitroprusside (SNP), angiotensin II (Ang II), cytochrome c, and NADPH were purchased from Sigma (St. Louis, MO, USA). The composition of Krebs solution was as follows (in mM): NaCl 118.3, KCl 4.7, CaCl2 2.5, KH2PO4 1.2, MgSO4 1.2, NaHCO3 25, and glucose 11.1.
Animals and diet
Male SD rats (240 ± 20 g, obtained from Zhejiang Province Experimental Animal Center, certificate number of the breeder: SCXK (zhe) 20080033) were maintained under controlled conditions of light, temperature, and humidity. In this study, all rats received humane care in accordance with the Guide for the Care and Use of Laboratory Animals published by the United States National Institutes of Health (NIH publication no. 85-23, revised 1996). An HFS diet was prepared as described in our previous study 19 and was composed of 4% sodium chloride, 21.1% protein, 20% fat (containing 15% lard oil and 1% cholesterin), 33% carbohydrate, and a standard vitamin and mineral mix.
Experimental design
As described in our previous study,18,19 rats were fed the HFS diet and 20% sucrose solution for 16 consecutive weeks except for the control group (C, n = 11, normal diet and water). After eight weeks of receiving the abnormal diet, the rats (except the C group) were randomly assigned to four groups, namely, MS model group (M, n = 11), losartan treatment group (L, MSD Pharmaceutical Co., 20 mg/kg, n = 10), pioglitazone treatment group (P, Takeda Pharmaceutical Co., 10 mg/kg, n = 10), and combined treatment group (received losartan plus pioglitazone at the doses described above, L+P, n = 10). The dosages of drugs and duration of treatment were selected according to previous studies.26,27 The drugs were administered orally at 9:00–10:00 a.m. by gavage for the following eight weeks on a daily basis. Untreated groups received an equal volume of distilled water.
During the entire period of the experiment, body weight (BW) was measured weekly. Systolic blood pressure (SBP) was measured on a monthly basis in conscious rats using the tail-cuff method (ALC-NIBP, Shanghai, China) as described in our previous studies.19,28 The rats were placed in metabolic cages to collect 24-hour urine at the 16th week. At the end of the study, the rats were fasted overnight and anesthetized by an intraperitoneal injection of sodium pentobarbital. Invasive carotid BP was recorded according to a previous report. 29 Briefly, a sterile polyethylene cannula (internal diameter: 0.9 mm) pre-filled with heparinized normal saline (0.5 IU/ml) and coupled to a YP-100 pressure transducer (Xinhang Electric Apparatus, China) was inserted through the right common carotid artery and placed in the middle of the lumen for intra-arterial BP measurement. Mean arterial pressure (MAP) was calculated as diastolic pressure plus one-third pulse pressure.
Then, blood samples were drawn from the abdominal aorta instantly, centrifuged to obtain plasma, and kept at −20°C until assayed. Aortas of rats were carefully removed, placed in cold oxygenated (95% O2−5% CO2) Krebs solution and cleaned of adhering tissue. Two transverse rings (3 mm in length) were cut from part of the descending thoracic aortas and used for vascular reactivity experiments. The remaining aortas were quickly frozen in liquid nitrogen and stored at −80°C until processed. Visceral fat mass was excised and weighed. Visceral fat weight/BW (VisF/BW) ratio was calculated. Obesity index was calculated as the cubic root of BW (g) divided by body length (cm) × 103.
Vascular reactivity studies
As mentioned in our previous study, 28 the aortic ring was suspended by a pair of stainless steel hooks in a water-jacketed organ bath containing 10 ml Krebs solution at 37°C, and continuously bubbled with carbogen. Changes in isometric tension were detected by a JZ101 force transducer (Xinhang Electric Apparatus, China). Preload (1.5 g) was applied to the rings, and the vessels were allowed to equilibrate for 60 minutes (with four washouts). In some preparations, the vascular endothelium was deliberately removed by gently rubbing the lumen side of the aorta rings. At the beginning of the in vitro vascular function experiments, all rings were stimulated with norepinephrine (3×10−7 M) to evaluate their viability. The subsequent treatment with ACh (10−6 M) was to test the integrity of the endothelium. Then, all rings were serially washed to the baseline and equilibrated again.
The concentration-relaxation response curves to ACh (10−8 to 10−4 M) and SNP (10−10 to 10−6 M) were performed in endothelium-intact aortic rings, which were precontracted by Phe (10−6 M). The relaxant responses to ACh and SNP were calculated as a percentage of the response to Phe.
The concentration-contraction response curves to Ang II (10−10 to 10−6 M) and Phe (10−9 to 10−5 M) were constructed by cumulative addition of the agents in endothelium-denuded rings. At the completion of the contraction study, the rings were blotted dry and weighed, and the lengths were measured to calculate tension as normalized for cross-sectional area by the formula: cross-sectional area (mm2) = weight (mg)/[length (mm) × density] with the density of vascular smooth muscle being 1.05 mg/mm3 as previously described.25,30
In addition, nonlinear regression was used to fit sigmoidal curves to individual concentration-response curves for the purpose of determining agonist potency (EC50), the dose of all agents giving half the maximum response. Area under the curve (AUC) was calculated from the individual concentration-response curve plots using the trapezoidal integration method.
Determination of physiological indices
Levels of plasma glucose, total cholesterol (TC) and triglycerides (TG) were determined using commercial kits (Rongsheng Biotechnology Co., Shanghai, China). Sodium content in the urine was determined by an electrode method. Plasma insulin level was measured using a radioimmunoassay (RIA) kit (North Institute of Biotechnology, Beijing, China). Homeostatic model assessment of insulin resistance (HOMA-IR) was calculated using the following equation: HOMA-IR = [fasting glucose (mmol/l) × fasting insulin (μIU/ml)/22.5]. 31
Determination of aortic NADPH oxidase and eNOS activity
Part of the vascular tissues were minced and homogenized in lysis buffer (10 mM Tris pH 7.1, 340 mM sucrose, 1 mM ethylenediaminetetraacetic acid (EDTA), 25 mM β-glycerophoshate, 1 mM Na3VO4, and 1% protease inhibitor cocktail). The homogenate was centrifuged (6,000 g at 4°C for five minutes) to obtain supernatant. The activity of NADPH oxidase in aortic tissues was determined by using SOD-inhibitable cytochrome c reduction assay as described previously.32,33 In brief, the supernatant samples were incubated in reaction buffer containing ferricytochrome c (80 μM), NADPH (100 μM), with or without SOD (400 U/mL) at 37°C for 45 minutes. Then, the absorbance was measured at 550 nm. NADPH oxidase activity was quantified from the absorbance with or without SOD, and results were normalized against the mean value of control and expressed as fold changes.
Aortic eNOS activity was determined using an NOS activity assay kit as previously described.34,35 Briefly, the supernatant samples were added to the reaction buffer containing L-arginine, NADPH, calcium (not present in iNOS assay buffer), calmodulin, tetrahydrobiopterin, nitroblue tetrazolium (NBT), and phenazine methosulfate (PMS). Fifteen minutes after incubation at 37°C, reaction was stopped by adding a termination buffer. Formazan, the reaction product of NBT/PMS with NADPH in the presence of NO, was quantified spectrophotometrically at 530 nm. The activity of eNOS was obtained by subtracting iNOS activity from the total NOS activity, and results were normalized against the mean value of control and expressed as fold changes.
Determination of aortic nitrotyrosine level
Parts of the vascular tissues were homogenized in lysis buffer (50 mM Tris pH 7.4, 150mM NaCl, 1% NP-40, 0.5% sodium deoxycholate, 0.1% SDS, 1 mM EDTA, 25 mM β-glycerophoshate, 1 mM Na3VO4, 2 mM sodium pyrophosphate, and 1% protease inhibitor cocktail) and centrifuged (12,000 g at 4°C for 15 minutes). The supernatant was used for ELISA and Western blot analysis.
Aortic nitrotyrosine level was measured using a commercial ELISA kit according to the manufacturer’s instructions. Briefly, the supernatant samples were transferred to a 96-well plate. The plate was incubated at room temperature for 10 minutes. The diluted anti-nitrotyrosine antibody was added to each well and incubated at room temperature for one hour. After three washes, the diluted secondary antibody-enzyme conjugate was added and further incubated for one hour. After washing a further three times, a warm substrate solution was added and further incubated for 10 minutes, a stop solution was added to each well, and the absorbance was measured at 450 nm. The values of each sample were normalized, with the protein concentrations measured using the bicinchoninic acid assay.
Western blot analysis
As described in our previous study,18,28 equal amounts of protein (40 μg protein/lane) were separated by electrophoresis on a sodium dodecyl sulfate polyacrylamide gel. The proteins were transferred electrophoretically to nitrocellulose membranes. After blocking with 5% skim milk in Tris-buffered saline at room temperature for one hour, the membrane was incubated with the primary antibodies against eNOS, phosphorylated eNOS, p47phox, AT1 or β-actin overnight and with the correspondent secondary peroxidase-conjugated anti-rabbit or mouse antibodies. The blots were visualized using an enhanced chemiluminescence kit (Pierce, Rockford, IL, USA) and evaluated by densitometry using image J software. The intensity of the bands was normalized to that of β-actin.
Statistical analysis
Data were expressed as mean ± SD. Comparisons among the five groups were determined by the use of analysis of variance (ANOVA) followed by a Newman-Keuls test. Changes in the BW, SBP, and dose-response curves over time were analyzed by two-way repeated-measures ANOVA followed by a Newman-Keuls test. p < 0.05 was considered statistically significant.
Results
Physiological indices
As shown in the Table 1, the BW, BW gain and obesity index in the MS group were significantly increased (p < 0.05 versus control group) and not obviously influenced by any of the drug therapies. The VisF/BW ratio, plasma levels of insulin, TC and TG, and HOMA-IR in the MS group were conspicuously enhanced compared with those in the control group. Pioglitazone treatment reduced the VisF/BW ratio (p < 0.05 versus MS group). Treatments with pioglitazone alone and the combination of losartan and pioglitazone both resulted in decreased plasma insulin, TC and TG levels, and HOMA-IR (p < 0.05 versus MS group). There were no marked differences in the glucose concentration and sodium excretion among all experimental groups.
Comparison of physiological indices in different experimental groups.

Body weight (BW) gain was calculated by subtracting the initial weight from the final weight. Obesity index was calculated as the cubic root of BW (g) divided by body length (cm) × 103. VisF/BW: visceral fat weight/BW. Values are expressed as mean ± SD, n = 10–11 per group; ap < 0.05 vs. control group; bp < 0.05 vs. model group; cp < 0.05 vs. L+P group; C: control; M: metabolic syndrome (MS) model; L: losartan (20 mg/kg per day); P: pioglitazone (10 mg/kg per day); L+P: losartan (20 mg/kg per day) plus pioglitazone (10 mg/kg per day); HOMA-IR: homeostatic model assessment of insulin resistance.
Non-invasive and invasive techniques for the monitoring of rat BP were employed in our study to measure the SBP and MAP, respectively. As shown in Figures 1(a) and 1(b), after receiving the abnormal diet for 16 weeks, the SBP and MAP in the MS model group were obviously higher than those in the control group. After eight weeks of treatment, the SBP and MAP in the losartan (115 ± 5 and 106 ± 6 mmHg), pioglitazone (130 ± 6 and 118 ± 6 mmHg), and combination therapy (105 ± 6 and 98 ± 5 mmHg) groups were lower than those in the MS group (150 ± 8 and 136 ± 9 mmHg). Treatment with the combination yielded further reductions in the SBP and MAP than administration of either drug alone.

Effects of losartan (L), pioglitazone (P), and their combination (L+P) on systolic blood pressure (SBP) and mean arterial pressure (MAP).
In vitro vascular activity
As shown in Figure 2, the vascular endothelial-dependent relaxation induced by ACh was obviously impaired in the MS rats. The Emax and AUC values of ACh were significantly lower in aortic rings obtained from the MS rats than those from the control rats. Treatments with losartan alone, pioglitazone alone, and their combination ameliorated the impairment of endothelial-dependent relaxation, as the Emax and AUC values were markedly increased in all treatment groups (p < 0.05 versus MS group). Combination therapy resulted in a further improvement of vasorelaxation than either monotherapy. As shown in Figure 3, the endothelium-independent vasorelaxation induced by SNP was not different from the control and MS model groups, and not affected by any drug treatment.

Effects of losartan (L), pioglitazone (P), and their combination (L+P) on endothelial-dependent relaxation induced by acetylcholine.

Effects of losartan (L), pioglitazone (P), and their combination (L+P) on endothelium-independent vasorelaxation induced by sodium nitroprusside.
As shown in Figure 4, Ang II induced a concentration-dependent contraction in endothelium-denuded aortic rings. The Emax and AUC values of Ang II were evidently higher in aortic rings obtained from the MS rats than those from the control group. Either monotherapy or combination therapy reduced the value of Emax and AUC (p < 0.05 versus MS group). Combination therapy contributed to further reductions in Emax and AUC than either monotherapy. The vascular contraction induced by cumulative concentrations of Phe in endothelium-denuded rings was alike among all experimental groups (Figure 5).

Effects of losartan (L), pioglitazone (P), and their combination (L+P) on vascular contractile responses induced by angiotensin II.

Effects of losartan (L), pioglitazone (P), and their combination (L+P) on vascular contractile responses induced by phenylephrine.
Aortic eNOS expression and activity
As shown in Figures 6(a) and 6(b), neither abnormal diet nor any drug treatment had a significant effect on aortic protein expression of eNOS. Nevertheless, the phosphorylated level and activity of eNOS were obviously reduced in the MS group, and these abnormal changes were essentially reversed by either losartan monotherapy or combination therapy. Combination therapy did not result in further enhancements than monotherapy (Figures 6(a), 6(d) and 7(c)).

Effects of losartan (L), pioglitazone (P), and their combination (L+P) on the protein expression of total endothelial nitric oxide synthase (eNOS), phosphorylated-eNOS and p47phox in aortic tissues by Western blotting.

Effects of losartan (L), pioglitazone (P), and their combination (L+P) on the aortic activity of nicotinamide adenine dinucleotide phosphate (NADPH) oxidase and endothelial nitric oxide synthase (eNOS) as well as nitrotyrosine level.
Aortic p47phox expression, NADPH oxidase activity and nitrotyrosine level
As shown in Figures 6(a), 6(c), 7(a), and 7(b), the aortic protein expression of p47phox, NADPH oxidase activity as well as nitrotyrosine level in the MS model group were markedly higher than those in the control group. These abnormal changes were partially reversed by treatment with either losartan or pioglitazone alone. Combination therapy further reversed these abnormal changes as compared with administration of either drug alone.
Aortic AT1 expression
As shown in Figure 8, protein expression of AT1 was significantly increased in the MS rats (p < 0.05 versus control group). Treatment with either losartan or pioglitazone alone downregulated the aortic AT1 expression. In addition, combination therapy resulted in a synergistic effect on reducing aortic AT1 expression to almost the normal level as compared with either monotherapy.
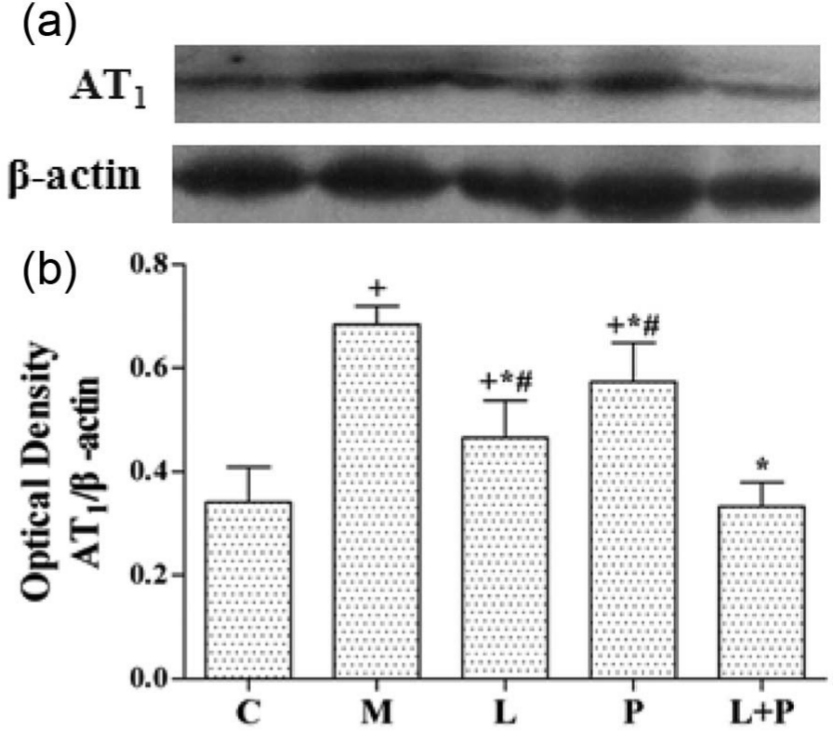
Effects of losartan (L), pioglitazone (P), and their combination (L+P) on the protein expression of angiotensin type 1 receptor (AT1) in aortic tissues by Western blotting.
Discussion
The major conclusions to be drawn from this study were that chronic combination therapy of losartan and pioglitazone showed synergistic effects on lowering BP, improving endothelial dysfunction and decreasing Ang II-induced vasoconstriction. These synergistic effects might be attributed, at least in part, to further amelioration of aortic nitric oxide (NO) inactivation and suppression of AT1 protein expression.
Establishing a valuable MS model accompanying stable hypertension
To date, the length of the time that it takes to form stable hypertension in rats fed a high-fat, high-sucrose diet remains controversial. Both one month21,36 and time longer than six months37,38 have been reported. In addition, the causes of hypertension in humans are not fully understood, but the fact that hypertension is correlated with sodium intake is widely acknowledged; however, data in regards to high-salt intake alone leading to hypertension in SD rats are somewhat controversial because data of both increased24,39,40 and unchanged41,42 BP have been reported. It is of interest that the high-fat, high-sucrose diet-fed rats were associated with marked salt sensitivity, as evidenced by a significant rise in BP after the intake of a high-salt diet. 43 Therefore, in this study, sodium was added to the diet for the purpose of establishing the MS model with stable hypertension. To clarify our points, we set up another group of SD rats (n = 6) with normal sodium content in their diet. At the fourth week, it turned out that the SBP in this group, which is obviously lower than that of the MS model group, had no significant difference from that of the control group (data not shown). To sum up, an addition of high sodium to the high-fat, high-sucrose diet could yield an MS rat model with stable hypertension in the early phase as expected.
Effects of pioglitazone on BP levels
As far as we are concerned, pioglitazone (especially at higher doses, ≥10 mg/kg/day) has been revealed to lower BP in Dahl salt-sensitive and one-kidney, one-clip SD rats, 1 Ang II-infused SD rats, 2 fructose-overloaded SD rats, 3 Wistar rats receiving chronic treatment of cyclosporine, 4 Zucker obese rats fed a high-protein diet, 5 SD rats receiving a high-fat diet, 6 sucrose-fed or normal-fed spontaneously hypertensive rats,7–9 and streptozotocin-induced diabetic SD rats. 10 Consistent with these reports, in our experimental conditions, daily oral administration of pioglitazone (10 mg/kg) for eight weeks induced a progressive reduction in the BP of MS rats. However, the BP-lowering effect of pioglitazone has not been reported in a few other animal studies.44–46 The reasons for this discrepancy are not clear, but such variability is generally attributed to differences in the dose of pioglitazone, the animal models and duration of hypertension, the BP measuring techniques, and the experimental conditions.
Synergistic effect on improving endothelial dysfunction
In accordance with our previous hypothesis, endothelial dysfunction occurred in our MS rats and was characterized by depressed endothelium-dependent vasorelaxation. Combination therapy of losartan and pioglitazone improved endothelial dysfunction of MS rats to a greater extent than either monotherapy. Among the various endothelium-derived molecules, NO has been demonstrated to play a key role in the regulation of vascular tone and BP. Reduction in production and/or oxidative inactivation of NO lead to diminished NO bioactivity. Accordingly, several potential mechanisms would be involved in the combination therapy-induced synergistic effect on improving endothelial dysfunction, such as changes in the expression and/or activity of eNOS, and in the oxidative inactivation of NO.
It is well known that eNOS is the major contributor to NO production. However, the protein expression of aortic eNOS was not obviously altered in our MS rats. As revealed by Roberts et al.,38,43 a high-fat, high-sucrose diet reduces aortic eNOS expression in the early phase (before onset of hypertension), whereas eNOS may be upregulated in the latter phase as a possible compensatory attempt to reestablish BP homeostasis. Hence, we speculate that there may be a time-dependent alteration in the eNOS expression, and high salt may speed up this compensatory expression because the stable hypertension of our MS rats is generated in the early phase. Although the expression of eNOS protein did not change, phosphorylation level and activity of eNOS were significantly decreased in the aortas of MS rats. These findings were in accordance with previous data from others showing depressed p-eNOS protein expression and attenuated eNOS activity in SD rats with high-salt or high-fat intake.22,35 In this study, the activity and phosphorylation of eNOS were obviously increased by treatment with either losartan or combination therapy, while these beneficial effects were not observed in monotherapy of pioglitazone. All of these data were similar with others reports.47–49 Hence, the synergistic effect on improving endothelial function by combination therapy may occur through other mechanisms.
NADPH oxidase has been shown to be a major source of superoxide anion (O2−, the main species of reactive oxygen species) in cardiovascular tissues. 50 O2− reacts avidly with NO to generate peroxynitrite, which reacts with tyrosine residues to produce nitrotyrosine (a stable marker for oxidative inactivation of NO). 51 Consistent with an earlier finding, 52 the elevations of aortic nitrotyrosine level, p47phox protein expression as well as NADPH oxidase activity were also found herein. Accordingly, the increased p47phox expression and NADPH oxidase activity would raise O2− production, and thereby contribute to the elevation of nitrotyrosine expression and NO inactivation in our MS model. Combination therapy further reversed these abnormal changes as compared with administration of either drug alone. These data indicate that the synergistic effect on improving endothelial dysfunction by combination therapy might be attributed to a further amelioration in the oxidative inactivation of NO.
Synergistic effect on reducing Ang II-induced vasoconstriction
High-salt intake, which causes hypertension in some individuals, induces a decrease in the activity of the circulating renin–angiotensin system, and this is thought to be involved in the accompanied upregulation of AT1 receptor expression. Previous studies have shown that cardiovascular AT1 expression and Ang II-induced vasoconstriction are increased in SD rats fed a high-salt diet.24,40 The enhanced Ang II-induced contraction occurs in the thoracic aortas of SD rats receiving a high-fat 25 or high-fructose diet. 23 However, current evidence suggests that aortic contraction induced by Phe is not altered in SD rats fed a high-fat, high-sucrose, 21 high-salt 53 or high-fructose diet. 23 In accordance with these results, we also observed an increase in the vascular response to Ang II and an upregulation of aortic AT1 expression in MS rats, while the Phe-evoked contraction was not obviously changed. The evidence obtained suggests that AT1-mediated Ang II signaling is essential for the maintenance of systemic BP in the states of insulin-resistant and high-salt intake.
Zanchi et al. revealed that pioglitazone decreases the expression of AT1 and abolishes the increased responsiveness to Ang II induced by a high-salt diet in obese Zucker rats. 54 In the Ang II-infused rats, pioglitazone attenuates the development of hypertension and prevents upregulation of AT1. 2 In this study, treatment with either losartan or pioglitazone alone also reduced the vascular contraction induced by Ang II and depressed the aortic AT1 expression, but the levels were still higher than those in the control group. Moreover, combination therapy yielded more improvements in these abnormal changes than either monotherapy. These results suggest that combination therapy generates a synergistic effect on reducing the Ang II-induced vasocontraction through a further downregulation of AT1.
In vitro experiments confirmed that Ang II (10−10 to 10−6 M) elicits concentration-dependent relaxation in normal and diabetic rat thoracic aortas incubated with losartan (10 −4 M). 55 Concentration-response curves to Ang II (10−10 to 3 × 10−7 M) are almost abolished in the carotid arteries of losartan-treated (15 mg/kg/day, 16 weeks) spontaneously hypertensive rats (SHRs). 56 However, Cosentino et al. showed that the Ang II contractile effects in aortas from the losartan treatment group (20 mg/kg/day, three weeks) are lower than those measured in aortas from streptozotocin-induced diabetic SD rats, but the values remained higher than those in normal group. 57 Failli et al. reported that Ang II (10−6 M) also induces contraction in aortas isolated from SHRs treated with losartan (30 mg/kg/day, eight weeks). 58 Taken together, we suppose that chronic treatment with losartan alone (20 mg/kg/day, eight weeks) could not abolish the aortic contraction induced by Ang II completely, while combination therapy of losartan and pioglitazone could reduce the contraction to below normal level in SD rats fed with the HFS diet and 20% sucrose solution.
The principal mechanism of synergistic antihypertensive effect
On the one hand, hydralazine (a vasodilator agent) obviously lowers BP but does not have any effect on aortic endothelial dysfunction in Ang II-infused SD rats. 2 On the other hand, combination treatment with low doses of candesartan and pioglitazone (without BP-lowering effect) also exerts a further improvement of endothelial dysfunction in stroke-prone SHRs. 47 As we can see from the above mentioned, there does not seem to be a strong relationship between synergistic BP-lowering effect and amelioration of endothelium-dependent vasorelaxation. Therefore, we suppose that the main cause of synergistic antihypertensive effect provided by combination therapy of losartan and pioglitazone is its further reduction on Ang II-induced vasoconstriction. However, the question whether this synergistic effect could be expanded to other animal models with hypertension calls for further investigation.
Study limitations
There are two limitations that need to be acknowledged and addressed regarding the present study. First, the dose of pioglitazone used in the experiment is much higher than that used in usual clinical practice. Further studies should be carried out to evaluate the effects of the recommended dose of pioglitazone. Second, we assessed the effects of drugs on rat aortic responsiveness. In fact, resistant vessels play a more important role during the development of hypertension. Therefore, whether combination therapy of losartan and pioglitazone also improves the reactivity of resistance vessels (tail and mesenteric arteries) requires further investigation.
Conclusions
In summary, our results suggest that pioglitazone enhances the antihypertensive effect of losartan through synergistic attenuation of Ang II-induced vasoconstriction in SD rats fed an HFS diet and 20% sucrose solution.
Footnotes
Acknowledgements
The authors thank MSD Pharmaceutical Co, Hangzhou, China, for the gift of losartan.
Conflict of interest
None declared.
Funding
This research was supported by the Natural Science Foundation of Education Department of Anhui Province (grant number KJ2010B466) and the National Natural Science Foundation of China (grant number 81000332).
