Abstract
Background:
Several polymorphisms in the angiotensin-converting enzyme (ACE) and ACE2 genes are associated with the development of end-stage renal disease (ESRD). Certain genetic polymorphisms may modify the deleterious effects of environmental factors such as cigarette smoking and may also modify the inherited risk. We investigated the association of six ACE and ACE2 polymorphisms with ESRD to determine whether a relationship exists between gene–smoking interactions and ESRD.
Materials and methods:
We performed a case-control association study and genotyped 683 ESRD patients and 653 healthy controls. All subjects were genotyped for ACE (I/D, G2350A and A-240T) and ACE2 (G8790A, A1075G and G16854C) gene polymorphisms by polymerase chain reaction–restriction fragment length polymorphism (PCR-RFLP) analysis.
Results:
Significant associations were observed between ACE I/D and G2350A polymorphisms and ESRD. There was no difference in ACE2 genotype distribution between ESRD patients and healthy controls. Haplotype analysis showed that DAA and DAT haplotypes were risk factors for ESRD. Moreover, a gene–environment interaction was observed between ACE I/D polymorphism and cigarette smoking.
Conclusion:
ACE I/D and ACE G2350A polymorphisms were associated with the development of ESRD. The interaction between ACE I/D polymorphism and smoking is also associated with an enhanced risk of ESRD.
Keywords
Introduction
Taiwan has the third highest incidence rate and the highest prevalence rate of end-stage renal disease (ESRD) in the world. 1 The high prevalence and incidence of ESRD not only burdens healthcare resources but also has a major impact on patients and their families. ESRD is a multifactorial and polygenic disorder predisposed by genetic, environmental and lifestyle factors. 2 Among the predisposing genetic factors, disruption of the renin–angiotensin system (RAS) is clearly associated with the development of ESRD.3,4
RAS is a key regulator of both blood pressure and kidney function and may play a role in their interaction. Angiotensin-converting enzyme (ACE) is a key enzyme in the RAS and has been widely used for association studies on ESRD. ACE is a carboxyl terminal dipeptidyl exopeptidase that converts angiotensin I to angiotensin II. ACE2, a homologue of ACE, is a monocarboxypeptidase that preferentially removes carboxy terminal amino acids from various substrates, including Ang II, Ang I and apelin. Approximately 42% of residues in the catalytic domain of ACE2 are also present in endothelial ACE. 5 Recent studies have shown that ACE2 is also a key modulator of RAS in renal diseases.6,7 Several recent studies have identified mutations and polymorphisms in ACE and ACE2 genes that affect host susceptibility to a range of diseases including hypertension, 8 myocardial infarction, 9 type 2 diabetes mellitus, 10 diabetic nephropathy, 11 and ESRD. 12 Some studies have also indicated that single nucleotide polymorphisms (SNPs) in ACE and ACE2 genes can alter homeostasis to an abnormal state.13,14
Smoking is a known risk factor for the development and progression of ESRD;15,16 however, little is known of the effects of smoking exposure on genetic variants influencing kidney traits in the general population. Moreover, the exact mechanism by which ACE and ACE2 polymorphisms affect ESRD progression is unclear. We hypothesized that interactions between multiple genetic variants of ACE and ACE2 and smoking status are associated with ESRD. Therefore, in this study, we investigated the association of ACE and ACE2 gene polymorphisms, and interactions between these gene polymorphisms and smoking status, with ESRD susceptibility.
Material and methods
This case-control study included 683 ESRD patients (368 females and 315 males; mean age 64.27 ± 14.69 years) visiting the Cardinal Tien Hospital and five haemodialysis centres in Taipei, Taiwan. The enrolled patients were stable (without clinical complications), aged over 20 years and had undergone haemodialysis (HD) for more than 6 months. Patients with autoimmune diseases, malignancy and acute or chronic infections were excluded. The cause of ESRD was diabetes mellitus in 295 patients (43.2%), chronic glomerulonephritis in 184 patients (27.0%), hypertensive nephropathy in 83 patients (12.2%), systemic nephropathy in 64 patients (9.4%) and unknown in 57 patients (8.2%). The study also included 653 healthy control subjects (381 females and 272 males; mean age 65.79 ± 13.59 years) from the Center of Physical Examination of the Cardinal Tien Hospital, with an estimated glomerular filtration rate (eGFR) of ≥ 60 ml/min/1.73 m2 and no proteinuria. The healthy controls had no kidney damage, proteinuria, haematuria or abnormalities on abdominal ultrasonography. Clinical information and biochemical parameters were obtained retrospectively from hospital records. The study was reviewed and approved by the institutional ethical committee of the Cardinal Tien Hospital (CTH-100-3-5-025). Written informed consent was obtained from all participants after giving them a complete explanation of the study.
Cigarette smoking
Cigarette smoking status (current, former, never) was ascertained at the time of enrollment. Individuals who had not smoked more than 100 cigarettes in their lifetime were classified as never-smokers, based on common conventions in epidemiologic research. 17 Former smokers were those who had smoked more than 100 cigarettes in their lifetimes but did not currently smoke. 18 Current smokers were defined as those who had smoked more than 100 cigarettes in their lifetimes and still smoked at the time of the survey. Current and former smokers were grouped together in the smoker’s group, and their data were compared with those of individuals in the never-smoker’s group to determine the effect of smoking on development of ESRD.
Genomic DNA extraction and genotyping
Genomic DNA was extracted from peripheral blood samples by standard procedures with proteinase K (Invitrogen, Carlsbad, CA, USA) digestion and phenol/chloroform extraction. 19 The ACE [I/D (rs1799752), G2350A (rs4343) and A-240T (rs4291)] and ACE2 [G8790A (rs2285666), A1075G (rs1978124) and G16854C (rs4646142)] polymorphisms were screened using polymerase chain reaction–restriction fragment length polymorphism (PCR-RFLP). Primers were designed using published sequences 20 or primer Z software (http://genepipe.ngc.sinica.edu.tw/primerz/beginDesign.do). The PCR program included the following steps: initial denaturation at 95°C for 5 min, followed by 35 cycles of denaturation at 95°C for 30 s, annealing at 58°C for 30 s and extension at 72°C for 30 s, and a final extension at 72°C for 10 min. The PCR products were digested with the respective restriction endonucleases (New England Biolabs, Ipswich, MA, USA), and the resulting fragments were separated by electrophoresis on a 3.0% agarose gel containing 0.5 g/mL ethidium bromide at 100 V and visualised under UV light. Genotyping was performed by blinding the case or control status. Two independent investigators interpreted the pictures from each gel, and all ambiguous samples were analysed twice. To validate genotyping results, at least 10% samples were randomly selected for repeated genotyping.
Statistical analysis
For each SNP, deviation from the Hardy–Weinberg equilibrium (HWE) was assessed using the standard χ 2 test. Genotypic and allelic frequencies were compared between ESRD patients and healthy controls using the χ 2 test or Fisher’s exact test, when appropriate. The demographics were evaluated using Student’s t-test for continuous variables and expressed as mean ± standard deviation (SD). Logistic regression analysis was used to estimate the crude and adjusted (age, gender, body mass index and smoking) odd ratios (ORs) and 95% confidence intervals (CIs) as measures of association with the risk of ESRD. Linkage disequilibrium (LD) and haplotype analyses were performed using Haploview software (http://www.broad.mit.edu/mpg/haploview/). 21 The interaction was tested using a term of the product of two variables in a multiple logistic regression model. All statistical analyses were performed using SPSS 18.0 for Windows (SPSS Inc., Chicago, IL, USA). A p value <0.05 was considered statistically significant.
Results
Basic characteristics of the study population
Table 1 shows the characteristics and biochemical and clinical data of the studied individuals. No significant differences were observed in gender, age and diastolic blood pressure between ESRD patients and healthy controls. However, the two groups showed a significant difference with respect to other variables (p<0.05).
Characteristics of subjects with ESRD and control subjects.

Association analyses of ACE and ACE2 gene polymorphisms with susceptibility to ESRD
The genotype distributions of ACE (I/D, G2350A and A-240T) and ACE2 (G8790A, A1075G and G16854C) genes were compatible with the Hardy–Weinberg expectation for both cases and controls (p>0.05). This indicates that the study subjects were representative of the study field. The genotypic and allelic distributions of the three SNPs in the ACE gene and their association with ESRD risk are shown in Table 2. The genotypic and allelic distributions of ACE I/D polymorphism were significantly different between the ESRD cases and healthy controls (p<0.05). In ACE I/D polymorphism, when the II genotype was used as the reference, ID and DD genotypes were associated with a higher risk of ESRD development (adjusted OR 1.38, 95% CI 1.07–1.79, p=0.01 and adjusted OR 1.59, 95% CI 1.07–2.37, p=0.02, respectively). The association between ACE G2350A polymorphism and ESRD risk showed borderline significance, with heterozygous and mutation carriers being at a higher risk of developing ESRD (adjusted OR 1.46, 95% CI 1.30–2.07, p=0.05 and adjusted OR 1.31, 95% CI 1.01–1.71, p=0.04, respectively). ACE A-240T polymorphism showed no significant genotypic and allelic associations in either ESRD patients or healthy controls (p>0.05).
Genotype distributions and allele frequencies for the ACE gene in ESRD patients and healthy controls.
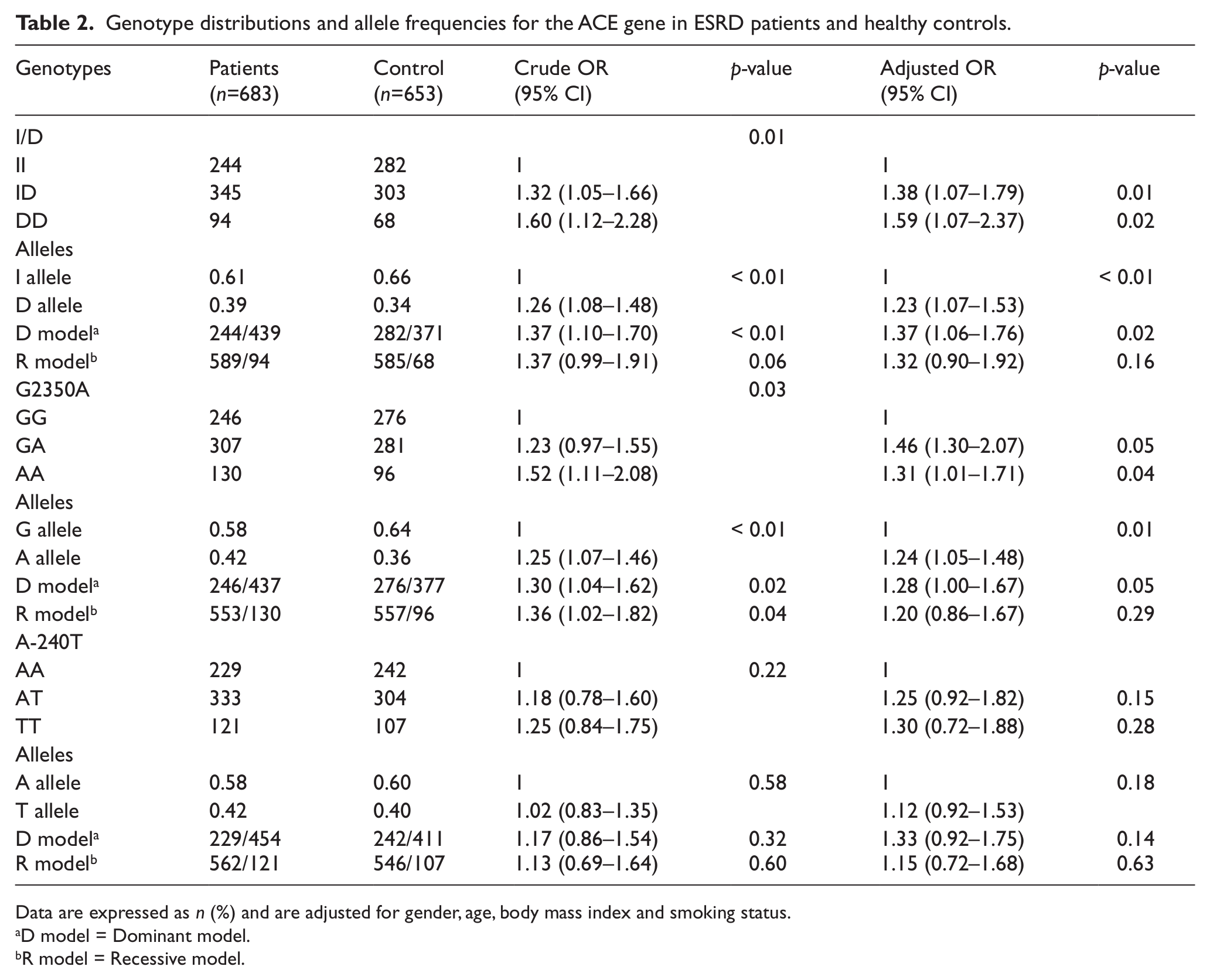
Data are expressed as n (%) and are adjusted for gender, age, body mass index and smoking status.
D model = Dominant model.
R model = Recessive model.
As the ACE2 gene is on the X chromosome, data for each gender were analyzed separately. The genotypic and allelic frequencies of ACE2 polymorphisms between ESRD patients and healthy controls are shown in Table 3. For males, the allelic frequencies of ACE2 polymorphisms (G8790A, A1075G and G16854C) showed no significant difference between ESRD patients and healthy controls (p>0.05). No associations were observed between ESRD and ACE2 polymorphisms in females.
Association analyses for ACE2 gene polymorphisms with ESRD.

Data adjusted for age, body mass index and smoking status.
MAF: minor allele frequency.
Haplotype analysis of ACE
The haplotype analysis of ACE polymorphisms in ESRD patients and healthy controls is shown in Table 4. The frequency of haplotype DA was 0.24 in ESRD patients compared with 0.14 in healthy controls (OR 2.01, 95% CI 1.60–2.53). In addition, haplotypes DAA and DAT were more common in ESRD patients (0.13 and 0.11, respectively) than in healthy controls (0.08 and 0.06, respectively) giving an OR of 1.79 and 1.98, respectively (95% CI 1.33–2.40 and 1.43–2.72, respectively). Other haplotypes showed no significant change.
Haplotype frequencies in ACE between ESRD and control subjects.

p < 0.05.
ACE gene–smoking interaction analysis
Potential interactions between ACE (I/D and G2350A) genotype and smoking status are shown in Figure 1. When never-smokers with wild-type genotype (ACE II) were used as a reference, smokers with a variant genotype (ACE DD) showed the greatest risk of ESRD development (adjusted OR 12.35, 95% CI 3.44–44.37). Moreover, multivariate logistic regression analysis showed evidence of interactions between ACE I/D polymorphism and smoking status (p for interaction 0.04). No statistically significant interaction was observed between ACE G2350A polymorphism and smoking status (p for interaction 0.17).

Effects of cigarette smoking and ACE gene polymorphisms on risk of ESRD. OR adjusted for age, gender and BMI.
Discussion
In this study, we investigated the association of ACE and ACE2 gene polymorphisms with ESRD and showed that ACE I/D and G2350A gene polymorphisms were significantly associated with ESRD. Our study also presents statistical evidence of significant association between ACE I/D SNP and smoking status interaction and the risk of developing ESRD. These findings support our hypothesis that ACE gene polymorphisms and potential gene–smoking interactions may contribute to ESRD susceptibility.
Previous studies showed that the ACE I/D polymorphism was expressed phenotypically by significant variations in ACE serum levels, with DD individuals having the highest levels. 22 The increased level of ACE is associated with the pathogenesis of renal disease. ACE I/D polymorphism has been studied extensively in relation to ESRD23,24; however, the findings of these studies remain controversial. Previous studies have indicated that a significant association exists between the DD gene and ESRD risk in Caucasians25,26 and East Asians27,28; however, this association is not observed in the Turkish population. 29 Our results are consistent with those of studies in Caucasians and East Asians, which indicate that the ACE DD genotype is associated with a significantly elevated risk of ESRD. This suggests the presence of a geography- or ethnicity-specific difference in the ACE gene and the association of gene polymorphisms and ESRD risk.
Several studies have reported that serum and plasma levels of ACE are influenced by ACE G2350A 30 and A-240T 31 polymorphisms. In this study, we also found an association between ACE G2350A polymorphism in a Han Chinese population; however, no association was observed with ACE A-240T polymorphism. Carriers of the A allele of ACE polymorphism were more frequent in the patient group than in the control group. Our results are consistent with those of previous studies, which indicate that the ACE 2350A allele is associated with a significantly elevated risk of chronic kidney disease (CKD) and hypertension in the Han Chinese population.
Many genes of the RAS pathway have been assessed for their associations with ESRD. However, research on the ACE2 gene is limited. The ACE2 gene is located on the Xp22 chromosome, contains 18 exons and is mainly expressed in the kidney. The ACE2 gene has been implicated in kidney disease, hypertension and cardiac function. 32 Previous studies have also shown that expression of the ACE2 gene is altered in the diabetic rat kidney 33 and in humans with renal diseases. 34 This suggests that an association exists between ACE2 gene variants and ESRD. However, no associations were observed between ESRD and ACE2 gene polymorphisms in this study. ACE2 polymorphisms may have a small effect on susceptibility to ESRD, but we were unable to detect such an effect.
The biological consequences of gene–smoking interactions have been investigated extensively over recent years. Smoking is one of the established risk factors of ESRD, but its exact pathogenesis has not yet been clarified. Several studies have already reported that smoking status may influence the effects of the ACE genotype on peripheral arterial disease (PAD) 35 and endothelial function. 36 In the present study, the interaction between ACE I/D polymorphism and cigarette smoking had an additive effect on the risk of developing ESRD. Smokers with the variant genotype (ACE DD) had the highest risk of developing ESRD. Our results are consistent with those of previous studies. Butler et al. reported a significant blunting of the endothelial function in DD carriers, especially in smokers. 37 Another study showed that the D allele was associated with increased systolic blood pressure in smokers. 38 Sayed-Tabatabaei et al. showed a combined effect of smoking and the ACE gene on intima media thickness (IMT), with a significant increase in the mean carotid IMT in current smokers carrying the D allele. 39
Several studies indicate that high levels of LD exist across the ACE gene.40,41 Haplotype analysis performed in a previous study showed an association between the ACE haplotype and major depressive disorder. 42 Wetmore et al. showed that the ACE haplotype is associated with survival in a cohort with prevalent ESRD. 43 Our data showed that ESRD patients carrying the DAA or DAT haplotype had a higher risk of developing ESRD than those not carrying these haplotypes. To test the potential of the genes highlighted by haplotype research more completely, a follow-up study that explores the role of haplotypes on genetic susceptibility to ESRD might be desirable.
In conclusion, this case-control study in a Han Chinese population showed that ACE I/D and G2350A polymorphisms may modulate the risk of development of ESRD. No association was observed between ACE2 polymorphism and ESRD susceptibility. ACE polymorphism and smoking interactions confer an increased risk of developing ESRD, indicating that ACE gene polymorphisms might identify smokers who are more susceptible to ESRD. However, these results must be confirmed in future studies.
Footnotes
Conflict of interest
None declared.
Funding
This study was supported by grants from the Cardinal Tien Hospital and Bureau of Health Promotion, Taiwan, ROC (CTH- NC10007, CTH101-1-2C03 and DOH100-HP-1102).
