Abstract
Introduction:
Rats exposed to angiotensin II (AII) receptor antagonists during lactation present progressive disturbances in renal development that lead to progressive alterations in renal function and structure. This study evaluates the role of oxidative stress in the renal changes induced by exposure to losartan, a type 1 AII receptor antagonist, in rats during lactation.
Materials and methods:
Male Wistar pups were divided into: Control, pups of dams that received 2% sucrose solution; Control-tempol, pups of dams that received tempol (0.34 g/l), a superoxide dismutase mimetic compound; Losartan, pups of dams that received losartan (100 mg/kg/day), and Losartan-tempol, pups of dams that received losartan and tempol. Losartan and/or tempol were administered during lactation. Blood and urine samples were collected at 21 or 60 days, and the kidneys were removed.
Results:
Losartan-treated pups exhibited disturbances in renal function and structure that persisted into adulthood. Tempol treatment reduced oxidative stress and attenuated the changes induced by losartan in the glomerular filtration rate, desmin expression at the glomerular edge, vimentin in tubular cells, as well as apoptosis and inflammatory infiltration in the renal cortex.
Conclusion:
Oxidative stress contributes at least in part to the renal injury observed in pups exposed to losartan during lactation.
Introduction
Rat nephrogenesis begins on embryonic day 12 and is completed between 10 and 15 days after birth.1,2 Clinical and experimental evidence indicates that the renin–angiotensin system (RAS) participates in renal development.3–6 All RAS components are expressed early at a high intensity in embryogenesis.7,8 Angiotensin II (AII) stimulates cultured mesangial cells to produce various extracellular matrix (ECM) components 9 and promotes cell proliferation and differentiation. 10 ECM formation is a key event in renal cell differentiation.11,12
Mutations in genes that encode RAS components are associated with autosomal recessive renal tubular dysgenesis. 5 It was observed that human fetal kidneys from the offspring of mothers that received type 1 AII receptor (AT1) antagonists during gestation have poorly developed tubules, increased renal mesenchymal tissue, decreased numbers of proximal tubules, abnormal arterial and arteriolar walls, and poorly developed vasa recta and hyperplasic juxtaglomerular apparati. 4 The inhibition of the RAS by administering AII antagonists to rats during postnatal development causes abnormalities in renal structure, including reduced glomerular volume, tubular dilation, papillary atrophy and increased interstitial relative area of the renal cortex.1,3,13,14 These changes lead to a progressive loss of renal function. 13 It was proposed that the structural abnormalities in this experimental model might result in part from the developmental disruption of vascular and tubular kidney compartments. 1 We previously showed that losartan administration during lactation caused changes in renal function and structure in pups, and these changes were associated with increased type 2 AII receptor (AT2) expression and reduced AT1 expression. 3 The greater AT2 expression was associated with increased numbers of apoptotic cells in the renal cortex of these animals. The blockade of AT1 can interfere with the balance between proliferation and apoptosis in various ways. 15 The fibrogenic effects of AII are mediated at least in part by AT2. Saward and Zahradka showed that AII-induced hypertrophy and fibrosis are mediated by AT2 in vascular muscle cells. 16
Several studies have shown that oxidative stress is involved in the pathogenesis of a variety of renal and systemic diseases.17–22 Reactive oxygen species (ROS) also have an important role in neonatal morbidity because antioxidant defenses may be altered during that period.18,23 Tempol (4-hydoxy-2,2,6,6-tetramethylpiperidine-1-oxyl), a member of a family of nitroxide compounds with a superoxide dismutase (SOD) mimetic action, has been extensively studied in animal models of increased ROS. 24
The aim of our study was to evaluate the role of oxidative stress in the pathogenesis of renal disorders induced by exposure to losartan during lactation.
Material and methods
Animals and experimental protocols
Male Wistar rats from 57 pregnant females provided by the Animal House of the Campus of Ribeirão Preto, University of São Paulo (Ribeirão Preto, SP, Brazil) were used in this study. The animals were housed in polycarbonate cages at standard room temperature (22°C) and a 12-hour (h) light/dark cycle with free access to standard rat chow. For mating, each male was housed with three females, and the first gestation day (GD1) was determined based on the presence of copulatory plugs. Within 24 hours of birth, the litters were reduced to six male pups to ensure equal feeding and divided into the following groups: Control (C) (n = 65), pups from dams that received a 2% sucrose solution; Control-tempol (CT) (n = 27), pups from dams that received 0.34 g/l tempol (Sigma-Aldrich-Chemie, Germany) diluted in a 2% sucrose solution; Losartan (LO) (n = 128), pups from dams that received losartan (All Chemistry, Brazil) diluted in a 2% sucrose solution (40 mg/dl, corresponding to a daily ingestion of ~100 mg/kg); and Losartan-tempol (LOT) (n = 42), pups from dams that received losartan plus tempol diluted in a 2% sucrose solution. Losartan, an AT1 antagonist, and tempol, a SOD mimetic compound, were administered daily in drinking water during lactation (21 days) to block renal AII effects and inhibit oxidative stress, respectively. We selected this dose of losartan based on previous studies that showed developmental toxicity was evident in the newborns from mothers that received doses ≥ 100 mg/kg/day.25,26 At this dose, losartan was detected in breast milk in a concentration close to 50% of the maternal serum in 75% of dams. The combination of losartan with tempol was based on a study by Dobrian et al. 27 Tempol is a small molecule that is able to cross biological membranes, and the treatment of rats with this compound was able to decrease both cytosolic and mitochondrial ROS production. 28
At 21 and 60 days old, the pups were anesthetized, blood and urine samples were collected to analyze renal function, and the kidneys were perfused via the left cardiac ventricle (21 days) or aorta (60 days) with phosphate-buffered solution (PBS; 0.15 M NaCl and 0.01 M sodium phosphate buffer, pH 7.4). One kidney was removed for Western blotting, lipid peroxidation and glutathione quantification. The other kidney was perfused with 4% phosphate-buffered paraformaldehyde for 10 min and fixed in Bouin’s solution for four h or formalin for 24 h. Then, this kidney was rinsed in 70% ethanol and embedded in paraffin. Urine and plasma samples were frozen for subsequent analysis of osmolality, sodium, potassium, creatinine, inulin and albumin excretion. Liquid intake and food consumption were measured in dams during lactation (days seven to 15) and in pups during days 40 to 46. Plasma levels of thiobarbituric acid-reactive substances (TBARS) were measured in dams at the end of lactation. Body weight was also evaluated during the study period.
All the experimental procedures were conducted in accordance with the principles and procedures outlined in the United States National Institutes of Health (NIH) Guide for the Care and Use of Laboratory Animals, and the Animal Experimentation Committee of the University of São Paulo at Ribeirão Preto School of Medicine approved the study protocol.
Systolic blood pressure (SBP)
SBP was measured in pups using the tail-cuff method (IITC Model 31; Life Science Instruments, Woodland Hills, CA, USA) at 30 and 60 days old under conscious conditions.
Renal function studies
At 21 days old, the pups were anesthetized with an intraperitoneal (i.p.) injection of tribromoethanol, urine was collected by puncture of the urinary bladder, and a blood sample was taken from the left cardiac ventricle. Renal function was evaluated by serum creatinine levels using a colorimetric method. 29 When the pups reached 60 days old, they were placed in metabolic cages, and urine samples were collected to measure 24-h urinary volume; osmolality was determined by freezing point depression (Fiske OS Osmometer, Norwood, MS, USA). The glomerular filtration rate (GFR) was evaluated by inulin clearance. Briefly, the animals were anesthetized with an i.p. injection of 40 mg/kg thionembutal. The body temperature was maintained at 37°C during the entire experimental period. After tracheostomy, the femoral artery and vein were cannulated to collect blood samples and inject fluids, respectively. The ureters were cannulated to collect urine. Inulin was administered in a priming dose (8 mg/100 g), followed by a maintenance dose (30 mg/100 g/h). After a stabilization period of approximately 60 min, urine was collected for one hour, and blood samples were collected at 30 min and one hour. These samples were used to assess levels of sodium, potassium, albumin and inulin. Inulin was measured in plasma and urine samples using the method described by Führ et al. 30 Albumin was quantified in urine samples by an electroimmunoassay using a specific antibody against rat albumin. 31 Plasma and urine sodium and potassium were measured by flame photometry (Micronal, model 262, São Paulo, Brazil).
Histological analysis
Renal tissue sections (3-μm thick) were stained with Masson’s trichrome and examined by light microscopy. Tubulointerstitial damage was defined as tubular lumen dilation or atrophy and an increase in the relative interstitial area with fibrosis and inflammatory cell infiltration. Renal cortex damage was graded on the following scale: 0 = normal; 0.5 = small focal areas; 1 = involvement of < 10% of renal cortex; 2 = 10–25%; 3 = 25–75%; 4 = extensive damage involving > 75% of cortex. 32
Immunohistochemical analysis
Renal tissue sections were deparaffinized and incubated with the following antibodies: anti-desmin (1:100; Dako Corporation, Glostrup, Denmark), anti-ED1 (1:1,000; Serotec, Oxford, UK. This antibody reacts only with a cytoplasmic antigen present in macrophages and monocytes of rats 33 ), anti-α-smooth-muscle actin (anti-α-SMA, 1:1,000; Dako Corporation), anti-vimentin (1:500; Dako Corporation), anti-phospho-p38 (p-p38; 1:1,000; Sigma-Aldrich, St. Louis, MO, USA), which reacts specifically with the active phosphorylated form of p38 kinase, and anti-nitrotyrosin (1:100; Upstate, Lake Placid, NY, USA) for 60 min at room temperature, or anti-phospho-JNK (p-JNK; 1:13; Santa Cruz Biotechnology, Santa Cruz, CA, USA) and anti-phospho-p44/p42 MAP kinase (p-ERK; 1:30; Cell Signaling Technology, Beverly, MA, USA) overnight at 4oC.
The reaction products were detected using avidin-biotin-peroxidase complex (Vector Laboratories, Burlingame, CA, USA), and the sections were counterstained with methyl green or hematoxylin, dehydrated and mounted. Non-specific protein binding was blocked by incubation with 20% goat serum in PBS for 20 min. Negative controls were created by replacing the polyclonal or monoclonal primary antibody with equivalent concentrations of normal rabbit IgG or mouse IgG, respectively. For the evaluation of desmin, vimentin, α-SMA, nitrotyrosin, p-JNK, and p-ERK staining, each glomerulus or cortical field (measuring 0.245 mm2 each) was semiquantitatively graded, and the mean score per kidney was calculated. 34 Each score depended on the percentage of positive staining: 0 indicated absent or < 5% staining; I, 5–25%; II, 25–50%; III, 50–75%; and IV, >75% staining. The numbers of ED1-positive cells (macrophages/monocytes) in each glomerulus or cortical interstitial grid field and of p-p38-positive cells in each cortical interstitial grid field were determined in all renal cortex, and the mean counts were calculated for each kidney.
In situ detection of apoptotic cells
Renal tissue was stained with terminal deoxynucleotidyl transferase (TdT)-mediated deoxyuridine 5-triphosphate-biotin nick end labeling using a commercial kit to evaluate apoptosis (Oncogene Research Products, Boston, MA, USA). 35 Tissues treated with DNase were used as positive controls, and sections stained without terminal nucleotidyl transferase were used as negative controls. The mean numbers of apoptotic cells in all grid fields from the renal cortical tubulointerstitium (measuring 0.245 mm2 each) were evaluated.
Quantification of lipid peroxidation
Lipid peroxidation was determined in plasma samples from dams and in renal tissue from pups by the thiobarbituric acid (TBA) assay. 36 Briefly, 500 µl of plasma or homogenate tissue combined with 1 ml of TBA + trichloroacetic acid (TCA) reagent (3.7 g/l TBA + 15% TCA in 0.25 mol/l HCl) were mixed in a 10-ml test tube and heated for 15 min in boiling water. After cooling in an ice bath, the tubes were centrifuged at 3000 rpm for 10 min. The optical density of the supernatant was read at 535 nm, and the quantity of TBARS was calculated using a molar extinction coefficient of 1.56 × 105 M−1 cm−1.
Determination of reduced glutathione
Glutathione levels were measured by the method of Sedlak and Lindsay. 37 A 0.6 ml aliquot of whole kidney homogenate was added to 1.4 ml of water and 0.1 ml of 50% TCA and centrifuged at room temperature for 10 min at 3,000 rpm. A 0.5 ml aliquot of supernatant was added to 2 ml of Tris buffer (0.2 M, pH 8.9) and 0.1 ml of 0.01 M 5,50-dithiobis-(2-nitrobenzoic acid) (DTNB) in methanol. After mixing the samples and allowing them to stand for 15 min, light absorbance was spectrophotometrically determined at 412 nm and compared with a standard curve consisting of α-cysteine at concentrations of 0.02, 0.04 and 0.1 µmol ml−1.
Levels of superoxide dismutase
Superoxide dismutase levels were evaluated in whole kidney by Western blotting. Briefly, renal tissue was homogenized in lysis buffer (50 mM Tris-HCl, pH 7.4, 150 mM NaCl, 1% Triton X-100, 0.1% sodium dodecyl sulfate (SDS), 1 µg/ml aprotinin, 1 µg/ml leupeptin, 1 mM phenylmethylsulfonyl fluoride, 1 mM sodium orthovanadate, pH 10, 1 mM sodium pyrophosphate, 25 mM sodium fluoride, and 0.001 M ethylenediaminetetra-acetic acid (EDTA), pH 8) and centrifuged at 4°C for 15 min at 10,000 rpm. Renal tissue protein was loaded (30 µg per well) and separated by electrophoresis on a 12% SDS polyacrylamide gel. 38 Protein estimations were performed using the Hartree method. 39 Gels were transferred to nitrocellulose membranes, incubated for one h in 50 ml of blocking buffer (PBS, 5% skim milk), washed in buffer (PBS, 0.1% Tween 20, pH 7.6) and incubated with anti-extracellular SOD (EC-SOD; SOD3; 1:500; Stressgen, Ann Arbor, MI, USA) or anti-α1-tubulin (1:20,000; Sigma-Aldrich, St. Louis, MO, USA) antibodies overnight at 4°C). The EC-SOD is the most widely expressed isoform in the kidney and in the vasculature. 40 Blots were washed and incubated with horseradish peroxidase-conjugated goat anti-rabbit IgG (1:20,000; Dako Corporation) for one h at room temperature. The membranes were then washed, and membrane-bound antibodies were detected using Supersignal West Pico Chemiluminescent Substrate (Pierce Chemical, Rockford, IL, USA) and captured on X-ray film. A densitometry analysis was performed using the ImageJ computer program (NIH, Bethesda, MD, USA). We also incubated all membranes with α1-tubulin antibody in order to correct protein loading and/or transfer.
Statistical analysis
The data that were not normally distributed were statistically analyzed using the nonparametric Kruskal-Wallis test followed by the Dunn post-test. Those data were expressed as medians and interquartile ranges (25–75%). For the normally distributed data, we used analysis of variance with the Newman-Keuls multiple comparisons test. Those data were expressed as the mean±standard error of the mean (SEM). In all cases, the level of significance was set at p < 0.05.
Results
Body weight, fluid intake, food consumption and plasma lipid peroxidation of the dams
There was no difference in body weight, fluid intake, food consumption and plasma lipid peroxidation among all dam groups during lactation. Body weight was also similar in all the pup groups during the study period (data not shown). Liquid intake was higher in both pup groups exposed to losartan during lactation compared to control groups. However, there was no significant difference observed in food consumption (Table 1).
Liquid intake and food consumption from days 40 to 46 in the pups of the control and losartan-treated groups.

Data are expressed as the mean ± SEM. ap < 0.001 compared with Control; bp < 0.05 compared with Control-tempol.
Renal function studies
Tables 2 and 3 show the functional parameters at 21 and 60 days of age, respectively. At 21 days old, there was no difference in plasma creatinine levels among the groups. However, at 60 days of age, the GFR, evaluated by inulin clearance, was reduced in losartan-treated pups. These animals showed increased fractional excretions of sodium and potassium and albuminuria compared to age-matched controls. Tempol treatment attenuated the increased fractional excretion of potassium observed at 21 and 60 days old and prevented the decrease in the GFR in 60-day-old rats treated with losartan. At 60 days of age, the pups exposed to losartan had normal blood pressure, and tempol treatment alone did not affect this parameter. Urinary volume was increased and urine osmolality was decreased in both groups exposed to losartan compared to the controls.
Albumin/creatinine ratio, serum creatinine and fractional excretions of sodium (FENa+) and potassium (FEK+) in 21-day-old pups from the control and losartan-treated groups.

Albumin is expressed as a ratio of the albumin concentration to creatinine in each urine sample; these data and FENa+ and FEK+ are expressed as the median and 25% and 75% percentiles. Pcreat = plasma creatinine; these data are expressed as the mean ± SEM. ap < 0.05 and bp < 0.01 compared with Control; cp < 0.05 compared with Control-tempol.
Systolic blood pressure (SBP), albuminuria, 24-h urine volume (V), urinary osmolality (U), glomerular filtration rate (GFR) and fractional excretions of sodium (FENa+) and potassium (FEK+) in 60-day-old pups from the control and losartan-treated groups.
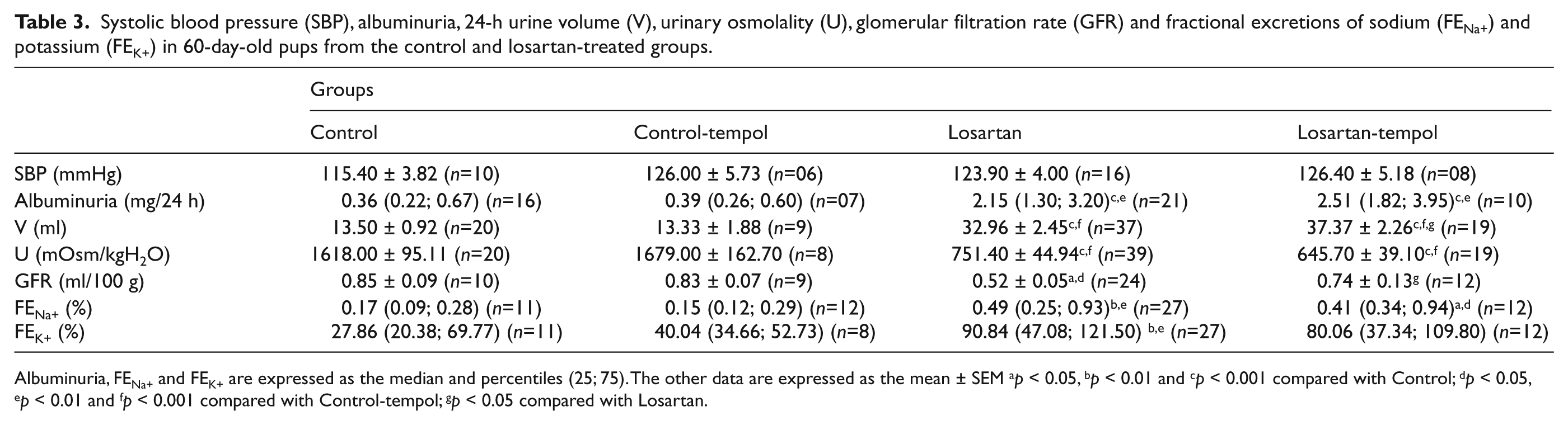
Albuminuria, FENa+ and FEK+ are expressed as the median and percentiles (25; 75). The other data are expressed as the mean ± SEM ap < 0.05, bp < 0.01 and cp < 0.001 compared with Control; dp < 0.05, ep < 0.01 and fp < 0.001 compared with Control-tempol; gp < 0.05 compared with Losartan.
Histological analysis
Histological analysis of the renal cortex of the pups from dams exposed to losartan during lactation showed tubular lumen dilation, tubular cell atrophy with loss of the brush border, and an increase in the relative interstitial area with inflammatory cell infiltration and fibrosis (Figure 1C, D) compared to the control animals (Figure 1A, B) at 21 and 60 days of age. At 60 days of age, the losartan-treated pups had an increased number of histological lesions compared to the 21-day-old pups. The histological scores of the groups treated only with losartan or losartan plus tempol were similar in animals of the same age (Figure 1E, F).

Representative Masson’s trichrome-stained histological sections from Control (A, B) and Losartan (C, D) groups, at 21 (A, C) or 60 days of age (B, D) and score for tubulointerstitial lesions (E, F). The data are expressed as the median and 25th and 75th percentiles. *** p < 0.001 compared with Control; ### p < 0.001 compared with Control-tempol. Bars indicate 50 µm.
Immunohistochemical studies
Losartan-treated pups (Figure 2E, F, G, H) exhibited higher desmin expression at the glomerular edge compared to controls (Figure 2A, B, C, D) at 21 and 60 days of age, which is suggestive of podocyte lesions. Tempol supplementation attenuated this losartan-induced alteration in 60-day-old pups (Figure 2J).

Representative immunostaining for desmin in the renal cortex of Control (A, D), Losartan- (B, E), and Losartan-tempol (C, F) groups, at 21 (A, B, C ) or 60 days of age (D, E, F) and score for desmin expression in the renal cortex (G, H). The horizontal line represents the median. * p < 0.05 and ** p < 0.01 compared with Control; # p < 0.05 and ### p < 0.001 compared with Control-tempol; • p < 0.05 compared with Losartan. Bars indicate 50 µm.
Vimentin expression in tubular cells in the renal cortex of 21- (Figure 3C, E) and 60-day-old rats (Figure 3D, F) was observed in losartan-treated pups, suggesting that these animals had recent tubular cell lesions or a lack of tubular cell differentiation. Tempol treatment attenuated the increase of vimentin immunostaining in 60-day-old losartan-treated animals.

Representative immunostaining for vimentin in the renal cortex of Control (A, B) and Losartan (C, D) groups, at 21 (A, C) or 60 days of age (B, D). Note the expression of vimentin in the tubular cells (C, D). Scores are for vimentin expression in the renal cortex (E, F). The horizontal line represents the median. ** p < 0.01 and *** p < 0.001 compared with Control; ## p < 0.01 and ### p < 0.001 compared with Control-tempol. Bars indicate 50 µm.
Immunohistochemical analysis also showed that the pups from dams exposed to losartan during lactation presented increased α-SMA expression, predominantly in the interstitium and vascular walls of the outer medulla, showing disturbances in organization of the vasa recta bundles (Figure 4B). The expression of α-SMA in the control rats was restricted to medullary vasa recta bundles and the edges of artery and arteriole walls (Figure 4A). Tempol supplementation did not affect the increase of α-SMA expression in the renal cortex of the 21-day-old rats exposed to losartan (Figure 4C). However, α-SMA expression was less intense in the 60-day-old rats treated with losartan plus tempol compared with the rats treated with losartan alone (data not shown).
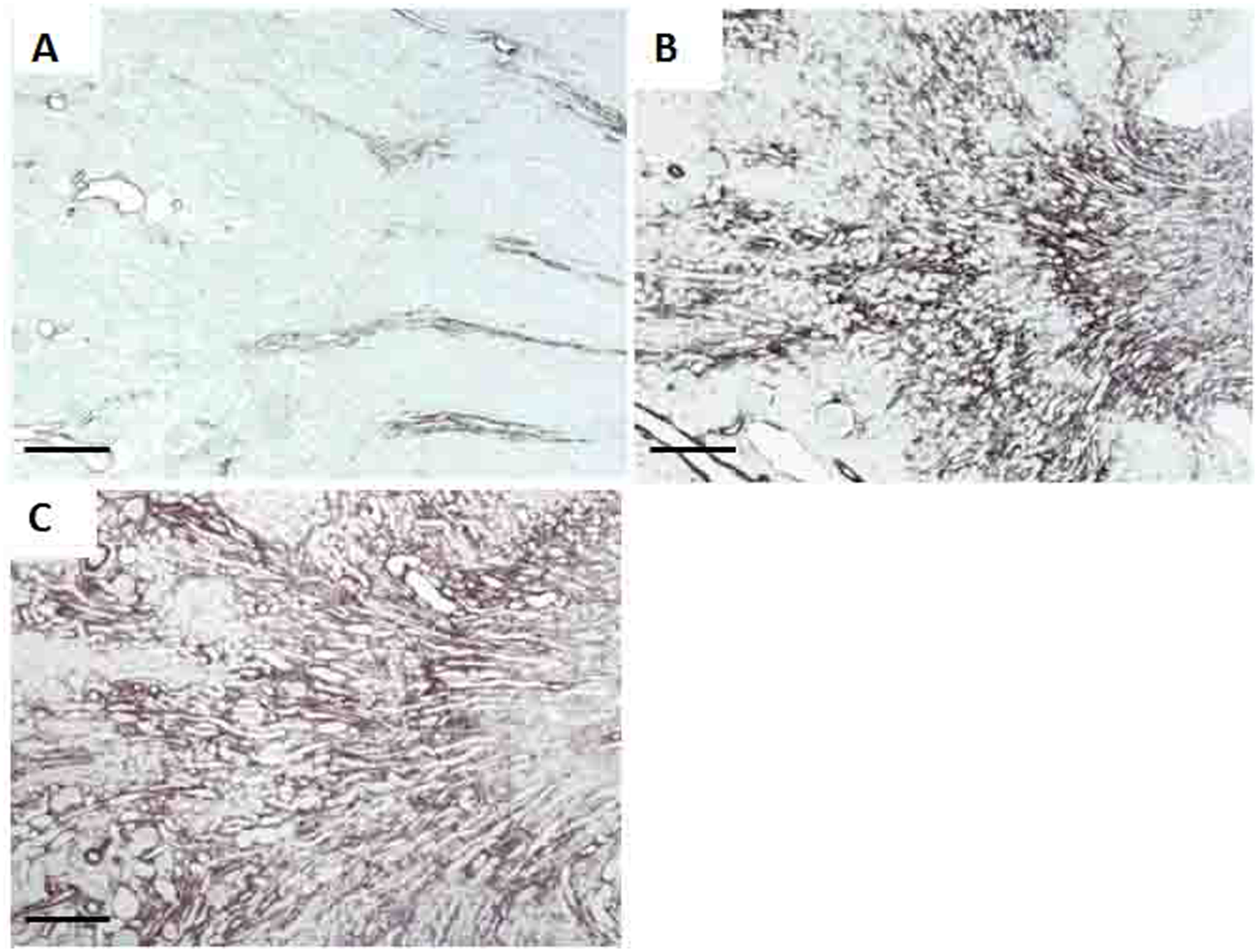
Representative immunostaining for α-smooth-muscle actin (α-SMA) in the renal outer medulla of Control (A), Losartan (B) and Losartan-tempol (C) groups, at 21 days of age. Bars indicate 200 µm.
Immunohistochemical studies performed at 21 and 60 days old revealed a marked increase in the number of ED1-positive cells (macrophages/monocytes) in the tubulointerstitial compartment of the losartan-treated rats (Figure 5C, D), indicating tubulointerstitial inflammation. At 60 days of age, the number of ED1-positive cells in the glomeruli was also higher in the losartan-treated animals compared to controls (Figure 5A, B). Tempol treatment prevented macrophage infiltration in the tubulointerstitial compartment and glomeruli caused by exposure to losartan during lactation at both ages.

Representative immunostaining for ED1-positive cells (macrophages/monocytes) in the Control (A, B), Losartan (C, D) and Losartan-tempol (E, F) groups, at 21 (A, C, E) or 60 days of age (B, D, F). Number of ED1-positive cells per tubulointerstitial area of 0.245 mm2 (G, H) or in the glomerulus (I, J). The data are expressed as the mean ± SEM. ** p < 0.01 and *** p < 0.001 compared with Control; ## p < 0.01 and ### p < 0.001 compared with Control-tempol; ••• p < 0.001 compared with Losartan. Bars indicate 50 µm.
Apoptosis evaluation
The number of apoptotic cells was significantly higher in the renal cortex tubulointerstitium of the losartan-treated pups (Figure 6B) compared to the controls (Figure 6A) at 21 days of age (Figure 6C), and tempol treatment attenuated this effect. There was no difference among the groups in the number of apoptotic cells at 60 days of age (Figure 6D).

Apoptotic cells (arrows) in the renal cortex from a control rat (A) and a losartan-injected rat (B). Number of apoptotic cells at 21 (C) and 60 days of age (D) per grid field (measuring 0.245 mm2) of the renal cortex of Controls and Losartan-treated rats. The horizontal line represents the median.
Oxidative stress markers and mitogen-activated protein kinase (MAPK) expressions
Oxidative stress markers analyzed at the end of lactation in 21-day-old rats showed that the treatment of dams with losartan during lactation caused a significant increase in oxidative stress in the renal tissue from losartan-treated pups, characterized by increased TBARS (Figure 7A), glutathione levels (Figure 7B), SOD (Figure 7C-D) and nitrotyrosine expressions (Figure 8A–C and J). Tempol treatment during lactation reduced all parameters of oxidative stress observed in the 21-day-old losartan-treated animals. Increased expression of p-JNK and p-p38 were observed in the renal cortex of the animals from the Losartan group, which were less intense in the rats treated with losartan plus tempol (Figure 8D–I and K–L). We did not observe a difference in the expression of p-ERK in the renal cortex among the groups (data not shown).

Oxidative stress markers evaluated in pups at 21 days of age: thiobarbituric acid-reactive substances (TBARS, A) and glutathione (GSH, B) levels in the renal tissue. The horizontal line represents the median. Extracellular superoxide dismutase (SOD) expression in the renal tissue from the Control (first two), Control-tempol (second two), Losartan (third two) and Losartan-tempol (last two) groups evaluated by Western blotting analysis (C). Densitometry analysis of extracellular SOD (D) from the different groups. The data are expressed as the mean ± SEM. * p < 0.05; ** p < 0.01 and *** p < 0.001 compared with Control; # p < 0.05 and ## p < 0.01 compared with Control-tempol and • p < 0.05 compared with Losartan.

Renal cortical expressions of nitrotyrosine (A, B, C), p-p38 (D, E, F) and p-JNK (G, H, I) mitogen-activated protein kinases (MAPKs) in the renal cortex of Control (A, D, G), Losartan (B, E, H), and Losartan-tempol (C, F, I) groups, at 21 days of age. Score for nitrotyrosin (J), number of p-p38-positive cells (K), and score for p-JNK (L) in the renal cortex. Data are expressed as median and interquartile range (J, L) or mean ± SEM (K). * p < 0.05, ** p < 0.01, and *** p < 0.001 compared with Control; # p < 0.05, ## p < 0.01, and ### p < 0.001 compared with Control-tempol; ••• p < 0.001 compared with Losartan.
Discussion
Pups exposed to the AT1 blocker losartan during lactation had disturbed renal development that led to progressive changes in renal function and structure. Machado et al. 13 followed up losartan-treated animals and observed a change in serum creatinine only after 10 months of age. In our study, at 60 days of age, pups exposed to losartan already had a significant reduction in GFR evaluated by inulin clearance, which was prevented by tempol supplementation.
The fractional excretions of sodium and potassium were increased in losartan-treated pups of both ages (21 and 60 days). This can be explained by both the direct effect of losartan on sodium transport in the renal tubules as well as by losartan-induced lesions in the renal tubulointerstitium in these animals. However, the changes persisted in the 60-day-old animals, showing that the tubulointerstitial injury observed in the kidneys of these animals likely contributes to the increased renal fractional excretion of sodium in losartan-treated rats. This change was associated with increased urinary volume and decreased urinary osmolality at 60 days of age. The decrease in urinary osmolality can also be explained, at least in part, by the washout in the inner medulla provoked by higher sodium excretion and by the decreased expression of aquaporins in the luminal membrane of the cells from the collector tubules in the urine osmolality of losartan-treated rats.
The liquid intake, measured from 40 to 46 days old, was also increased in pups exposed to losartan. It could be a consequence of the higher urinary volume and loss of sodium and potassium in urine from these rats, leading to decrease of extracellular volume. Possibly, animals exposed to losartan during this period also have thirst stimulus other than the central dipsogenic action of AII that are responsible for the overall drinking response, as a compensatory mechanism in order to counterbalance the increased renal excretion of water and sodium by functional kidneys. 41 Studies have shown that the inhibition of the RAS during kidney development has been linked with challenges in physiological hydromineral balance in adulthood. 42 Butler et al. 41 also found that AT1 blockage with losartan during gestation in rats provokes an increase in thirst in their adult offspring.
Urinary albumin excretion was increased at the end of lactation in pups exposed to losartan, and this increase was higher at 60 days compared to 21 days of age, which demonstrates the age-dependent progression of renal injury in this experimental model. Tempol treatment did not prevent this change.
The histological analysis of the renal cortex showed that the 21-day-old pups whose dams received losartan showed changes in the tubulointerstitial compartment, such as flattening of tubular cells, increased interstitial area, tubular cell atrophy and lumen dilation with brush border loss. These histological changes were more intense in the 60-day-old rats. Antioxidant treatment did not attenuate the histological changes induced by losartan. However, these histological changes were evaluated by scores that have a large range, and small differences could not be detected. 32 The immunohistochemistry performed with antibodies that react specifically with the components of the cytoskeleton such as desmin, vimentin and α-SMA is a more sensitive way to detect structural changes than the histological scores.
The pups of losartan-treated dams also showed increased desmin expression at the glomerular edge, suggesting podocyte lesions, once podocytes only express desmin when injured. 34 In the control rats, desmin expression was restricted to mesangial cells. Tempol treatment attenuated desmin expression at the glomerular edge in the 60-day-old rats, indicating that tempol had some protection against the renal podocyte lesions observed in this model. Glomerular diseases can lead to progressive involvement of the tubulointerstitial compartment, and podocyte injury may have an important role in this process. 43 Proteinuria caused by filtration barrier changes may damage the tubulointerstitial compartment, causing loss of renal function. 44 Although proteinuria can induce interstitial lesions, the level of proteinuria and the desmin expression observed in the pups of the losartan-treated dams were mild, and could not be responsible for the marked tubulointerstitial changes observed in the renal cortex of these animals.
The immunohistochemical studies also demonstrated that the animals exposed to losartan exhibited increased vimentin expression in tubular cells from the cortical tubulointerstitium at 21 and 60 days of age. Tubular cells express vimentin only when they are proliferating and during renal development; therefore, this finding suggests recent cell injury or a lack of cell differentiation.34,45 The hypothesis of epithelial-mesenchymal transition should be considered. Vimentin and α-SMA are also a marker of epithelial-mesenchymal transition. However, this is a less probable hypothesis because the study performed in the 21-day-old rats showed that tubule cells did not differentiate. Tempol treatment at 21 days old did not attenuate the increased expression of vimentin induced by losartan, possibly because at this age, the pups were still exposed to losartan via breast milk. However, at 60 days of age, the animals that had tempol supplementation showed decreased expression of vimentin in renal tubular cells compared to age-matched rats exposed to losartan alone.
We also observed an increased expression of α-SMA in the tubulointerstitial compartment from the renal cortex of the losartan-treated pups. Mesangial, tubular and interstitial cells can express α-SMA protein under pathological conditions, when these cells begin to produce more collagen and other ECM components. 46 During normal postnatal renal development in rats, the α-SMA cytoskeletal protein is expressed more intensely in the glomeruli, interstitium and peritubular capillaries, but α-SMA expression is restricted to arteriole and artery walls when the above mentioned structures mature. 47 Madsen et al. 48 showed that the treatment of rats with the AT1 antagonist candesartan for two weeks after birth reduced the total length, volume and surface area of capillaries in both cortex and medulla and disrupted vasa recta bundle organizations. These animals exhibited α-SMA-positive spindle-shaped cells that predominantly populated the outer medullary interstitium and the cortical medullary rays. In our study, the same pattern of α-SMA expression was observed with increased α-SMA expression predominantly in the outer medulla and vascular walls. Taken together, these data demonstrate that treatment with losartan during lactation disturbs the postnatal development of the postglomerular capillaries and vasa recta bundle organization, which can lead to changes in renal plasma flow. 48 Tempol supplementation did not change α-SMA expression at 21 days of age, but was able to reduce its increased expression in the renal cortex of 60-day-old losartan-treated rats.
The number of apoptotic cells was increased at 21 days of age in the tubulointerstitial compartment of the kidneys from the pups exposed to losartan, and this alteration was less intense in the animals that received the antioxidant together with losartan. The apoptosis can also be provoked by oxidative stress and tissue hypoxia caused by the vascular changes provoked by the neonatal losartan exposition. On the other hand, there was no difference in the number of apoptotic cells in the tubulointerstitial compartment of the renal tissue from the 60-day-old rats, which can be explained by the end of the development period when proliferation and apoptosis are both reduced.
Losartan-treated pups also exhibited intense tubulointerstitial macrophage infiltration in the renal cortex at 21 days of age. This increase persisted into adulthood at 60 days of age, when it was observed in both the glomerular and tubulointerstitial compartments. Machado et al. 13 followed pups up to 10 months of age and observed that the number of macrophages correlated with the number of AII-positive cells and the progression of changes in kidney function and structure. Macrophage infiltration is a common feature of human chronic kidney disease, 49 and its activation may result from hypoxia and oxidative stress due to the disturbed renal vasculature development caused by losartan exposure during kidney development, which leads to ROS generation and nitric oxide, and release of pro-inflammatory cytokines. 50 Therefore, interstitial macrophage accumulation in the kidneys of animals exposed to losartan may be a consequence of oxidative stress and tissue hypoxia caused by vascular changes, which generate stimuli to trigger a wide range of cellular responses, ranging from cell proliferation to apoptosis, and exacerbate the inflammatory process.51,52
In our study, tempol treatment associated with losartan during lactation attenuated the increase of macrophage infiltration in the tubulointerstitial area in the 21- and 60-day-old rats and in the glomeruli at 60 days of age. These results demonstrate an anti-inflammatory effect of tempol related to its antioxidant action. Increased levels of TBARS, glutathione, EC-SOD and nitrotyrosin expressions were found in the renal tissue from pups exposed to losartan during lactation at 21 days of age. These changes reflect increased oxidative stress in the tissues of these animals. Tempol treatment during lactation was able to reduce the oxidative stress observed in the 21-day-old losartan-treated animals. We observed that renal glutathione expression was higher in the rats exposed to losartan alone than in the rats exposed to both losartan and tempol. Other studies also show increased SOD expression in conditions that led to oxidative damage, which was considered by the authors as a compensatory mechanism.53–55 The expression of nitrotyrosine was increased in the renal cortex from losartan-treated animals. The peroxynitrite anion is produced by the reaction between the superoxide anion and nitric oxide, and protein tyrosine nitration is considered a consequence of peroxynitrite formation. 56 The treatment with tempol, as an antioxidant, attenuated this increased expression. Tempol has been found to enter rapidly in the cells and to be widely distributed in the body. 57 It reacts with cellular components in the cytoplasm and in the mitochondria. Beside this, the fact that the oxidative stress observed in pups from losartan-treated dams was not present in pups from dams treated with losartan plus tempol, showed that tempol should be present in the breast milk from these dams.
Our data showed that the increased oxidative stress may significantly contribute to the renal development disturbances that lead to the structural and functional changes observed in this model. It was observed that oxidative stress plays a crucial role in pathological conditions during the early neonatal period.18,23 Newborns are more susceptible to oxidative damage because of their high metabolic rates and low levels of antioxidant enzymes. We found increased expression of p-JNK and p-p38 in the renal cortex of losartan-treated animals that was prevented by tempol treatment. The MAPK pathways can be activated by oxidative stress, leading to the inflammation processes, 58 apoptosis and the development of renal lesions in losartan-treated pups. 3 However, the effect of tempol treatment in these animals was limited probably because AII also has several effects that are not mediated by oxidative stress such as the proliferation and differentiation of the tubular, interstitial and vascular cells.4,5,14,15 It can explain the abnormalities in renal vasculature and tubule interstitial compartment observed in both groups of losartan-treated rats, considering that cell proliferation and differentiation are important events in development.
In summary, the present study provides evidence that oxidative stress is involved in renal injury caused by exposure of offspring to losartan during lactation. Blocking oxidative stress by tempol attenuated the changes in GFR, fractional excretion of potassium, the increased phosphorylation of the MAPKs JNK and p38, apoptosis and inflammatory responses in the renal cortex induced by losartan.
Footnotes
Acknowledgements
The authors are grateful to Flávio Henrique Leite, Guilherme de Paula Lemos and Rubens Fernando de Melo for their expert technical assistance.
Conflict of interest
None declared.
Funding
This work was supported by a grant from the Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP, Foundation for the Support of Research in the State of São Paulo), Grant number 07/55816-4. Heloísa D.C. Francescato, Cleonice G.A. da Silva, Roberto S. Costa and Terezila M. Coimbra were recipients of fellowships from Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq).
