Abstract
Introduction:
This study was performed to address the pathological roles of the skeletal renin-angiotensin system (RAS) in type 1 diabetes-induced osteoporosis and the effects of the angiotensin II type 1 receptor blocker losartan on bones in diabetic mice.
Materials and methods:
Bone histomorphology was detected by H&E staining, Safranin O staining and X-ray radiography. Micro-CT was performed for the analysis of bone parameters. Gene and protein expression were determined by RT-PCR and immunoblotting.
Results:
Type 1 diabetic mice displayed osteopenia phenotype, and losartan treatment had no osteoprotective effects on diabetic mice as shown by the reduction of bone mineral density and microarchitectural parameters at the proximal metaphysis of the tibia. The mRNA expression of AGT, renin receptor and ACE, and protein expression of renin and AT1R were markedly up-regulated in the bones of vehicle-treated diabetic mice compared to those of non-diabetic mice. The treatment with losartan further significantly increased the expression of AGT, renin, angiotensin II and AT1R, and reduced the expression of AT2R receptor as compared to those of diabetic mice.
Conclusion:
Local bone RAS functionally played a role in the development of type 1 diabetic osteoporosis, and losartan had no bone-sparing function in diabetes mice because of enhance skeletal RAS activity.
Introduction
The renin-angiotensin system (RAS) is a hormonal cascade that is thought to act as a master controller of blood pressure and fluid balance within the body. 1 In addition to the systemic RAS, there is a fully functional RAS in local tissue that is postulated to participate in various physiological and pathological processes such as insulin secretion, 2 glomerular sclerosis, 3 renal inflammation, 4 atherosclerosis, 5 and cardiac hypertrophy. 6 Recent studies showed that the components of RAS, such as renin, angiotensin-converting enzyme (ACE), and angiotensin II (Ang II) receptors, are expressed in the local milieu of bone.7,8 Functional studies revealed that Ang II could stimulate the differentiation and activity of osteoclasts in vivo 9 and in vitro. 10 Moreover, the local RAS in bone was involved in age-related osteoporosis of aging mice 11 and the process of fracture healing in a mouse femur fracture model. 12
Osteoporosis, as a metabolic bone disease, is one of the most important complications of diabetes mellitus. The well-known causes for diabetic osteoporosis include the direct effects of insulin deficiency or resistance and hyperglycemia on bone and the bone marrow microenvironment. Additionally, advanced glycation end products of bone matrix proteins, and some biological factors like cytokines and adipokines, could exert their detrimental effects on bone cells. 13 A growing body of studies has demonstrated that diabetic complications, such as cardiovascular disease, 14 nephropathy, 15 and retinopathy, 16 are due to the high activity of RAS and the increased production of Ang II in local tissues, and that RAS inhibitors could significantly improve these pathological alterations as revealed by clinical practice.14 –16 However, whether the local RAS is involved in the skeletal deteriorations associated with diabetes is not known.
The common problem for all classes of RAS inhibitors is the appearance of a compensatory renin increase after RAS blocker use because of the disruption of the feedback inhibitory loop in renin production. This increase in renin activity stimulates the production of angiotensin I and ultimately Ang II, leading to a reduction in the efficacy of RAS inhibition. 17 We have reported these observations on losartan, one of the Ang II type 1 receptor (AT1R) blockers (ARBs), when applying it for the treatment of nephropathy in hyperglycemia mice with type 13,4 and type 2 diabetes. 18 Clinical evidence confirms that ARBs are effective reagents in reducing the risk of tissue-specific disorders associated with diabetes. 19 Therefore, we have a concern whether ARBs would produce any influence on the bones of diabetic patients, especially those associated with secondary osteoporosis, when the drug is usually used as the first-line treatment for diabetes complications.
We recently performed an animal study to address the effects of losartan on bone tissue of DBA/2J mice with type 1 diabetes induced by streptozotocin (STZ). The aim of the present study is to identify the pathophysiological role of local RAS in bone disorders induced by diabetes and, above all, to elucidate the impact of the AT1R blocker losartan on the bones of diabetic mice.
Materials and methods
Animal treatment
Ten-week-old male DBA/2J mice (Slac Laboratory Animal, Shanghai, China) were allowed to acclimate to their environment for one week before inducing diabetes. Mice were fasted for 4 hours (h) and then given an intraperitoneal injection of freshly prepared STZ at 40 mg/kg dissolved in 10 mM citrate buffer (pH 4.2, n = 25) or vehicle (non- diabetic control mice, n = 10) daily for five consecutive days. One week post-STZ injection (Week 0), fasting blood glucose (FBG) levels were measured with a blood glucose monitoring system (Roche). The mice with FBG more than 9 mmol/l were randomly divided into a diabetic osteoporosis group (DOP, n = 10) and a losartan-treated group (10 mg/kg, intragastrically (i.g.), n = 10). FBG was monitored at four and eight weeks post-drug treatment, and body weight was measured every two weeks during the experimental period. Eight weeks after drug administration, spot urine of each mouse was collected. Serum, tibias and femurs were immediately harvested for a variety of biochemical, histological and molecular analyses. The animal study protocol was reviewed and approved by the institution’s Animal Ethics Committee at the University of Shanghai for Science and Technology.
Serum and urine chemistries
Calcium (Ca) and creatinine (Cr) concentrations of serum and urine were measured by standard colorimetric methods using a micro-plate reader (Bio-Tek, VT, USA). The level of urine Ca was corrected by the concentration of urine Cr. Serum levels of intact parathyroid hormone (PTH 1-84) were detected using mouse bioactive PTH enzyme-linked immunosorbent assay (ELISA) assay (Immutopics, Inc., San Clemente, CA, USA). Serum tartrate-resistant acid phosphatase 5b (TRACP-5b) and bone-specific alkaline phosphatase (ALP-B) levels were determined using a sandwich ELISA kit.
X-ray analysis
Tibias and femurs were examined by Digital Radiography (GE AdvantX, USA) with the parameters as 50 mA and 32 mS.
Bone histology
The tibias were fixed in 4% formaldehyde/phosphate-buffered saline (PBS) (pH 7.2), decalcified in 0.5 M ethylenediaminetetraacetic acid (EDTA) (pH 8.0), and embedded in paraffin by standard histological procedures. Serial sections of 3 µm were cut and stained with hematoxylin and eosin (H&E). Safranin O (Sigma-Aldrich, St. Louis, MO, USA) staining was performed, combining with fast green and counter stain by hematoxylin. Stained slides were visualized under a microscope (Leica DM 2500, Germany).
Micro-computed tomography (CT) analysis
The hind limb without sample preparation or decalcification was fixed in a cylindrical plastic tube to prevent movement of the limb during scanning. The whole tibia of each animal was scanned to obtain an image, and then the proximal tibial metaphysis underneath the growth plate was examined on a 1.81 mm slab, corresponding to 173 slices, with a high-resolution micro vivaCT 40 system (Scanco Medical, Bassersdorf, Switzerland). The scanning parameters used were 70 kVp, 111 µA, and 1000 projections per 180°, resulting in a 10.5 μm isotropic voxel size and a total scan time of 13.2 minutes. The trabecular bone was determined by a fixed threshold. Hand-drawn contours were used to isolate the metaphyseal region of interest and trabecular compartments based on 100 consecutive slices. Bone microarchitecture was assessed with direct three-dimensional (3D) methods by µCT Evaluation Program (Image Processing Language v. 5.0A, Scanco). Morphologic 3D parameters for tibial metaphyseal trabecular bone were obtained as follows: (1) the mean mineral density of total volume (BMD/TV); (2) bone volume/total volume (BV/TV); (3) trabecular thickness (Tb.Th); (4) the geometric degree of anisotropy (DA), defined as the ratio between the maximal and minimal radius of the mean intercept length ellipsoid. Additionally, two-dimensional (2D) and 3D images for the 100 slices were reconstructed.
Reverse transcription-polymerase chain reaction (RT-PCR)
The tibia of each animal was crushed under liquid nitrogen condition, and RNA extraction was performed according to the TRIzol manufacturer’s protocol (Invitrogen, Carlsbad, California, USA). RNA integrity was verified by agarose gel electrophoresis. Synthesis of cDNAs was performed by RT reactions with 4 µg of total RNA using Moloney murine leukemia virus reverse transcriptase (Invitrogen) with oligo dT(15) primers (Fermentas) as described by the manufacturer. The first-strand cDNAs served as the template for the regular PCR performed using a DNA Engine (ABI). Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) as an internal control was used to normalize the data to determine the relative expression of the target genes. The PCR primers used in this study were as previously described. 11
Western blotting
The femurs were homogenized and extracted in Laemmli buffer (Boston Bioproducts, Worcester, MA, USA), followed by 5 min boiling and centrifugation to obtain the supernatant. Samples containing 40 µg of protein were separated on 10% sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) gel and transferred to nitrocellulose membranes (Bio-Rad Laboratories, Hercules, CA, USA). After saturation with 5% (w/v) nonfat dry milk in tris-buffered saline (TBS) and 0.1% (w/v) Tween 20 TBS with Tween 20 (TBST), the membranes were incubated with one of the following antibodies at dilutions ranging from 1:1000 to 1:2000 at 4°C overnight: mouse anti-renin monoclonal antibody, goat anti-angiotensin II polyclonal antibody, anti-angiotensin II type 1 receptor monoclonal antibody, and anti-angiotensin II type 2 receptor polyclonal antibody. All the above primary antibodies were purchased from Santa Cruz Biotechnology (USA). After three washes with TBST, membranes were incubated with secondary immunoglobulins conjugated to IRDye 800CW Infrared Dye (LI-COR Biotechnology, USA), including donkey anti-goat IgG and donkey anti-mouse IgG with the dilution of 1:15000. After 2 h incubation at room temperature, the membranes were washed three times with TBST. Blots were visualized by the Odyssey Infrared Imaging System (LI-COR). Signals were densitometrically assessed (Odyssey Application Software version 3.0) and normalized to the β-actin signals to correct for unequal loading using the mouse monoclonal anti-β-actin antibody (Bioworld Technology, USA).
Statistical analysis
The data from these experiments were reported as mean ± standard error of mean (SEM) for each group. All statistical analyses were performed using PRISM version 4.0 (GraphPad). Inter-group differences were analyzed by one-way analysis of variance (ANOVA) and followed by Tukey’s multiple comparison test as a post-test to compare the group means if overall p < 0.05. Differences with a p value of less than 0.05 were considered statistically significant.
Results
Basic parameters
The body weight of normal mice was steadily increased (Figure 1A) and the FBG level was maintained between the normal range of 7~8.5 mmol/l in the control group (Figure 1B) during the experimental period. One week after STZ injection, the mean body weight of STZ-treated mice was 3 g less than that of the control group mice; even though there is a slight increase of body weight at Week 2 and Week 4 post-STZ injection, the body weight in the DOP group and the losartan group was significantly reduced at Week 8 as compared to that of control group. The FBG value of DOP mice and losartan-treated DOP mice climbed up from 12 mmol/l at week 0 to 28 mmol/l at Week 8, significantly higher than that of the control group at each time point.

Body weight and fasting blood glucose. One week after STZ injection (Week 0), diabetic mice were treated with losartan for eight weeks. Body weight (A) and fasting blood glucose (B) were measured every two weeks and four weeks, respectively, during the experimental period. Values are expressed as means ± SEM, n = 10. *p < 0.05, **p < 0.01, ***p < 0.001, vs control group. STZ: streptozotocin; DOP: diabetic osteoporosis group.
Serum and urine chemistries
Serum calcium level was comparable in the three experimental groups (Table 1). Losartan significantly reduced the serum level of PTH 1-84 in DOP mice (p < 0.05) when compared with controls. The serum creatinine level of DOP mice was significantly higher (p < 0.05) than that of control mice, and the treatment with losartan did not improve this parameter. Similarly, STZ injection induced high urine calcium excretion (p < 0.05), and losartan caused an insignificant decrease in urine calcium content. Additionally, serum concentrations of bone turnover markers, like TRACP-5b as bone resorption marker and ALP-B as bone formation marker, were determined. The results showed that the serum TRACP-5b level in the losartan group was higher (p < 0.05) than that of the controls, and the serum ALP-B level was largely elevated (p < 0.05) in the DOP group and the losartan group.
Chemistries in serum and urine.

Values are expressed as means ± SEM, n = 10 in each group. ap < 0.05, compared to control. DOP: diabetic osteoporosis group; Ca: calcium; Cr: creatinine; PTH: parathyroid hormone; TRACP-5b: tartrate-resistant acid phosphatase 5b; ALP-B: bone-specific alkaline phosphatase.
Radiography
X-ray radiogrammetry was performed to qualitatively observe bone mass of the whole bones including the femur (Figure 2A) and tibia (Figure 2B). The pictures clearly showed the low bone mass in the DOP group and losartan group when compared to those of controls with much more brightness. Moreover, losartan did not show any benefit on the whole bone mass of diabetic mice.

X-ray and histomorphological analysis on bones. X-ray radiogrammetry was performed to qualitatively observe the changes of bone mass in the femur (A) and tibia (B). The high brightness level in bone tissue represents more bone mineral content. Hematoxylin & Eosin (H&E) staining was conducted on the proximal end of the tibia (C). Safranin O staining was carried out on the proximal end of the tibia (D) and the distal end of the femur (E). The cartilage adjacent to the growth plate of femoral distal metaphysis are shown by the arrows with solid line. Magnification (C, D, E) ×100. DOP: diabetic osteoporosis group.
Histomorphology
H&E staining (Figure 2C) and Safranin O staining (Figures 2D and 2E) were performed to observe the epiphyseal structures of tibias and femurs. The H&E and Safranin O stainings showed the loss of cartilage network connections and cartilage mass at the tibial proximal metaphysis of DOP mice. The thickness of cartilage adjacent to the growth plate at the femoral distal end was reduced in the DOP group (shown by arrow) when compared to that of the control group, suggesting the occurrence of delayed epiphyseal ossification caused by diabetes. These phenotypes of epiphyseal microstructures were unchanged in the losartan-treated group when compared to the vehicle-treated DOP group.
Micro-CT analysis
The loss of trabecular bone mass at proximal metaphysis of the tibia was clearly demonstrated in the profiles of 2D (Figure 3A) and 3D images (Figure 3B) where osteopenia occurred in DOP mice and was exacerbated in losartan-treated diabetic mice, and these observed changes were quantitatively reflected by the 3D bone parameters (Figure 3C). Type 1 diabetes triggered a significant decrease in BV/TV by 33%, Tb.Th by 20%, and BMD/TV, p < 0.05 by 32%, while causing a corresponding 22% increase in the DA at tibial proximal metaphysis when compared to those parameters of the control. Notably, the treatment with losartan for diabetic mice resulted in a further reduction in BV/TV by 17%, Tb.Th by 25% and BMD/TV by 14%, and an elevation of 15% in DA when compared to those of the DOP group. Furthermore, the decrease in BV/TV (p < 0.05), Tb.Th (p < 0.01) and BMD/TV (p < 0.01) was significantly different between the losartan group and the control group. However, the difference of these parameters between the DOP group and control group was not statistically significant.
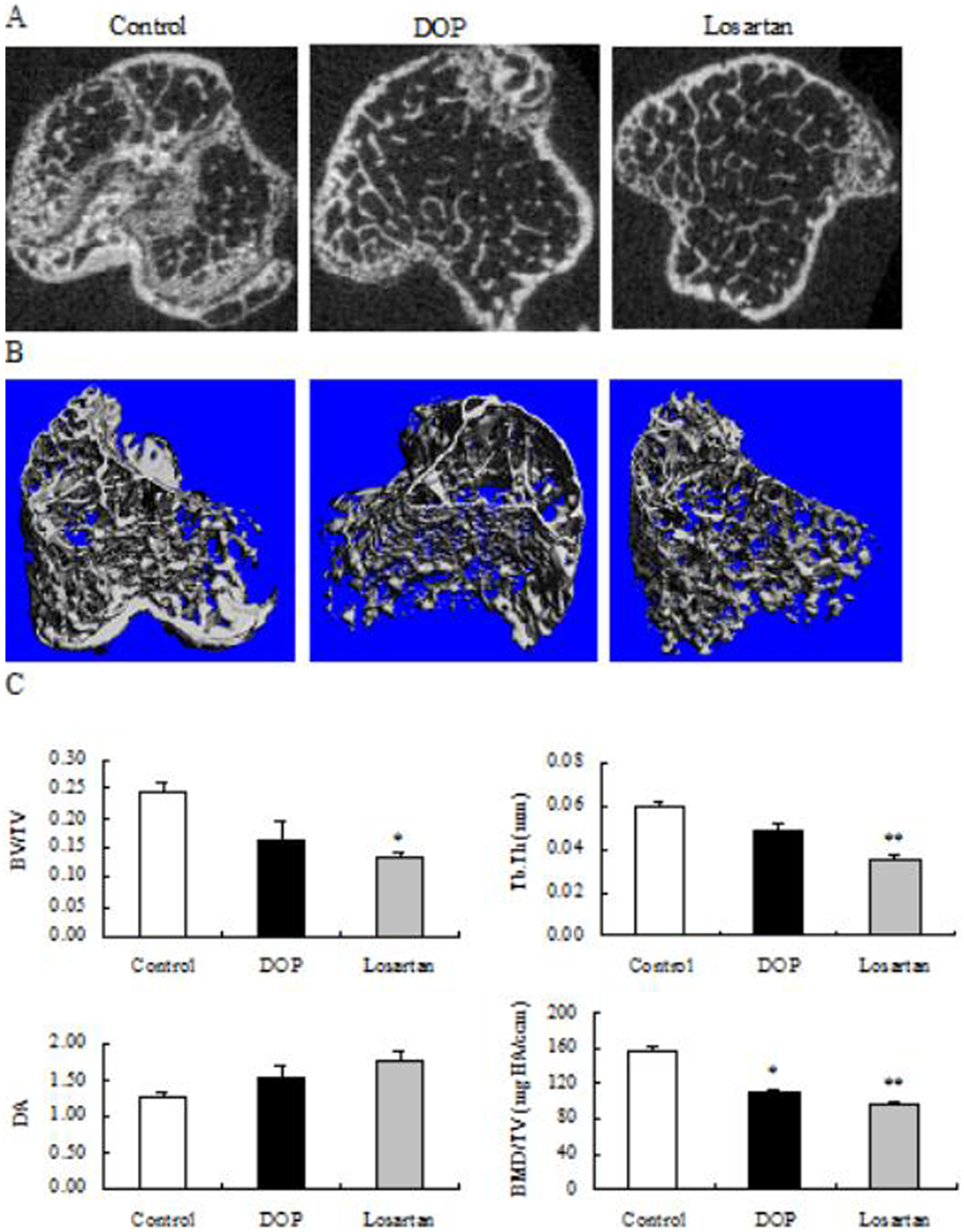
Micro-CT analysis on the trabecular architecture of proximal tibial metaphysis. (A) Two-dimensional images of trabecular bone. (B) Three-dimensional images of trabecular bone. (C) Trabecular bone morphological parameters: bone volume over total volume (BV/TV), trabecular thickness (Tb.Th.), geometric degree of anisotropy (DA), bone mineral density over total volume (BMD/TV). Values are expressed as means ± SEM, n = 6. *p < 0.05, **p < 0.01, vs control group. Micro-CT: micro-computed tomography; DOP: diabetic osteoporosis group.
mRNA and protein expression of RAS components
The mRNA expression of angiotensinogen (AGT), renin receptor and ACE was significantly higher (p < 0.01, Figure 4B) in tibias of DOP mice and losartan-treated DOP mice than those of control mice. The increased expression of AGT and AT1R in tibias of the losartan group was statistically significant (p < 0.05) compared to those of the DOP group, while there was no difference in mRNA expression level of renin receptor and ACE between the DOP group and the losartan group.

mRNA expression of RAS components in the tibia. (A) RT-PCR examination of angiotensinogen (AGT), renin receptor (R-R), angiotensin-converting enzyme (ACE) and angiotensin II type 1 receptor (AT1R). (B) Densitometric quantification of the RT-PCR data. Values are expressed as means ± SEM, n = 8. **p < 0.01, ***p < 0.001, vs control group; #p < 0.05, ##p < 0.01, vs DOP group. mRNA: messenger RNA; RAS: renin-angiotensin system; RT-PCR: reverse transcriptase-polymerase chain reaction; DOP: diabetic osteoporosis group; GAPDH: glyceraldehyde-3-phosphate dehydrogenase.
The protein levels of renin (p < 0.001, Figure 5B) and AT1R (p < 0.05) were markedly elevated in femurs of DOP mice and losartan-administered DOP mice as compared to those of control mice. The treatment with losartan stimulated the up-regulation of renin and Ang II protein expression (p < 0.01) and down-regulation of AT2R protein expression (p < 0.05) compared to those of the DOP group.
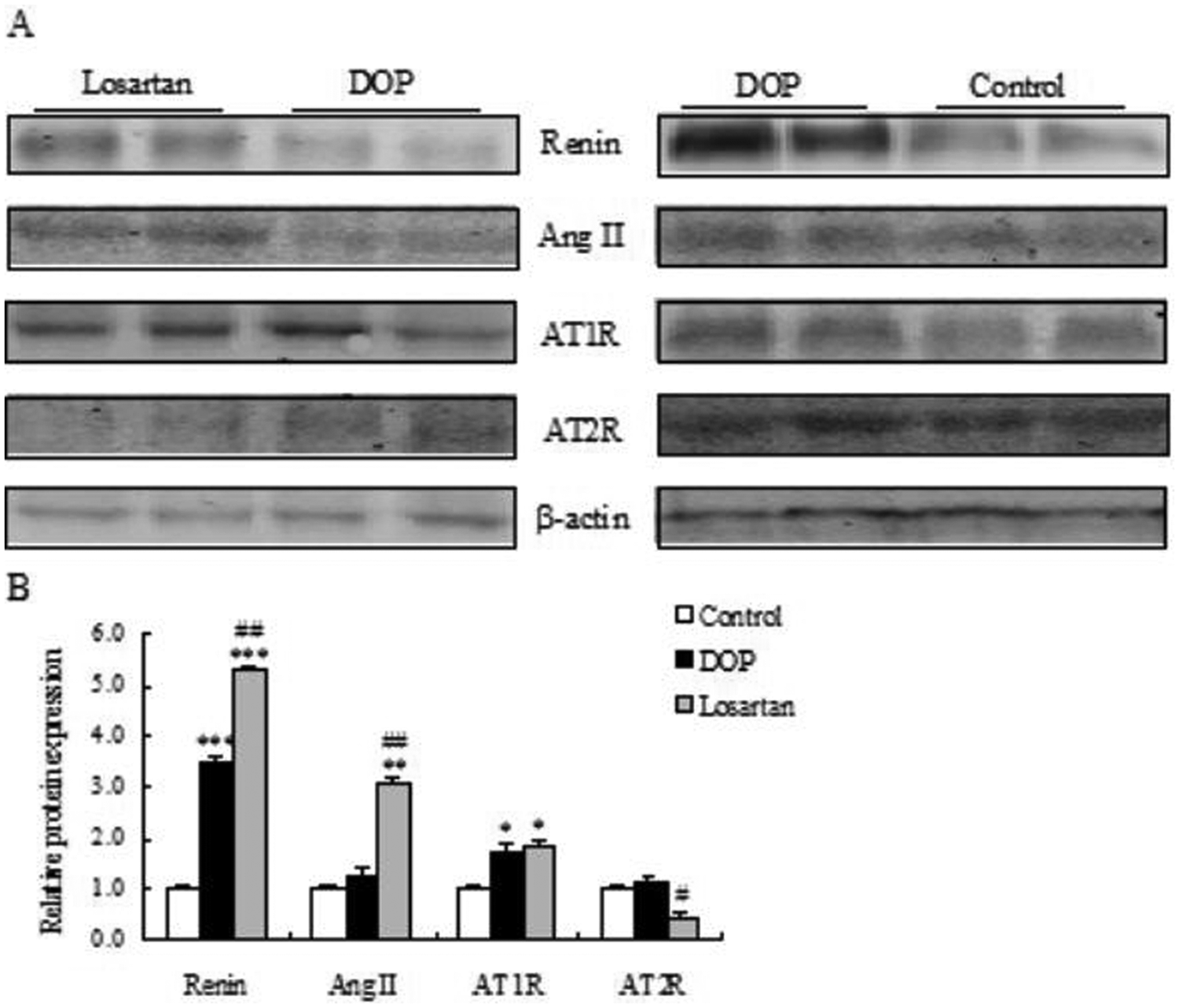
Protein expression of RAS components in the femur. (A) Representative Western blot of renin, angiotensin II (Ang II), and its type 1 receptor (AT1R) and type 2 receptor (AT2R). (B) Densitometric quantification of the Western data. Values are expressed as means ± SEM, n = 8. *p < 0.05, **p < 0.01, ***p < 0.001, vs control group; #p < 0.05, ##p < 0.01, vs the DOP group. RAS: renin-angiotensin system; DOP: diabetic osteoporosis group; Ang II: angiotensin II; AT1R: angiotensin II type 1 receptor; AT2R: angiotensin II type 2 receptor.
Discussion
Diabetic osteoporosis is a long-term complication of diabetic mellitus, and effective blockade of the progression of osteoporosis remains a medical challenge. Because the activation of the local RAS in the skeleton is one of the major contributors for osteopenia revealed by recent in vivo7,11 and in vitro studies,8,9 we investigated the pathological roles of skeletal RAS in type 1 DOP in the present study.
In pharmacological studies, it has been proposed that inhibition of RAS with ACE inhibitors (ACEIs) or ARBs has beneficial effects beyond those resulting from lowering blood pressure alone, such as renoprotective effects3,4,18 and cardiovascular outcomes. 20 These small-molecule inhibitors targeting RAS are currently used for treatment of diabetic nephropathy. 21 The efficacy of RAS-targeting drugs is often compromised by the reactive renin increase caused by disruption of the renin feedback inhibition. 22 High renin buildup increases the risk of Ang II-dependent and -independent organ damage. Therefore, this study was performed to identify the effects of losartan (ARBs) on bones of diabetic mice. This work has important clinical significance when applying ARBs in the treatment for diabetes nephropathy.
As expected, in contrast to the non-diabetic mice, the mice in the DOP group exhibited the typical features of type 1 diabetes as shown by the basic physiological parameters, such as decreased body weight and sustained elevation of FBG level. Losartan treatment did not result in any improvements in body weight and FBG level of diabetic mice, in accordance with our previous reports.3,4
Diabetes induced the loss of bone minerals of the whole femur and tibia, the disconnection of cartilage network, and the decrease of trabecular bone mass at the proximal metaphysis of the tibia and distal end of the femur as demonstrated by X-ray radiography, H&E staining, Safranin O staining, and 2D and 3D images reconstructed from micro-CT analysis in this study. These results are in agreement with our previous findings in type 1 diabetic mice where the deteriorations of bone microstructure and loss of BMD occurred because of hypercalciuria, which was attributed to the alteration of vitamin D metabolism and the concomitant decrease in renal calcium transporters expression. 23
To explore whether local RAS is involved in the progression of DOP in the current study, the expression of RAS components in bone was determined. The results showed that mRNA expressions of AGT, ACE and renin receptor in the tibia as well as protein expressions of AGT and AT1R in the femur were dramatically up-regulated in the DOP group, suggesting AT1R-involved Ang II signaling and renin receptor-involved renin signaling might functionally stimulate bone deterioration in type 1 diabetes. To our knowledge, this study is the first to reveal the alterations of skeletal RAS activity in type 1 diabetes, even though these changes have been demonstrated in the cardiovascular system, 14 the kidney, 15 and the retina 16 associated with diabetes mellitus.
In an attempt to explore the influences of ARBs on bone health when they are utilized to treat diabetes complications, the current study applied radiogrammetry, histomorphological stainings, and micro-CT technique in evaluating the effects of losartan on long bones of type 1 diabetic mice. We found that losartan treatment for eight weeks appears to have no protective effects in tibias and femurs as compared to those of the vehicle-treated diabetes group. The quantitative data, as demonstrated by the decrease of trabecular BV, Tb.Th and BMD, were in accordance with the histomorphological observations. These findings supported the earlier pharmacological result that treatment of Tsukuba hypertensive mouse with losartan resulted in exacerbation of the low bone mass phenotype. 7 The effects of losartan on bones of diabetic mice in the current study could be attributed to its modulations on bone metabolism shown by biochemical markers and local RAS components shown by gene and protein expressions.
This study showed that losartan treatment accelerated the decrease in circulating intact PTH level of type 1 diabetic mice. Similar results were reported in humans by Koiwa et al. that a significant decrease in serum intact PTH level was seen in males with diabetes mellitus when treated with ARBs. 24 However, there is likely a discrepancy between low serum PTH level and high bone turnover rate as demonstrated by an increase in the serum level of ALP-B and TRACP 5b in the losartan group of this study. One of the possible reasons could be the potential activation of losartan on the peroxisome proliferator-activated receptor γ pathway, 25 which is known to be a critical factor in regulating bone metabolism as reported in recent studies. 26 On the other hand, since sympathetic tone has been shown to be an important regulator of bone remodeling, we suspect that it might be related to the capability of Ang II to induce the release of norepinephrine from sympathetic nerve endings. 27 Future study will be needed to determine whether the two proposed pathways are involved in mediating the effects of losartan on bone in diabetic mice.
The molecular basis underlying the regulation of losartan therapy of bone is the stimulation of RAS activation within the local bone tissue. Consistent with the theory of compensatory renin increase, losartan treatment dramatically increased renin as well as Ang II levels in bone. Additionally, we noted that losartan has biphasic effects on the expressions of skeletal Ang II receptors as demonstrated by a significant up-regulation of AT1R and a down-regulation of AT2R in diabetic mice. Based on the study of AT2R knockout mice and AT2R antagonist-treated mice, 8 Izu et al. concluded that AT2R blockade increased bone mass. In contrast to the results of Izu that there was no pathological change in bone in AT1R-deficient mice, 8 Shimizu et al. reported that Ang II accelerated osteoporosis by AT1R pathway, 9 and Kaneko et al. found male and female AT1R null mice exhibited an increase in trabecular bone volume, trabecular number, and connectivity. 28 However, studies indicated that there is a cross-talk between AT1R and AT2R pathways and functional interactions exist between the two receptors. 29 Thus, it is possible that the counter-regulatory functions of AT1R and AT2R are involved in the effects of losartan on bones in this study.
Overall, this study indicates that high local bone RAS activity contributes to type 1 diabetic osteoporosis, while the treatment with the ARB losartan for diabetic mice could not exert bone-sparing function because of the compensatory stimulation of bone RAS activation. These findings suggest that the indicators monitoring bone health need to be measured when ARBs are clinically utilized in treating diabetes complications. Additionally, whether the RAS inhibition with ACEIs to block the Ang II synthesis could produce beneficial effects on bone health in diabetes should be further investigated even though cross-sectional studies on elderly Chinese populations 30 as well as hypertensive menopausal women 31 and a prospective cohort study on older American men 32 showed an association of ACEIs use with higher BMD.
Footnotes
Conflict of interest
None declared.
Funding
This work was sponsored by the Shanghai Pujiang Program (10PJ1407700) and Innovation Program of Shanghai Municipal Education Commission (11ZZ137) and was partially supported by the Hong Kong Scholars Program (XJ2011022) and the China Postdoctoral Science Foundation (2012M511115).
