Abstract
Introduction:
The development of left ventricular remodelling after acute myocardial infarction is a predictor of heart failure and mortality. The purpose of the present study was to assess whether the polymorphism of angiotensinogen (AGT) gene with threonine (T) instead of methionine (M) at amino acid 235 in exon 2 (M235T) had effects on cardiac remodelling after acute myocardial infarction.
Methods:
One hundred and forty-one patients (mean age 56.4±11.1 years) with a first acute myocardial infarction were enrolled. Within 24–72 hours of the onset of the symptoms and at a four month period two-dimensional echocardiography was performed. Remodelling was defined as a 20% increase from the baseline in left ventricular end-diastolic volume. The genotypes of the study group were compared with the reference group (n=1010) genotypes. AGT M235T polymorphism was determined using polymerase chain reaction amplification.
Results:
At follow-up, 49 patients (34.7%) were classified as having left ventricular remodelling. Anterior localization of the infarct (p=0.008), leucocyte count at admission (p=0.040), global left ventricular longitudinal strain (p=0.021) and MM genotype of AGT (p=0.024) were independent predictors of ventricular remodelling after myocardial infarction.
Conclusions:
Anterior wall infarction, increased leucocyte count, decreased longitudinal strain of left ventricular and polymorphism of AGT M235T may predict remodelling after myocardial infarction.
Introduction
Left ventricular (LV) dilatation after acute myocardial infarction (AMI) is a major predictor of further prognosis and development of heart failure. LV dilatation and remodelling starts immediately after AMI and is defined as changes in both infarcted and non-infarcted regions of the ventricle, leading to impaired contractile function, ventricular dilatation and heart failure.1,2
Previous studies demonstrated relations between the occurrence of LV remodelling after AMI and pre-existing hypertension, size of an infarct area, revascularization of the culprit artery, anterior location of the infarct, neurohumoral activation, including the sympathetic, beta-adrenergic and renin–angiotensin system (RAS).3–7 The effect of thrombolysis or early interventional revascularization of the infarct artery, β-blockade and angiotensin-converting enzyme (ACE) inhibition in the acute phase after AMI and in the chronic phase of consequent development of heart failure due to LV remodelling are widely discussed. However, significant heterogeneity exists in the pharmacological benefits of the standard treatment and prognostic outcomes to individual subjects.8,9 Because of that, great attention has been devoted to the evaluation of genetic variation of certain enzymatic systems, as a potential risk factor in the development of LV remodelling. RAS plays a central role in arterial blood pressure regulation and pathophysiology of heart failure and many of the key proven pharmacotherapies based on RAS enzymatic inhibition are used in patients with acute coronary syndromes and heart failure. 10
Angiotensinogen (AGT) is discussed as one of the potential components of RAS to have a significant role in LV remodelling and heart failure development. 11 AGT is a liver protein and is the substrate of renin. 12 The reaction between renin and AGT starts an enzymatic cascade that generates the decapeptide angiotensin I, which is later converted to angiotensin II by ACE and chymase. The human AGT gene consists of five exons and four introns and is located in the chromosome 1q42–43. The molecular variant (M235T) of the AGT gene, encoding a threonine (T) instead of a methionine (M) at residue 235 of the mature protein, has been associated with higher plasma AGT levels in patients homozygous for the T allele, which might have crucial effects on pathophysiology of acute coronary syndromes in acute and chronic phases regarding the development of LV remodelling.11,13,14
The aim of the present study was to assess whether the genetic background of the individual patient, particularly polymorphism of AGT gene, might predict changes of LV systolic function and volume size contributing to LV remodelling in patients with AMI.
Materials and methods
Study population
One hundred and eighty-two patients with a first AMI (with the symptoms from the onset of AMI not more than 72 hours) were enrolled into the study. All of them were treated at the Cardiology Department of the Lithuanian University of Health Sciences Hospital from October 2007 to March 2011 and underwent coronary angiography, following percutaneous intervention of the culprit artery. The mean age of the studied population was 56.4±11.1 years. AMI was defined according to the standard criteria based on clinical symptoms, ECG findings and cardiac enzyme abnormalities. 15 Exclusion criteria were as follows: prior myocardial infarction; arrhythmias, including atrial fibrillation; inadequate two-dimensional (2D) echocardiographic images for analysis; contraindication to coronary angiography/intervention (including renal failure, bleeding tendency, shock status, severe anaemia). Family history, cardiovascular risk factors and current treatment were obtained from each patient using a standard questionnaire. Hypertension was defined as the presence of elevated systolic (>140 mmHg) and/or diastolic (>90 mmHg) blood pressure or the current use of antihypertensive drugs. A patient was considered as a smoker if he was smoking at the current moment or was a smoker in the past. Diabetes mellitus was identified when dietary treatment and/or medical therapy was required to control blood glucose levels. Hypercholesterolaemia was defined as serum total cholesterol levels of 5.2 mmol/l or more, low density cholesterol more than 2.6 mmol/l, and triglycerides equal or more than 1.7 mmol/l or use of statin medication. 16
All the patients were invited for the follow-up visit at a four-month period and underwent a repeated echocardiographic examination. Data of 141 patients matching criteria of the study design were further evaluated.
Reference group genotypes were used for the comparison with the genotypes of the AMI population. The reference group (N=1010) consisted of a random sample of Kaunas population patients, taken from the epidemiological HAPIEE (Health, Alcohol and Psychosocial Factors In Eastern Europe) study, free from symptoms of ischaemic heart disease (IHD) and stroke.
Written informed consent was obtained from all patients, and the study protocol was approved by the ethics committee of the hospital.
Echocardiography
All AMI subjects were examined using 2D echocardiography within 48–72 hours after the symptoms of AMI. Patients were imaged in the left lateral decubitus position using a commercially available system (Vivid Seven, General Electric-Vingmed, Milwaukee, WI, USA). Standard images were obtained using a 3.5-MHz transducer, at a depth of 16 cm in the parasternal (long- and short-axis images) and apical (two-, three- and four-chamber images) views. Standard 2D and colour Doppler data, triggered to the QRS complex, were saved in a cine-loop format. Measurements were averaged from at least three consecutive beats. At baseline, 2D echocardiography was used to assess conventional parameters such as LV end-systolic diameter (LVESD), LV end-diastolic diameter (LVEDD), LV end-systolic volume (LVESV), LV end-diastolic volume (LVEDV), LV ejection fraction (LVEF), wall motion score index (WMSI), the mitral inflow peak early velocity (E)/mitral annular peak early velocity (Eʹ), or E/Eʹ ratio. 2D speckle-tracking imaging analysis for evaluation of deformation parameters (strain) was also performed. At four-month follow-up, 2D echocardiography was made repeatedly to reassess LV volumes and LVEF. The LV volumes (end-systolic and end-diastolic) and LVEF were calculated from the conventional apical two- and four-chamber images, using the biplane Simpson’s technique. 17 All other echocardiographic dimensions, severity of mitral regurgitation, assessment of regional contraction and evaluation, and longitudinal strain of LV assessed by speckle-tracking imaging were evaluated as described previously. 18
LV remodelling was defined as a ≥20% increase in LV end-diastolic volume (LVEDV) at four-month follow-up compared with the baseline, respectively. 19
Genetic analysis
Genomic DNA was extracted from 3 ml EDTA venous blood samples (white blood cells) using a commercial DNA isolation kit – Sorpoclean Genomic DNA Extraction Module kit (Sorpo Diagnostics, Vilnius, Lithuania) – according to the manufacturer’s instructions. Aliquots of purified DNA were stored at −20°C until use in real time-PCR analysis. Subjects were genotyped for AGT gene M235T polymorphism. Samples were genotyped with TaqMan allelic discrimination Assay-By-Design (Applied Biosystems, Foster City, CA, USA). Cycling conditions were preceded by a denaturing step at 95°C for 10 min, followed by 40 cycles of denaturation at 95°C for 30 s, annealing at 60°C for 1 min. Allele-specific fluorescence was then analysed on an ABI 7900HT Sequence Detection System with SDS v. 2.1 (Applied Biosystems, Foster City, CA, USA).
Statistical analysis
All statistical analysis was carried out using Software Package for Social Sciences (SPSS) version 17.0 (SPSS, Chicago, IL, USA).
Continuous variables were expressed as means ± standard deviations (SDs) when normally distributed, and as medians (25th, 75th percentiles) when not normally distributed. Continuous variables were assessed using the unpaired Student’s t test and Mann–Whitney U test, as appropriate. Categorical variables are presented as absolute numbers and percentages and are compared using the χ2 test. Differences in echocardiographic variables (baseline vs. four months) were assessed by paired Student’s t test. Allelic and genotypic frequencies were analysed with the χ2 test. In order to predict LV remodelling in an individual patient, clinical, echocardiographic and genetic variables were put into logistic regression analysis. Variables with statistical significance at univariable analysis were further analysed at multivariable analysis. A p value < 0.05 was considered statistically significant.
Results
Feasibility
A total of 182 consecutive AMI patients matching the inclusion criteria were screened initially. Eight subjects did not complete the follow-up because of their decision not to participate in the repeated evaluation. One AMI patient died during a four-month follow-up (non-cardiac cause of death) and seven patients were withdrawn from the study because of the recurrent symptoms of angina/infarction with following percutaneous coronary intervention percutaneous coronary intervention (PCI). Twenty-six patients (14.3%) with poor quality echocardiographic images were excluded from the further evaluation after the first echocardiographic evaluation.
Clinical and echocardiographic patient characteristics
At follow-up, 49 patients (34.7%) were classified as having LV remodelling. Age, gender, time from AMI symptoms to revascularization, body surface area, ischaemic heart disease risk factors, use of medications at follow-up, and number of involved coronary arteries showed no significant differences between the two groups (Table 1). Patients with LV remodelling were more frequently prone to anterior wall MI (p<0.01), higher leucocyte count value at admission (p<0.01), lower LVEF (p<0.05), increased LVESV (p<0.05) and higher WMSI (p<0.01) at the acute episode of MI. Parameters of myocardial deformation such as global systolic longitudinal strain of LV and global systolic longitudinal strain rate of LV were lower at baseline in AMI patients who developed LV remodelling at follow-up. There were no differences between the groups in mitral regurgitation (MR) grade or E/Eʹ ratio.
The clinical and echocardiographic characteristics of the patient group with and without left ventricular remodelling.
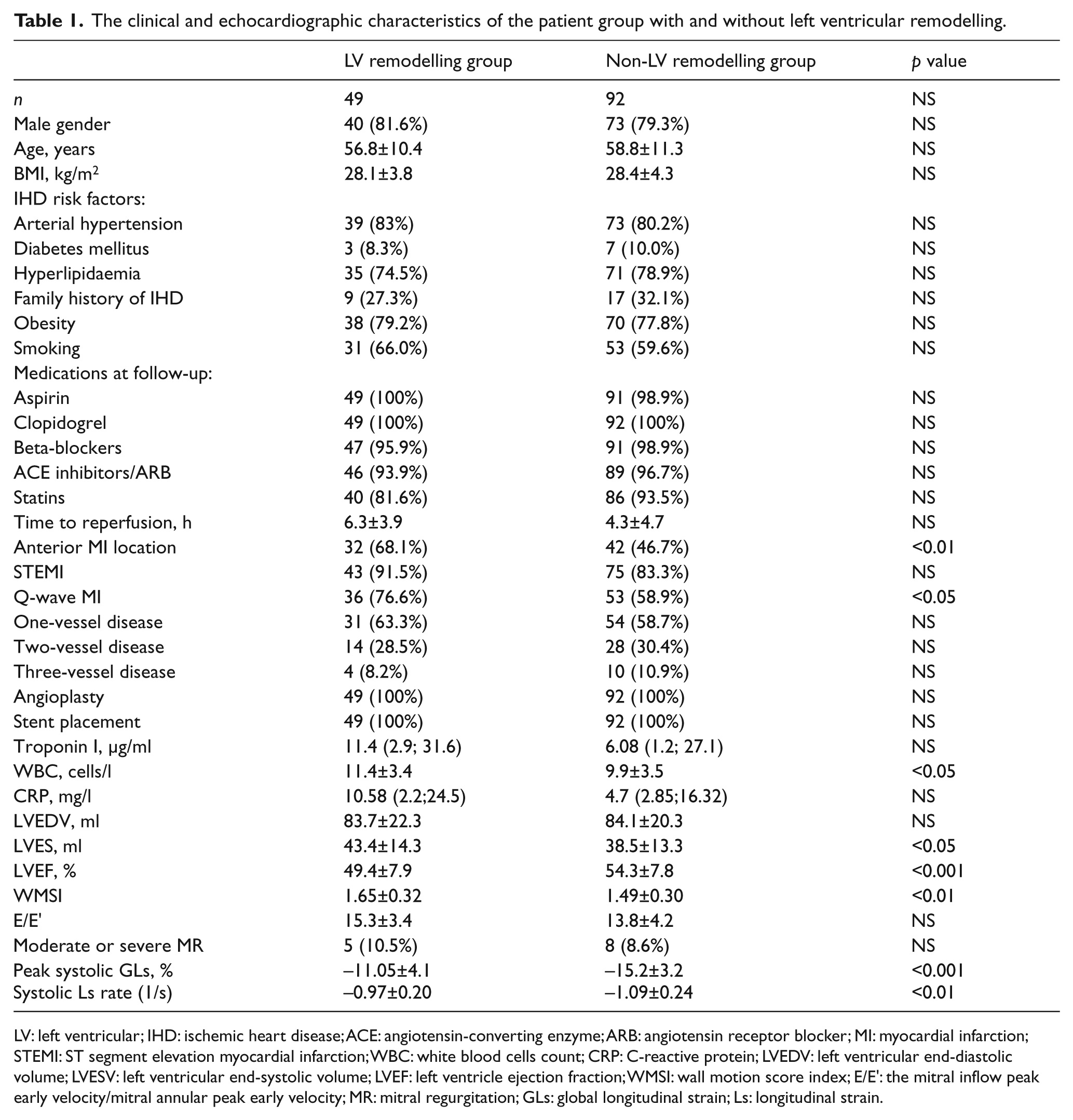
LV: left ventricular; IHD: ischemic heart disease; ACE: angiotensin-converting enzyme; ARB: angiotensin receptor blocker; MI: myocardial infarction; STEMI: ST segment elevation myocardial infarction; WBC: white blood cells count; CRP: C-reactive protein; LVEDV: left ventricular end-diastolic volume; LVESV: left ventricular end-systolic volume; LVEF: left ventricle ejection fraction; WMSI: wall motion score index; E/Eʹ: the mitral inflow peak early velocity/mitral annular peak early velocity; MR: mitral regurgitation; GLs: global longitudinal strain; Ls: longitudinal strain.
Frequencies of AGT gene genotypes
AGT genotype and allele frequency distribution of LV remodelling and non-LV remodelling groups were according to the Hardy–Weinberg equilibrium (p>0.05).
The frequencies of the genotypes of AGT gene from the studied AMI population are presented in Table 2. Patients with LV remodelling significantly more often were carriers of the MM genotype when compared with non-LV remodelling group patients (p=0.02). When genotype frequencies were compared between AMI patients and reference group patients, patients from the non-LV remodelling group more rarely were carriers of the MM genotype (p=0.0005), while LV remodelling group patients have not shown any significant differences in genotypes compared with the general population.
Genotype frequency in patients with and without left ventricular remodelling compared with the reference group.

AGT: angiotensinogen; LV: left ventricular.
p<0.05 between LV remodelling and non-LV remodelling groups.
p<0.001 between non-LV remodelling and reference groups.
p<0.05 between non-LV remodelling and reference groups.
At follow-up, patients with MM genotype had larger LVESD (34.6±4.8 mm vs. 30.3±4.8 mm, p<0.05), LVEDV and LVESV (99.81±28.5 ml vs. 88.1±25.1 ml, p<0.05 and 49.7±19.9 ml vs. 41.2±17.7 ml, p<0.05, respectively) and lower LVEF (48.6±6.5% vs. 52.46±9.3%, p<0.05) than patients with the TM or TT genotype (Table 3). Patients with the MM genotype revealed decreased LVEF at follow-up when compared with the initial parameters (48.6±6.5% vs. 53.2±7.6%, p<0.05), while carriers of the TM+TT genotype showed a significant improvement of LVEF at follow-up (52.46±9.3% vs. 49.3±8.6%, p<0.05).
Conventional echocardiographic parameters in acute myocardial infarction patients with MM genotype and acute myocardial infarction patients with TM+TT genotype.

p<0.05 between parameters of MM genotype group and TM+TT genotype group.
AMI: acute myocardial infarction; LVEDD: left ventricular end-diastolic diameter; LVESD: left ventricular end-systolic diameter; LVEDV: left ventricular end-diastolic volume; LVESV: left ventricular end-systolic volume; LVEF: left ventricular ejection fraction; LA: left atrium; E/Eʹ: the mitral inflow peak early velocity/mitral annular peak early velocity; MR: mitral regurgitation.
Clinical and genetic predictors of LV remodelling
When clinical, echocardiographic and genetic variables were put into the logistic regression analysis, significant predictors of LV remodelling were revealed: anterior localization of the infarct (p=0.012), leucocyte count at admission (p=0.038), LVESV (p=0.041), baseline LVEF (p=0.011), global longitudinal strain (p=0.001) and MM genotype (p=0.037) (Table 4). Multivariable analysis revealed that anterior localization of the infarct, leucocyte count at admission, global longitudinal strain and MM genotype were independent predictors of LV remodelling after AMI (Table 4).
Logistic regression analysis to predict left ventricular remodelling. a
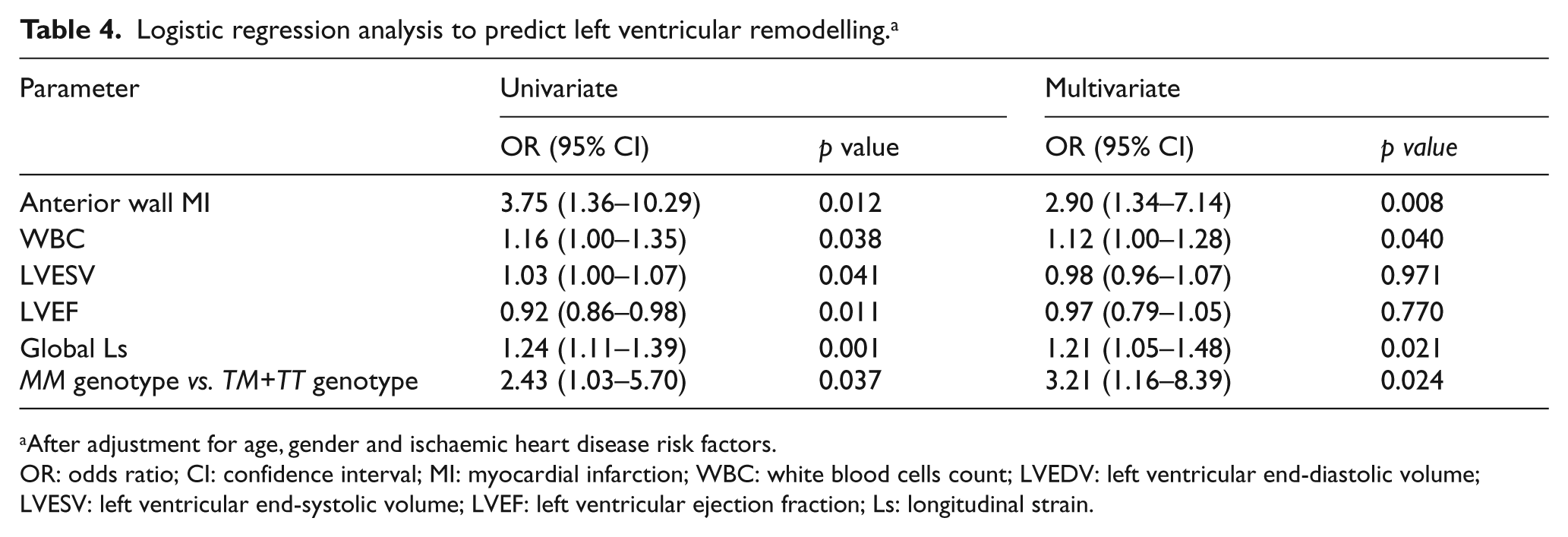
After adjustment for age, gender and ischaemic heart disease risk factors.
OR: odds ratio; CI: confidence interval; MI: myocardial infarction; WBC: white blood cells count; LVEDV: left ventricular end-diastolic volume; LVESV: left ventricular end-systolic volume; LVEF: left ventricular ejection fraction; Ls: longitudinal strain.
Discussion
In the present study we have analysed the possible impact of several clinical, echocardiographical and genetic factors, particularly the polymorphism of the AGT gene on ventricular remodelling following AMI. Results of our study revealed that predominance of the anterior wall MI, increased white blood cell count at admission, reduced global systolic longitudinal strain assessed by speckle-tracking imaging and polymorphism of the AGT gene independently had an impact on LV remodelling development after AMI.
LV remodelling occurs in a quite significant number of AMI patients and is associated with development of heart failure, cardiac arrhythmias and a poor survival rate, therefore early identification of this consequence of AMI is clinically very important.3,4,20 The process of LV remodelling begins immediately after the onset of acute MI. Along with the changes of the infarct zone and non-infarct area of the LV, activation of the neurohumoral system after AMI plays an important role in post-infarct remodelling development.1,21
The RAS deserves to be pointed out in the pathophysiology of the post-AMI remodelling process.11,22,23 Post-infarction activation of that system stimulates the intracellular signalling pathway, resulting in the increase of myocytes and fibroblasts protein synthesis, leading to the cell hypertrophy and fibrosis of the ventricle. Other effects such as increased vessel permeability, growth factor and metalloproteinases activation, haemodynamic overload through vasoconstriction and water retention, increased oxidative stress and direct cytotoxic effect leading to cell death through necrosis or apoptosis are observed.23 –25 Furthermore, effects of the RAS in the remodelling development can be demonstrated by effectiveness of the RAS blocking treatment on post-infarction remodelling, which was demonstrated in previous studies and trials.8,9
In our study, we have analysed the impact of AGT polymorphism on prediction of LV remodelling. Pathophysiologically, AGT is one of the key components of RAS, playing an important role in the RAS enzymatic cascade. 13 AGT is converted by renin into angiotensin I and further cleaved to angiotensin II by ACE. Accordingly, AGT and AGT gene polymorphism become important determinants of angiotensin II level in both serum and tissues and have crucial effects in the remodelling process after AMI. It is possible that carriers of the particular genotype of AGT M235T gene may have different levels of circulating AGT or different pathophysiological effects on the vessels, tissues, enzymatic reactions or other pathophysiological processes. The results of our study showed associations between the genetic polymorphism of AGT and post-infarct remodelling. Impairment of the echocardiographic parameters such as LV end-systolic and end-diastolic volumes and LVEF at follow-up in carriers of MM genotype suggested a hypothesis of the genetic background on LV remodelling progression after myocardial infarction. In previous studies, the impact of T allele of AGT M235T on the etiopathogenesis of cardiovascular diseases has been much discussed. However, data from the different studies revealed some disagreement. In several meta-analyses, the TT genotype was associated with increased risk for hypertension;12,13,26 in a few studies an association between AGT gene polymorphism and LV remodelling and hypertrophy in healthy subjects, mainly athletes, 27 dialysis patients 28 and hypertensive patients was demonstrated. 29 Although some authors have found the T allele to be a risk factor for MI or severity of coronary heart disease,11,30,31 others found no association. 7 Sekuri et al. showed that the risk of premature coronary heart disease in subjects with the AGT MM genotype was significantly higher when compared with individuals not carrying the M allele. 32 Similar results were published by Fernández-Arcás et al., where M allele was reported as an independent risk factor for MI. 33 In the analysis of our results of genotype distribution between the LV remodelling group and non-LV remodelling group, MM genotype demonstrated predominance in the LV remodelling group, suggesting that patients with MM genotype have a higher susceptibility to LV remodelling after AMI. Furthermore, the echocardiographic parameters of the group with MM genotype also demonstrated the dilatation of LV and impairment of systolic function of LV at follow-up. Interestingly, combined analysis of the general population and AMI patients’ genotypes demonstrated that prevalence of MM genotype is the same in the overall population and patients with LV remodelling, and T allele is significantly rare in patients with LV remodelling; T allele is much more common in patients without LV remodelling. This suggests that T allele may have a protective role in LV remodelling etiopathogenesis and unfavourable LV remodelling is seen less in carriers of T allele. Regarding our data, at least in the patient population with AMI the possibility of M235T polymorphism of AGT as an additional risk factor for the occurrence of acute coronary syndromes could not be excluded.
The importance of localization of the infarct and the increase of the inflammatory markers at the onset of AMI on the LV remodelling development have been described in our previous study 18 and are supported by some other authors.34,35 The present study also revealed a predictive value of anterior wall infarction and increased count of leucocytes while troponin I and C-reactive protein did not reveal statistical significance. Anterior localization of AMI contributing to the larger size of infarction involving apical segments of the LV has been demonstrated to be a significant predictor of worse outcome after AMI.4,35,36 Despite the increase in white blood cell count, C-reactive protein, cardiac necrosis markers, which have been demonstrated as predictors of ventricular remodelling, and also the other serum markers such as BNP, tenascin, and homocysteine have been demonstrated to have a role in post-infarct remodelling,34 -38 although these markers have not been evaluated in our study.
The early initial evaluation of systolic function of the LV is very important in post-infarct LV dysfunction assessment and is a marker in the further prognosis statement after AMI. 39 Furthermore, the diagnosis of ventricular remodelling is mainly based on the echocardiographic evaluation at follow-up. Our results revealed that initial decrease of LVEF and enlarged end-systolic volume may predict LV remodelling. A novel echocardiographic parameter of myocardial deformation such as longitudinal strain of the LV (assessed by speckle-tracking imaging) may be suggested as an independent echocardiographic predictor for LV remodelling in regard to our results. Lower values of the ventricular longitudinal strain when measured at an acute episode of MI have been demonstrated to be a prognostic marker for patients surviving AMI. The diagnostic reliability of longitudinal strain is still under evaluation and it has been reported as a novel sensitive diagnostic echocardiographic parameter in some studies, while showing no exceptional importance in the others.18,19,39,40
Limitations
The main limitation of our study is the modest number of patients in a few genotype groups, but despite that, statistical analysis showed significant differences in the variables we have analysed and allowed us to suggest a few predictors of LV remodelling including AGT genotype. To measure the impact of genetic factors in such a multifactorial disease as AMI, in which many risk factors and heterogeneous mechanisms are implicated, continuing studies are needed.
Conclusions
Results of our study revealed that predominance of the anterior wall infarction, increased white blood cell count at admission, reduced longitudinal strain of left ventricular and polymorphism of AGT M235T may be presented as predictors of left ventricular remodelling after myocardial infarction.
Footnotes
Conflict of interest
None declared.
Funding
This work was supported by the Lithuanian Science Council (grant number LIG-10009). The HAPIEE study was funded by the Wellcome Trust (grant number 064947/Z/01/Z), the US National Institute on Aging (grant number IR0I AG23522-01) and the MacArthur Foundation (Health and Social Upheaval network).
