Abstract
Introduction:
Left ventricular mass (LVM) is a strong predictor of various heart diseases. We examine the association between the G(-6)A AGT, I/D ACE, A1166C AGTR1, T(-344)C CYP 11β2, A538G MR and A10631G REN polymorphisms and LVM and blood pressure in newborn infants.
Material and methods:
The study included 211 healthy newborn infants. Two-dimensional M-mode echocardiography was used to assess LVM between days 3–4 after birth. Polymorphisms were determined by polymerase chain reaction - restriction fragment length polymorphism (PCR-RLFP).
Results:
AGTR1 genotype was significantly associated with neonatal systolic blood pressure (≥90 percentile). LVM indexes (LVMIs) were tested for association with genotypes in multivariate analysis. The carriers of the A allele of the AGT polymorphism had significantly higher LVM/body length (BL) values when compared with newborn infants homozygous for the G allele (padjusted=0.03). The higher LVM/BL values were seen in the carriers of the A alleles of the AGTR1 polymorphism (padjusted=0.046). All examined indexes (LVM/body surface area (BSA), LVM/BL, LVM/bodyweight (BW)) were associated with CYP11B polymorphism. The newborn infants homozygous for the T allele had significantly higher values of LVM/BSA, LVM/BL, and LVB/BW compared to non-TT-homozygous neonates (padjusted=0.003; padjusted=0.003; padjusted=0.004 respectively).
Conclusion:
The AGT, AGTR1, CYP11β polymorphisms are associated with increased LVMIs in newborns. This observation indicates that genetic factors may be modulating LVM at birth.
Introduction
Variation in left ventricular mass (LVM) is a strong predictor of various heart diseases. Left ventricular hypertrophy (LVH) is a major independent risk factor for cardiovascular morbidity and mortality.1,2 LVM variation results from a complex interaction between genetic, environmental, and lifestyle factors. 3 One of these genetic factors is the renin-angiotensin system (RAS), which contributes not only to blood pressure regulation, renal hemodynamics, fluid and electrolyte homeostasis, but also particularly influences the development of the heart and cardiovascular system through effects mediated by circulating angiotensin II (Ang II) as well as by local activation of the RAS in the myocardium. 4 The presence of common functional polymorphisms in genes encoding components of the RAS can enhance RAS activation and/or receptor function. 4 In previous studies in adults, the G(-6)A AGT, I/D ACE and A1166C AGTR1 gene polymorphisms have been associated with LVM. 5 However, other studies have failed to demonstrate a consistent association between the RAS genotype and LVM. 6 LVM is a continuous trait influenced by interaction between genetic, environmental and lifestyle factors. There is a lack of research concerning the factors influencing heart development during embryogenesis, when external environmental factors have not yet had a marked impact. In addition, despite the fact that healthy newborns born at full term seem to be well suited for such studies due to exclusion of confounding environmental influences such as diet, lifestyle, smoking or diseases, 7 there are no reports which have focused on the association of RAS functional polymorphisms with LVM in this population group. Changes in LVM may be an important diagnostic or prognostic finding in children but there are no full data on normal LVM from echocardiography in children or newborns. Therefore the aims of the present study were to identify some possibilities for the genetic basis of LVM and left ventricular mass indexes (LVMIs) that excluded the influences of environments (lifestyle, diet, disease). It has been proposed that the RAS has a pivotal role in a fetal development and growth which is contained in the placentas shown by the identification of receptors in trophoblast. 8 Studies have shown that the RAS is connected with the modulation of cardiac growth as shown in a group of healthy adults, where it was proven that LVM is positively associated with the level of a potential stimulator of cardiac growth, plasma Ang II and its precursor angiotensinogen.9,10 For this reason we hypothesize that RAS polymorphisms are possibly some of the gene-candidates, which are involved in modulating LVM in newborns. We therefore analyzed the relation between RAS gene polymorphisms: G(-6)A AGT, I/D ACE, A1166C AGTR1, T(-344)C CYP 11β2, A538G MR and A10631G REN with LVM, LVMIs, and additionally potential correlates of LVM, systolic blood pressure (SBP) and diastolic blood pressure (DBP) in a cohort of healthy, full-term newborns.
Material and methods
Study subjects
The study was approved by the Pomeranian Medical University Ethics committee. The population in this study included 211 healthy aboriginal-European newborns born after the end of the 37th week of gestation (gestational age from 37– 40 weeks), recruited at the Department of Neonatology, Pomeranian Medical University in Szczecin, Poland. The mothers in this study were healthy without any complications such as hypertension, pre-eclampsia or eclampsia, fetal growth restriction and the scientists identifying the RAS genotypes were blinded to the clinical characteristics of subjects. Infants in this study were appropriately grown for their gestational age (defined as birthweight above 10th centile). Exclusion criteria were: twins, diabetes, intra-uterine growth restriction, chromosomal aberrations and/or congenital malformations, or ‘small for gestational age’ i.e. below the 10th centile body length (BL), birth weight (BW) or head circumference. At birth, cord blood (500 μL) of neonates was obtained for isolation of genomic DNA. The gender of the newborn, BL, BW and head circumference were taken from standard hospital records. Body surface area (BSA) was calculated as the square root of (BL (cm) x BW (kg)/3600) according to Mosteller. 11
Blood pressure measurements
A Diascope oscillometer (Artema) was used to determine SBP and DBP, and one of the investigators (BŁoniewska) performed all of the blood pressure (BP) measurements using a standardized protocol. The smallest cuff size that covered at least two-thirds of the right upper arm and encompassed the entire arm was selected. BPs were measured in the supine position on the third day after delivery. Measurements were taken at least one and a half hours following their last feeding or medical intervention. An appropriately sized cuff was applied to the right upper arm and the newborn was then left undisturbed for at least 15 minutes or until the newborn was sleeping or in a quiet awake state. Three successive BP recordings were taken at three-minute intervals.
Echocardiographic measurements
Echocardiographic measurements in neonates on the third day after delivery were made by one pediatric cardiologist. Two-dimensional M-mode echocardiography was performed using Acuson Sequoia 512 unit (USA) equipped with a 2–4 MHz imaging transducer. Measurement techniques were consistent with American Society of Echo-cardiography conventions. 12 LVMs were calculated from echocardiographic left ventricular dimension measurements using the Penn convention with equation modified by Huwez et al. 13 as follows: LVM=1.04((IVST+LVPWT+ LVID) 3 -LVID 3 ) where IVST, LVPWT and LVID denote interventricular septal thickness, left ventricular posterior wall thickness and left ventricular internal dimension, respectively. LVM was indexed for body length, body weight and body surface area.
Genotyping
Genomic DNA from cord blood was isolated with the QIAamp Blood DNA Mini Kit (QIAGEN, Germany). The I and D alleles were identified on the basis of polymerase chain reaction (PCR) amplification from intron 16 of the ACE gene according to the method introduced by Lindpaintner et al. 6
DNA fragments that contained the G(-6)A AGT, A1166C AGTR1, T(-344)C CYP 11β2, A538G MR and A10631G REN polymorphism were amplified by PCR by using pairs of primers (Table 1). The PCR product of G(-6)A AGT (193 bp) was digested with the restriction enzyme Mva I, for the (-6)G allele restriction fragment 110 bp, 83 bp, for the (-6)A of 83, 55 and 55 bp were obtained. The PCR product of A1166C AGTR1 (201 bp) was digested with the restriction enzyme Hae III, for the 1166A allele the fragment remained undigested, while the 1166C allele was digested into 171bp and 30 bp restriction fragments. The PCR product of T(-344)C CYP 11β2 (153 bp) was digested with the restriction enzyme Hae III, for the (-344)T allele the fragment remained undigested, while the (-344)T allele was digested into 97 and 56 bp restriction fragments. The PCR product of A538G MR (460 bp) was digested with the restriction enzyme Nco I, for the 538A allele restriction fragments 229 bp, 209 bp, 26 bp for the 538G of 251 bp and 209 bp fragments were obtained. The PCR product of A10631G REN (250 bp) was digested with the restriction enzyme Mbo I, for the 10631G allele the fragment remained undigested, while the 10631A allele was digested into 170 bp and 80 bp restriction fragments. The obtained amplicons and restriction fragments were electrophoretically separated and visualized in ethidium bromide-stained 3% agarose gels. Results were recorded with photographs of gels under UV light.
Characteristics of the primers.

Statistical analysis
The divergences of RAS genotype frequencies from Hardy-Weinberg equilibrium were assessed using a χ2 test. The distribution of each quantitative variable was tested for skewness. The quantitative (presented as means±SD) and categorical variables were analyzed either by Student’s t-test/one-way analysis of variance (ANOVA) or χ2/Fisher’s exact tests, respectively. The Pearson’s correlation test was used to analyze LVM or LVMIs in relation to SBP and DBP. The SBP, DBP, LVM and LVMIs in regard to dominant and recessive mode of inheritance of the minor allele (D ACE, G AGT or C AGTR1, C CYP 11β2, G MR and G REN, respectively) When comparing the LVMIs between groups, p values were adjusted in an analysis of covariance (ANCOVA). Statistical significance was defined as p<0.05. All data were analyzed with STATISTICA (version 8.0, StatSoft, Inc., USA, 2007, www.statsoft.com).
Results
Characteristics of the newborn cohort (n=211) are shown in Table 2. The distribution of these characteristics in our cohort approached normality (skewness<2 for all variables). Mean BL, BW and BSA values in boy newborn infants were significantly higher as compared to girls. There were no significant differences in RAS genotype. The ACE, AGT, AGTR1, MR, REN, CYP 11β2-genotype distributions conformed to the expected Hardy-Weinberg equilibria (Table 3).
Clinical and echocardiographic characteristics of the newborns in regard to gender.

BL: body length; BSA: body surface area; BW: birth weight; DBP: diastolic blood pressure; IVS: intraventricular septum; LVPW: left ventricular posterior wall; LVD: ; LVM : left ventricular mass; LVV: left ventricular volume; MAP: mean arterial pressure; SBP: systolic blood pressure.
adjusted for SBP, DPB.
Genotype and allele frequencies.

There were no significant differences in the values of LVM and LVMIs in regard to ACE, AGT, AGTR1, MR, REN, CYP 11β2 genotypes assessed by one-way ANOVA. The SBP presented positive but weak correlations with LVM (r=0.121, p=0.079), LVM/BL (r=0.142, p=0.040), LVM/BW (r=0.082, p=0.232), LVM/BSA (r=0.114, p=0.098), LVM/BL1.506 (r=0.153, p=0.026), LVM/BW0.669 (r=0.094, p=0.171), and LVM/BSA1.038 (r=0.112, p=0.105) and the correlations for DBP were: LVM (r=0.155, p=0.024), LVM/BL (r=0.142, p=0.040), LVM/BSA (r=0.157, p=0.022), LVM/BW (r=0.154, p=0.025), LVM/BL1.506 (r=0.149, p=0.031), LVM/BW0.669 (r=0.153, p=0.021), and LVM/BSA1.038 (r=0.156, p=0.024). There were no significant differences in values of SBP and DBP or LVM and LVMIs assessed by multiple regression analysis for dominant and recessive models of inheritance of the minor allele with adjustment for gender (for SBP and DBP analysis) or gender, SBP and DBP (for LVM and LVMIs) (data not shown).
ACE genotype in the newborns showed a significant correlation with birth weight, neonatal body length and neonatal head circumference. AGTR1 and AGT genotype were significantly associated with neonatal SBP (≥90 percentile) and MAP (≥90 percentile), respectively. Additionally, AGTR1 was significantly correlated with maternal history of hypertension. MR, REN and CYP11B showed no association with any of neonatal and maternal parameters (Tables 4 and 5).
Overview of results depending on fetal genotypes: observation data of the study population according to ACE, AGTR1 and AGT genotype.
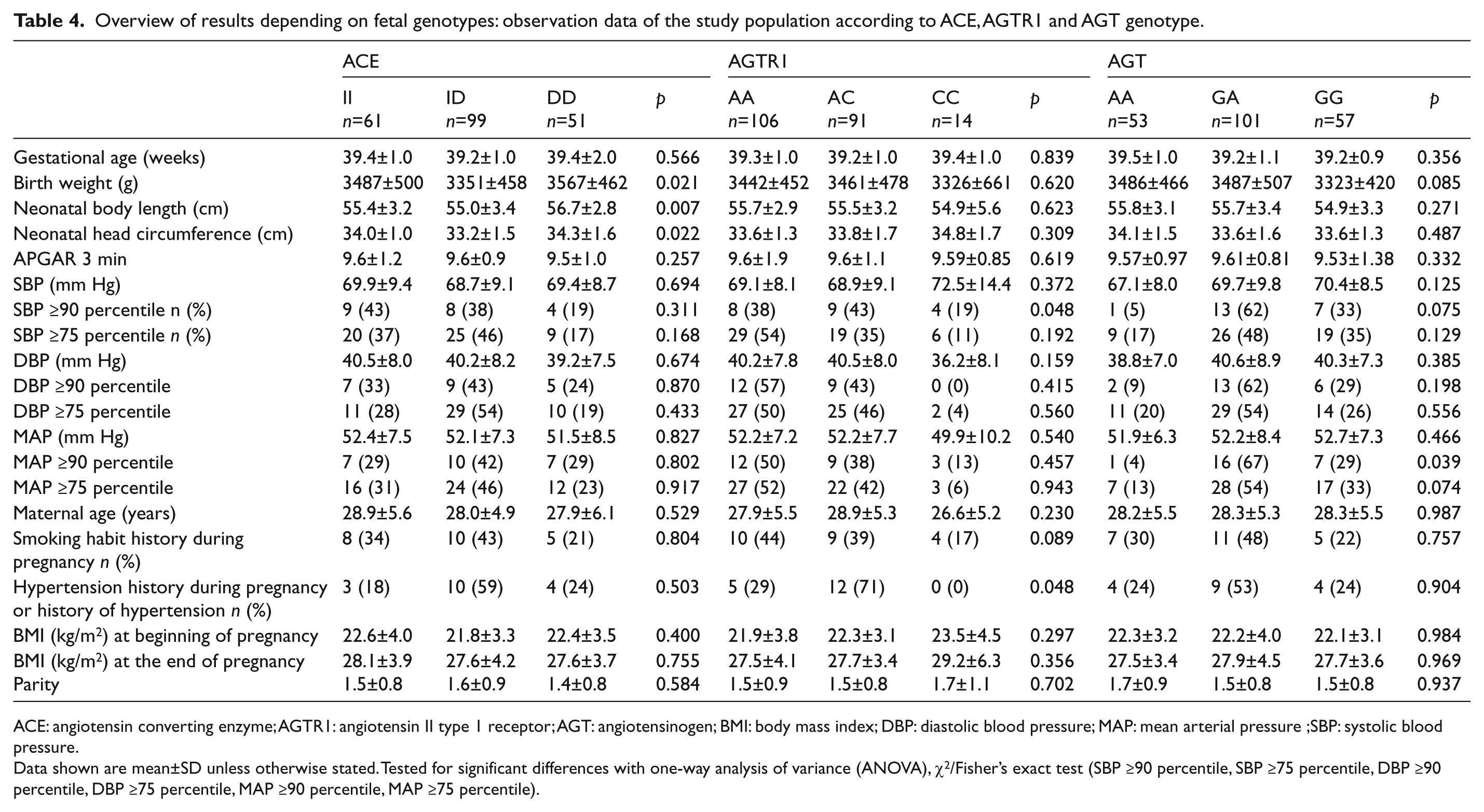
ACE: angiotensin converting enzyme; AGTR1: angiotensin II type 1 receptor; AGT: angiotensinogen; BMI: body mass index; DBP: diastolic blood pressure; MAP: mean arterial pressure;SBP: systolic blood pressure.
Data shown are mean±SD unless otherwise stated. Tested for significant differences with one-way analysis of variance (ANOVA), χ2/Fisher’s exact test (SBP ≥90 percentile, SBP ≥75 percentile, DBP ≥90 percentile, DBP ≥75 percentile, MAP ≥90 percentile, MAP ≥75 percentile).
Overview of results depending on fetal genotype: observation data of the study population according to MR, REN and CYP11B genotype.

BMI: body mass index; CYP11B: aldosterone synthase.DBP: diastolic blood pressure; MAP: mean arterial pressure; MR: mineralocorticoid receptor; REN: renin; SBP: systolic blood pressure.
Data shown are mean±SD unless otherwise stated. Tested for significant differences with one-way analysis of variance (ANOVA) or Student’s t-test (MR), χ2/ Fisher’s exact test (SBP ≥90 percentile, SBP ≥75 percentile, DBP ≥90 percentile, DBP ≥75 percentile, MAP ≥90 percentile, MAP ≥75 percentile).
LVMIs were tested for association with genotypes in multivariate analysis (ANCOVA) in order to adjust for possible confounding factors. After adjusting for neonatal (gestational age, gender, APGAR at three minutes) and maternal (age, BMI at beginning of pregnancy, BMI at the beginning and end of pregnancy, smoking status and hypertension status) parameters, we revealed a significant association between LVMIs (LVM/BSA, LVM/BL and LVM/BW) and the three polymorphisms genotypes (AGT, AGTR1, CYP11B). The carriers of the A allele of the AGT polymorphism had significantly higher LVM/BL values when compared with newborns homozygous for the G allele (18.6 g/m versus 15.8 g/m, padjusted=0.03) (Figure 1). Likewise, the higher LVM/BL values were seen in the carriers of the A alleles of the AGTR1 polymorphism (18.4 g/m versus 14.7 g/m, padjusted=0.046) (Figure 2). All examined indexes (LVM/BSA, LVM/BL, LVM/BW) were associated with CYP11B polymorphism (Figure 3). The newborns homozygous for the T allele had significantly higher values of LVM/BSA, LVM/BL, LVB/BW compared to non-TT-homozygous neonates (51.2 versus 42.3, padjusted=0.003; 21.4 versus 17.3, padjusted=0.003; 3.5 versus 2.8, padjusted=0.004, respectively).

Left ventricular mass (LVM) indexes according to AGT genotype.BL: body length; BSA: body surface area; BW: birthweight. Mean and standard error of the mean (SEM) are shown. a: p=0.055; b: p=0.030; c: p=0.106; a, b, c: AA+GA versus GG.
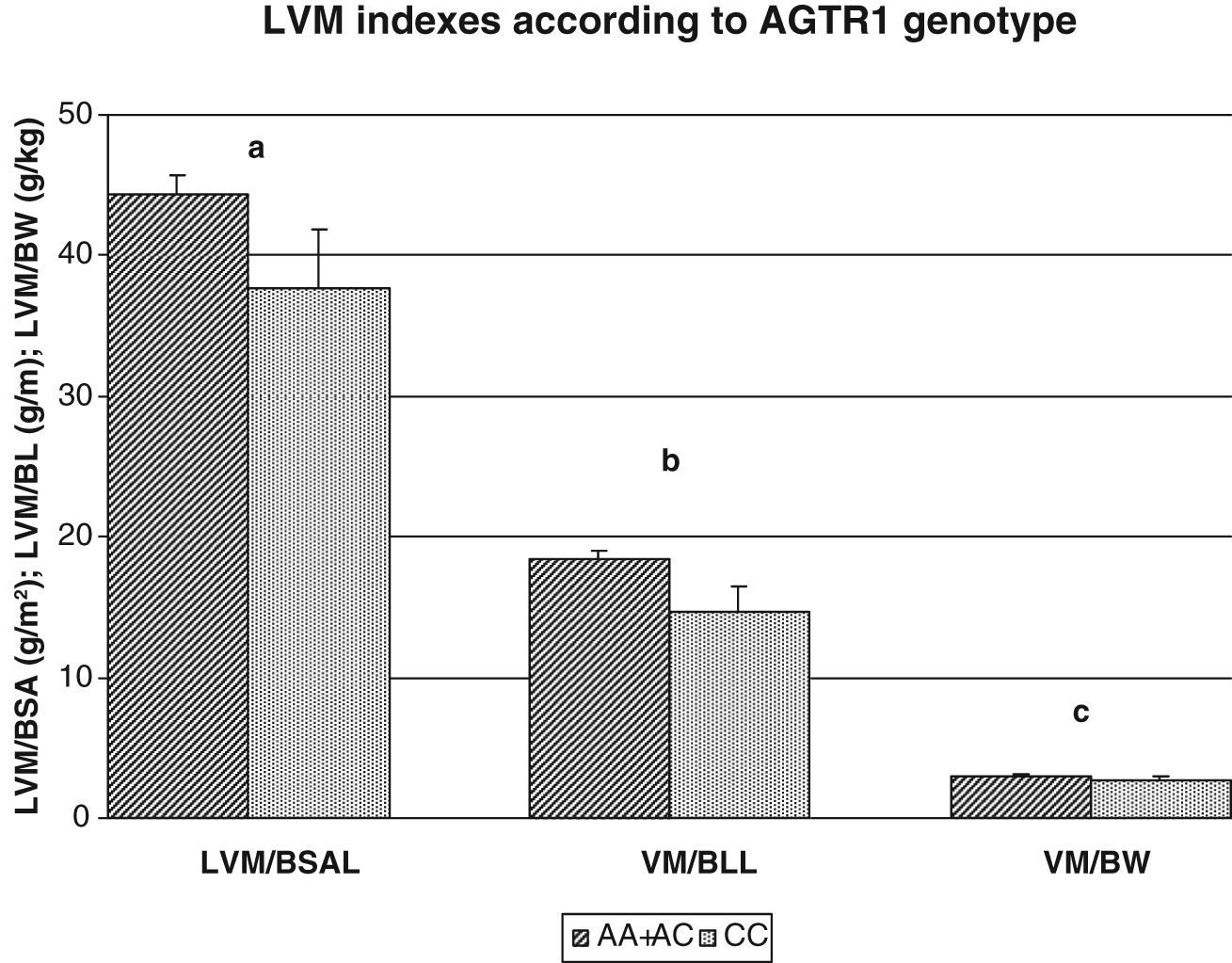
Left ventricular mass (LVM) indexes according to AGTR1 genotype.

Left ventricular mass (LVM) indexes according to CYP11B genotype.
Discussion
To our knowledge this is the first cohort study that has examined the association of RAS genes and LVM in newborns.
In this study we estimated BP and the parameters of LVM in newborns. We assessed the interaction between I/D ACE, G(-6)A AGT, A1166C AGTR1, A538G MR, A10631G REN and T(-344)C CYP 11β2 polymorphisms and blood pressure (SBP, DBP, MAP) in newborns. We found no significant interactions between blood pressure (SBP, DBP, MAP) and the RAS gene; and only between SBP ≥90 percentile and AGTR1 gene polymorphism was a significant association (p=0,048) observed. In our study the LVM was determined by routine M-mode echocardiography. These data are updates for a newborn infant group because previously published normative values collected in a small pediatric sample were acquired for subjects of different ages (0–18 years).14,15 In other studies data obtained by Vogel et al. 16 in a small study showed a higher LVM/m2 in newborns, however Kozak-Barany et al. found a lower LVM/BSA in a small group of healthy newborns. 17 This discrepancy could result from different techniques of assessing left ventricular parameters and the differences between populations. It should also be noted that echocardiography methods are still developing which allow high-precision testing and more precise measurement of cardiac dimensions. 18 However, M-mode echocardiography is one of the most commonly performed routine tests. It is also important that the echocardiography examination in our group was conducted by one cardiologist. Also, the problem of proper assessment of the size of the LVM may be very important which has caused so much controversy especially in newborns. The predominant view suggests that the most objective parameter for LVM evaluation in children is BSA. However, new methods of determining the LVM in relation to (BW, BL and BSA of the newborn are reported to be more appropriate. 19 That is why we have used all of the suggested methods to accurately transform and index the determined LVM. The original hypothesis of our study was that RAS polymorphism may influence the LVM in newborns. We have found a significant association of AGT, AGTR1, CYP11β and increases in LVMIs. This finding is in accordance with data obtained in adults.20–22 For -6A allele AGT we observed significant increase in LVM/BL and borderline LVM/BSA. Also 1166A allele of AGTR1 is significantly associated with increases in LVM/BL, while T-344 allele CYP11β is associated with increase in LVM/BSA, LVM/BL, LVM/BW. Foster et al. observed that the assessment of LVM with height is a better method of normalizing LVM for body size in children than LVMI. 15 A few authors have observed that height as the indexing variable has a number of attractive aspects: The correlation of height with LVM is close to that of BSA with LVM; the use of height is also less ‘forgiving’ of obesity, which may be associated with an increase in LVM. 23 Although The Bogulusa Heart Study demonstrates that BMI measured in childhood (4–17 years) and BMI and SBP measured in adulthood (20–38 years) as cumulative burden since childhood are independent predictors of LVMI in young adulthood, also a previous study showed a cross-sectional association between LVM and BMI in children and adolescence. 24 In the current study, we detected positive but weak correlation between SB and LVM/BL or trend with LVM, also positive correlation between DBP with LVM, LVM/BL, LVM/BW and LVM/BSA.
Ang II and its precursor angiotensinogen are thought to play a central role not only in etiology to hypertension but also in the pathophysiology of cardiac hypertrophy, remodeling heart failure and atherosclerosis. 25 Ang II binds primarily to AGTR1 to promote cell growth and hypertrophy. In addition Ang II is involved in cardiac remodeling and apoptosis. 26 The RAS may also play an important role in the pathogenesis of cardiovascular disease including left ventricular hypertrophy (LVH) later in life. Previously, studies have indicated the pronounced developmental changes in RAS activity during the first days of life27,28 which may lead to increased Ang II production. The connection between RAS and LVM in adults is well known. The -6A allele AGT in young adults, 1166C allele AGTR1 and -344T allele CYP11β in adults associated with LVH.20–22 However the effect of RAS on fetal development is not clear. Schlemm et al. in a large study, showed that the fetal AGT M235T polymorphism is associated with low birth weight and elevated fetal total glycated hemoglobin at birth. 29 Han et al. observed that the deletion allele of ACE was associated with relatively impaired insulin sensitivity in healthy newborn infants. 7 Nobilis et al. noted association between ACE polymorphism circulatory failure in very-low-birthweight neonates. 30 Also the Multiethnic Youth Study examined the effect of AGT M235T, AGTR1 C-521T, L191L, A1166C polymorphisms on the blood pressure and LVM development from childhood into early adulthood in a group of healthy African Americans and European Americans. In this study significant interaction were found between the M235T AGT and ethnicity, gender and BP levels, also haplotype analysis identified a protective haplotype (C-521, 191L, A1166) AGTR1 for LVM levels. 31 The CYP11β gene encodes a key enzyme of aldosterone biosynthesis, aldosterone synthase. It was shown that genetic variation in the CYP11β affects left ventricular size and mass in young adults free of clinical heart disease and that this polymorphism may also affect response of the left ventricle to increases in dietary salt.32,33 However, the association of the -344T variant with cardiac hypertrophy in adults remains controversial, other studies have also concluded that it is associated with higher values of LVM in young adults,33,34 but some other reports found no association between LVM and CYP11B variant. 35 The meta-analysis demonstrated that a higher LVM in TT homozygous was only observed in the in hypertension subject and not in normotensive individuals. 22 In our study we found the correlation between CYP11β and LVM/BL, LVM/BW and LVM/BSA. The mechanisms underlying these findings have yet to be clarified. Also very important is the fact that changes in left ventricular size and structures of the heart can develop with age, even without the influence of risk factors. 1 Healthy newborn infants at the beginning of their life are not burdened by the influence of external factors which could cause excessive activation of the RAS, maternal factors may be partially related in fetal development, and therefore lead to an excessive increase in LVM. So far, the relationship between RAS polymorphisms, LVM and blood pressure are controversial. More studies have been reported, mainly in adults whose life style and habits are important determinants, which could show a relationship with strong genetic components.36,37 Moreover,one study does not support this correlation. 38 Genetic factors may have a weaker potency, and their impact is part of a combined operation of genetic and environmental factors that have an influence on LVM during a human’s life. It is known that the RAS system is a plausible candidate for the modification of LVM.39,40 In fetal life the tissues and organs of the body go through critical periods of development 41 and these critical periods may coincide with periods of rapid cell division: a stimulus or insult at the critical period of development has lasting or lifelong effect.42,43 Therefore, we have hypothesized that newborns, in whom the impact of RAS is observed in such an early stage of life, may have a greater predisposition to LVH development during their life.
However, our study has several limitations. It is the first report demonstrating the genetic association of the RAS polymorphism with development of LVM in newborn infants, but there is still a lack of studies which could show this association in other countries. Additionally this study involved a relatively small sample size, and only healthy newborns were included, the observations need to be followed-up with much larger sample sizes to make any causal inference.
Conclusions
We showed that the AGTR1 and AGT genotype were significantly associated with neonatal SBP (≥90 percentile) and MAP (≥90 percentile) respectively. We revealed a significant association between LVMIs (LVM/BSA, LVM/BL and LVM/BW) and the three polymorphism genotypes (AGT, AGTR1, CYP11B). These findings support the hypothesis that RAS gene polymorphisms might modulate LVM at birth. We believe that our results will be a small step to a better understanding of cardiac structures and will lead to further exploration of genetic factors that modulate them. Follow-up studies are needed to examine whether these differences persist or change in later life.
Footnotes
Conflict of interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
