Abstract
Polycyclic aromatic hydrocarbons (PAHs) are believed to be abundant in the dense regions of the interstellar medium (ISM) and are hypothesized to play a role in the origin of diffuse interstellar bands (DIBs). Among these, corannulene C₂₀H₁₀ stands out due to its unique bowl-shaped structure, which not only makes it an intriguing candidate for understanding interstellar chemistry but also holds potential applications in nanotechnology and materials science. To provide low-temperature laboratory benchmarks for PAH–H₂/D₂ interactions, we employed high-resolution mass spectrometry to investigate H₂/D₂ tagging of helium-doped C₂₀H₁₀ oligomers. We emphasize that the large solvation numbers accessible in helium droplets represent a model cryogenic environment and should not be interpreted as a direct prediction for the low-density ISM. H₂ tagging changes the system's stability and chemical properties, providing important information for chemistry studies. A notable peak for corannulene-monomer at 32-H₂ molecules indicates the formation of an ordered solvation layer. Theoretical studies determine that adsorption of hydrogen and deuterium on C₂₀H₁₀ clusters is enhanced by cluster size, due to enhanced bonding and electron changes such as the Highest Occupied Molecular Orbital HOMO- Lowest Unoccupied Molecular Orbital LUMO gap shrinkage. Deuterium is found with tighter bindings and smaller dipole moments, as a result of its smaller zero-point energy, with certain clusters exhibiting exceptionally robust ‘magic number’ structures.
This is a visual representation of the abstract.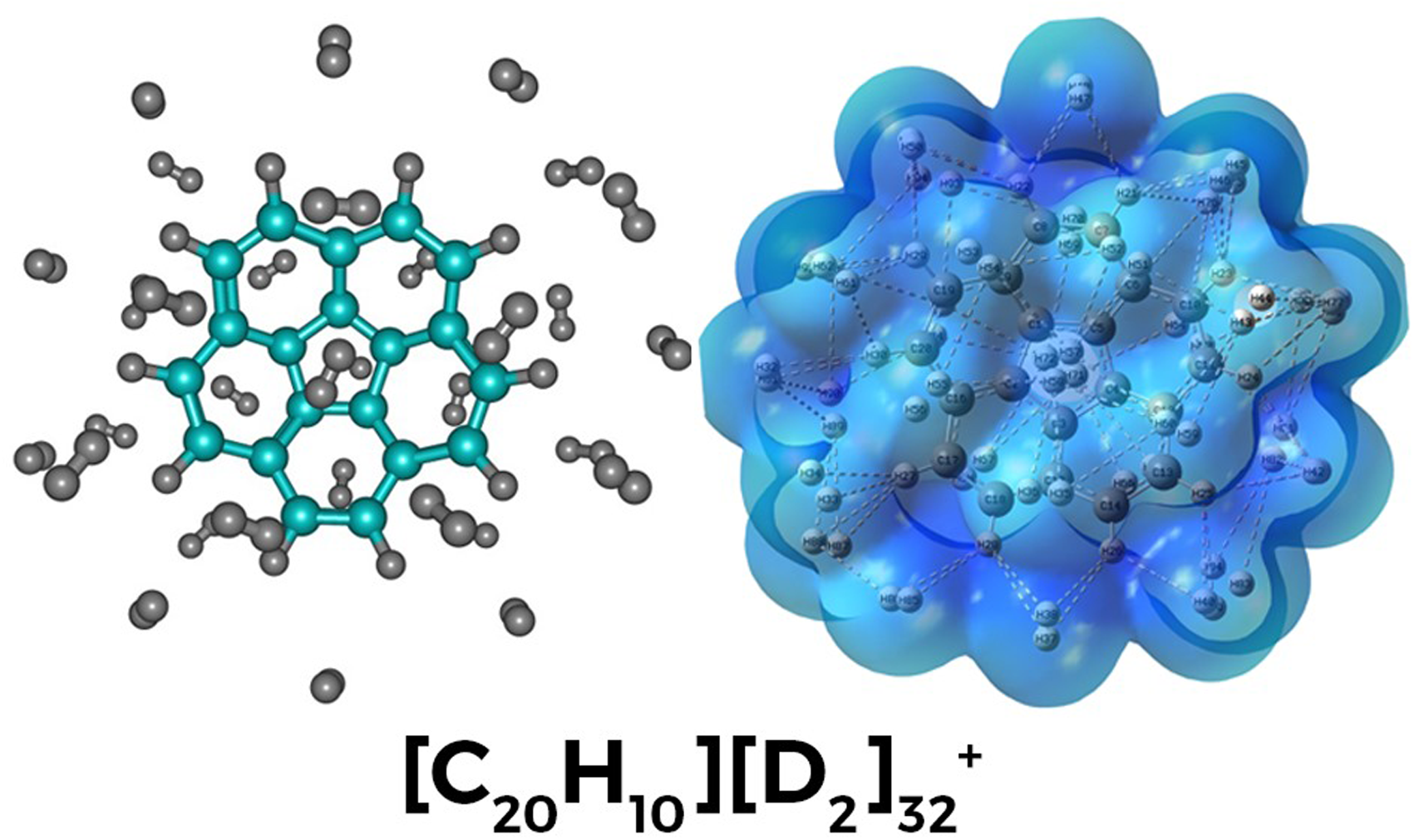
Keywords
Get full access to this article
View all access options for this article.
