Abstract
To further understand the complexation and fragmentation during the extraction process, the formation of 2,9-bis(5,5,8,8-tetramethyl-5,6,7,8-tetrahydro-12,4-benzotriazin-3-yl)-1,10-phenanthroline (CyMe4-BTPhen) complexes with lanthanides (Ln = La, Ce, Nd, Sm, Eu, Yb) and actinides (UO22+, Th4+) was observed by electrospray ionization mass spectrometry (ESI-MS) technique and density functional theory (DFT) calculations. Mass spectrometry titrations showed the variation relationship of different complexes in acetonitrile. For lanthanides, the major complexes were 1:2 species ([Ln(L)2]3+ and [Ln(L)2(NO3)]2+) with a ratio of 1:2, which were observed at the initial addition of Ln3+, whereas the species ([Ln(L)(NO3)2]+) with a ratio of 1:1 was detected when the [Ln]/[L] concentration ratio reached 1.0. For UO22+ and Th4+ complexes, 1:1 or 1:2 species ([UO2L(NO3)]+, Th(L)2(NO3)3+ and Th(L)2(NO3)22+) were formed. The fragmentation chemistry of both the ligand and the complex cations was characterized in detail by collision-induced dissociation. The fragmentation process of CyMe4-BTPhen was unfolded sequentially on both sides of the ligand by cleavage of C–C and C–N bonds. DFT calculations provided a detailed analysis of the structures and thermodynamics of those complexes, which indicated that the stable complexes formed in acetonitrile solution were consistent with the ESI-MS results.
Introduction
The reprocessing of spent nuclear fuel (SNF) aims to separate and recover uranium and plutonium, in order to maximize the utilization of resources and minimize the environmental impacts. 1 However, the long-lived minor actinides and fission products are still left in the high-level liquid waste (HLLW). For the treatment of the HLLW, researchers proposed the “Partitioning and Transmutation” strategy in the 1970s to minimize the radiotoxicity,2,3 which is to separate long-lived radionuclides from the HLLW and then convert them into short-lived or stable nuclides.4,5 Since the neutron-capturing cross sections of the lanthanides are bigger than those of actinides, it is necessary to separate actinides and lanthanides before transmutation.
Considering the similarities of the chemical properties of two series of elements, the highly efficient ligands for the separation are very desirable. In recent studies, heterocyclic ligands with soft N-donors based on triazinyl-pyridine have already shown the efficient extraction potential application value to separate the minor actinides from the lanthanides in recent years, 6 such as 6,6′-bis-(5,5,8,8-tetramethyl-5,6,7,8-tetrahydro-12,4-benzotriazin-3-yl)-2,2′-bipyridine (CyMe4-BTBP) and 2,9-bis(5,5,8,8-tetramethyl-5,6,7,8-tetrahy-dro-12,4-benzotriazin-3-yl)-1,10-phenanthroline7–10 (CyMe4-BTPhen) (Figure 1). As their structures only contain C, H, O, and N elements, which conform to the CHON principle and are conducive to waste management. Therefore, the research and development of N-donor ligands have received growing attention. As a triazinyl-based N-donor extractant for the separation of actinides and lanthanides, CyMe4-BTPhen exhibits good extraction abilities with relatively low extraction kinetics. For large-scale applications, phase transfer agents are required to improve the extraction kinetics. CyMe4-BTPhen is obtained by introducing the phenanthroline into the ligand structure, exhibiting a cis–cis conformation. The cavity of CyMe4-BTPhen facilitates its chelation coordination with metal ions. In recent studies, the CyMe4-BTPhen ligand was reported to exhibit higher extraction kinetics and separation factors (SFs), Distler et al. 11 found that the equilibrium time of Am3+ is 45 min and 15 min for 1 mmol/L CyMe4-BTBP and CyMe4-BTPhen with a fluorinated carbonate diluent (BK-1), respectively. CyMe4-BTBP also exhibits an SFAm(III)/Eu(III) over 1000. 12 Benay et al. 13 found that the distribution ratio of CyMe4-BTPhen is two orders of magnitude higher than that of CyMe4-BTBP ligand in liquid–liquid extraction. It has also shown better tolerance toward acids. Lewis et al. 16 found that BTPhens can work with a 4.0 mol/L HNO3 solution. 11
Molecular structures of the CyMe4-BTBP and CyMe4-BTPhen.
The coordination mode of the complexes conduces to understanding the coordination chemistry of ligands in the extraction process. In recent studies, multiple techniques, such as electronic absorption spectroscopy, luminescence spectroscopy, nuclear magnetic relaxation dispersion titration, time-resolved laser fluorescence spectroscopy, and single-crystal X-ray diffraction, 11 were used to characterize the complexes in both solid state and liquid state, which proved that the prevalent complex is 1:2 species. By contrast, electrospray ionization mass spectrometry (ESI-MS) enables the determination of complexes quickly, intuitively, and accurately. 14 The characterization of metal–ligand coordination by mass spectrometry titration would provide evidence for the extraction process. Previous reports revealed that the fragmentation by mass spectrometry is similar to the radiolytic degradation to some extent, 15 which could provide a reference for the study of identifying radiation products. To our knowledge, there are no systematic studies of the coordination or the fragmentation chemistry of CyMe4-BTPhen ligand with lanthanides and actinides that have been performed by electrospray mass spectrometry yet.
In this work, we focused on the coordination and fragmentation chemistry of CyMe4-BTPhen complexes with some representative lanthanide cations Ln3+ (Ln = La, Ce, Nd, Sm, Eu, Yb) and actinide cations (UO22+, Th4+). The complexation was studied by mass spectrometry titrations under different metal–ligand concentration ratios, and the fragmentation of ligands and complexes was analyzed by collision-induced dissociation (CID) technology. Then density functional theory (DFT) calculations were used to probe the structural characters and stability of the complexes. It is expected that this work could provide a further understanding of the extraction process and a reference for the radiation products under strong radiation fields.
Experimental
Materials and sample preparation
The CyMe4-BTPhen (M = 558.3 g/moL) ligand was synthesized according to the procedures reported by Lewis et al. 16 Chemical reagents such as La(NO3)3·6H2O, Ce(NO3)3·6H2O, Nd(NO3)3·6H2O, Sm(NO3)3·6H2O, Eu(NO3)3·6H2O, and Yb(NO3)3·5H2O were of analytical grade and purchased from commercial sources. Stock solutions (2 × 10−3 M) of CyMe4-BTPhen ligand and metal nitrate were dissolved in HPLC-grade acetonitrile provided by Thermo Fisher. Solutions containing UO22+ and Th4+ nitrate were diluted by the stock solutions in our laboratory.
Solutions for the mass spectrometry titration were prepared prior to the experiments. Stock metal nitrate solution and stock ligand solution were mixed together in 2.0 mL plastic centrifuge tubes and shaken thoroughly in a shaker. Aliquots of 50 µL were diluted with acetonitrile 10 times, and this step was repeated twice before analysis.
ESI-MS measurements
The mass spectrometric measurements were performed using a triple-quadrupole spectrometer (TSQ Quantum Access, Thermo Scientific, USA). The instrument was equipped with an electrospray ion source (ESI) for measurements, a TSQ software for data acquisition, and an Xcalibur software for data processing. The pressure of high-purity nitrogen gas was set to 120 psi and operated at 10 arb as the sheath gas. The spray voltage was set to 3500 V. The evaporation temperature was set to 275 °C. In full scan positive ion detection mode, MS spectra were acquired at 1 s/scan over a mass range of m/z 50–1000 with the mass width (full width half maximum) of m/z = 0.4. A syringe infusion pump delivering solvent at 5 μL/min was connected to the ESI probe, and a 500 μL glass syringe with a stainless steel needle (Hamilton) was used to inject the sample directly. For MS/MS measurements, the high-purity argon with pressure set to 30 psi was used as the collision gas. MS/MS spectra were acquired by CID in full scan mode. Ten scans were averaged in the data acquisition process. The complex ion peak was simulated by the simulation function from the Xcalibur software and compared with the spectrogram of the laboratory to determine the attribution of the complex ion peak (as shown in Figure 2). The assignment of the charge number was determined by the partially resolved isotopic patterns arising from adjacent isotope peaks. The amount of charge in the spectrum can be determined by the difference in the mass-to-charge ratio of its adjacent isotope peaks. 17
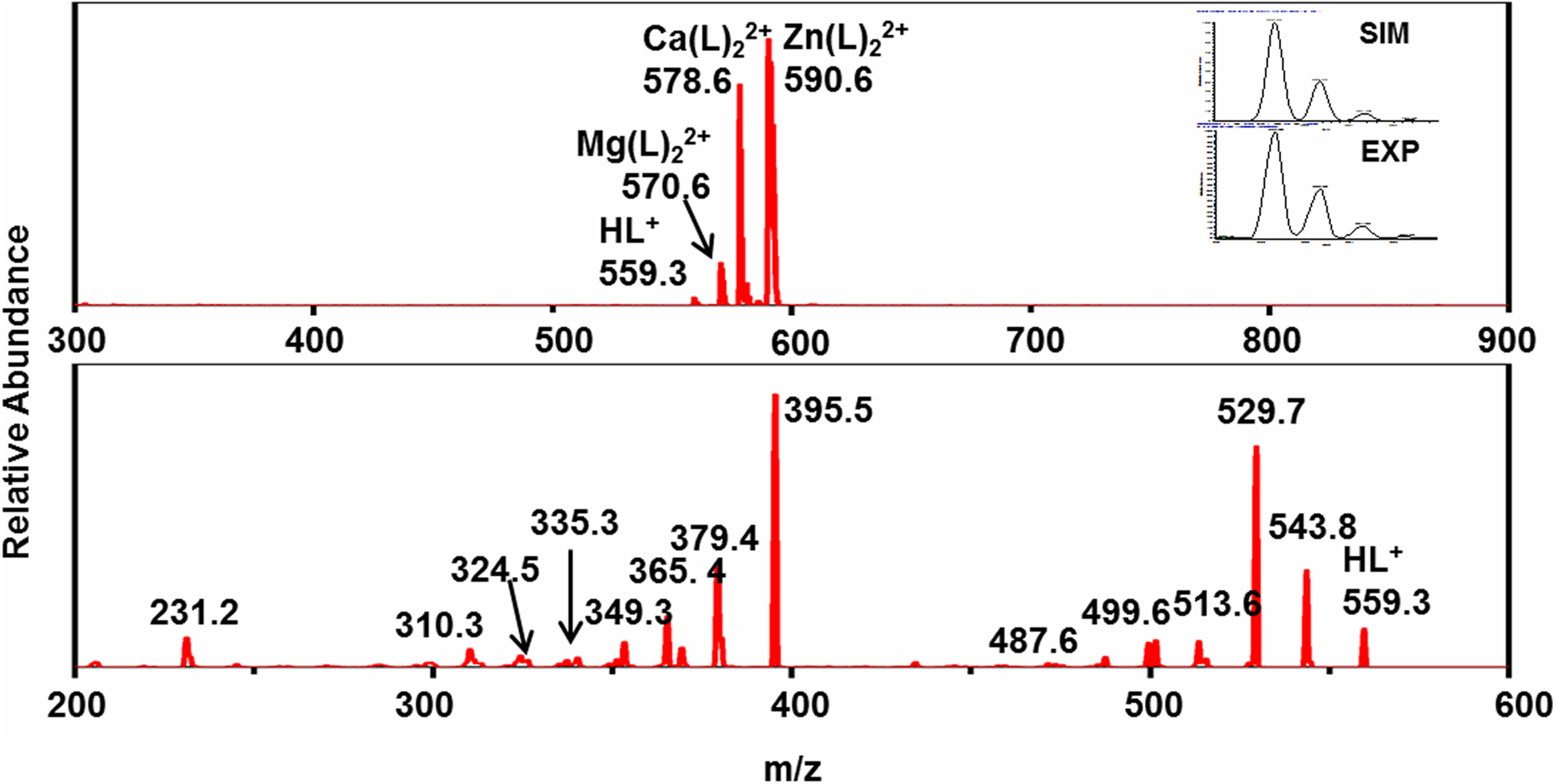
Positive ESI-MS spectrum of CyMe4-BTPhen (top). Inset: comparison between EXP and SIM patterns of [HL]+. MS/MS spectrum of [HL]+ (bottom), where L represents the CyMe4-BTPhen ligand.
Theoretical calculation
The structures of the La3+, Eu3+, Th4+, and UO22+ complexes were fully optimized in the gas phase using the hybrid DFT functional, B3LYP 18 with the Gaussian 16 program package. 19 The relativistic effects were considered by using the quasi-relativistic effective core potentials (quasi-RECPs), which substitute 60 core electrons for the Th and U atoms and 28 core electrons for the La and Eu atoms.20,21 Meanwhile, the corresponding ECP60MWB-SEG valence basis set was used to describe Th and U atoms, whereas the ECP28MWB-SEG valence basis set was for the La and Eu atoms. The 6-31G(d, p) basis sets were adopted for H, C, N, and O atoms. For the La3+, Th4+, and UO22+ complexes, the ground state was considered a singlet state, whereas the sextet spin state was for the Eu3+ complexes. At the B3LYP/6-31G(d, p)/RECP level of theory, vibration frequency analysis was conducted to confirm the true minima of the species.
The natural atomic charges and the Wiberg bond indices (WBIs) were also calculated by the natural bond orbital (NBO 6.0)22,23 analysis at the B3LYP/6-31G(d, p)/RECP level of theories. In order to obtain more accurate energy, thermodynamic calculations were carried out at a higher theoretical level (B3LYP/6-311G(d, p)/RECP). To modify the possible solvent extraction process, the solvation effect was taken into account by the conductor-like screening model (COSMO)24,25 in the acetonitrile solution.
Results and discussion
Coordination chemistry research
Mass spectrometry titrations of lanthanide and actinide nitrate with CyMe4-BTPhen in acetonitrile were performed to identify the complexes, in particular, the composition and the intensity of each complex. An initial experiment was carried out with pure CyMe4-BTPhen ligand in acetonitrile to observe the possible complexes of pure ligands in electrospray mass spectrometry (Figure 2). The results show that the dominant MS peaks can be attributed to the protonated ligand ([HL]+, m/z 559.3) and the dimeric adducts ([Mg(L)2]2+ at m/z 570.6, [Ca(L)2]2+ at m/z 578.6, [Zn(L)2]2+ at m/z 590.6). [Mg(L)2]2+, in which [Ca(L)2]2+ and [Zn(L)2]2+ are the common impurities in ESI-MS experiments. 26 ESI-MS spectra of Ln3+ (Ln = La, Ce, Nd, Sm, Eu, Yb), UO22+, Th4+ with CyMe4-BTPhen ligand in acetonitrile are recorded at different [M]/[L] concentration ratios, as shown in Figures 3 to 5, Supplemental Figures S1 to S5, and Table 1. The results show that basically there is no difference in the coordination mode between different lanthanide ions. Both 1:1 and 1:2 species are observed. Only 1:2 Ln3+/CyMe4-BTPhen species appear with the initial addition of lanthanide ions. Due to the presence of nitrate ions and solvent molecules involved in the coordination, there are three complexes for La3+, Ce3+, and Nd3+, namely Ln(L)23+, Ln(L)2(CH3CN)3+, and Ln(L)2(NO3)2+. There is no Ln(L)2(CH3CN)3+ complex for Sm3+, Eu3+, and Yb3+. With the addition of Ln3+, a small amount of 1:1 species is observed with a concentration ratio of 1.0. The only exception during the experiment is Yb3+, for which the only species is 1:2. All these results indicate that the 1:2 species is dominant in the extraction process, while the 1:1 species is present in low abundance, if at all. Notably, a series of low abundance peaks of lanthanide compounds are attributed to the Ln–L complexes by the simulated isotopic pattern (Supplemental Figure S6), which are speculated to be caused by the loss of methylene groups. The mass spectrometry titration experiment of UO22+ was carried out with the range of metal–ligand concentration ratio from 0 to 2.0. As shown in Figure 4, only 1:1 species is generated, which can be manifested as dipositive ions [UO2(L)]2+, [UO2(L)(CH3CN)]2+, and a small amount of monopositive complex ions [UO2(L)(NO3)]+. The mass spectrometry titration experiments of Th4+ are shown in Figure 5. With the range of metal–ligand concentration ratios growing from 0 to 1.5, the 1:2 complex ions [Th(L)2(NO3)2]2+ and Th(L)2(NO3)]3+ generate at m/z 470.6 and m/z 736.7. The results show that [Th(L)2(NO3)2]2+ has a relatively higher abundance.
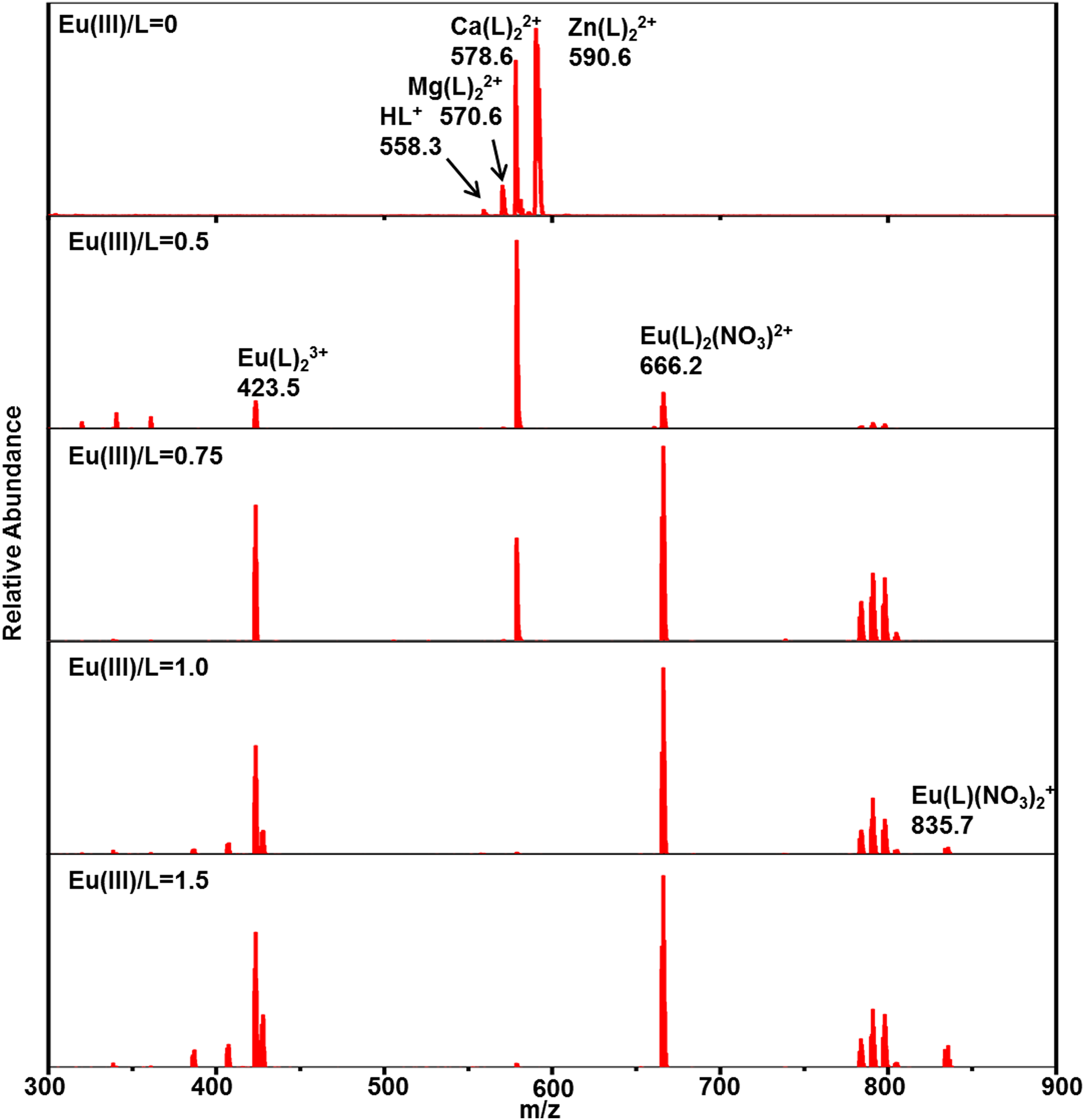
Positive electrospray ionization mass spectrometry (ESI-MS) spectra of the Eu3+ complexes at various [Eu3+]/[L] ratios.
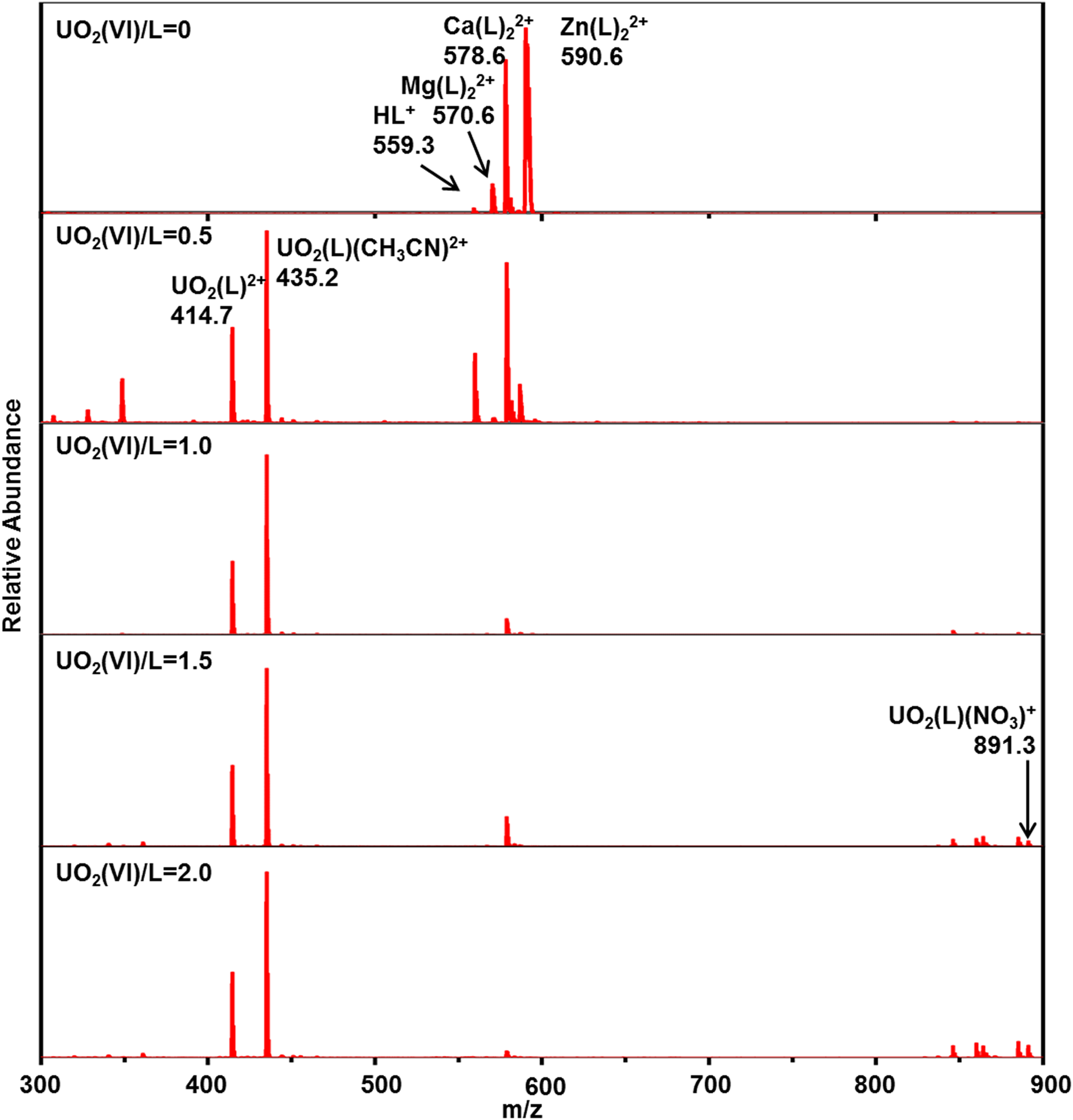
Positive electrospray ionization mass spectrometry (ESI-MS) spectra of the UO22+ complexes at various [UO22+]/[L] ratios.
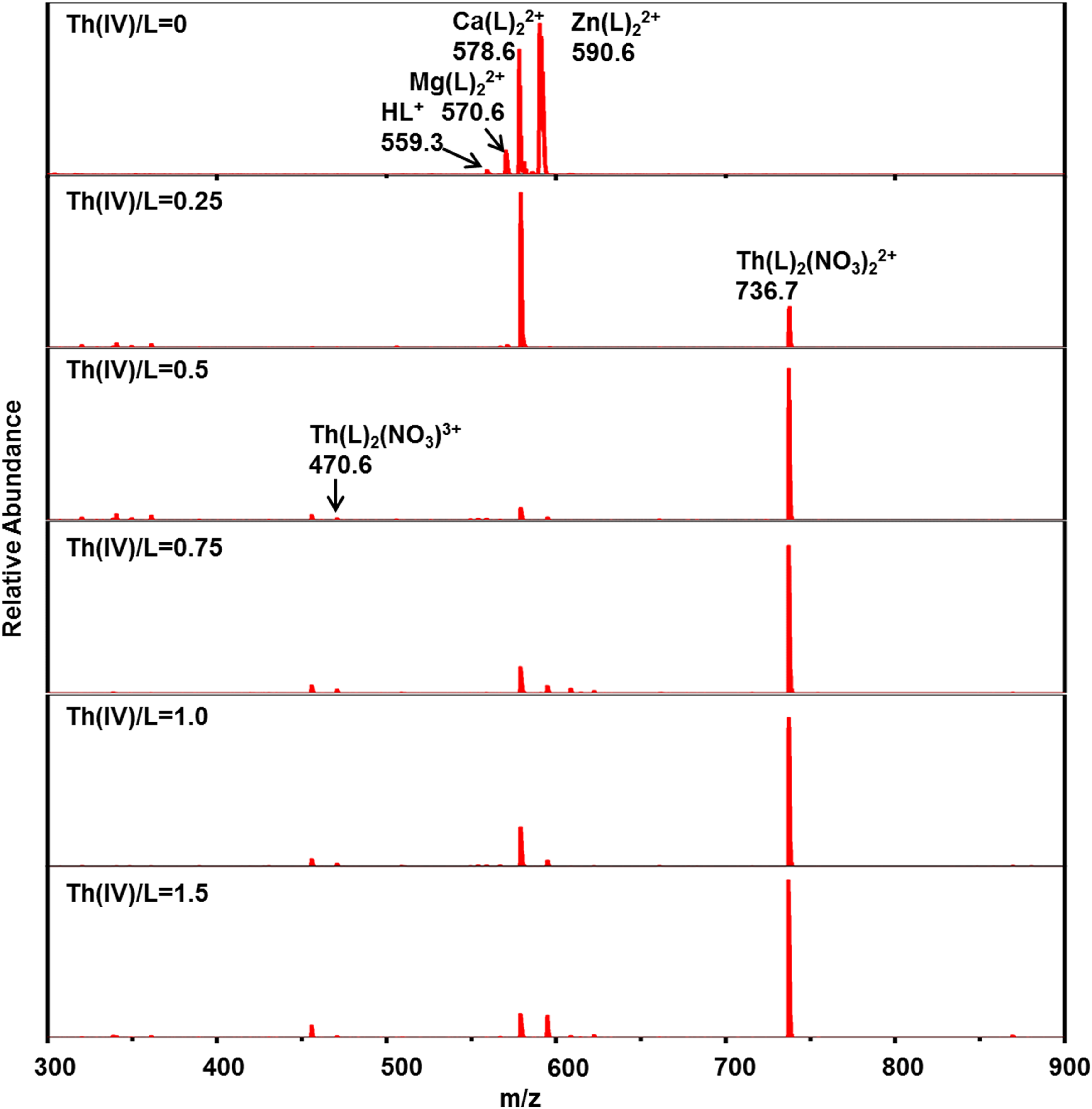
Positive electrospray ionization mass spectrometry (ESI-MS) spectra of the Th4+ complexes at various [Th4+]/[L] ratios.
Stoichiometry ratio and assignment of M–L species detected in ESI-MS.
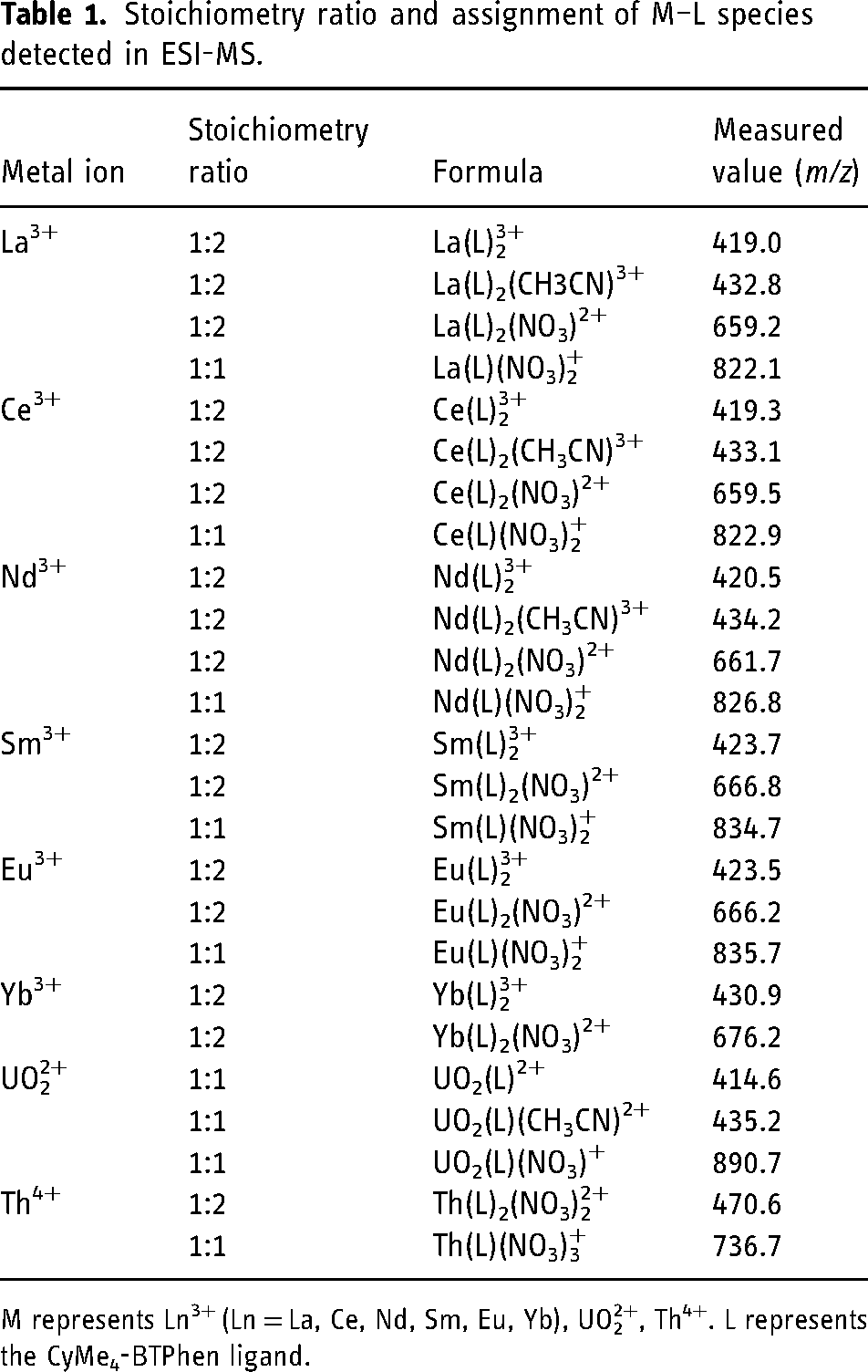
M represents Ln3+ (Ln = La, Ce, Nd, Sm, Eu, Yb), UO22+, Th4+. L represents the CyMe4-BTPhen ligand.
To sum up, Ln3+ complexes show two coordination modes with CyMe4-BTPhen. The 1:1 complexes occur when the metal ions are excessive. The 1:2 species seen by Retegan et al. 27 for CyMe4-BTBP is observed in our experimental condition. Therefore, it can be deduced that the dominant complex is 1:2 species for CyMe4-BTBP and CyMe4-BTPhen, representing L2M(NO3)2+ and L2M3+ in the gas phase. UO22+ and Th4+ complexes have only one coordination mode of 1:1 or 1:2. The nitrate ion and acetonitrile molecule widely participate in the coordinated process.
Fragment analysis
To further understand the structures and stability of CyMe4-BTPhen and its complexes, the CID technique was used to investigate the relationship of fragmentation patterns between CyMe4-BTPhen ligand and its complexes. Recent reports revealed that the fragmentation pattern of metal complexes showed metal dependence. 28 Complexes of [HL]+, [Ln(L)2]3+, and [Ln(L)2(NO3)]2+ were chosen as the precursor ions. They were isolated and reacted at a suitable collision energy. A low collision energy will leave the reaction incomplete, while a higher collision energy will lose some product ions. During this experiment, we chose the collision energy at which the relative abundance of the parent ions was about 30%.
The CID results of [HL]+ revealed regular cleavage behavior. As shown in Figure 6, the fragmentation pathway is inferred based on the fragment ions. The main fragment ions are shown in Supplemental Table S1. With the collision energy set to 55 V, the fragmentation unfolded sequentially from both sides of the structure, which mainly split into two parts. In the first part, the independent monoalkyl groups are extremely unstable and very easy to lose. MS peaks at m/z 543.8, m/z 529.7, m/z 513.6 and m/z 499.6 represent the continuous loss of CH4, CH2, CH4, and CH2. The abundance of fragment ion m/z 529.7 is higher than the others. Hence, we can deduce that the methyl groups on the same carbon site are more likely to be lost simultaneously. The cleavage of alkyl groups results in the formation of two double bonds in the alkyl ring. The fragment ions of m/z 486.2 and m/z 474.2 correspond to the cleavage of the C–C bond in the alkyl ring. The species at m/z 462.2 and m/z 448.2 correspond to the loss of alkyl group fragments. The fragment ion at m/z 394.2 is obtained from the ring-opening reaction by the triazine ring to generate the monocyano compound. The second part is the same cleavage as the first part on the other side of the CyMe4-BTPhen ligand. MS peaks at m/z 378.2, 364.1, 348.1, and 334.1 refer to the continuous loss of alkyl groups. Finally, the dicyano compound is generated at m/z 230.1. Notably, we compared the synthesis process of CyMe4-BTPhen with those of the observed fragments, which have a high correlation. The synthesis procedure of CyMe4-BTPhen includes intermediate products such as 1,10-phenanthroline-2,9-dicarbonitrile and 1,10-phenanthroline-2,9-dicarbohydrazonamide. This is consistent with the instability of the C–C bond and C–N bonds in the ligand and reasonably explains the final formation of the dicyano complex.
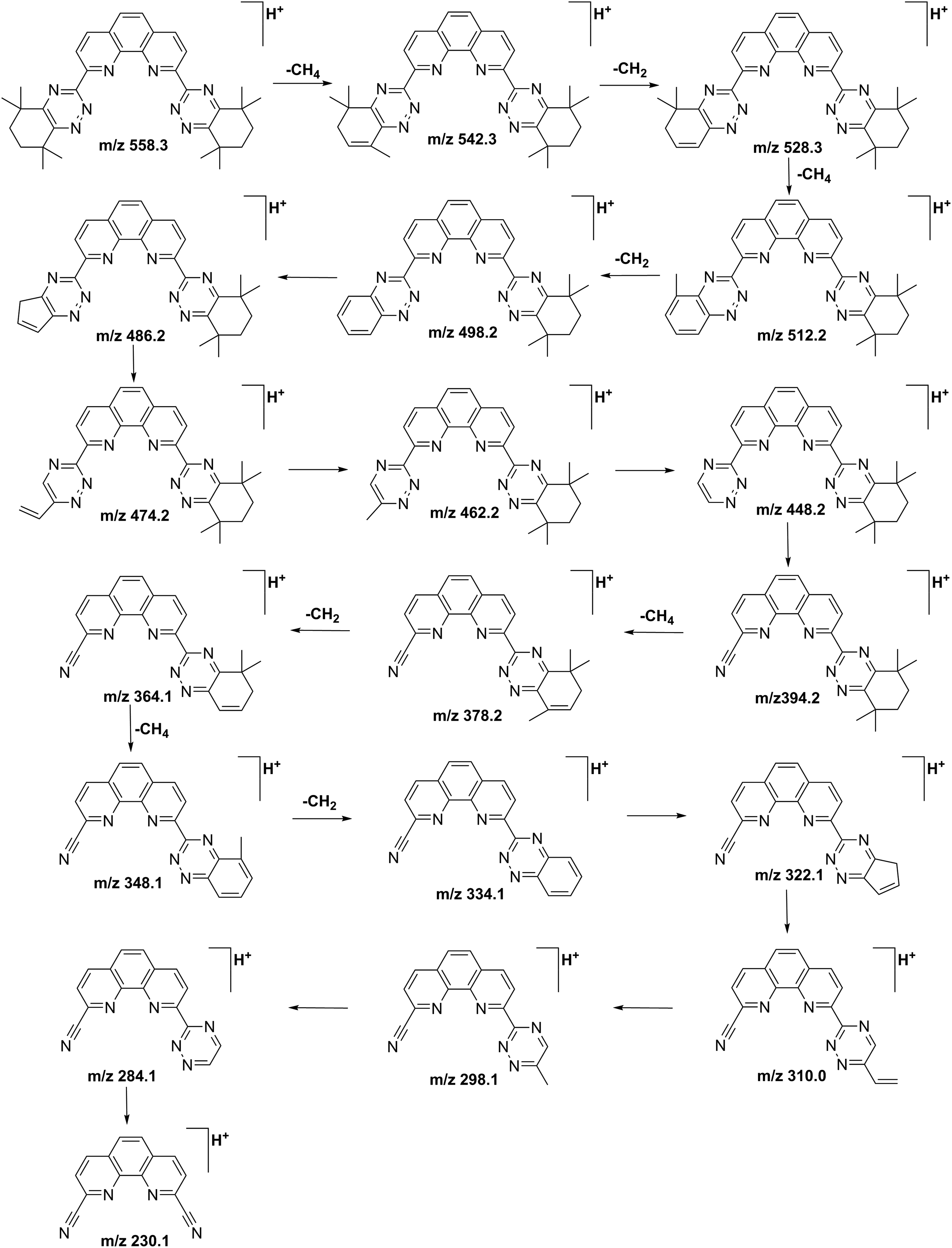
Proposed fragmentation pathways of CyMe4-BTPhen obtained from the ESI-MS/MS.
The CID results show that the fragment ions produced by M(L)23+ (Figure 7) are consistent with the fragmentation pathway of [HL]+. In terms of La(L)23+, the fragment ions of M–L species detected in ESI-MS/MS are shown in Supplemental Table S1. The fragmentation rises from the cleavage of the ligand. By sequential loss of CH4 and CH2 groups of [M(L)2]3+, a series of fragment ions are generated ([La(L)2]3+, m/z 419.0, 413.2, 408.8, 403.5, and 398.6). Due to the weak degree of dissociation, monocyano complex ([La(L)2]3+: m/z 347.8) is the only product generated in this process. The MS/MS spectrum of [Ln(L)2(NO3)]2+ shows two primary competitive fragmentation pathways: direct occurrence of the cleavage of the ligand or loss of a molecule of HNO3 or –NO2 before the cleavage of CyMe4-BTPhen ligand 29 (Figure 8). For [La(L)2(NO3)]2+ (m/z 659.2), MS peaks at m/z 651.1, 644.1, 635.8, and 628.8 can be attributed to the consecutive loss of CH4 and CH2 groups. MS peaks at m/z 610.0, 603.2, 596.2, 588.2, and 580.3 can be attributed to the loss of HNO3 and the subsequent successive loss of CH4 and CH2 groups. MS peaks at m/z 546.0 and m/z 463.8 represent the monoacyano and biscyano compounds. The CID results for [Ce(L)2(NO3)]2+ (m/z 659.5) and [Sm(L)2(NO3)]2+ (m/z 666.8), which are different from the fragmentation pathway of [La(L)2(NO3)]2+, are presented in Figure 8 and Supplemental Table S1. The fragmentation pathway is different from other lanthanides with a loss of –NO2 before the ligand cleavage. MS peaks at m/z 636.4, 629.0, 622.6, 614.8, and 605.9 might be attributed to the loss of –NO2 and subsequent successive losses of CH4 and CH2 groups. MS peak at m/z 553.6 represents the formation of a monocyano compound. All these CID data show that the 1,10-phenanthroline-2,9-dicarbonitrile group is the most stable structure and has not been eliminated during the CID experiments, which corresponds to the previous research. 27 In addition, more detailed investigations of actinides and CyMe4-BTPhen ligands in the gas phase are being undertaken. The MS/MS spectrum of [UO2(L)(NO3)]+ (m/z 890.7) shows the same way as [Ln(L)2(NO3)]2+ (Supplemental Figure S7). MS peaks at m/z 874.5, 860.8, 843.7, and 829.6 represent the loss of alkyl groups. MS peak at m/z 826.6 represents the loss of HNO3. MS peaks at m/z 664.5 and m/z 500.2 correspond to the monocyano and dicyano compounds, respectively. The MS/MS spectra of Th(L)2(NO3)]3+ at m/z 470.6 show the same way as [Ln(L)2(NO3)]2+ (Supplemental Figure S8). MS peaks at m/z 464.4, 460.0, 454.6, and 450.7 represent the loss of alkyl groups. MS peak at m/z 449.8 represents the loss of HNO3. The MS/MS spectra of [Th(L)2(NO3)2]2+ at m/z 736.7 show the same way as [Ce(L)2(NO3)]2+ (Supplemental Figure S8). MS peaks at m/z 713.3 and 690.3 might be attributed to the loss of –NO2 and –2NO2. MS peaks at m/z 704.8, 698.9, 682.4, 675.6, 668.5, and 661.6 might be attributed to the subsequent successive losses of CH4 and CH2 groups. In summary, the cleavage process of the complex with nitrate ion has another competitive fragmentation pathway compared with the binary complexes without nitrate ion, which is the loss of HNO3 or –NO2 before the cleavage of the ligand.
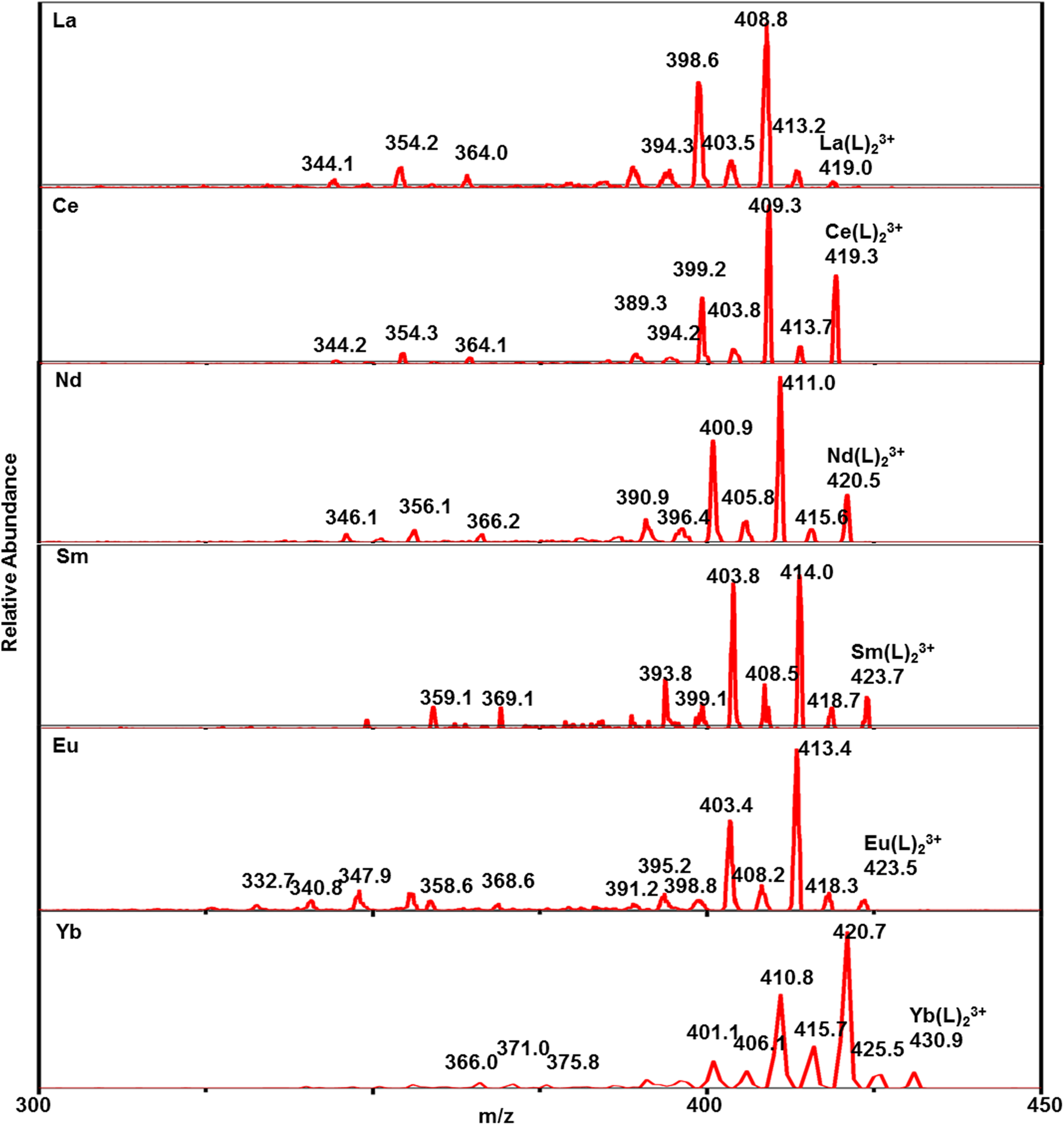
Tandem mass spectrometry (MS/MS) spectra of [Ln(L)2]3+ (Ln = La, Ce, Nd, Sm, Eu, Yb).
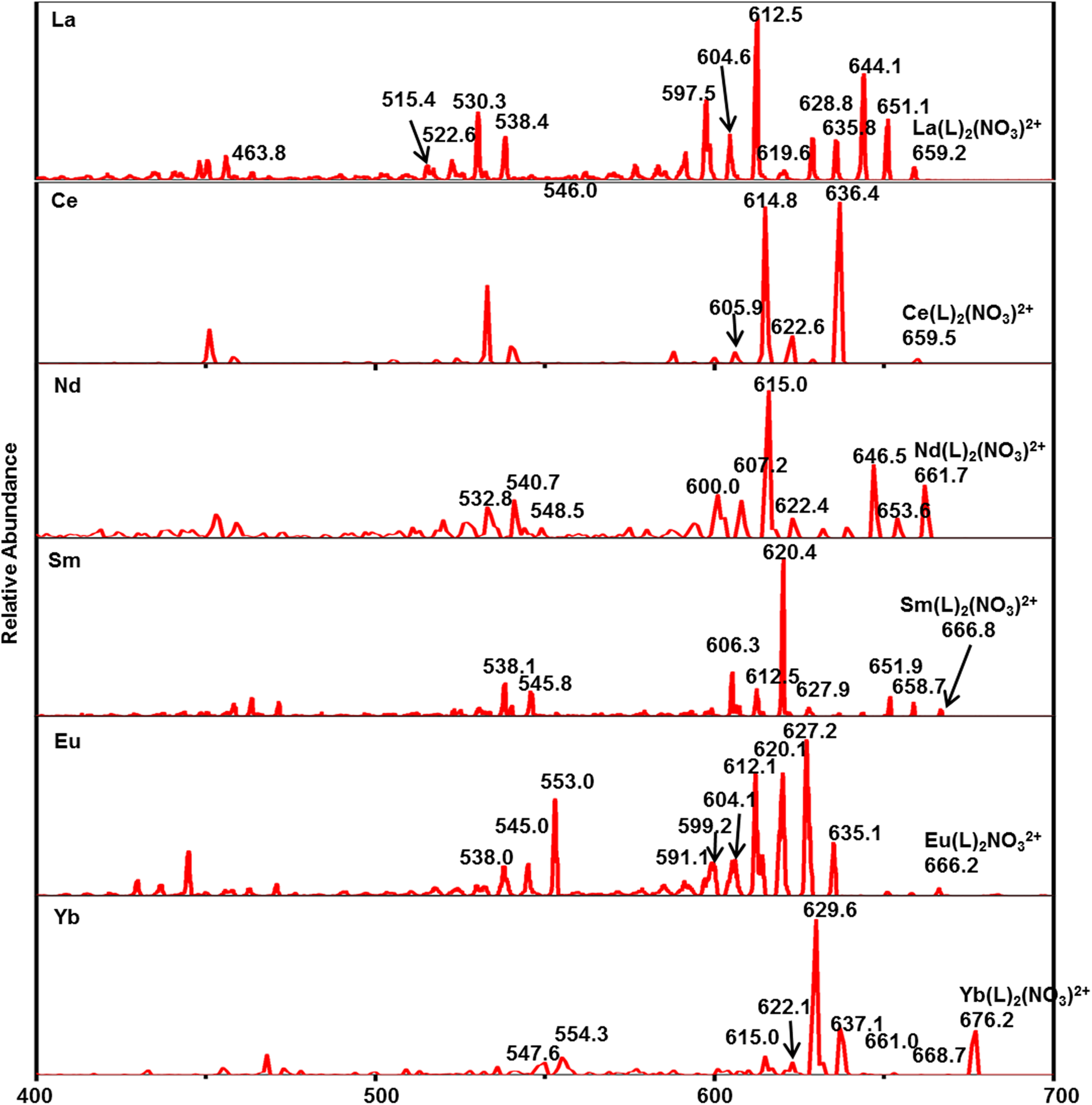
Tandem mass spectrometry (MS/MS) spectra of [Ln(L)2(NO3)]2+ (Ln = La, Ce, Nd, Sm, Eu, Yb).
DFT calculations
The molecular structures of the La3+, Eu3+, Th4+, and UO22+ complexes observed in the mass spectra have been optimized at the B3LYP/6-31G(d, p)/RECP level of theory (Figures 9 and 10). For all the complexes, the tetradentate ligand CyMe4-BTPhen coordinates with metal ions via two nitrogen atoms (N1) of the phenanthroline group and two nitrogen atoms (N2) of the triazine group. In addition, the nitrate anions are both bidentate ligands while acetonitrile is a monodentate ligand in the coordination process.
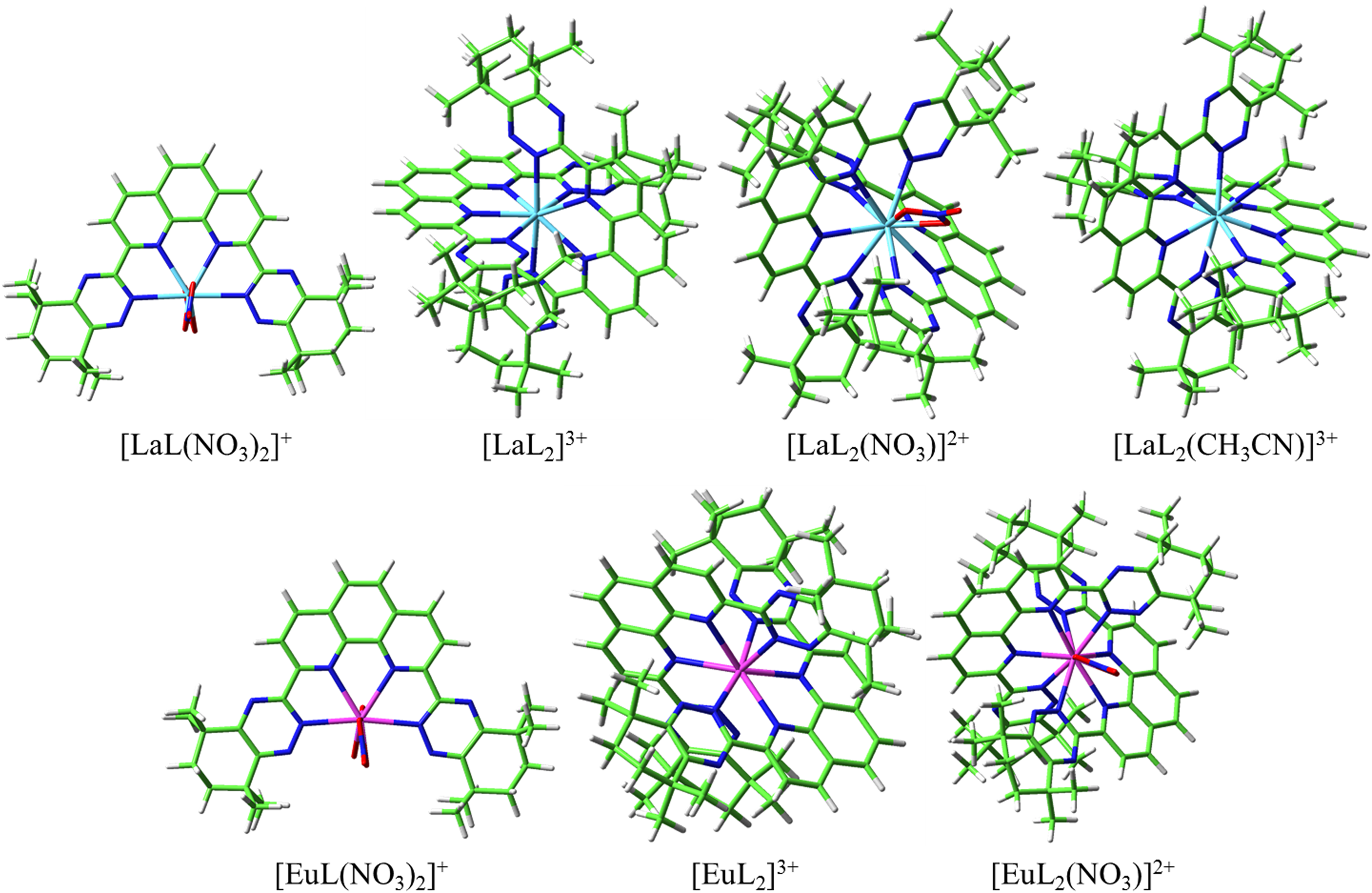
The optimized geometrical structures of La3+ and Eu3+ complexes at the B3LYP/6-31G(d, p)/RECP level of theory in the gas phase. The H, C, N, O, La, and Eu atoms are represented by white, green, blue, red, wathet, and crimson sticks, respectively.
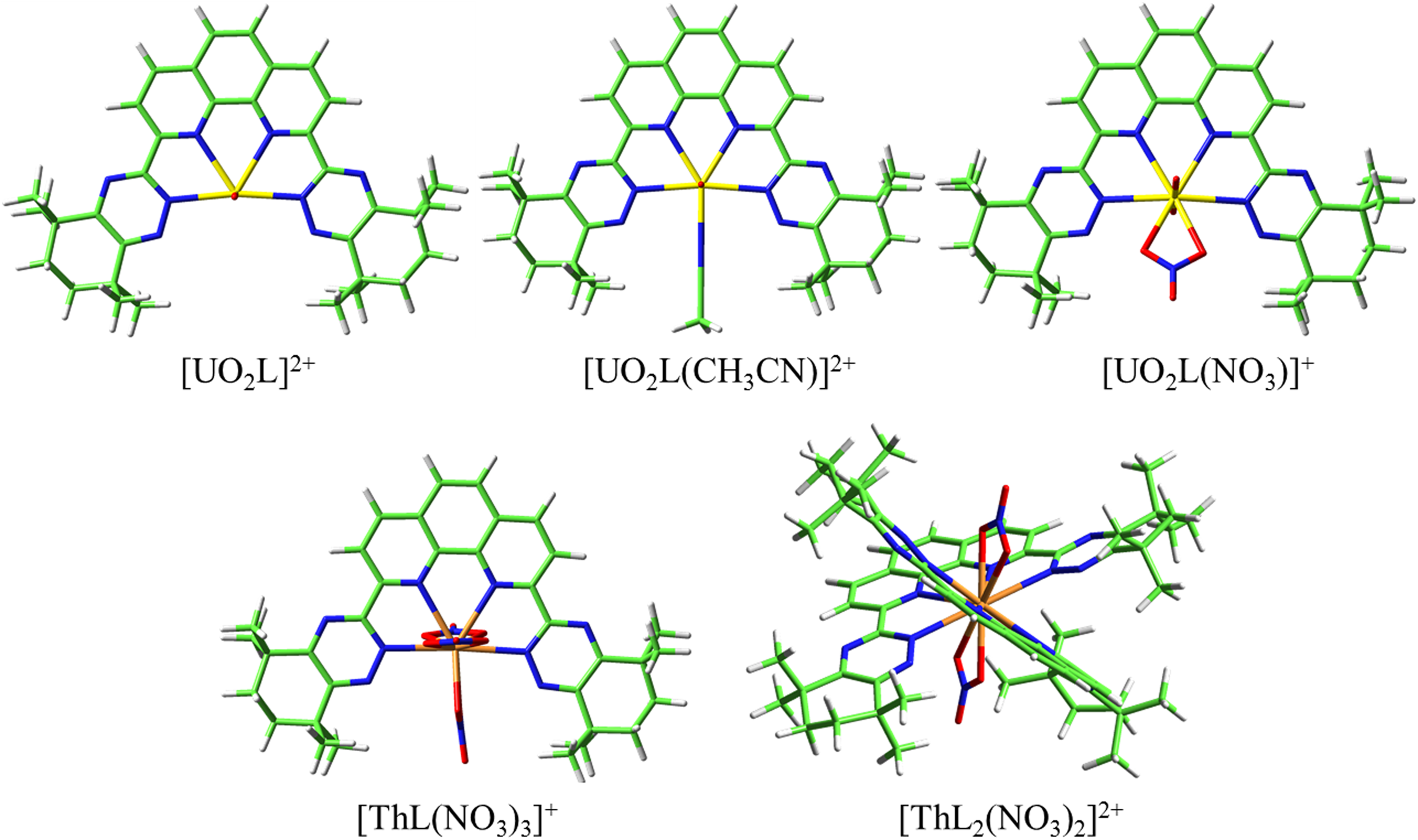
The optimized geometrical structures of Th4+ and UO22+ complexes at the B3LYP/6-31G(d, p)/RECP level of theory in the gas phase. The H, C, N, O, Th, and U atoms are represented by white, green, blue, red, yellow, and orange sticks, respectively.
The lanthanum and europium were used as representatives to analyze the structure and properties of Ln(III) complexes. For La(III) complexes, four structures [La(L)2]3+, [La(L)2(CH3CN)]3+, [La(L)2(NO3)]2+, and [La(L)(NO3)2]+ were optimized, which were observed in the mass spectra. As shown in Supplemental Table S2, [La(L)2]3+ complexes have the shortest La–N1 and La–N2 bond distances because of the lower coordination number, while the [La(L)2(NO3)]2+ complex has the longest La–N bond distance due to its high coordination number, which results in a steric hindrance effect. In addition, the La–N2 bond distance is shorter than La–N1 in all complexes except for the [La(L)2(CH3CN)]3+ complex, indicating that the affinity between the N2 atom and metal ion is stronger than the N1 atom. For Eu(III) complexes, three structures [Eu(L)2]3+, [Eu(L)2(NO3)]2+, and [Eu(L)(NO3)2]+ were optimized. As relevant crystal structures and computational studies have been reported for the [Eu(L)2(NO3)]2+ complex, the comparisons of bond distance are listed in Supplemental Table S3. Comparing the optimized structure with the crystal structure, it is found that the difference is small (<0.08 Å), indicating that the calculated structure is reasonable. Compared with reported theoretical studies, 30 it is found that the difference in Eu–-N bond distance is relatively close (<0.04 Å). Similarly, the 8-fold [Eu(L)2]3+ and [Eu(L)(NO3)2]+ complexes have a shorter Eu–N bond distance than the [Eu(L)2(NO3)]2+ complex. Furthermore, the Eu–N1 bond distance of the 1:2 complex is shorter than that of the Eu–N2 bond distance, although the difference is within 0.02 Å, it is not consistent with that of La(III) complexes. The difference in metal ion radius between 9-coordination La and Eu complexes is 0.096 Å. The bond distance difference between La–N2 and Eu–N2 is within 0.096 Å, while the bond distance difference between La–N1 and Eu–N1 is opposite, indicating that the interaction between Eu–N1 is stronger than that between La–N1.
The NBO analysis results are shown in Table 2. The WBIs of Ln–N1 and Ln–N2 are between 0.17 and 0.23, indicating that these bonds have less covalent characteristics. The WBIs of the Ln-N2 bond are higher than those of the Ln–N1 bond for most complexes. Compared to the 1:1 complexes, the WBIs of the 1:2 complexes are larger, suggesting that the binding strength of the Ln–N bond in these 1:2 species may be stronger than that in the 1:1 species. For the [La(L)2(CH3CN)]3+ complex, the WBI of La–N(CH3CN) is smaller than that of La–O(NO3−), indicating that the coordination of CH3CN with La(III) is much weaker than that of the nitrate ion. The natural population analysis (NPA) showed that the natural charges on the Ln atoms are smaller than those on the free Ln3+ ions, suggesting that significant ligand-to-metal charge transfer occurs in these complexes. The calculated charge transfer from the ligand to metal ions (ΔQM) of [Ln(L)2(NO3)]2+ complexes is larger than the other complexes. Taking WBI and NPA analyses into consideration, 1:2 complexes are more advantageous.
Average WBIs of Ln–N1(L), Ln–N2(L), Ln–N(CH3CN) and Ln–O(NO3−) for [Ln(L)2]3+, [Ln(L)2(CH3CN)]3+, [Ln(L)2(NO3)]2+, and [Ln(L)(NO3)2]+ complexes at the B3LYP/6-31G(d, p)/RECP level of theory.
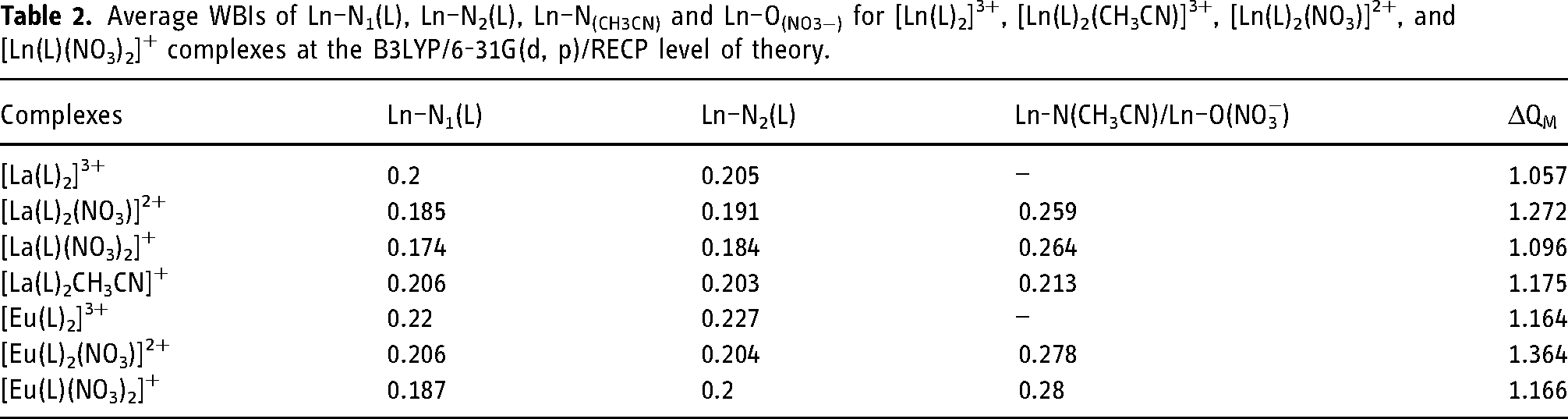
All the uranyl complexes were 1:1 species, and three structures [UO2(L)]2+, [UO2(L)(CH3CN)]2+, and [UO2(L)(NO3)]+ were optimized at the B3LYP/6-31G(d, p)/RECP level of theory. As shown in Supplemental Table S4, the U–N bond distance of the [UO2(L)]2+ complex is the shortest compared with the other two complexes because of the lower coordination number, especially the [UO2(L)(NO3)]+ complexes. The bond distance of U–N2 is much shorter than that of U–N1 for all the uranyl complexes, indicating that the coordination between the N2 atom and actinide ion is stronger than that with the N1 atom. Correspondingly, the WBIs of the U–N2 bond of the uranyl complex are greater than that of the U–N1 bond, indicating that the WBI results are in good agreement with the bond distance results. Compared with the other uranyl complexes, the ΔQM of [UO2(L)(NO3)]+ complexes is greater, suggesting the [UO2(L)(NO3)]+ complexes might be more dominant.
Two structures [Th(L)2(NO3)2]2+ and [Th(L)(NO3)3]+ of Th4+ complexes were optimized at the B3LYP/6-31G(d, p)/RECP level of theory. The Th–N1 and Th–N2 bond distances of the 12-fold [Th(L)2(NO3)2]2+ complex are longer than that of the 10-fold [Th(L)(NO3)3]+ complex due to the steric hindrance caused by coordination difference. As shown in Table 3, the WBIs of Th–N1 and Th–N2 bonds in the [Th(L)2(NO3)2]2+ complex are greater compared with the [Th(L)(NO3)3]+ complex, indicating that the Th–N bond strength in the [Th(L)2(NO3)2]2+ complex is higher than that in the [Th(L)(NO3)3]+ complex. Similarly, the ΔQM of [Th(L)2(NO3)2]2+ complexes is larger than that of the [Th(L)(NO3)3]+ complexes. Taking both WBI and NPA analyses into account, 1:2 complexes are more advantageous.
Average WBIs of An–N1(L), An–N2(L), An–N(CH3CN) and An–O(NO3−) for [UO2(L)]2+, [UO2(L)(CH3CN)]2+, [UO2(L)(NO3)]+, [Th(L)2(NO3)2]2+, and [Th(L)(NO3)3]+ complexes at the B3LYP/6-31G(d, p)/RECP level of theory.

Since the preparation of La3+, Eu3+, Th4+, and UO22+ stock solutions involves the dissolution of the corresponding nitrate in acetonitrile solution, the coexistence of metal ions, nitrate ions, and CH3CN may be present in the substrate. The complexes [Ln(CH3CN)9]3+, [Th(CH3CN)9]4+, and [UO2(CH3CN)5]2+ conduct the reactants for the complexation reactions (Supplemental Table S5) to explore the stability of the La3+, Eu3+, Th4+, and UO22+ complexes with CyMe4-BTPhen. The ΔG values of all complexation reactions are negative, indicating that all reactions can take place spontaneously at room temperature. For the Ln3+ complexes, the ΔG values of 8-fold [Ln(L)2]3+ complexes are larger than those of [Ln(L)2(NO3)]2+ and [Ln(L)(NO3)2]+ complexes, suggesting that the complexion involving nitrate is more favorable. Furthermore, uranyl complexes show similar results compared to Ln3+ complexes, proving that the [UO2(L)(NO3)]+ complex is more advantageous than the [UO2(L)]2+ complex. The Th4+ complex has the lowest ΔG values, which can be known that the Th4+ complex is favorable to the extraction reactions. In addition, complexes containing CH3CN ligands are disadvantageous compared to the complexes containing nitrates.
Conclusion
In this work, we elucidated the complexation of CyMe4-BTPhen complexes with lanthanides and actinides using ESI-MS. The speciations of Ln3+ (Ln = La, Ce, Nd, Sm, Eu, and Yb), UO22+, and Th4+ with CyMe4-BTPhen in acetonitrile were investigated by mass spectrometry titrations. The species were consistent with the previous studies. The mass spectra showed that the species and abundance of the complexes varied with the different [M]/[L] ratios. The MS peaks are dominated by 1:2 Ln3+/L complexes when the ligand is excessive. And 1:1 species is formed in small amounts when the metal ion is in excess. Therefore, it can be inferred that the 1:2 species is easier to form. There is only one type of UO22+ and Th4+ complexes, namely 1:1 and 1:2 species. Furthermore, it is worth mentioning that most species shown in the ESI-MS spectra contain one or two nitrate ions or a solvent molecule. The complexes with nitrate ions are more advantageous than the other species, as the nitrate ion is very easy to participate in the coordination to form a stable structure. We deduce the fragmentation pathway of the ligand from the CID results, which occurred side by side. The fragmentation pathway is closely related to the synthesis process of CyMe4-BTPhen. This can provide a reference for the judgment of unconventional products during the extraction process. The fragmentation pathway of different metal complexes shows slight differences, the loss of HNO3 or –NO2 varies depending on the center of a metal ion. The geometrical structures, NBO analysis, and thermodynamic stability of the complexes were studied by DFT calculations. From the view of applications point, this work has shown an accurate analysis method for the research of the complexation behavior of CyMe4-BTPhen complexes, which is also beneficial to the understanding of real extraction and separation processes in spent fuel reprocessing.
Supplemental Material
sj-docx-1-ems-10.1177_14690667231206035 - Supplemental material for Coordination and fragmentation chemistry of CyMe4-BTPhen complexes with lanthanides and actinides: A combined investigation by ESI-MS and DFT calculations
Supplemental material, sj-docx-1-ems-10.1177_14690667231206035 for Coordination and fragmentation chemistry of CyMe4-BTPhen complexes with lanthanides and actinides: A combined investigation by ESI-MS and DFT calculations by Qiqi Zhang, Yang Liu, Shuping Tan, Yan Chen, Xinyue Liang, Weiqun Shi and Yonggang Zhao in European Journal of Mass Spectrometry
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
