Abstract
The reactions of 5- and 7-substituted isatin derivatives (H, Me, Cl, Br) with piperidine in 80% aqueous methanol gave the corresponding 1-(2’-amino-5’- or 3’- substituted phenyl)-2-(piperidin-1-yl) ethane-1,2-dione derivatives. The reaction is proceeded through nucleophilic attack on C-2 followed by ring opening process. The reaction showed a second order and the isokinetic temperature is 169 K indicating that the reaction is entropy controlled. The constant ∆G# value, the linear plot of ∆H# versus ∆S#, plot of log kN values at high temperature versus those at low temperatures indicated that a unified mechanism of the reactions for all substituents. The σo-Taft's constants are linearly correlated with log kN values and the ρ values are (2.01–1.86) pointing out that inductive effect of substituent is predominant and the rate-limiting is the breakdown of the tetrahedral intermediate T− and the ring opening is catalyzed by water.
Introduction
Isatin is an indole derivative (1H-indole-2,3-dione) and found in many plants and humans as it is a metabolic derivative of adrenaline. 1 It was reported that isatin participated a broad range of synthetic leading to its use as aprecursor molecule in medicinal chemistry.2–5 Isatin has special properties due to the fact that it possess many reactive centers, so it undergoes electrophilic aromatic substitution (SEAr) at carbon atoms on positions five and seven of the phenyl ring., It has been reported that isatin underwent condensation with primary amine and with active methylene compounds such as malononitrile CH2(CN)2 or ethyl cyanoacetate (NCCH2COOC2H5) on C-3 carbonyl group. 6 Additionally, isatin showed ring opening via nucleophilic attack on C-2 amide group by secondary amine or negatively charge ions such as HO− in which H2O must be present,7,8 or ring expansions as well as spiro-annulations. The rates of hydrolysis of isatins derivatives showed a complex dependence upon pH indicative of subtle changes in mechanisms and rate-limiting step. 9 Most reactions of hydroxide ions and secondary amines with isatins and N-alkyl isatin in aqueous cosolvents undergo nucleophilic substitution reactions at the amide linkage with ring opening process. The rate and mechanism of ring opening of isatin and N-substituted isatin derivatives showed great dependence on water molecules present in the reaction mixture.10,11
The aim of this work is to use the kinetic study as a tool to suggest the mechanisms for the reaction of some five- and seven- substituted isatin with piperidine in 80% aqueous methanol. Furthermore, the effect of a remote substituent on the sensitivity of the electronic character of the amide carbonyl group toward piperidine will be discussed.
Experimental
Melting points were measured by MEL-TEMP II melting point apparatus in open glass capillaries and are uncorrected. The UV spectra were carried out on a 160-A UV-VIS recording spectrophotometer Shimadzu. The IR spectra were recorded for potassium bromide (KBr) discs on a Perkin-Elemer FT-IR, System spectrum ratio recording infra-red spectrophotometer. The 1H-NMR spectra were carried out at ambient temperature (∼25 C) on a (JEOL) 500 MHz spectrophotometer using tetramethylsilane (TMS) as an internal standard, Faculty of Science, Central Laboratory Unit, Alexandria University.
Preparation of five and 7-substituted isatin
Step 1: Preparation of isonitrosoacetanilide derivatives 1a-f
0.05 mol of chloral hydrate was taken into the round bottom flask and dissolved in 120 mL water to 0.09 mol of sodium sulphate, a solution of 0.05 mol of aniline derivatives in 30 mL of water containing 0.14 mol of concentrated hydrochloric acid (4.34 mL) to dissolve the amine and solution of 0.15 mol of hydroxylamine hydrochloride in 50 mL of water were added. Flask was then heated vigorously until the reaction was completed. Later, the solution containing beaker was cooled in running water followed by the filtration of reminder crystallized product with suction pump and air dried.
Step 2: synthesis of isatin derivatives 2a-f
Dry isonitrosoacetanilide derivatives 0.15 mol was added to 0.18 mol of concentrated sulphuric acid (10.0 mL) was warmed to 50°C in such a rate so as to keep the temperature between 60 and 70°C but not higher. External cooling was applied at this stage so that the reaction could be carried out more rapidly after the addition of isonitroso compound was finished. The solution was heated to 80°C and kept at this temperature for about 10 min to complete the reaction. Then the reaction mixture was cooled to room temperature and poured into 10 times its volume of cracked ice. After standing for 90 min, the final product was filtered with suction pump followed by washing with cold water to remove sulphuric acid and dried in air.12,13
Preparation of 1-(2’-amino substituted phenyl)-2-(piperidin-1-yl)ethane-1,2-dione
A solution (10 mL) of isatin derivatives
1-(2’-aminophenyl)-2-(piperidin-1-yl)ethane-1,2-dione,3a
Yield: 45%; m. p: 184°C; (lit 185). 14
1-(2’-amino-5’-methylphenyl)-2-(piperidin-1-yl)ethane-1,2-dione, 3b
Yield: 40%; m. p: 185°C; UV (MeOH: H2O) (2:8): λmax = 380 nm (ε = 4333), IR (KBr): 3429 (NH), 1630 (C = O), 1550 (C = O amide) cm−1, 2950(C-H aliphatic) cm−1. 1H NMR (DMSO-d6): δ 1.39 (m, 2H, J = 5.35, 5.35 Hz, H-4),1.54 (m, 2H, J = 8.4, 6.15 Hz, H-5), 1.58 (m, 2H, J = 6.9, 6.15 Hz, H-3), 2.11 (s, 3H, CH3), 3.15 (t, 2H, J = 5.35, 6.20, H-6), 3.53 (t, 2H, J = 5.35 Hz, H-2), 6.73 (d, 1H, J = 8.4 Hz, H-3’), 6.99 (s, 1H, H-6’), 7.1 (d, 1H, J = 1.5, 8.5 Hz, H-4’), 7.20(sbr,2H, NH2, D2O exchangeable). C14H18N2O2: (Calc.) 4.87 C%; 0.41 H%; 5.65 N%. (Found): 5.17 C%; 0.51 H%; 5.84 N%.
1-(2’-amino-5’-chlorophenyl)-2-(piperidin-1-yl)ethane-1,2-dione, 3c
Yield: 53%; m. p: 190°C; UV (MeOH:H2O) (2:8): λmax = 380 nm (ε = 4333), IR (KBr): 3420 (NH), 1624 (C = O), 1543 (C = O amide) cm−1.1H NMR (DMSO-d6): δ 1.40 (m, 2H, J = 3.05, 2.3 Hz, H-4), 1.51 (m, 2H, J = 6.90, 4.65 Hz, H-5), 1.59 (m, 2H, J = 6.1, 4.6 Hz, H-3), 3.17 (t, 2H, J = 5.35, 5.35, H-6), 3.55 (t, 2H, J = 6.1, 5.35 Hz, H-2), 6.81 (d, 1H, J = 8.4 Hz, H-3’),7.28 (d, 1H, J = 2.3 Hz, H-6’), 7.34-7.41 (dd, 1H, J = 2.3, 2.3, 6.1 Hz, H-4’), 7.52 (sbr, 2H, NH2, D2O exchangeable). C13H15N2O2Cl: (Calc.) 4.51 C%; 0.37 H%; 5.25 N%; 13.20 Cl %. (Found): 4.82 C%; 0.31 H%; 5.52 N%; 13.51 Cl%.
1-(2’-amino-5’-bromophenyl)-2-(piperidin-1-yl)ethane-1,2-dione, 3d
Yield: 38%; m. p: 200°C; UV (MeOH:H2O) (2:8): λmax = 380 nm (ε = 4000), IR (KBr): 3419 (NH), 1622 (C = O), 1541 (C = O amide) cm−1.1H NMR (DMSO-d6): δ 1.39 (m, 2H, J = 6.1, 5.35 Hz, H-4), 1.53 (m, 2H, J = 6.1, 4.60 Hz, H-5), 1.58 (m, 2H, J = 4.55, 6.85 Hz, H-3), 3.17 (t,2H, J = 5.35, 5.35, H-6), 3.53 (t, 2H, J = 6.1, 4.60 Hz, H-2), 6.86 (d, 1H, J = 9.15 Hz, H-3’), 7.16 (d, 1H, J = 2.3 Hz, H-6’), 7.33 (dd, 1H, J = 3.05, 2.3, 6.16 Hz, H-4’), 7.51 (sbr, 2H, NH2, D2O exchangeable). C13H15N2O2Br: (Calc.) 3.86 C%; 0.32 H%; 4.50 N%; 25.7 Br%. (Found): 3.51 C%; 0.36 H%; 4.21 N%; 25.1 Br%.
1-(2’-amino-3’-methylphenyl)-2-(piperidin-1-yl)ethane-1,2-dione, 3e
Yield: 56%; m. p: 175°C; UV (MeOH:H2O) (2:8): λmax = 380 nm (ε = 4333), IR (KBr): 3428 (NH), 1617 (C = O), 1560 (C = O amide) cm−1, 2941 (C-H aliphatic) cm−1.1H NMR (DMSO-d6): δ 1.325 (m, 2H, H-4), 1.51 (m, 4H, H-3,5), 2.09 (s, 3H, CH3), 3.102 (t, 2H, J = 5.35, 5.35 Hz, H-6), 3.476 (t, 2H, J = 5.35,5.35 Hz, H-2), 6.523 (t, 1H, J = 7.6, 7.65 Hz, H-5’), 7.117 (d, 1H, J = 8.4 Hz, H-4’),7.204 (sbr, 2H, NH2, D2O exchangeable), 7.22 (d, 1H, J = 6.85 Hz, H-6’). C14H18N2O2: (Calc.) 4.87 C%; 0.41 H%; 5.65 N%. (Found):4.30 C%; 0.38 H%; 5.12 N%.
1-(2’-amino-3’-chlorophenyl)-2-(piperidin-1-yl) ethane-1,2-dione, 3f
Yield: 52%; m. p: 180°C; UV (MeOH:H2O) (2:8): λmax = 380 nm (ε = 4666), IR (KBr): 3419 (NH), 1638 (C = O), 1585 (C = O amide) cm−1.1H NMR (DMSO-d6): δ 1.53 (m, 2H, H-4), 1.68 (m, 4H, H-3,5), 3.26 (t, 2H, J = 5.35, 5.35 Hz, H-6), 3.67 (t, 2H, J = 5.35,5.35 Hz, H-2), 6.5 (sbr, 2H, NH2, D2O exchangeable), 6.635 (t, 1H, J = 7.65, 7.6 Hz, H-5’),7.27 (d, 1H, J = 8.4 Hz, H-6’), 7.53 (d, 1H, J = 7.65 Hz, H-4’). C13H15N2O2Cl: (Calc.) 4.51 C%; 0.37 H%; 5.25 N%; 13.20 Cl %. (Found): 4.83 C%; 0.40 H%; 5.83 N%; 13.46 Cl%.
1-(2’-amino-3’-Bromophenyl)-2-(piperidin-1-yl) ethane-1,2-dione, 3g
Yield: 48%; m. p: 190°C; UV (MeOH:H2O) (2:8): λmax = 380 nm (ε = 5000), IR (KBr): 3415 (NH), 1637 (C = O), 1 (C = O amide) cm−1.1H NMR (DMSO-d6): δ 1.53 (m, 2H, H-4), 1.532 (m, 4H, H-3,5), 3.122 (t, 2H, J = 5.35, 5.35 Hz, H-6), 3.4495 (t, 2H, J = 5.35,5.35 Hz, H-2), 6.602 (t, 1H, J = 7.65, 7.65 Hz, H-5’),7.34 (sbr, 2H, NH2, D2O exchangeable), 7.37 (d, 1H, J = 8.4 Hz, H-6’), 7.47 (d, 1H, J = 7.65 Hz, H-4’). C13H15N2O2Br: (Calc.) 3.86 C%; 0.32 H%; 4.50 N%; 25.7 Br%. (Found):3.46 C%; 0.36 H%; 4.28 N%; 25.34 Br%.
Kinetic measurements
The reactions of isatin derivatives with piperidine in 80% water-methanol mixed solvent were followed spectrophotometrically. The optical densities of the solutions measured after the completion of the reactions agreed with the authentic samples of the corresponding products having the same concentrations in the same mixed solvents. The resultant change of absorbance with time was recorded by kinetic mode or time scan mode on UV-VIS Shimadzu 160-A spectrophotometer.
The reaction mixture was prepared by the transfer of 1 mL of the substrate solution (3 × 10−3 M) that gives a final concentration of 3 × 10−4 M to a 10 mL measuring flask and the volume is completed by a given volume of absolute methanol. The measuring flask and the stock solution of the piperidine are allowed to reach thermal equilibrium in a well stirred and thermostatic bath at the required temperature (±0.5°C). The reaction time started when the amine is mixed with the reaction mixture (final concentration of piperidine ranges from 0.3 × 10−2 to 12 × 10−2 M) and transfered quickly to a well thermostated chamber containing the UV cell. The absorbance At at the desired wavelength is recorded at several time intervals depending on the reaction rate. Measurements were usually carried out spectrophotometrically by following the increase in the reaction products absorbance with time at λ = 380 nm.
Results and discussion
Chemistry
The derivatives Synthesis of isatin derivatives 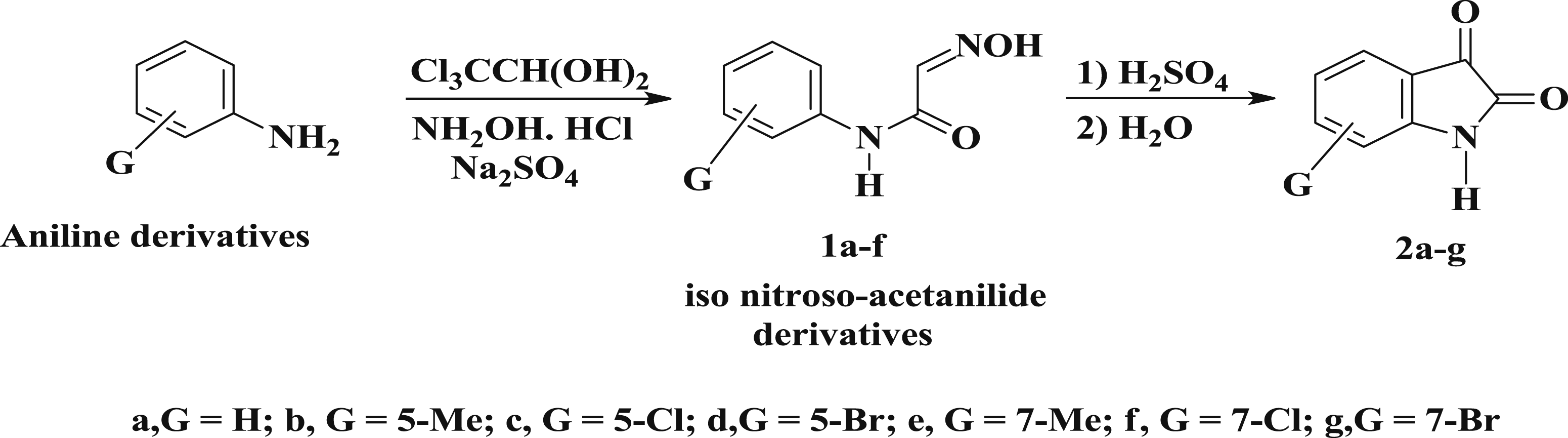
The reaction of five- and 7-substituted isatin with piperidine in 80% aqueous MeOH at 25°C gave 1-(2’-amino-5’- or 3’- substituted phenyl)-2-(piperidin-1-yl) ethane-1,2-dione derivatives 3a–g, Scheme 2. Elemental analysis, IR and 1H-NMR spectroscopy confirmed the formation of a ring opened 1-(2’-amino-5’- or 3’-substituted phenyl)-2-(piperidin-1-yl) ethane-1,2-dione derivatives Reaction of 5- and 7-substituted isatins with piperidine.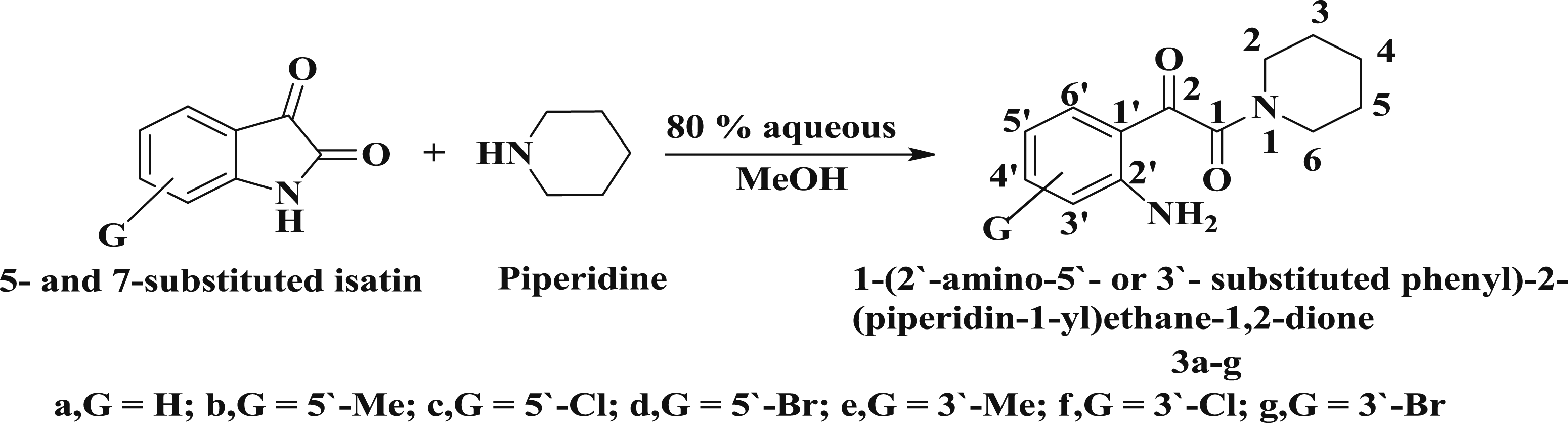
Kinetic study
The rate of the piperidinolysis was followed spectrophotometrically by observing the increase in the optical density near λmax = 380 nm due to the formation of 1-(2’-amino-5’- or 3’- substitutedphenyl)-2-(piperidin-1-yl) ethane-1,2-dione derivatives
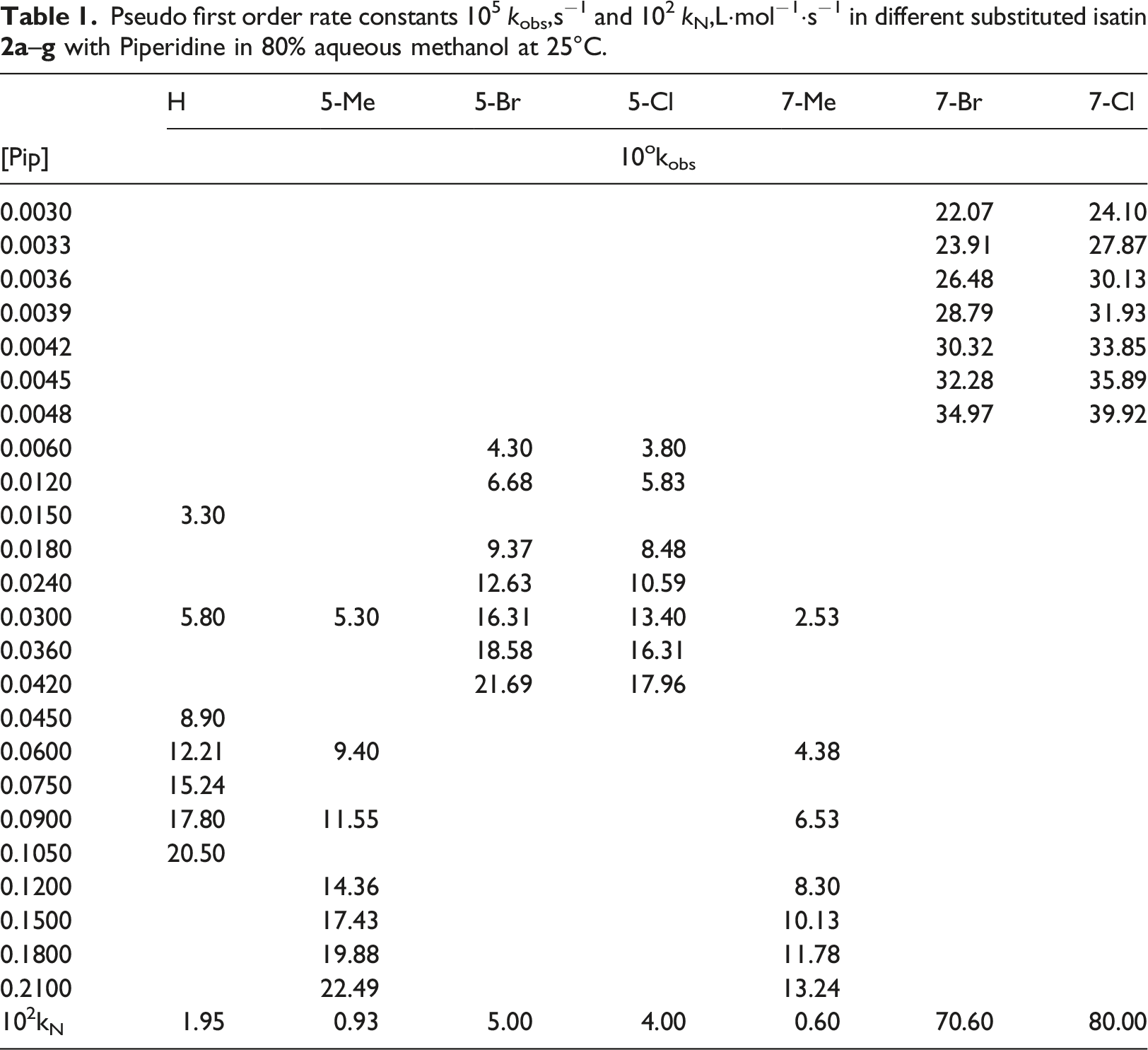
The pseudo first-order rate constants kobs at 25°C, increased linearly with piperidine concentration, Figure 1(a) and (b) and Table 1. This result indicates that the reaction is not catalyzed by piperidine and it is first order in substituted isatin and first order in piperidine. i.e. an overall second order kinetics. The slope of the linear plots obtained from k
obs
versus [Pip] gave the second-order rate constants (k
N
) at 25°C. While, the values of k
N
at different temperatures are obtained by dividing k
obs
by the corresponding piperidine concentration, Table 2. Plot of k
obs
against [Pip] at 25°C. (a) 4-Me isalin (b) 5-Bromo isatin. Pseudo first order rate constants 105 kobs,s−1 and 102 kN,L·mol−1·s−1 in different substituted isatin Second order rate constants kN l mol−1 s−1, reaction constant p and activation parameter values for the piperidinolysis of isatin derivatives 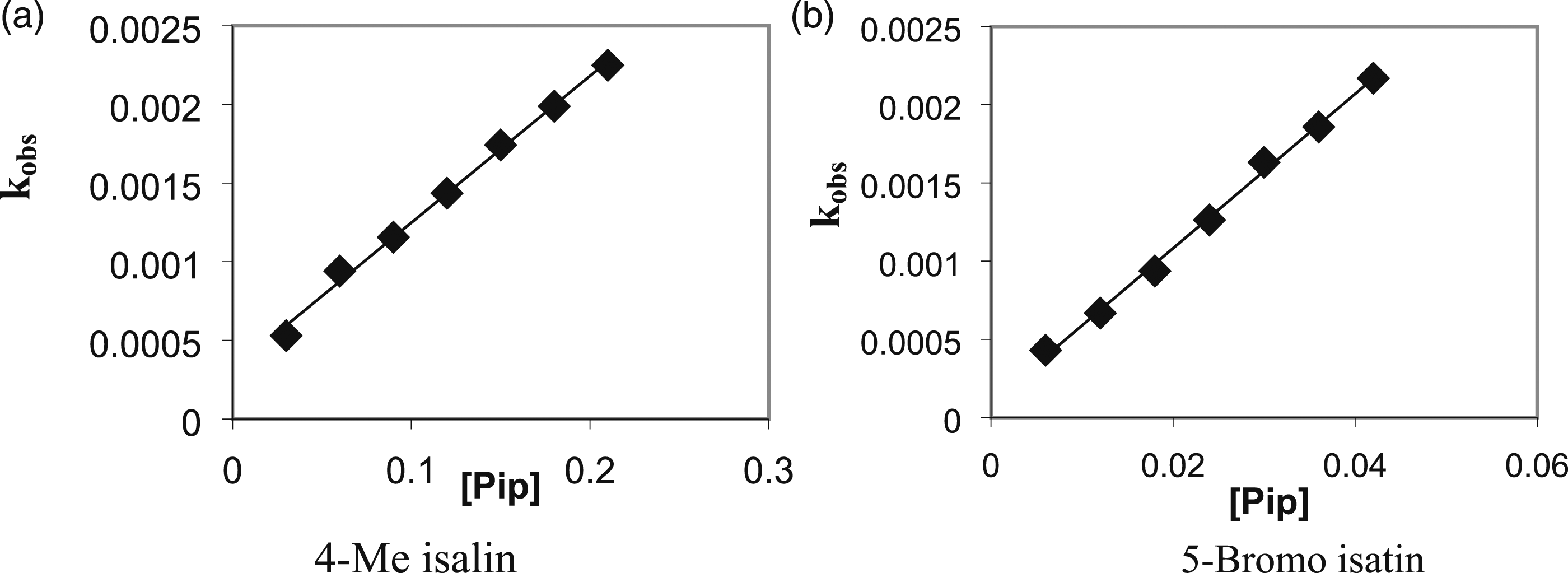
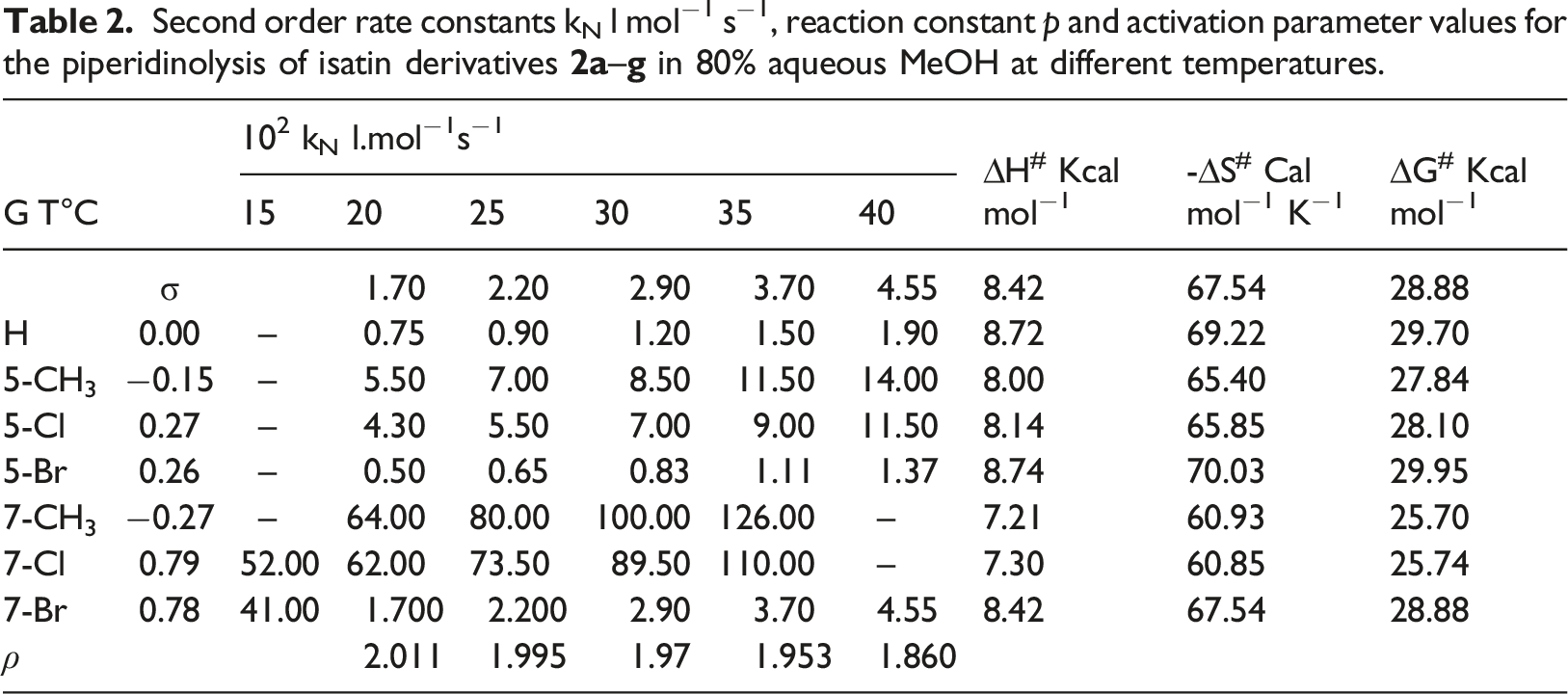
Mechanism of piperidinolysis of isatin derivatives
The reaction of isatin derivatives Piperidinolysis of isatin derivatives 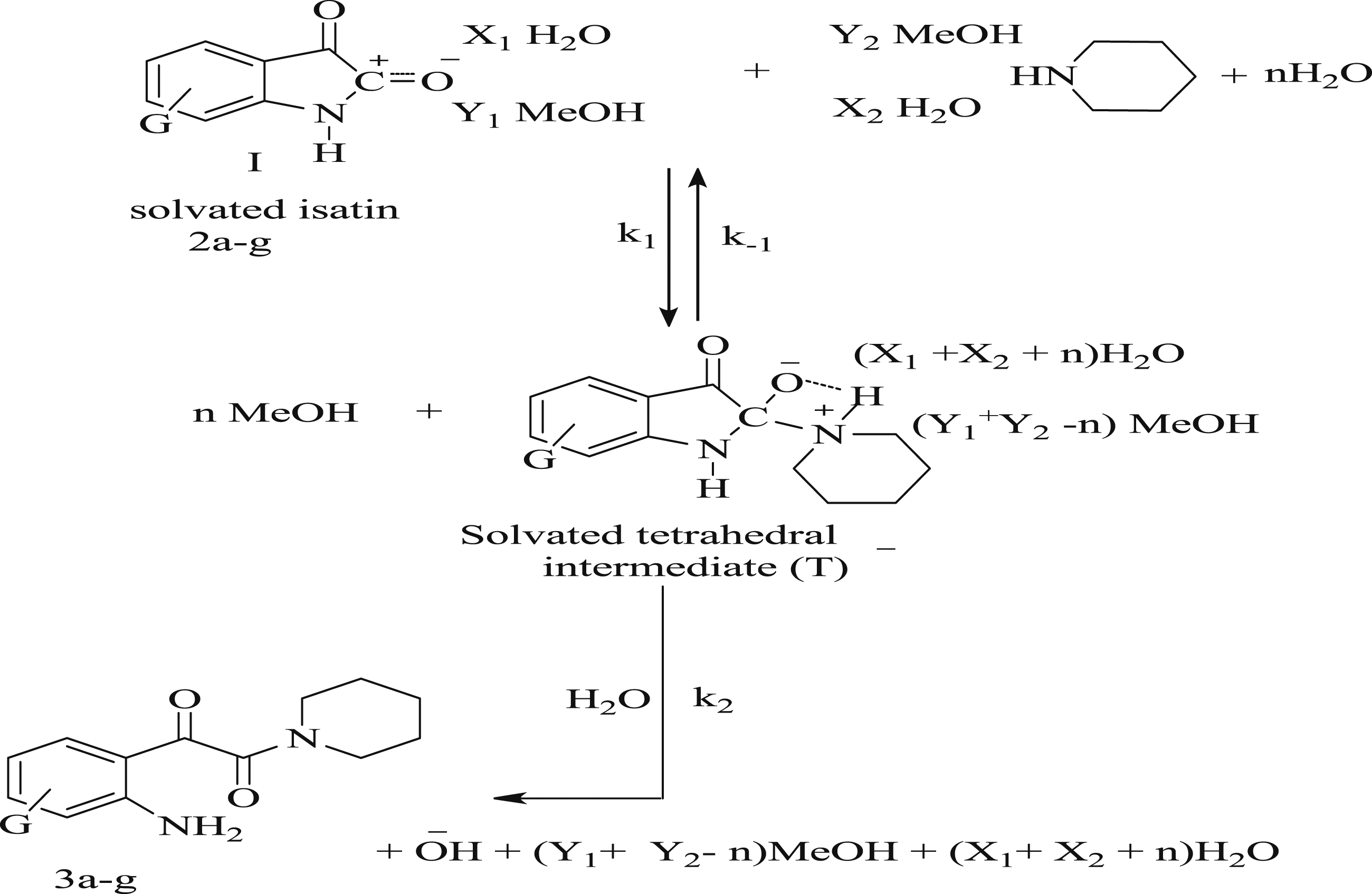
The mechanism is proceeded by an initial attack by piperidine (k1) to generate reversibly the anionic tetrahedral intermediate T‒ which spontaneously reverted to the reactants, k-1, or be converted to products
The kinetic expression for the reaction is derived with reference to the mechanism in Scheme 3 and the application of the steady-state assumption and the great dependence of H2O to complete the reaction as shown in equation (2)
If k−1 ≫ k2 and so k2 [H2O] can be neglected in the denominator, then

Thus, the rate varies directly proportional to [H2O]n+1 while it is inversely proportional to [S]n. Consequently, the ring opening process is the rate determining step.
If k2 ≫ k-1 and so k2 [S]n is neglected in the dominator, so equation (2) is reduced to equation (5)
This indicates that the formation of solvated tetrahedral intermediate is the rate determining step.
Therefore, the effect of temperature, nature and position of the substituent can be used to confirm what mechanism is suggested and determine the rate determining step if the reaction proceeded by stepwise mechanism as shown in Scheme 3 for the titled reaction.
Effect of temperature
The kN values for the titled carried out at different temperatures ranging from 15°C to 40°C (interval temperature is 5°C) are listed in Table 2. Thermodynamic parameters were calculated by least square fit using the equation In kN = a = b (1/T), where, a = ln A and b = −ΔE#/R. The trend in variation of the activation parameters (enthalpy of activation ∆H#, entropy of activation ∆S# and the free energy of activations ΔG#) showed a regular variation with changing substituents in five- and 7-positins in isatin derivatives, Table 2. The electron-donating substituents increase and electron–withdrawing groups decrease ΔH#. The ∆S# values indicate that the transition state is more solvated than the reactants. The relatively high ∆S# values are presumably due to occurrence of internal hydrogen bond in the tetrahedral zwitterion T−, Scheme 2. The constancy of ∆G# value indicates that a unified reaction mechanism operates for all substituents in piperidinolysis process.
Isokinetic relationship
The isokinetic temperature, β for the reactions of isatin derivatives
Effect of substituent on reactivity
Effect of 5- and 7- substituent G on the reactivity of amide carbon atom in isatin derivatives and feasibility of the leaving group is used to probe the reaction mechanism, Figure 2. Internal leaving group in isatin.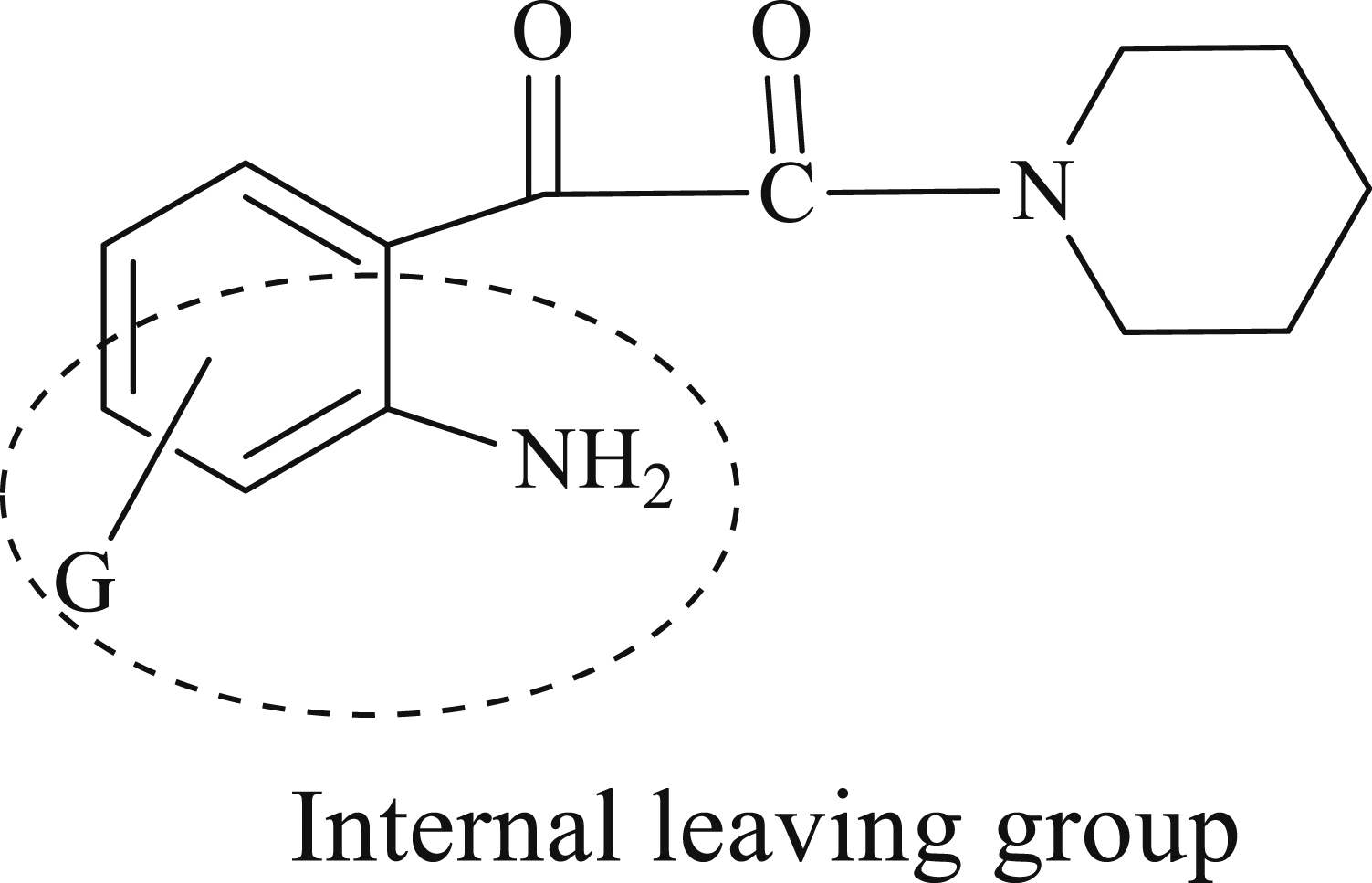
The electronic effect of the substituents in the leaving group moiety can be quantified by the use of a Hammett equation (6),
16
σ is the substituent constant and ρ is the reaction constant.
The value of ρ is a measure of the sensitivity of the rate constant for a given mechanism of a given reaction type to the influence of substituents.17–20 Table 2 reveals that electron withdrawing substituent enhances the reactivity while electron releasing substituent inhibit the rate9,21 [.and showed that the effect of substituents follows the order: 7-chloro > 7-bromo > 5-Chloro > 5-bromo > H > 7-CH3 > 5-CH3. The relative values of kN showed little dependence on the 5-substitutent and great dependence of 7-substituent.
This points out that the electronic effect of the five- and 7-substitutent is inductive rather than resonance. While the greater effect of 7-substituent than 5-substitutent is presumably due to proximity effect. The steric effect of the substituent can be ruled out because the G-substituents are placed too far away from the carbonyl carbon to provide a significant shielding effect.
The correlations of kN values with σ-Hammett-σ*-Taft’s (in one plot), σ-Hammett constants (for para-like substituent), σ*-Taft’s substituent constants (for ortho-like substituents) at various temperatures (20°–40°C) gave different ρ values as shown in Table 2. It shows that using these σ values is not suitable for the titled reaction due to (i) correlations between combined σ-Hammett and σ*-Taft’s and kN values for the reactions of isatin derivatives
The sign and magnitude of reaction constant ρ was used as mechanistic criteria to gain insight into the concerted or stepwise nature of the process.17,19,20 The sign of reaction constant ρ is invariably positive for stepwise reaction.24–26 When the ρ values are larger than 2, the reaction proceeds by a stepwise mechanism, Scheme 3 in which the rate-limiting is the expulsion of leaving group anion from T‒ (ρ ≈ 6.24 – 4.78).
27
i.e. rate-limiting breakdown of a tetrahedral intermediate T‒. When ρ is positive and smaller than 2 (ρ < 2) and more than 1, the reaction proceeds by a stepwise mechanism in which the rate-limiting step is the formation of the tetrahedral intermediate.
28
While for small ρ values (ρ < 1) indicated a concerted mechanism.
29
Scheme 4. For examples, ρ = 0.54 for the piperidinolysis of four- nitrophenyl X-substituted-benzoates
30
and ρ = −0.63 for aminolysis of aryl N-phenythio-carbamates.
31
It involved simultaneous bond breakage and bond formation in T±concerted, Scheme 4.
27
Concerted mechanism of piperidinolysis of isatin derivatives 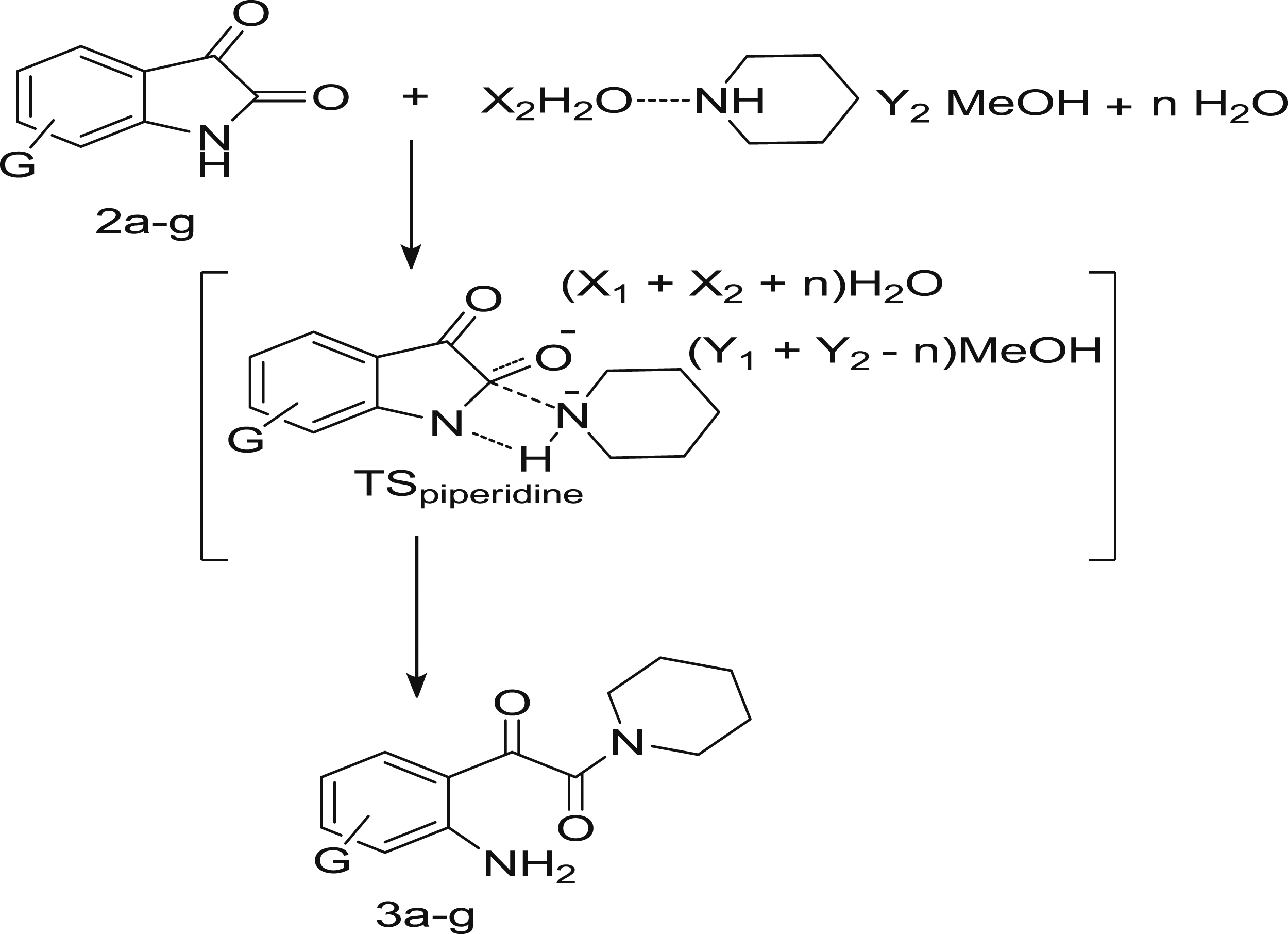
The ρ values of 2.01–1.86 for the titled reactions are compatible for a reaction mechanism shown in Scheme 3 with rate-limiting breakdown of the tetrahedral intermediate T¯. The relatively low ρ values can be explained by the responding to changes in the substituent on the leaving group. Such substituent can alter the partial positive charge on the carbonyl carbon and hence perturb the rate of bond formation between C-2 and nucleophile. He suggested mechanism is consistent with the explanation that an electron attracting substituents, for example, Cl, Br, would accelerate the rate of reaction and electron releasing group such as CH3 group inhibit the rate, while the rate determining step is the breakdown of tetrahedral intermediate, so the substituents will effect on the stability of the intermediate (I, and II). Thus, the inductive effect of Cl, and Br group increase the stability of the negative charge developed on the nitrogen atom of the intermediate (I) when it compared with the stability tetrahedral intermediate T‒. While electron donating substituent at the same positions such as CH3 group inductively decreases the stability of the intermediate (II) relative to the stability of tetrahedral intermediate T‒, (Figure 3). The effect of substituents is more significant on 7-positon than substituent on 5-position, that confirm that the main effect of the substituent is the inductive effect. Effect of electron withdrawing and donating groups on the intermediates (I) and (II).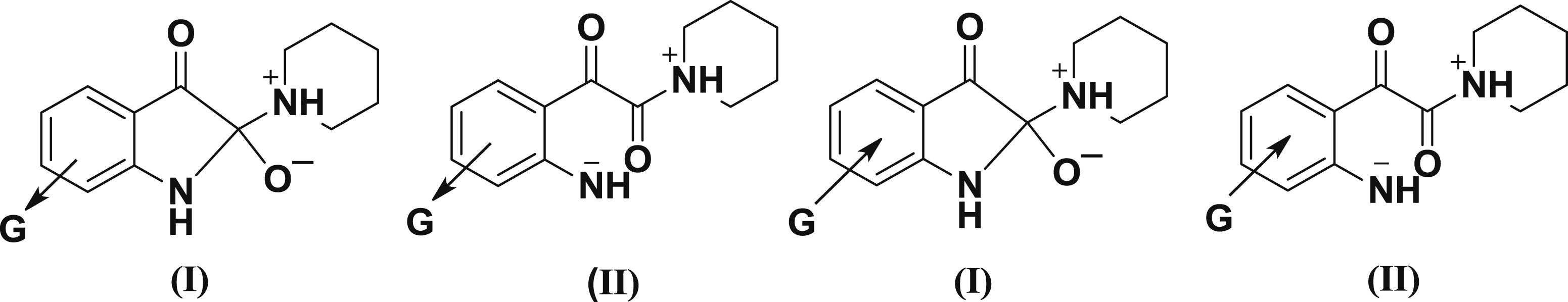
Conclusion
The five- and 7-substituted isatin derivatives (5- Me, 5-Cl, 5-Br, 7-Me, 7-Cl, 7-Br) were prepared by Sandmeyer methodology while isatin is commercially available. The reaction of 2a–g with piperidine in 80% aqueous MeOH at 25°C gave 1-(2’-amino-5’- or 3’- substituted phenyl)-2-(piperidin-1-yl)ethane-1,2-dione derivatives and their structures were confirmed by elemental analysis, IR and 1H-NMR spectroscopy. Thus, the reaction proceeds by nucleophilic attack at the carbonyl group at C-2 followed by ring opening process. The linear increase of pseudo first-order rate constants kobs at 25°C with negligible intercept against various excess of piperidine concentrations indicated that the reaction is not catalyzed by piperidine and second-order overall. Many facts are used to select the suitable mechanism for the titled reaction namely: (i) The need of water to complete the reaction of isatin derivatives with nucleophiles. (ii) The amine catalysis was rejected on the grounds that the reaction is second-order (iii) the kinetic dependence on nature of G-substituent are explicable in terms of the normal tetrahedral intermediates, Scheme 3. The previous facts, the activation parameter values and ρ values led to suggestion that the reaction of piperidine with of isatin derivatives proceeded by two step mechanism and the ring opening is catalyzed by water in the fast step.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

