Abstract
The reaction kinetics among isoquinoline, dimethyl acetylenedicarboxylate, and indole (as NH-acid) were investigated using ultraviolet (UV) spectrophotometry. The reaction rate equation was obtained, the dependence of the reaction rate on different reactants was determined, and the overall rate constant (kov) was calculated. By studying the effects of solvent, temperature, and concentration on the reaction rate, some useful information was obtained. A logical mechanism consistent with the experimental observations was proposed. Also, comprehensive theoretical studies were performed to evaluate the potential energy surfaces of all structures that participated in the reaction mechanism. Finally, the proposed mechanism was confirmed by the obtained results and the probable and logical reaction paths and also a correct product configuration were suggested based on the theoretical results.
Keywords
Introduction
Multicomponent reactions (MCRs) have been recognized as strong tools in modern drug discovery procedure since these types of reactions make it easy and rapid to access the structural variation and complexity of compounds through single-step conversions.1,2 Such reactions are rapid, well-diversified, efficient, atom-economic, and environment-friendly.3,4 Due to the advantages mentioned above, developing new MCRs with environmentally benign protocols has been recognized as one of the most important topics in synthetic chemistry.5–7 MCRs display many of the most desired features for an organic synthesis,8–10 and they have been intensively studied in recent years.11–14 Heterocyclic rings are the fundamental components in the skeleton of more than half of the biologically active compounds produced by nature. 15 As an example, aza-aromatic compounds that are activated by acyl chlorides or dimethyl acetylenedicarboxylate (DMAD) are important intermediates for the synthesis of a variety of biologically active nitrogen-containing alkaloids.16,17 This is why great efforts have been made to discover and optimize methods to construct new heterocycles.
Isoquinoline skeleton can be found in a large number of naturally occurring18–21 and biologically synthesized active heterocyclic compounds.22,23 In particular, 1,2-dihydroisoquinoline derivatives act as delivery systems that transport drugs through the highly impermeable blood−brain barrier.24,25 These compounds also exhibit sedative,
26
antidepressants,
27
antitumor, and antimicrobial activities.28,29 With wide kinetic studies been done in the synthesis of phosphorus ylides,30–34 the structural effect of the reactant (different NH-acids and dialkyl acetylenedicarboxylates) on the reaction rate has been well investigated. Changes in the reactant structure that participates in the reaction, eventuates changes in the overall reaction rate. Evaluating these changes could reveal many facts about the confirmation of the reaction mechanism. A number of our projects, including the theoretical and experimental studies of chemical reaction kinetics, have been completed.35–41 Now, by the synthesis of the novel compounds via a pseudo-three-component reaction, in the presence of isoquinoline
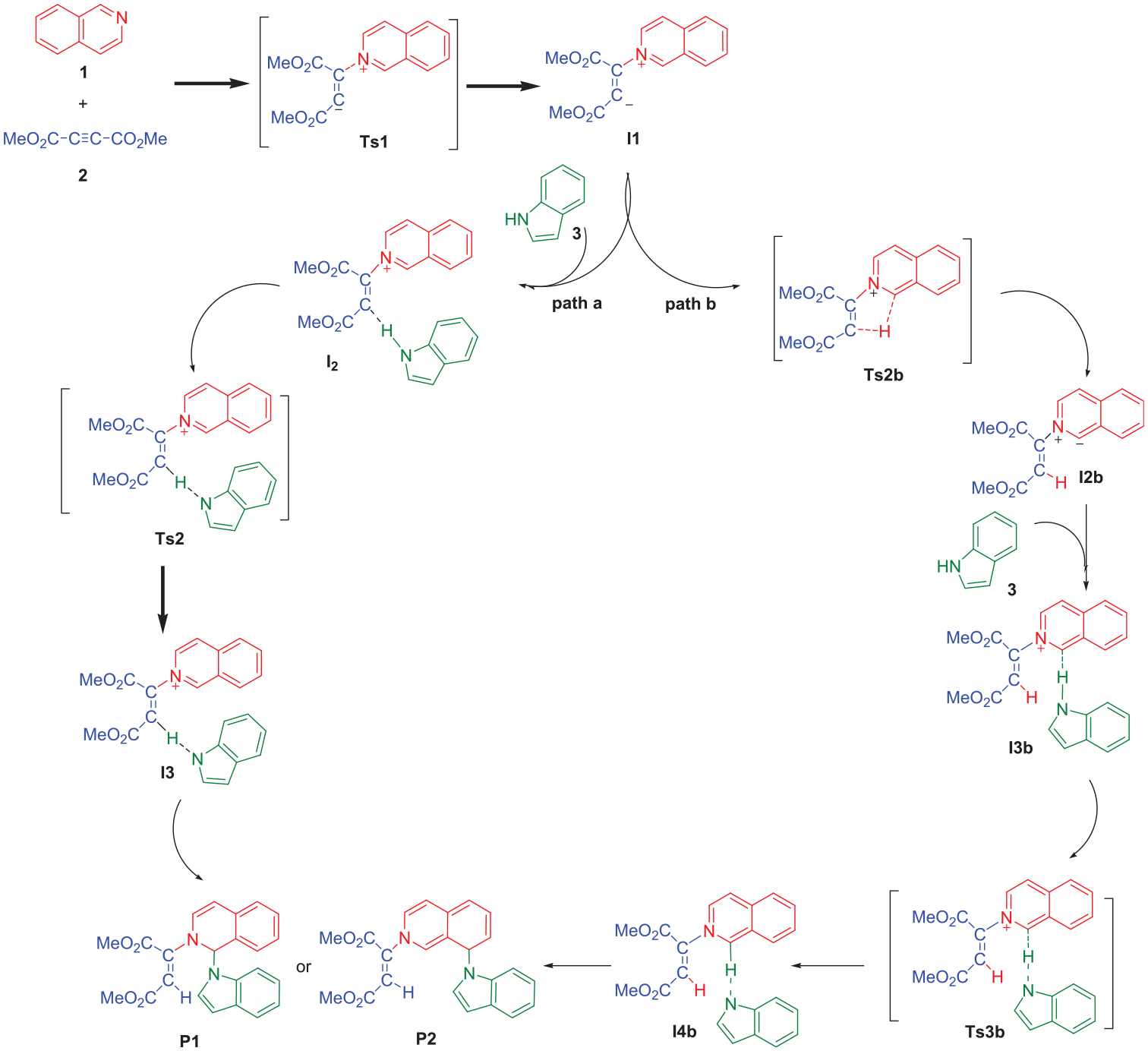
The reaction among isoquinoline
Materials and methods
DMAD, isoquinoline, and indole, purchased from Fluka (Buchs, Switzerland), were used without further purifications. All the other pure solvents including dichloromethane and acetone were purchased from Merck (Darmstadt, Germany). A Cary UV/Vis spectrophotometer model Bio-300 (Australia) was also applied throughout this work. All geometrical structures were optimized at B3LYP/6-311++G (2d, 2p) level of theory using the Gamess suite software package. 47 The corresponding frequencies of the structures were estimated at the same level of theory to check the stationary points without imaginary frequencies and the transition states with only one imaginary frequency. Also, the intrinsic reaction coordinate (IRC) approach48,49 was followed to ensure that the given transition state is related to the corresponding reactants and products. The solvent effect on complex stability was examined using the B3LYP/6-311++G (2d, 2p) method by applying the polarizable continuum model (PCM).
Results and discussion
Experimental studies
The first step in investigating the reaction kinetics is to determine the appropriate wavelength. So, for this purpose, the UV spectra of 3-mM dichloromethane solution of compounds
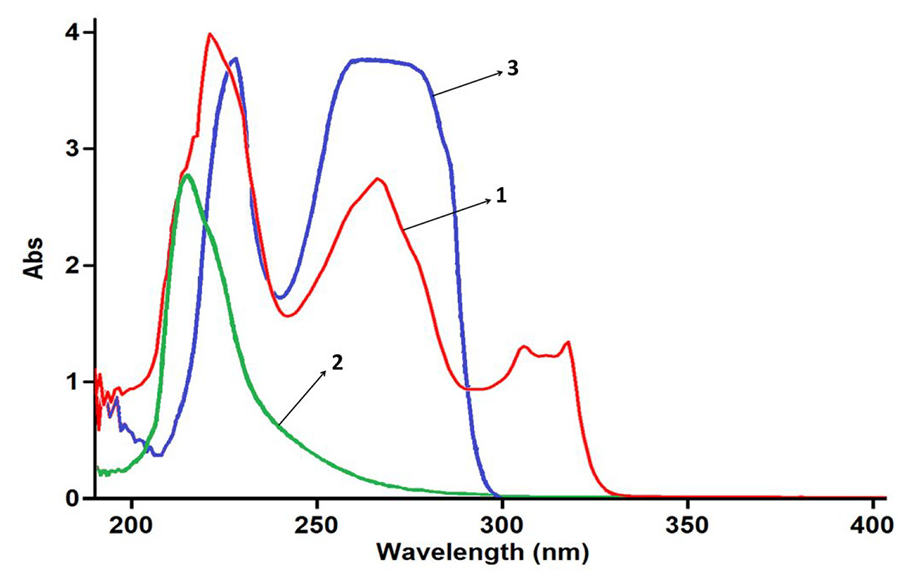
The plot of absorbance versus wavelength of the solution 3-mM isoquinoline
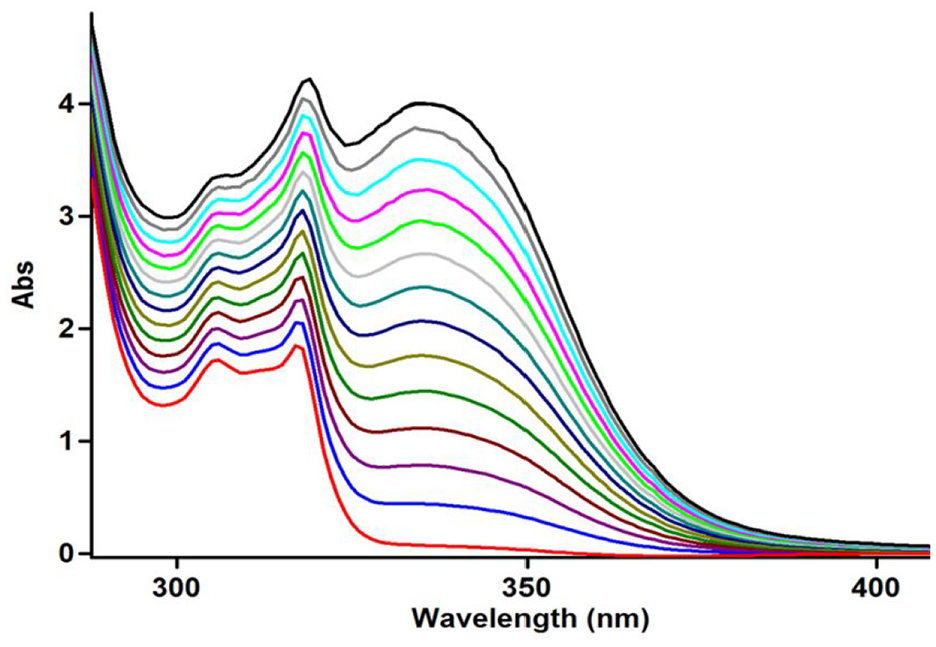
Expanded section of UV spectra (an increasing absorption) over the wavelength range 280–400 nm for the reaction among
The reaction kinetics were then followed by plotting UV absorbance versus time for the reaction among
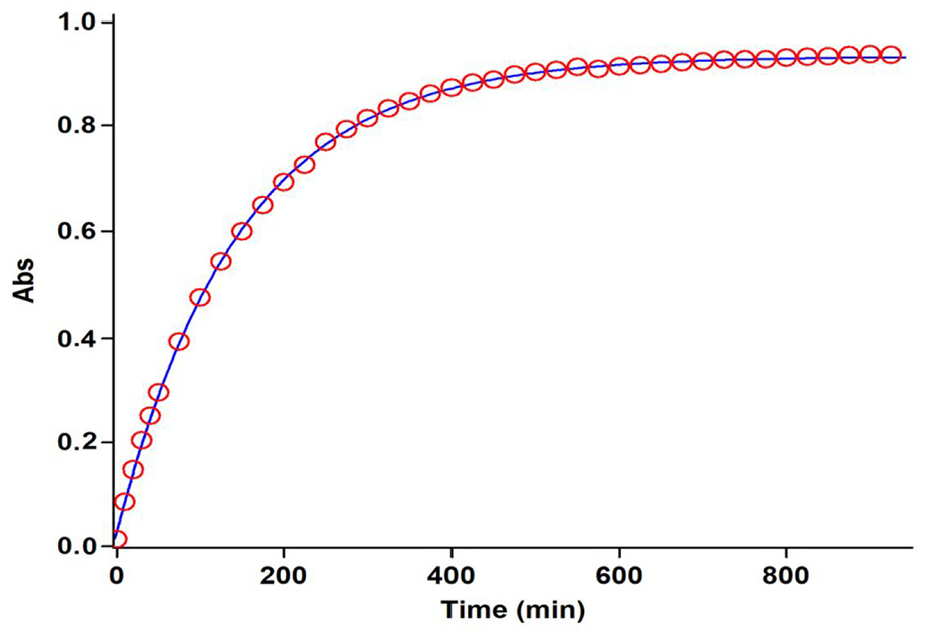
The experimental absorbance changes (dotted line) versus time accompanied by the second-order fit curve (solid line) for the reaction among
Using nonlinear regression analysis,
50
the data of original experimental absorbance versus time provided a pseudo-first-order curve (solid line) that exactly coincides with the experimental curve (dotted line), as shown in Figure 4. The pseudo-first-order rate constant (kobs) was also calculated at 15.0°C. The observed pseudo-first-order rate constant (kobs) indicates that the order of reaction concerning the sum of compounds
Continuing the experiments, the reaction among
The data of original experimental absorbance versus time were used to provide a pseudo-first order fit curve (solid line) at 370 nm, which exactly coincides with the experimental curve (dotted line). In this case, the observed pseudo-first order rate constant
To further confirm the above results, all kinetics were studied in the presence of isoquinoline
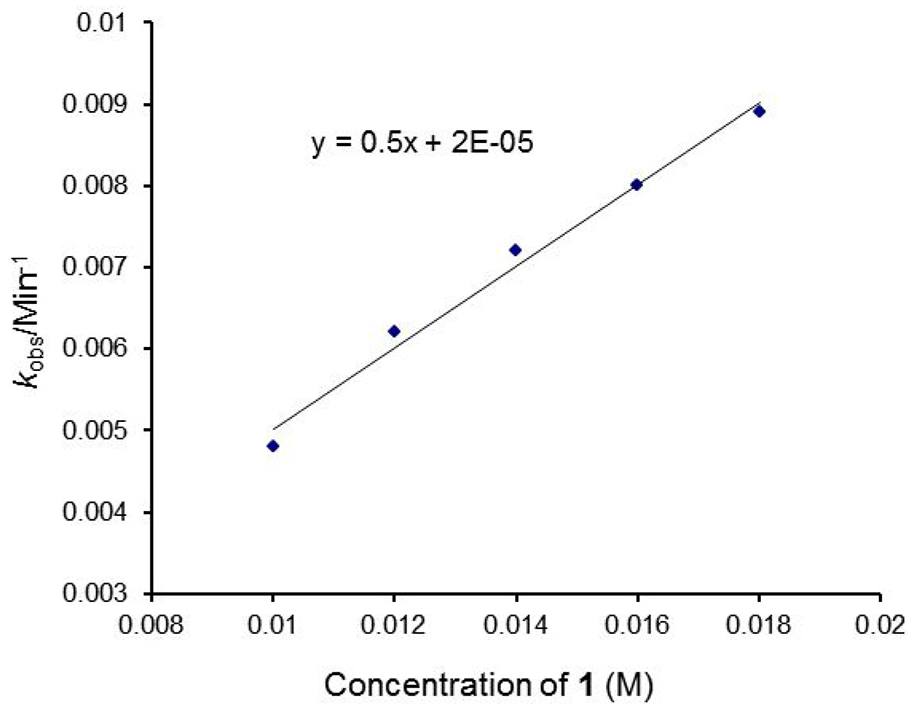
Observed rate constant
Values of the overall second-order rate constant (k2) for the reactions of

To determine the effects of temperature and solvent environment on the reaction rate, a set of experiments was conducted at different temperatures and solvent polarities with other conditions being the same as those of the previous experiments. For this purpose, acetone, with the dielectric constant of 20.7 was replaced by dichloromethane with a dielectric constant of 8.9. Based on the results, the rate of reaction was accelerated in an environment with a higher dielectric constant (acetone). Also, at all the investigated temperatures, the results indicate an increase in reaction rates at higher temperatures (Table 1). In the studied range of temperatures, the reaction activation energy was calculated from the slope of the pseudo-second-order rate constant (log kov) versus reciprocal temperature (1/T).
Rate equation
Based on the above results, a simplified proposed reaction mechanism is depicted in Figure 6. Unlike the experimental results, it may be assumed that the second step is the rate-determining step for the proposed mechanism. In this case, the rate equation can be expressed as follows

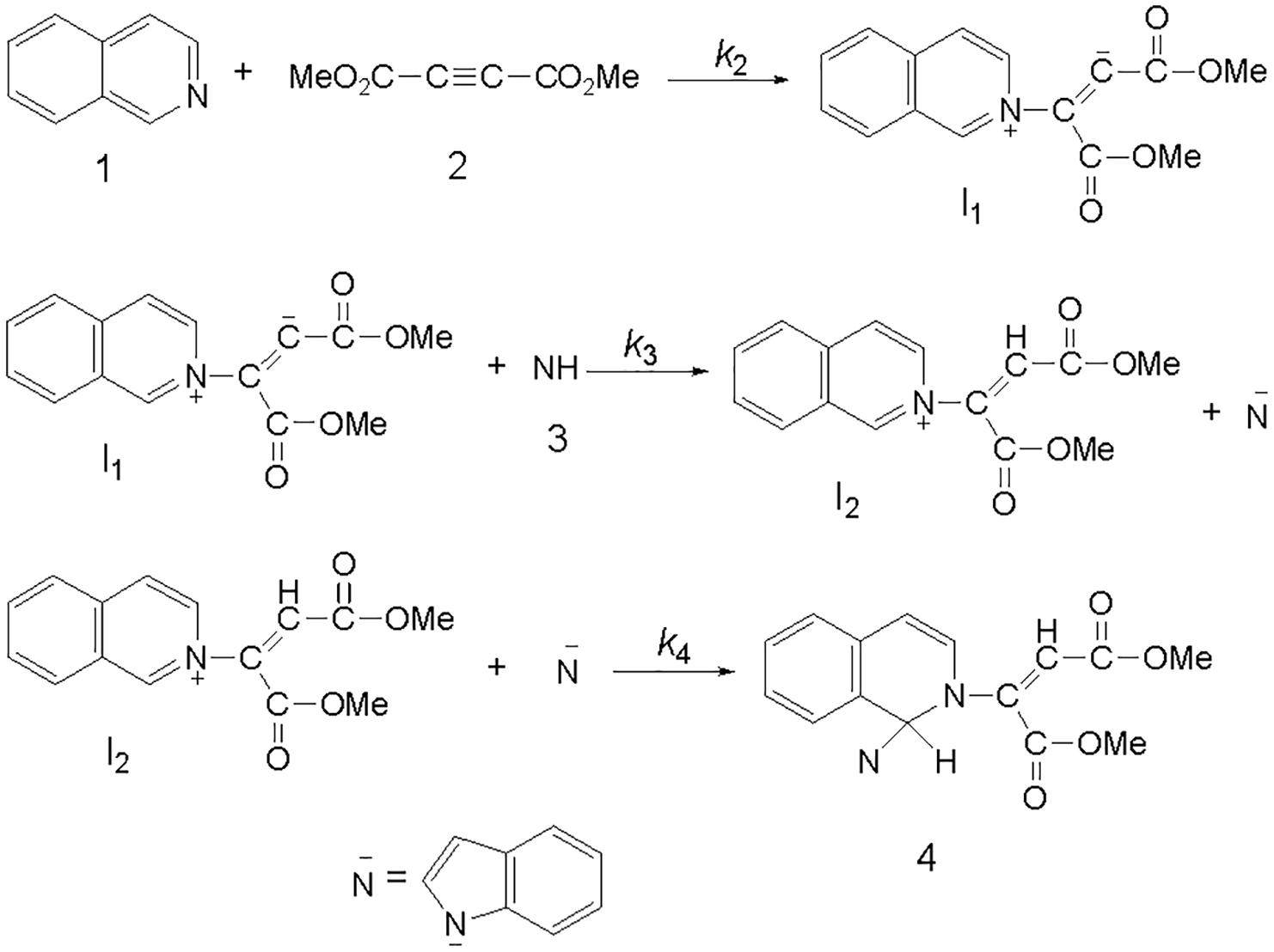
Proposed mechanism for the reaction among
The final equation indicates that the overall order of the reaction is three which is not compatible with experimental overall order of reaction (= 2). Besides, according to this equation, the order of reaction to indole
If we assume that the third step (rate constant k4) is the rate-determining step for the proposed mechanism, in this case, there are two ionic species to consider in the rate-determining step (I2 and N−) (Figure 6), have full positive and negative charges and form very powerful ion–dipole bonds to the acetone as a higher dielectric constant solvent. However, the transition state for the reaction between two ions carries a dispersed charge and bonding of solvent (acetone) to this dispersed charge would be much weaker than the reactants. The solvent, thus stabilizes the ionic species, more than it would the transition state. Therefore, the activation energy could be raised and slow down the reaction. However, in practice, acetone speeds up the reaction and for this reason, the third step could not be the rate-determining step. Furthermore, the rate law can be expressed as

The final equation also indicates that the overall order of the reaction is three, and the order of reaction for indole
If the first step (rate constant k2) was the rate-determining step, in this case, bonding of solvent to the dispersed charge in the transition state is much stronger than the uncharged reactants. The solvent thus stabilizes the transition state more than it does the reactants and speeds up the reaction. Our experimental results show that the solvent with higher dielectric constant exerts a powerful effect on the rate of reaction (see Table 1). Results indicate that the first step (k2) of the proposed mechanism could be the rate-determining step. However, a good kinetic description of the experimental result using a mechanistic scheme based upon the steady-state approximation is frequently taken as an evidence of its validity. By application of this, the rate of the formation of product

By replacing the value of [I1] and [I2]

This equation is compatible with the results obtained by UV spectrophotometry. For equation (6) that shows the overall reaction rate, the activation parameters involving ΔG#, ΔS#, and ΔH# could be now calculated for the first step (rate-determining step), as an elementary reaction, based on Eyring equation. The results are reported in Table 2.
Activation parameters for the reactions

The activation parameters (ΔG#, ΔS#, and ΔH#) of the first step (rate-determining step), as an elementary reaction, can be calculated based on the Eyring equation. The results are reported in Table 2.
Theoretical study
The overall order of reaction and the order of reaction to different reactants, solvent, and temperature effect and also the detection of rate-determining step, and finally, the reaction mechanism as well are determined in the experimental section. Now, to gain more insight into the reaction mechanism, we intend to consider the mechanism of this reaction using theoretical calculations.
In this section, we will evaluate the potential energy levels, different kinetic paths, and energy levels of different products. Also, we intend to determine the preferred thermodynamic product, the kinetic preferred path, and to confirm the rate-determining step, as outlined in the experimental section. All structures were optimized by density functional theory at B3LYP/6-311++G (2d, 2p) level of theory. Two different pathways (
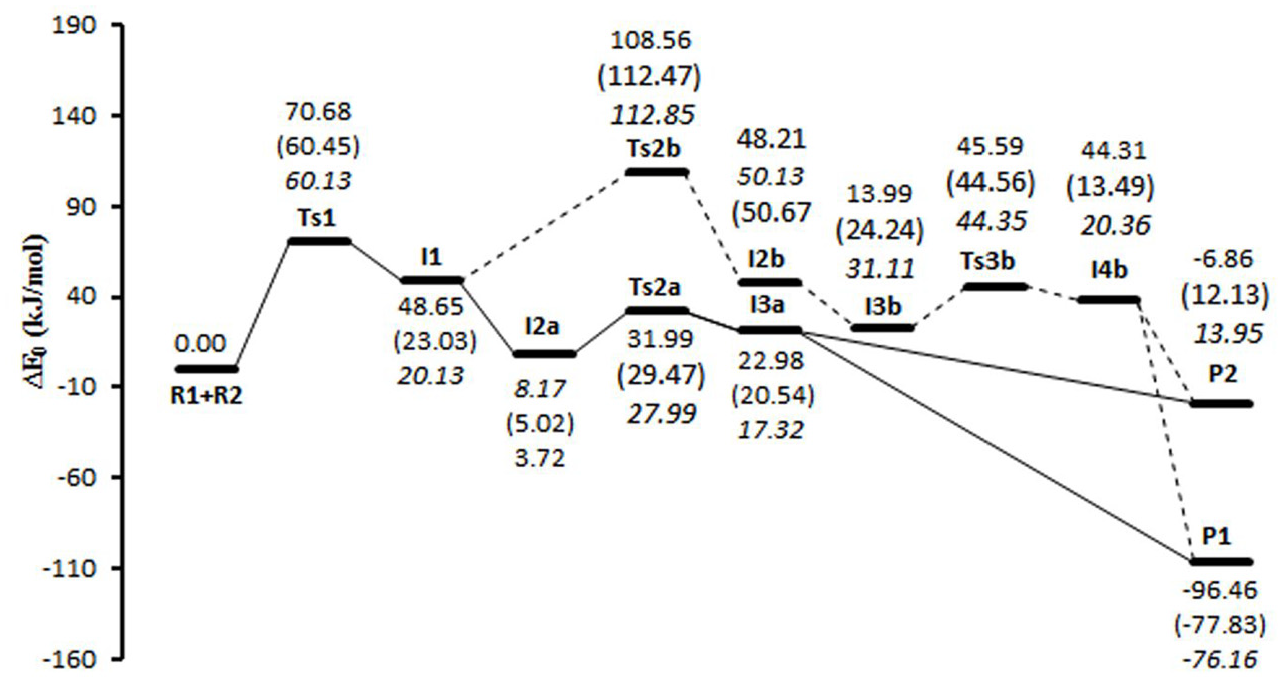
The potential energy profile of the reaction in paths
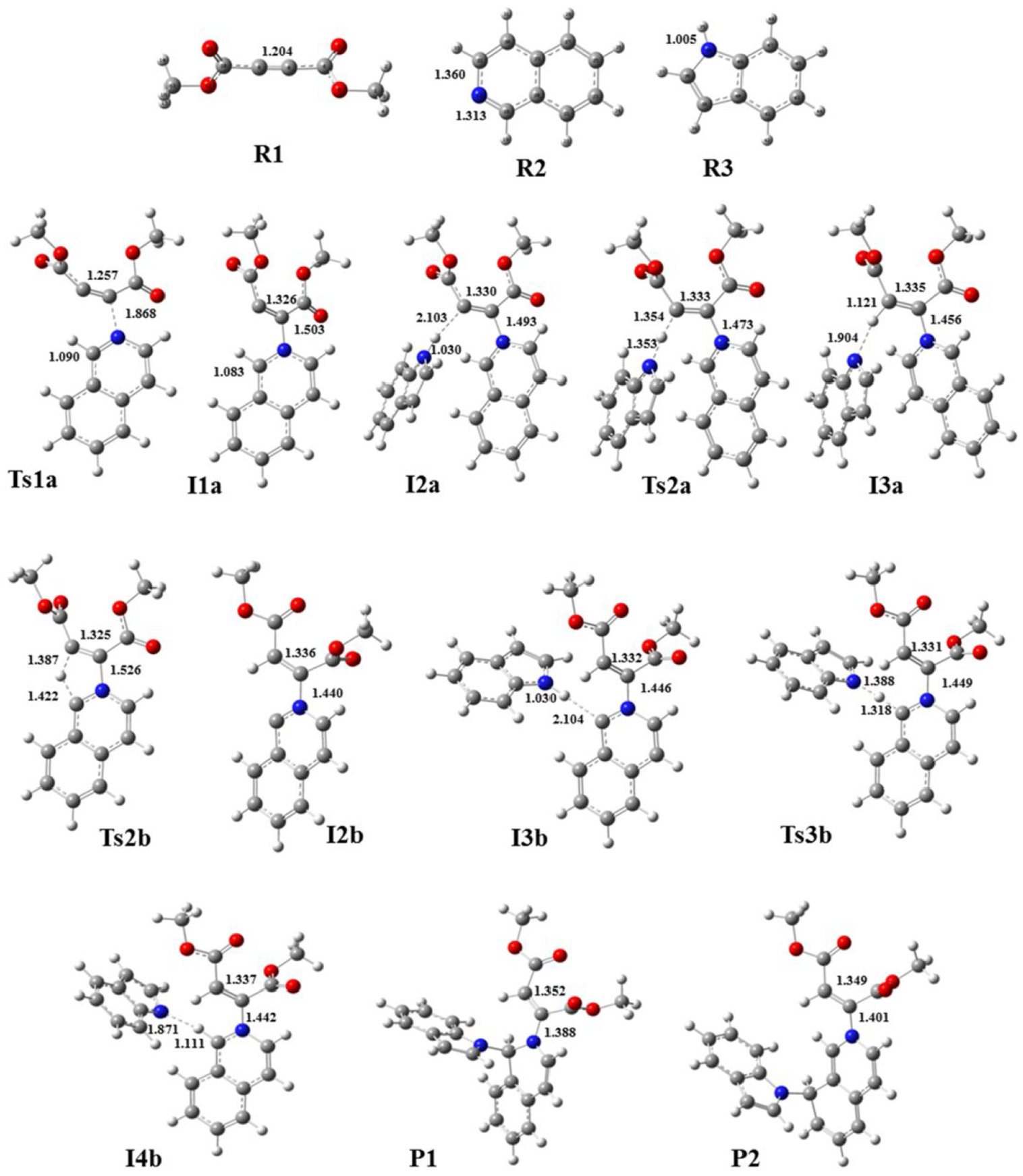
Optimized geometries of all structures in pathways
Investigation of the potential energy surfaces in the pathways
To check the effect of solvent on the potential energy surfaces, dichloromethane was subjected to condensed phase calculations carried out using the PCM. As shown in Figure 6, the energy barrier of the first and the third step of the pathway
Conclusion
Mechanistic investigation of the reaction among isoquinoline
The kinetics of the reaction were studied by measuring the increase in the product absorption rate. The overall reaction order follows the pseudo-second-order kinetics with the first-order dependent on the concentration of isoquinoline
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors sincerely thank Payame Noor University for providing financial support for this work.


