Abstract
The effects of metallic cobalt crystal phase on catalytic activity of cobalt catalysts in the Fischer–Tropsch synthesis were investigated in a continuous spinning basket reactor. The cobalt catalysts were prepared by impregnation of the cobalt active phase in a microemulsion system on multiwall carbon nanotube supports. A series of cobalt catalysts with different Co particle sizes was prepared by variation of the water-to-surfactant molar ratio from 2 to 12 in the microemulsion system. The X-ray diffraction results validate a complex composition of cobalt phases containing cobalt oxides and metallic cobalt with hexagonal close-packed and face-centered cubic phases. The results show that larger cobalt particles exhibit more face-centered cubic and less hexagonal close-packed metallic cobalt. The experimental results show that the catalysts with higher fractions of hexagonal close-packed phase exhibited higher conversion in the Fischer–Tropsch reaction.
Introduction
The Fischer–Tropsch synthesis (FTS) is a potential option for production of chemicals and fuels from coal and natural gas.1,2 Supported cobalt catalysts have considerable potential in the FTS reaction, due to their high activity and high selectivity toward heavier hydrocarbons.3,4 As reported in the literature, the cobalt phase composition in cobalt nanoparticles could also affect FTS catalytic performance.5,6 Recent improved experimental structure determinations showed that the cobalt hexagonal close-packed (hcp) phase shows higher activity in the FTS reaction than the face-centered cubic (fcc) phase, and the catalysts with higher fractions of hcp phase exhibited higher activity in the FTS reaction. 5
The cobalt-based catalysts with high specific rates in the FTS reaction require the synthesis of small metallic cobalt crystallites with high surface densities on the support and the utilization of supports that amplify the rate per surface of cobalt active phase (turnover rate).7,8 Nevertheless, interaction between the support and metallic cobalt active phases leads to a drop off of the catalyst reduction efficiency.9,10 Carbon nanotubes (CNTs), as a cobalt catalyst support, allow better metal dispersion control and minimize the metal phase interaction (formation of mixed compounds) with the support.11,12 In addition, the metallic cobalt particle size supported on CNTs may affect catalytic activity in the FTS reaction.13–15 Thus, CNTs as a new type of carbon material have shown attractive properties and high catalytic activity for FTS cobalt catalysts. In addition, CNT-supported cobalt catalysts are the best example for evaluation of particle size effects in supported cobalt catalysts because of the low interaction between support and cobalt nanoparticles.12,15–17
In the present work, five Co/CNT model catalysts with Co particle sizes in the range of 4.9–12.4 nm were prepared to examine the possible cobalt phase effect in FTS synthesis. Transmission electron microscopy (TEM) and X-ray diffraction (XRD) techniques were used to characterize the particle size distribution.
Experimental
Catalyst preparation
The cobalt catalysts were prepared with 15 wt% cobalt on CNT supports by incipient wetness impregnation and microemulsion techniques, as reported in our previous work. 18 In the microemulsion method, to complete a series of catalysts with different cobalt particle sizes, the surfactant-to-oil weight ratio was adjusted to a value of 0.3 and then the water-to-surfactant (W/S) molar ratio was varied from 2 to 12. The different catalysts prepared are listed in Table 1.
Average Co particle size of fresh catalysts.
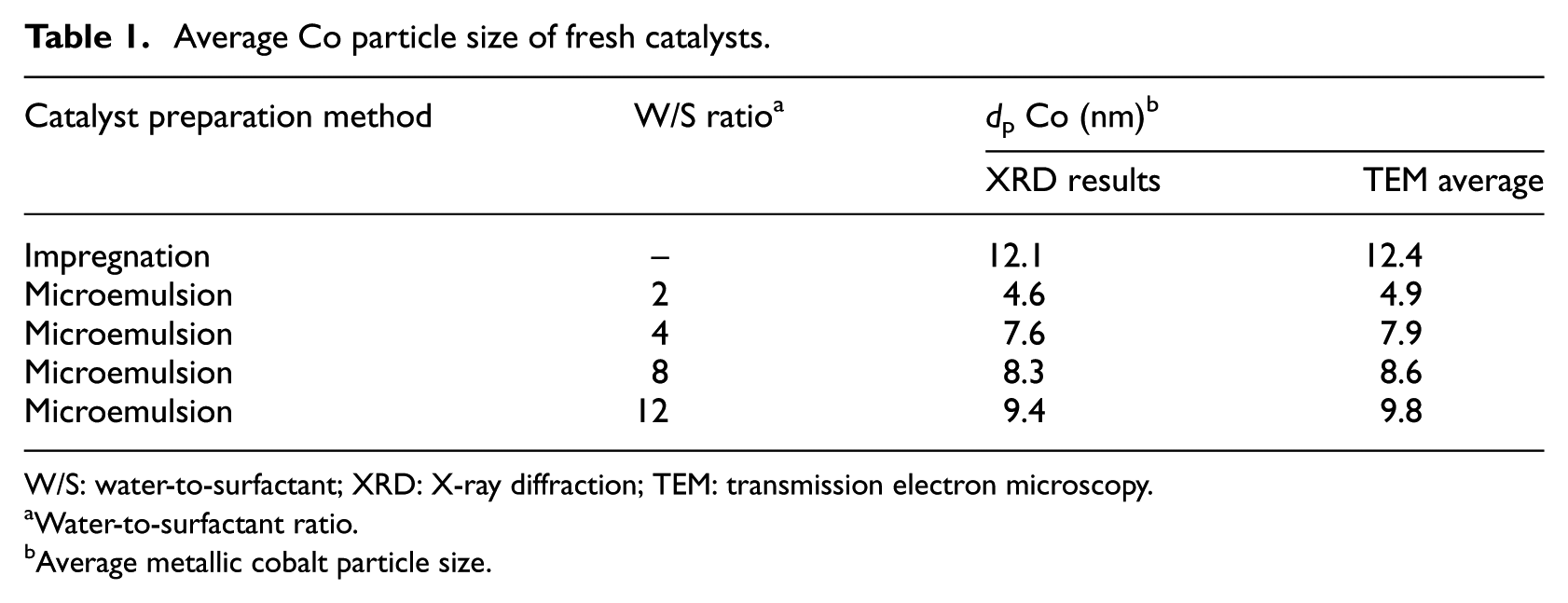
W/S: water-to-surfactant; XRD: X-ray diffraction; TEM: transmission electron microscopy.
Water-to-surfactant ratio.
Average metallic cobalt particle size.
Catalyst characterization
XRD was conducted on a Philips PW1840 X-ray diffractometer with monochromatized Cu/Kα radiation and the phase detection was made using the JCPDS database. The average cobalt oxide crystallite thickness in fresh samples was calculated from the Scherrer equation using the (311) Co3O4 peak placed at 2θ = 36.9° and Scherrer factor (K) of 0.89. The Co3O4 crystallite size was converted to the corresponding cobalt metal particle size according to the comparative molar volumes of metallic cobalt and Co3O4. The resulting conversion factor for the diameter of a given Co3O4 crystallite size (dXRD(Co3O4)) being reduced to metallic cobalt (dXRD(Co0)) is
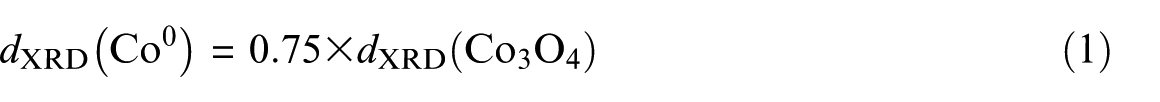
where dXRD (nm) is the crystallite diameter calculated from the Scherrer equation. After use in the reaction, the catalysts were passivated with a 1 vol% O2/He mixture at room temperature for 1 h, according to a standard method described elsewhere, and XRD determinations of used samples were made after passivation. 19
The morphology of prepared cobalt catalysts after calcination was observed with a transmission electron microscope (LEO 912 AB; Germany). A suitable quantity of catalyst suspension that was taken straight from the sonicator was dropped onto the carbon-coated copper grids for TEM observation. The average diameter of particles calculated from TEM images (dTEM) and particle size distribution were also determined by TEM images by counting more than 100 particles in each micrograph.
Catalyst activity
The FTS reaction rates were determined in a spinning basket reactor (stainless steel, H = 0.122 m, D0 = 0.052 m, Di = 0.046 m) with temperature controllers (WEST series 3800) and Brooks mass flow meters for hydrogen and carbon monoxide. A detailed description of the experimental setup and procedures has been provided in our previous work.20,21 The weight of the catalysts loaded was about 2.5 g, which was diluted with 30 mL inert silica (with the same mesh size range). The fresh catalysts were reduced in situ with purified hydrogen for 12 h (at 673 K, 1 bar, and 3.6 NL gcat−1 h−1, where NL is normal liter and gcat is the weight in grams of catalyst) before FTS evaluation. After pretreatment, the FTS tests were carried out at T = 493 K, 20 bar, H2/CO = 2 and 2.4 NL gcat−1 h−1 feed rate.
Throughout the complete runs, the reactor temperature was changed from 493 to 508 K, the pressure was 20 bar, the space velocity of the synthesis gas was 4.8 NL gcat−1 h−1, and H2/CO was 2. Conversion of carbon monoxide and hydrogen, and formation of various products were considered within a time of 24 h at each run. CO conversion and product selectivity were based on gas chromatographic analyses and carbon balance. Both total mass and atomic material balances were performed with the condition that a run could be accepted for further analysis if the carbon material balance was between 97% and 103%. This standard was adopted since compounds containing carbon and hydrogen may collect in the reactor in the form of high molecular weight hydrocarbons.
The products were analyzed using three gas chromatographs. A Shimadzu 4C gas chromatograph set with the two following associated packed columns: Porapak Q and Molecular Sieve 5A, and a thermal conductivity detector (TCD) with argon as carrier gas was used for hydrogen analysis. A Varian CP 3800 gas chromatograph with a Chromosorb™ column and a TCD was used for CO, CO2, CH4, and other non-condensable gases. A Varian CP 3800 with a Petrocol™ DH100 fused silica capillary column and a flame ionization detector was used for organic liquid products such that a full product distribution could be provided.
Results and discussion
Catalyst characterization
XRD patterns of the fresh Co/CNT catalysts after calcination are shown in Figure 1. In the XRD spectrum, the peaks at 2θ = 25° and 43° match up to the CNT support, the peak at 2θ = 36.8° relates to the (311) Co3O4 peak, and there are minor peaks at 44° (400), 59° (511), and 65° (440), associated with the cubic spinel structure of Co3O4, based on JCPDS 78-1970. 12 The average cobalt oxide crystallite size was calculated from the Scherrer equation using the (311) Co3O4 peak located at 2θ = 36.9° and is listed in Table 1. The crystallite sizes of Co3O4 particles calculated from the XRD pattern were changed to the equivalent cobalt metal particle sizes (according to equation (1)) and are shown in Table 1. As shown in Table 1, the average particle size of cobalt nanoparticles depended linearly on the respective W/S ratio in the microemulsion system.
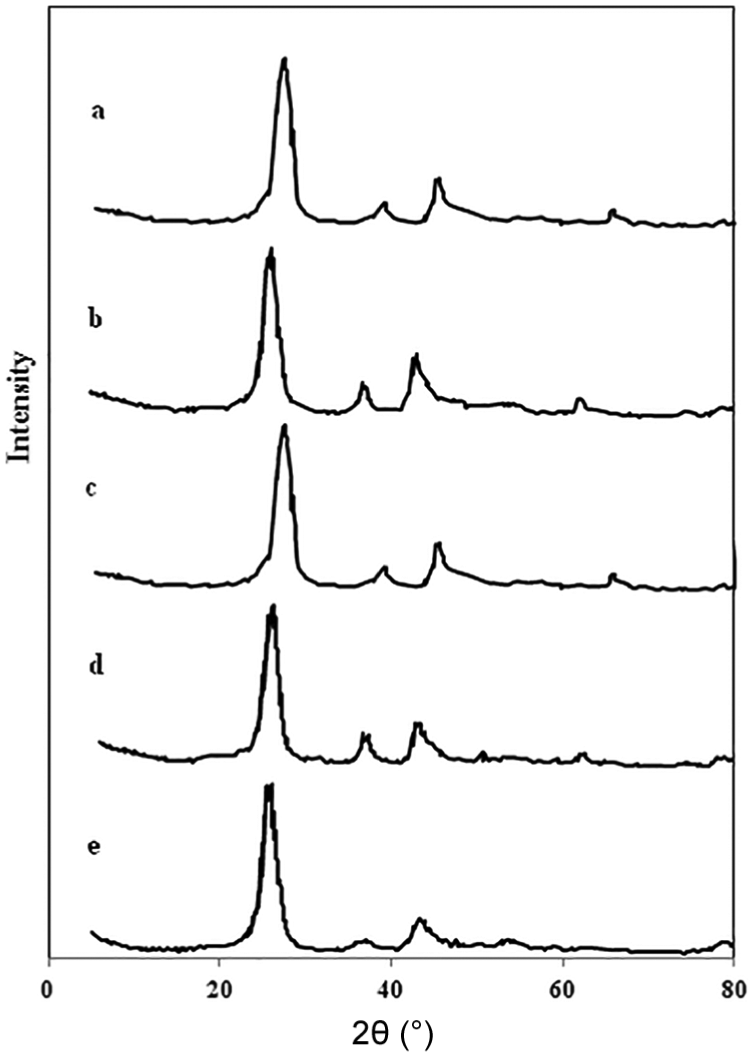
XRD patterns of fresh catalysts after calcination: (a) catalyst prepared by impregnation method, (b) catalyst prepared by microemulsion method and W/S = 12, (c) catalyst prepared by microemulsion method and W/S = 8, (d) catalyst prepared by microemulsion method and W/S = 4, and (e) catalyst prepared by microemulsion method and W/S = 2.
Figure 2 shows the XRD patterns of the Co/CNT catalysts reduced at 673 K after passivation. As shown in Figure 2, the XRD results verify a multipart composition of nanoparticles containing cobalt oxides and metallic cobalt with hcp and fcc phases. In the XRD spectrum of passivated samples, the peaks at 2θ values of 42°, 48°, 76°, and 92° correspond to the metallic cobalt with hcp phase based on JCPDS 05-0727, and the peaks at 2θ values of 45°, 52°, and 98° correspond to metallic cobalt with fcc phase based on JCPDS 01-1259.22,23 As shown in Figure 2, the samples with considerably larger particle size exhibit more fcc and less hcp metallic cobalt.
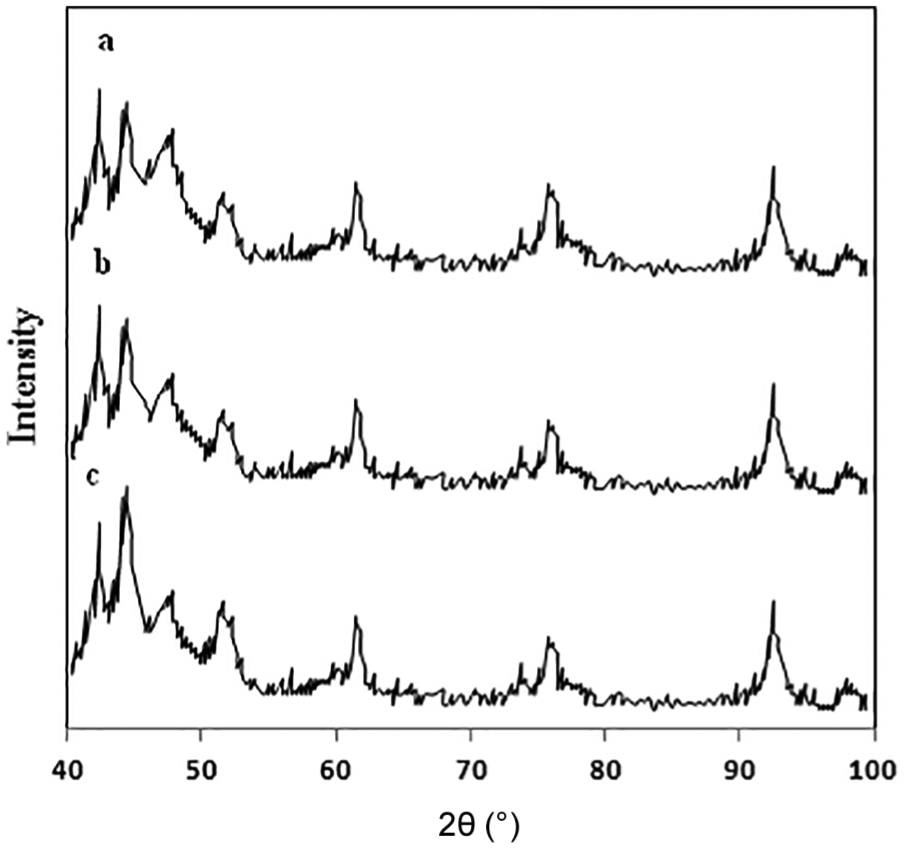
XRD patterns of catalysts reduced at 673 K after passivation: (a) catalyst prepared by microemulsion method and W/S = 4, (b) catalyst prepared by microemulsion method and W/S = 8, and (c) catalyst prepared by impregnation method.
The TEM image of the fresh catalyst sample prepared at W/S = 4 is shown in Figure 3. The average particle size (dTEM) was determined by counting more than 100 particles in TEM images using the average Feret diameter, as reported in previous work, and is listed in Table 1.4,17,18,24 The dXRD value provides information on particle size which was derived indirectly. However, the dTEM value is a direct measure of average particle size of cobalt nanoparticles and thus, it has been used in further consideration.
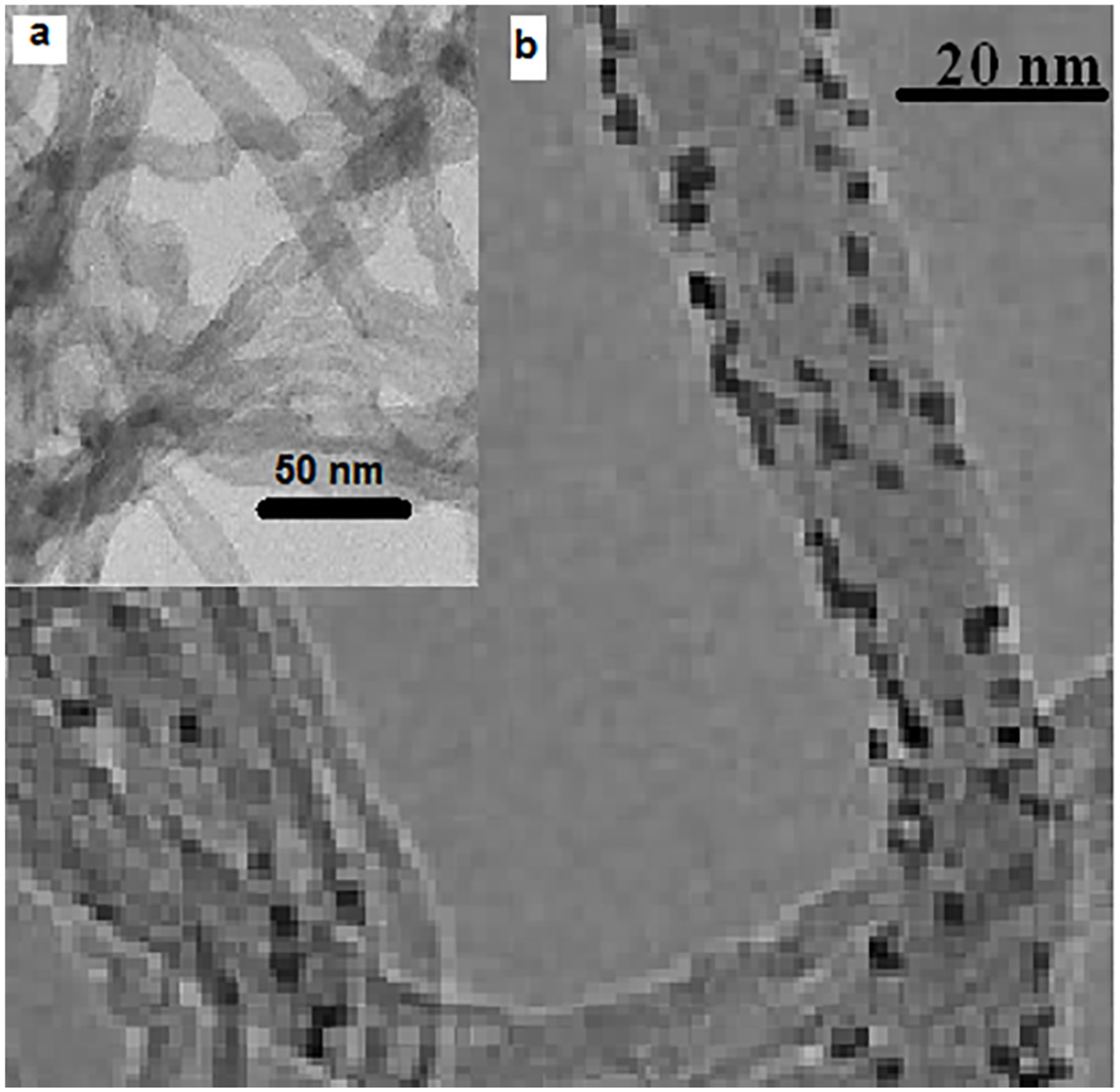
TEM images of (a) CNT support and (b) Co/CNT with W/S ratio of 4.
Fischer–Tropsch activity
The series of Co/CNT catalysts are excellent candidates to study particle size effects in the FTS reaction. Their catalytic activity in the FTS at T = 493 and 508 K, P = 20 bar, H2/CO = 2 and space velocity of the synthesis gas equal to 4.8 NL gcat−1 h−1 has been examined. The effects of temperature and catalyst particle size on FTS reaction rates are shown in Figure 4 where the catalyst particle size is the radius of metallic cobalt nanoparticles and RFTS is the rate of CO conversion to organic products. As shown in Figure 4, the FTS reaction rate was increased by increasing the reaction temperature and passed through a maximum on decreasing the catalyst particle size. These results show that the FTS reaction is a size-dependent reaction and is affected by the structure of the catalyst active sites. These complexities (a maximum in FTS activities against catalyst particle size) make it difficult to establish a simple correlation between catalyst activity and particle size. As reported in the previous section, larger particle size is associated with more fcc and less hcp metallic cobalt. As reported in the literature, the cobalt phase composition in cobalt nanoparticles could also affect FTS catalytic performance. Recent and more accurate experimental structure determinations clearly demonstrate that the cobalt hcp phase shows higher activity in the FTS reaction than fcc phase and the catalysts with higher fractions of the hcp phase exhibited higher conversion in the FTS reaction. 5 This suggests that cobalt sites situated on the cobalt hcp phase might have higher specific activity in the FTS reaction. The origin of this difference in FTS activities is not clear and can be explained by the FTS reaction mechanism. Thus, higher activities of smaller cobalt particle size are related to higher fractions of hcp metallic cobalt phase, but the origin of the lower activity of catalysts with cobalt particle size of 4.8 nm will require more attention.
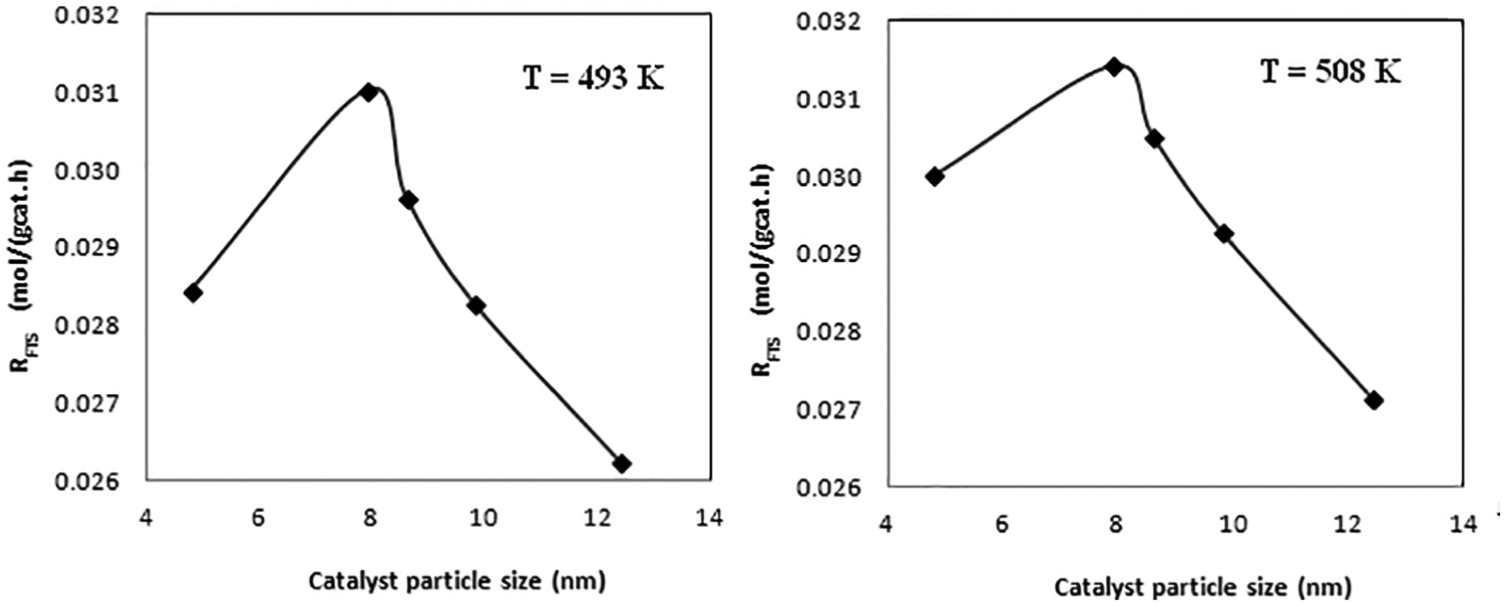
Effects of temperature and catalyst particle size on FTS reaction rates.
Conclusion
A series of Co/CNT catalysts with various particle sizes was prepared and used to examine the structure sensitivity of the FTS reaction. The catalyst activity results show that the FTS reaction rate increased on increasing the reaction temperature and passed through a maximum by decreasing the catalyst particle size. The higher activity of catalysts with small particle size may be related to the greater hcp metallic cobalt content in the catalyst active phase (XRD results).
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
