Abstract
Changing the mechanism of the widely used 1,3-dipolar cycloaddition reaction from its usual asynchronous one-step pattern to the rarely observed stepwise form leads to the emergence of intermediates, side products, and other impurities. Thus, it is crucial to determine the nature of the mechanism of the 1,3-dipolar cycloaddition reaction between a special 1,3-dipole and a specified dipolarophile (by theoretical methods) before using them for synthesizing a desired product. In this study, therefore, we have investigated the possibility of some probable intermediates emergence in the 1,3-dipolar cycloaddition reaction between cyclobutadiene and thioformaldehyde S-oxide. The results showed that emergence of Int (B) (−52.1 kcal mol−1) via transition state (B-1) is favorable both thermodynamically and kinetically (in comparison with all other stepwise routes). That is, developing probable impurities should not be neglected at least in the cases of the reactions between some thioformaldehyde S-oxide and some dipolarophiles.
Keywords
Introduction
In the catalyst-free 1,3-dipolar cycloaddition (1,3-DC) reactions, the diversity of the side products and stereoisomers strongly depends on the nature of the mechanism of the process, as the stereospecificity is lost when the mechanism of the 1,3-DC reactions is changed from the usual one-step model to the rarely observed stepwise form.1–4 In the recent decades, this individual fact has attracted the attention of dozens of researches all around the world in their rigorous efforts to find the exact mechanism of this reaction. Due to the fact that each dipole group could exhibit especial behaviors and show different behavior in the 1,3-DC process, it is particularly important to investigate the reaction of each dipole individually. 5
Since 1986 and the Huisgen’s discovery of the first stepwise example in the 1,3-DC reaction between a thioformaldehyde S-oxide and an electron-poor dipolarophile, 6 many efforts have been aimed at finding more stepwise examples. However, most reports on the stepwise cases belonged to the thioformaldehyde S-oxide7,8 and the nitrile oxides.9–11 Furthermore, it seems, save the type of the dipole group, there are some other alternatives that could effect on changing the mechanism of the reaction from one step to stepwise, and vice versa. For example, a report revealed that by introducing toluene as solvent for the 1,3-DC reaction between a highly electronegative gem-dinitroethene and (Z)-C, with N-diphenylnitrone as the dipole species, the reaction mechanism shifts from one step to stepwise. 12
In this regard, by investigating stepwise and some one-step reports, a researcher tried to find the parameters that could switch the mechanism of the 1,3-DC reaction from its usual one-step and stereospecific form to the stepwise model. 13 In addition, a reaction between a steric hindered nitrone and an alkene led mechanism to be stepwise, 14 while putting some large groups on a nitrile ylide and an olefin did not change the reaction mechanism into the stepwise model. 15
Thioformaldehyde S-oxide is a type of dipole that is subject of the researchers’ interest in organic synthesis processes for which reason, the mechanistic aspects of its 1,3-DC reaction have received further considerations. Also, the molecular mechanism of cycloaddition between thiocarbonyl ylides and π-deficient alkenes has been recently analyzed on the basis of density functional theory (DFT) calculations. 16 Also, stepwise zwitterionic mechanism has been explored for 1,3-DC processes involving other-type 1,3-dipoles, such as diazocompounds, azomethine ylides, as well as organic azides. 13 This study aims at understanding the nature of the mechanism of the 1,3-DC reaction between cyclobutadiene, as a classic defined antiaromatic dipolarophile (existing in many important compounds), and thioformaldehyde S-oxide, in order to observe the mechanistic behavior of this dipole species toward certain type of olefins. Several species with the potential of emergence in different orientations were detected in the calculation process.
Computational analysis
Several hypothetical structures with certain orientations were developed as input files for each predicated species and were optimized to give any possible state. Therefore, a number of stable and metastable states were found for the reactants, intermediates, and the transition states (TSs). The Gaussian 03 chemical quantum package 17 was used to perform all of the calculations, and the DFT procedure in B3LYP/6-311++G(d,p) theoretical level was applied for optimizing the possible structures.18–20 The TS structures were found using the synchronous transit-guided quasi-Newton (STQN) approach,21,22 and the frequencies of each optimized structure were extracted for calculating the thermodynamic energies of each possible state. Also, the intrinsic reaction coordinates (IRCs) calculations have confirmed the TS structures.23,24 In order to find the electrical charge of each atom in reactants, intermediates, and TSs, the natural bond orbital (NBO) analysis was used.25,26 The related partial bond order was then reached through Pauling relation (Relation 1). 27 The synchronicity relation28–30 was used to calculate the synchronicity31,32 of each considered pathway (Relation 2)
where the bond order nX for a bond length rx is a function of a standard bond of length r0, whose bond order is defined as n0
where ΔBi represents the relative variation of bond order index Bi at the transition state.
The following formula (Relation 3) was applied for calculating global electron density transfer (GEDT) 33
where qA is the net Mulliken charge, and the sum covered the entire atoms of dipolarophile species. In addition, the simulations were performed using the Born–Oppenheimer Molecular Dynamics (BOMD) method, which is widely used for the same calculations. 34
Results
Several geometries with different orientations and fragmental distances were designed as input files and were then optimized for providing any possible state which could appear during the progress of the reactions. According to our predictions and based on the reaction maps of the previous reports, several metastable stations were defined for TSs and intermediates of the parallel stepwise pathways, followed by developing a pre-reaction map containing the one-step and all stepwise channels. Indeed, the triplet (excited state) of such molecules, which would lead to emergence of stepwise-diradical intermediates, was reported for some dipolar cycloadditions (that is not rare).13,35 For this reason, we studied the reaction system under mild conditions.
During the optimization of a number of input files, the reliable geometrical structures were achieved for any station of the map. In addition, the accuracy of the nature and the position of each TS in the reaction map was examined by the hypothetical frequency vibrations. To find any possible species emerging during the reaction coordinate, we have developed the BOMD simulation initiated from certain detected species containing TS (A-1) (green curve), TS (Con) (blue curve), and Int (A) (containing a C–C bond formation; red curve; Figure 1). The simulation time was about 2700 fs. Also, we have optimized a hypothetical intermediate containing a new C–O bond which was not stable, and thus, it became a product structure (see Supplemental Material).
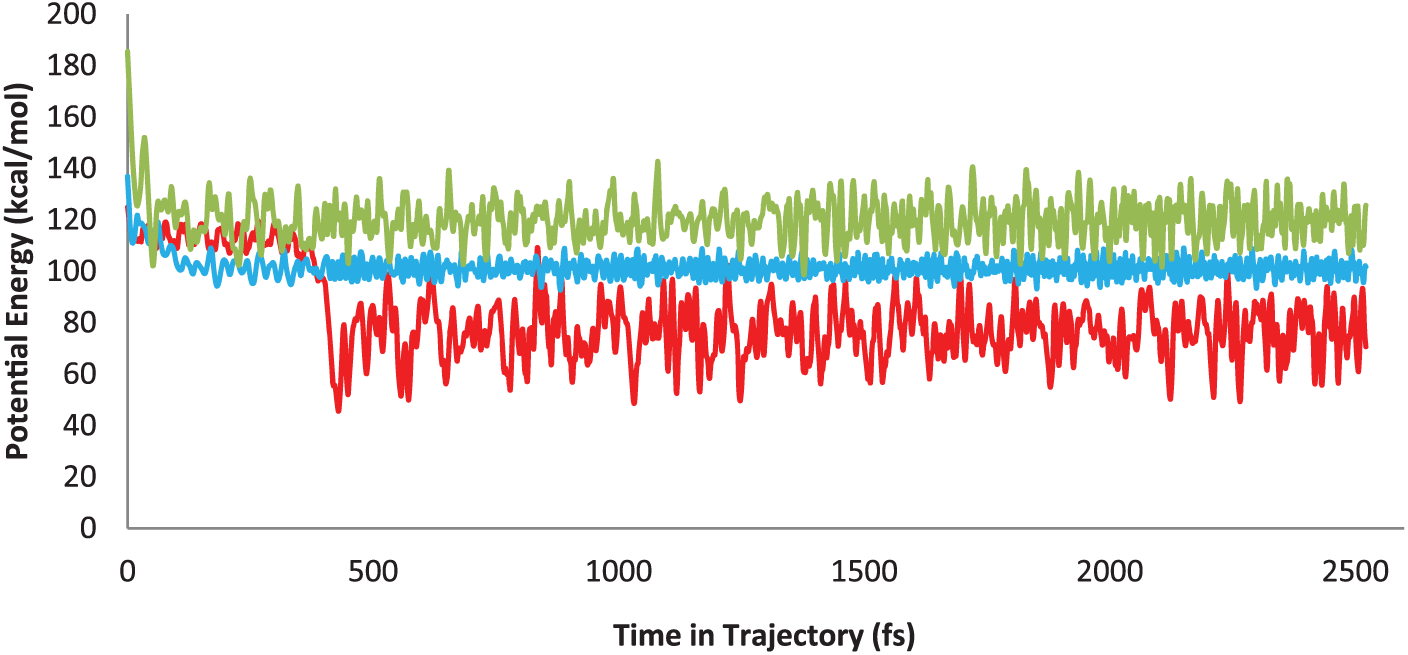
The BOMD simulation started from TS (A-1) (green curve), TS (Con) (blue curve), and Int (A) (red curve).
At the first step, both of the two reactants containing thioformaldehyde S-oxide and cyclobutadiene begin to move near to each other until a complex of those species (reactive complex, RC) forms via intermolecular interactions between them; at that situation, the reactants are at a distance of 2.95 Å and a relatively suitable orientation (Figure 2). As Table 1 shows, there are three ways which divide from RC containing TS (con) which links RC to the final product, TS (A-1) which produces Int (A), and TS (B-1) that creates Int (B). The lengths of the two forming bonds in TS (Con) are 2.55 and 2.85 Å for C6–C11 and C1–O10, respectively, showing an asynchronous one-step TS which is usually observed for one-step 1,3-DC reactions. The partial bond order calculated by the Pauling relation for the two forming bonds C6–C11 and C1–O10 are 0.645 and 0.993, respectively; also, the synchronicity value for the TS is 0.778, which all of these confirm the asynchronous one-step mechanism. In the case of the two other TSs which divide from RC, it should be noted that the main difference between those two is their orientations; somehow, unlike the orientation of thioformaldehyde S-oxide (D = 162°), the dipole fragment in TS (A-1) is in the exo-side compared to the butadiene (D = 16.7°). Moreover, the distances for the forming bonds C11–C6 are 2.16 and 2.45 Å for TS (A-1) and TS (B-1), respectively, which shows TS (B-1) is much near to RC compared to TS (A-1), and the energy surface of each TS may confirm this. After releasing the energy contents, TS (A-1) and TS (B-1) turn their structures into the related intermediates (Int (A) and Int (B), respectively; see Table 1).
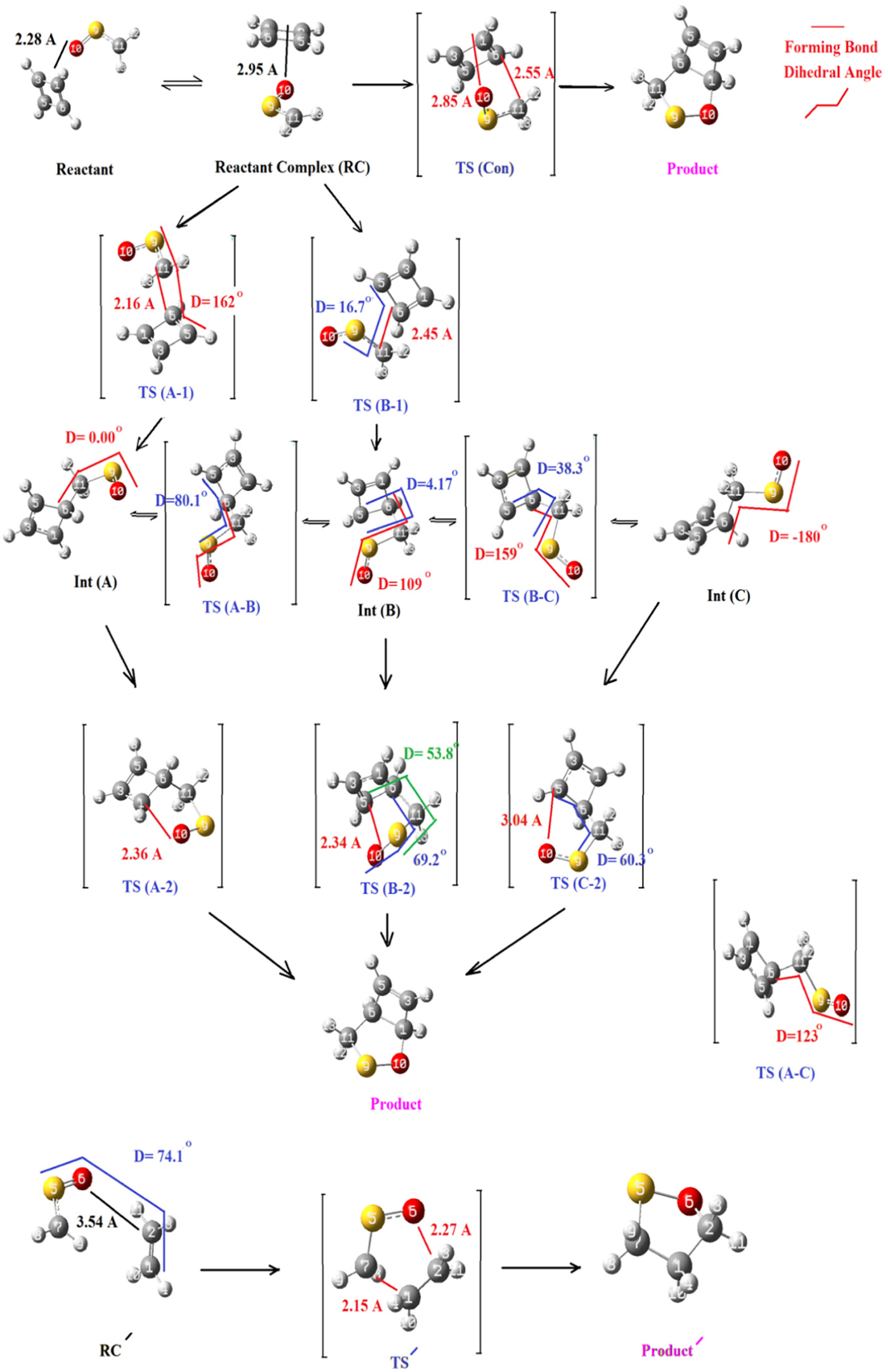
The geometrical structures for all of the detected species which could emerge during the progress of the reactions.
Key parameters for critical structures of the 1,3-dipolar cycloaddition reactions of thioformaldehyde S-oxide with cyclobutadiene and ethane.
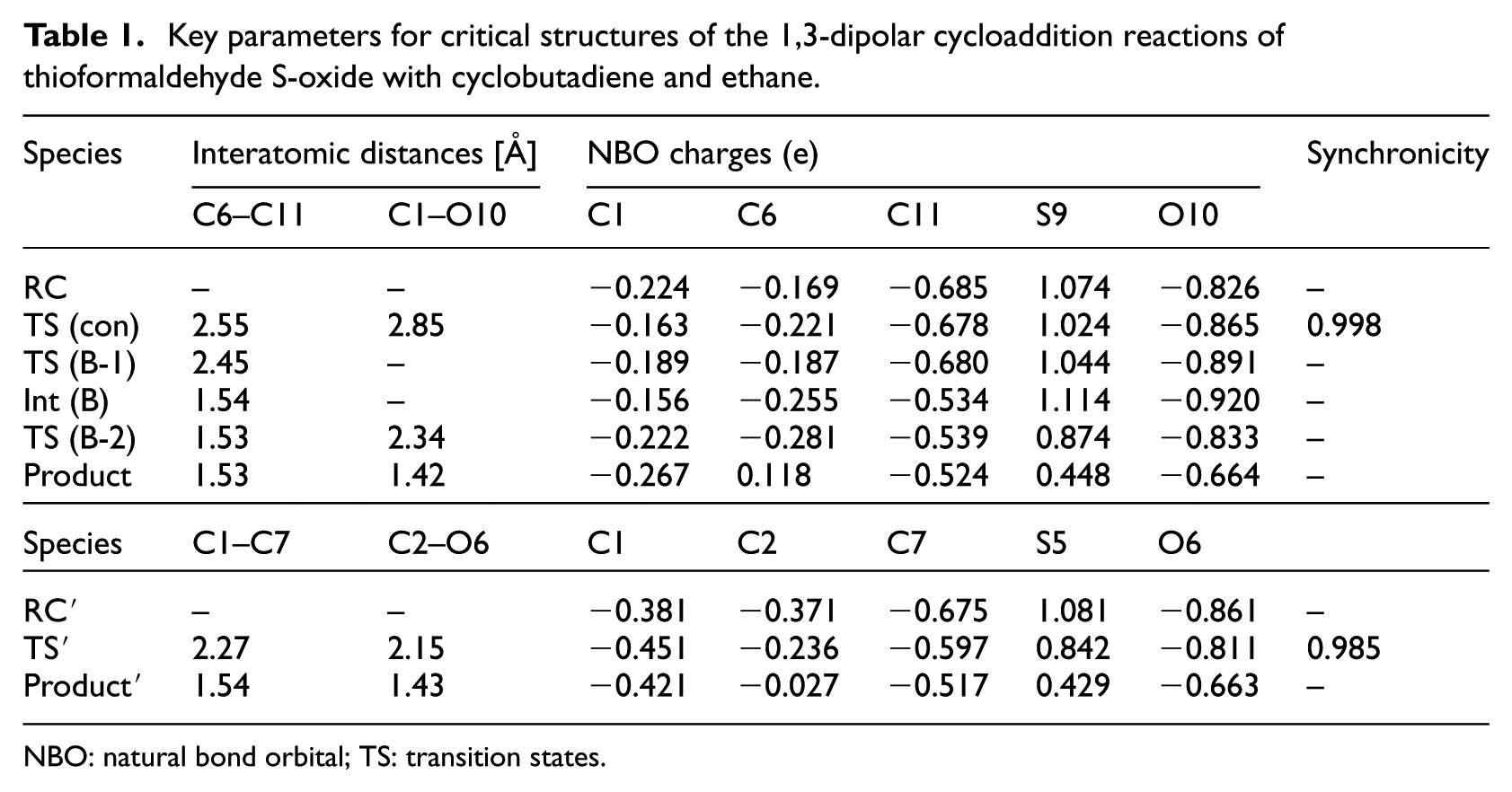
NBO: natural bond orbital; TS: transition states.
The NBO charge analysis reveals that the negative charge increase of −0.052 in C6 and charge decrease of −0.061 in C1 (from RC to TS (Con)) may show that the reaction may begin by the negative charge from C6 of cyclobutadiene to C11 of thioformaldehyde S-oxide. Also, the data show that in the case of the reaction between ethene and the thioformaldehyde S-oxide, the negative charge in C2 decreases from −0.371 to −0.236, and the charge in C1 increases from −0.381 to −0.451 (from RC′ to TS′ (Con)), which shows that the negative charge in C1 attacks to the C7 of thioformaldehyde S-oxide. The higher changes of the atomic charges in the reaction centers between the reactants complex and the transition states for the reaction ethene and thioformaldehyde S-oxide, compared to the reaction between cyclobutadiene and thioformaldehyde S-oxide, may confirm the lower value of synchronicity of that reaction.
From Int (B), two other routes containing TS (B-C) (with a relatively distorted structure and a high energy difference compared to its source, Int (B)) and TS (B-2) with a forming bond (C5–O10 with 2.34 Å) reaching to the product are created (Figure 2). Also, Int (A), with its main difference with Int (C) in the value of the dihedral angle of O10-S9-C11-C6 (0.00° for Int (A) and 180° for Int (C)), could directly transform into TS (A-2) (with a forming bond (C1–O10) of 2.36 Å) and form the product. Besides this, Int (A) could create TS (A-B) (S9-C11-C6-C5 is 80.1°) and TS (A-C) with a dihedral angle (O10-S9-C11-C6) of 123°, without forming bonds, to give Int (B) and Int (C), respectively. Also, for investigating the effect of cyclobutadiene in making the process stepwise, we have recalculated the 1,3-DC reaction between thioformaldehyde S-oxide with ethene at the same basis set of this work, in order to have a reference system, and the results showed, unlike butadiene, at least in our theory level and without fixing the geometries, that this sample reaction was not stepwise.
The synchronicity value for TS (Con) is about 0.998, which indicates that this reaction path is less synchronized than that for TS′ (Con) (0.985). The values for the GEDT indices which show that TS′ (Con) (−0.103) is more polar than TS (Con) (−0.058) may confirm the results for the synchronicity amounts (Table 2). As shown in Figure 3, beside TS (con) with an energy content of 0.831 kcal mol−1(that directly links RC to the final product), two stepwise channels, containing TS (A-1) (9.60 kcal mol−1) and TS (B-1) (−8.07 kcal mol−1), divide from RC (which produce Int (A) and Int (B)), respectively, while any attempts to find a TS between RC and Int (C) were failed. However, there was no direct way to produce Int (C) from RC, but that intermediate might emerge by transformation of Int (A) (via TS (A-C) with an energy surface of −9.83 kcal mol−1) and Int (B) (via TS (B-C) with an energy content of −8.05 kcal mol−1); however, due to the high energy barrier (43.6 kcal mol−1) of TS (B-C) compared to Int (B), turn of Int (B) to Int (C) seems to be impossible both kinetically and thermodynamically. Subsequently, Int (C) transforms to the product via TS (C-2) (−8.86 kcal mol−1; energy barrier: 3.04 kcal mol−1). In spite of detecting two ways dividing from Int (B), it could only transform to TS (B-2) (energy content of 42.5 kcal mol−1 and an energy barrier of 9.60 kcal mol−1) that links this intermediate to the product. Beside TS (A-B) and TS (A-C), Int (A) creates TS (A-2) with a related energy surface of −9.22 kcal mol−1 (and an energy barrier of 2.48 kcal mol−1) that links Int (A) to the final product (Table 2).
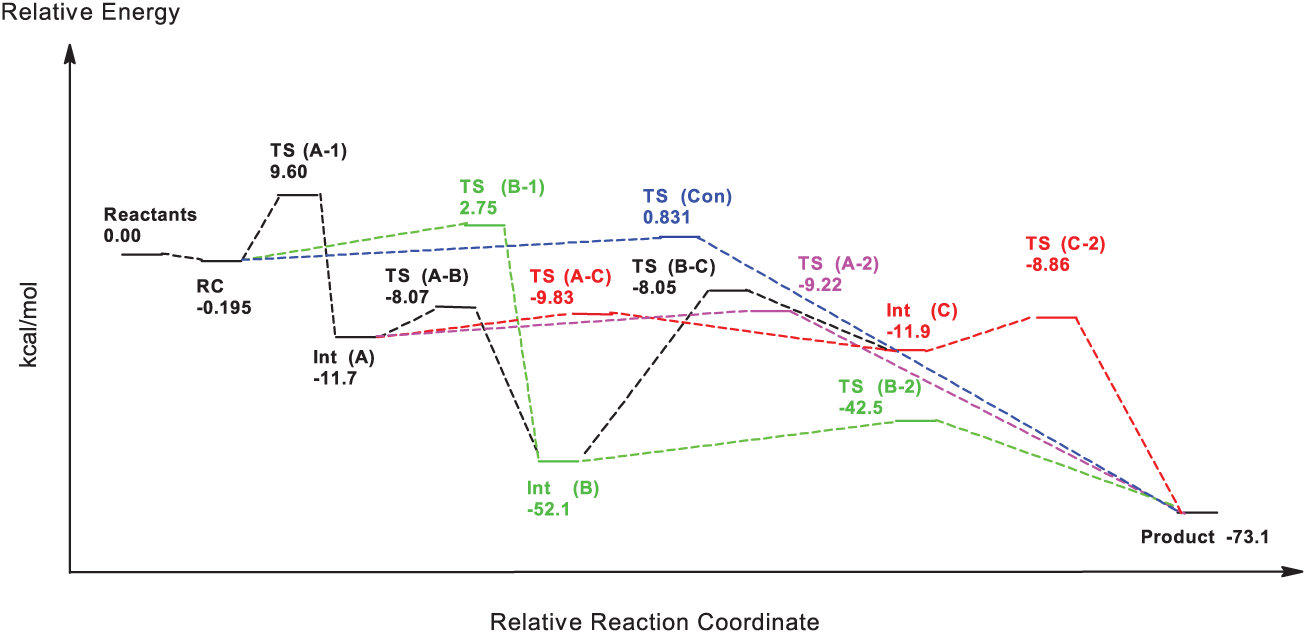
The potential energy surfaces for all of the detected species emerging during the reaction coordinate.
The relative potential energy (kcal mol−1) for all of the detected species which would emerge during the reaction coordinate.
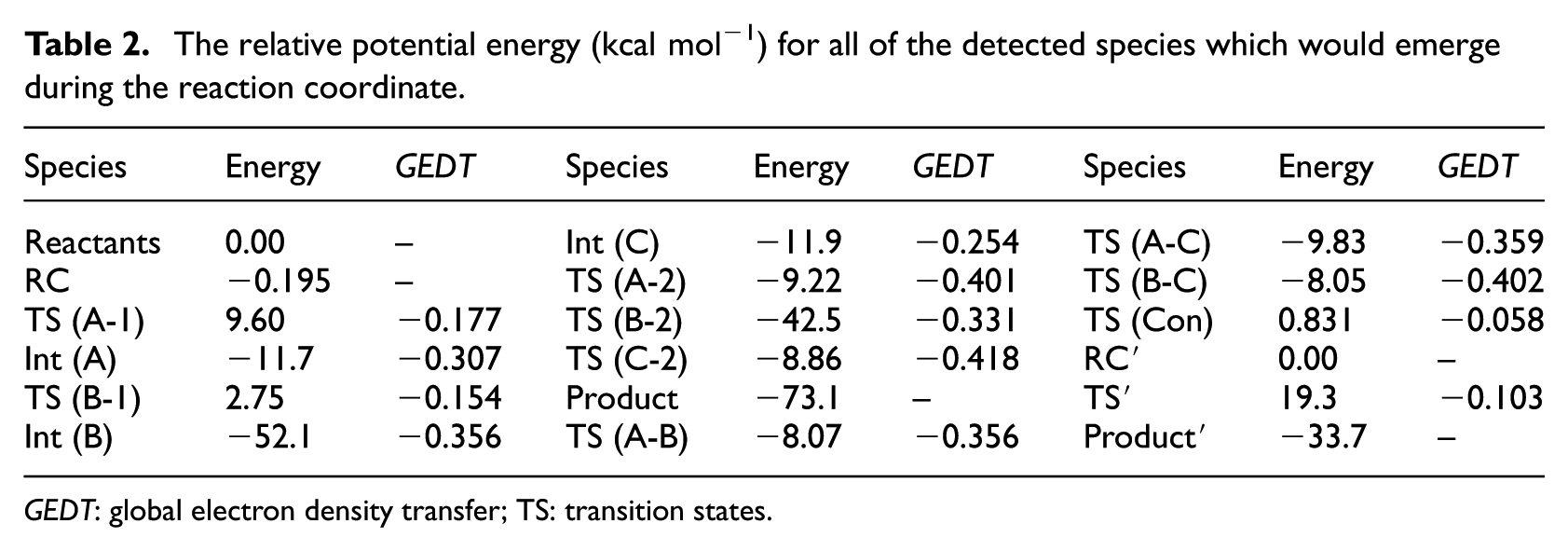
GEDT: global electron density transfer; TS: transition states.
Comparing the relative potential energy surfaces show that almost all reaction pathways are in a very close competition, while the pathway containing reactants (0.00 kcal mol−1) to RC (−0.183 kcal mol−1) to TS (A-1) (9.60 kcal mol−1) to Int (A) (−11.7 kcal mol−1) to TS (A-2) (−9.22 kcal mol−1) to the product (with the energy barriers of 9.60 and 2.48 kcal mol−1, respectively) is the most low-lying route between all stepwise ones. Finally, emergence of Int (B) (−52.1 kcal mol−1) via TS (B-1) is both kinetically and thermodynamically very favorable; that is, emergence of side products created by intermediates would be possible when using the catalyst-free 1,3-DC reaction of thioformaldehyde S-oxide and cyclobutadiene for synthesis plans.
Conclusion
The results of this study show that unlike ethene and thioformaldehyde S-oxide system, in the 1,3-DC reaction between thioformaldehyde S-oxide and cyclobutadiene, the parallel multi-station stepwise pathways and the one-step route are in a close competition. Our predictions for emergence of unwanted intermediates (and probable side products) during the reaction of thioformaldehyde S-oxide and an antiaromatic species like cyclobutadiene seem to be true, since several complicated routes supporting the stepwise mechanism detected in alignment to the one-step way. However, the hypothetical reaction between this dipole species and a simple olefin like ethene was not predicted to be stepwise. Emergence of Int (B) (−52.1 kcal mol−1) via TS (B-1) is highly favorable both thermodynamically and kinetically (in comparison with all other stepwise routes); that is, emergence of side products created by intermediates would be possible when using the catalyst-free 1,3-DC reaction of thioformaldehyde S-oxide and cyclobutadiene for synthesis plans. Thus, at least in the case of the reaction between some thioformaldehyde S-oxide and some dipolarophiles containing butadiene fragments, the possibility of developing probable impurities could not be ignored.
Supplemental Material
INT_(A) – Supplemental material for Understanding the mechanism of the 1,3-dipolar cycloaddition reaction between a thioformaldehyde S-oxide and cyclobutadiene: Competition between the stepwise and concerted routes
Supplemental material, INT_(A) for Understanding the mechanism of the 1,3-dipolar cycloaddition reaction between a thioformaldehyde S-oxide and cyclobutadiene: Competition between the stepwise and concerted routes by Bita Mohtat, Seyyed Amir Siadati and Mohammad A Khalilzadeh in Progress in Reaction Kinetics and Mechanism
Supplemental Material
INT_(B) – Supplemental material for Understanding the mechanism of the 1,3-dipolar cycloaddition reaction between a thioformaldehyde S-oxide and cyclobutadiene: Competition between the stepwise and concerted routes
Supplemental material, INT_(B) for Understanding the mechanism of the 1,3-dipolar cycloaddition reaction between a thioformaldehyde S-oxide and cyclobutadiene: Competition between the stepwise and concerted routes by Bita Mohtat, Seyyed Amir Siadati and Mohammad A Khalilzadeh in Progress in Reaction Kinetics and Mechanism
Supplemental Material
INT_(C) – Supplemental material for Understanding the mechanism of the 1,3-dipolar cycloaddition reaction between a thioformaldehyde S-oxide and cyclobutadiene: Competition between the stepwise and concerted routes
Supplemental material, INT_(C) for Understanding the mechanism of the 1,3-dipolar cycloaddition reaction between a thioformaldehyde S-oxide and cyclobutadiene: Competition between the stepwise and concerted routes by Bita Mohtat, Seyyed Amir Siadati and Mohammad A Khalilzadeh in Progress in Reaction Kinetics and Mechanism
Supplemental Material
Product- – Supplemental material for Understanding the mechanism of the 1,3-dipolar cycloaddition reaction between a thioformaldehyde S-oxide and cyclobutadiene: Competition between the stepwise and concerted routes
Supplemental material, Product- for Understanding the mechanism of the 1,3-dipolar cycloaddition reaction between a thioformaldehyde S-oxide and cyclobutadiene: Competition between the stepwise and concerted routes by Bita Mohtat, Seyyed Amir Siadati and Mohammad A Khalilzadeh in Progress in Reaction Kinetics and Mechanism
Supplemental Material
Product – Supplemental material for Understanding the mechanism of the 1,3-dipolar cycloaddition reaction between a thioformaldehyde S-oxide and cyclobutadiene: Competition between the stepwise and concerted routes
Supplemental material, Product for Understanding the mechanism of the 1,3-dipolar cycloaddition reaction between a thioformaldehyde S-oxide and cyclobutadiene: Competition between the stepwise and concerted routes by Bita Mohtat, Seyyed Amir Siadati and Mohammad A Khalilzadeh in Progress in Reaction Kinetics and Mechanism
Supplemental Material
RC- – Supplemental material for Understanding the mechanism of the 1,3-dipolar cycloaddition reaction between a thioformaldehyde S-oxide and cyclobutadiene: Competition between the stepwise and concerted routes
Supplemental material, RC- for Understanding the mechanism of the 1,3-dipolar cycloaddition reaction between a thioformaldehyde S-oxide and cyclobutadiene: Competition between the stepwise and concerted routes by Bita Mohtat, Seyyed Amir Siadati and Mohammad A Khalilzadeh in Progress in Reaction Kinetics and Mechanism
Supplemental Material
RC – Supplemental material for Understanding the mechanism of the 1,3-dipolar cycloaddition reaction between a thioformaldehyde S-oxide and cyclobutadiene: Competition between the stepwise and concerted routes
Supplemental material, RC for Understanding the mechanism of the 1,3-dipolar cycloaddition reaction between a thioformaldehyde S-oxide and cyclobutadiene: Competition between the stepwise and concerted routes by Bita Mohtat, Seyyed Amir Siadati and Mohammad A Khalilzadeh in Progress in Reaction Kinetics and Mechanism
Supplemental Material
Reactants – Supplemental material for Understanding the mechanism of the 1,3-dipolar cycloaddition reaction between a thioformaldehyde S-oxide and cyclobutadiene: Competition between the stepwise and concerted routes
Supplemental material, Reactants for Understanding the mechanism of the 1,3-dipolar cycloaddition reaction between a thioformaldehyde S-oxide and cyclobutadiene: Competition between the stepwise and concerted routes by Bita Mohtat, Seyyed Amir Siadati and Mohammad A Khalilzadeh in Progress in Reaction Kinetics and Mechanism
Supplemental Material
TS_(A-1) – Supplemental material for Understanding the mechanism of the 1,3-dipolar cycloaddition reaction between a thioformaldehyde S-oxide and cyclobutadiene: Competition between the stepwise and concerted routes
Supplemental material, TS_(A-1) for Understanding the mechanism of the 1,3-dipolar cycloaddition reaction between a thioformaldehyde S-oxide and cyclobutadiene: Competition between the stepwise and concerted routes by Bita Mohtat, Seyyed Amir Siadati and Mohammad A Khalilzadeh in Progress in Reaction Kinetics and Mechanism
Supplemental Material
TS_(A-2) – Supplemental material for Understanding the mechanism of the 1,3-dipolar cycloaddition reaction between a thioformaldehyde S-oxide and cyclobutadiene: Competition between the stepwise and concerted routes
Supplemental material, TS_(A-2) for Understanding the mechanism of the 1,3-dipolar cycloaddition reaction between a thioformaldehyde S-oxide and cyclobutadiene: Competition between the stepwise and concerted routes by Bita Mohtat, Seyyed Amir Siadati and Mohammad A Khalilzadeh in Progress in Reaction Kinetics and Mechanism
Supplemental Material
TS_(A-B) – Supplemental material for Understanding the mechanism of the 1,3-dipolar cycloaddition reaction between a thioformaldehyde S-oxide and cyclobutadiene: Competition between the stepwise and concerted routes
Supplemental material, TS_(A-B) for Understanding the mechanism of the 1,3-dipolar cycloaddition reaction between a thioformaldehyde S-oxide and cyclobutadiene: Competition between the stepwise and concerted routes by Bita Mohtat, Seyyed Amir Siadati and Mohammad A Khalilzadeh in Progress in Reaction Kinetics and Mechanism
Supplemental Material
TS_(A-C) – Supplemental material for Understanding the mechanism of the 1,3-dipolar cycloaddition reaction between a thioformaldehyde S-oxide and cyclobutadiene: Competition between the stepwise and concerted routes
Supplemental material, TS_(A-C) for Understanding the mechanism of the 1,3-dipolar cycloaddition reaction between a thioformaldehyde S-oxide and cyclobutadiene: Competition between the stepwise and concerted routes by Bita Mohtat, Seyyed Amir Siadati and Mohammad A Khalilzadeh in Progress in Reaction Kinetics and Mechanism
Supplemental Material
TS_(B-1) – Supplemental material for Understanding the mechanism of the 1,3-dipolar cycloaddition reaction between a thioformaldehyde S-oxide and cyclobutadiene: Competition between the stepwise and concerted routes
Supplemental material, TS_(B-1) for Understanding the mechanism of the 1,3-dipolar cycloaddition reaction between a thioformaldehyde S-oxide and cyclobutadiene: Competition between the stepwise and concerted routes by Bita Mohtat, Seyyed Amir Siadati and Mohammad A Khalilzadeh in Progress in Reaction Kinetics and Mechanism
Supplemental Material
TS_(B-2) – Supplemental material for Understanding the mechanism of the 1,3-dipolar cycloaddition reaction between a thioformaldehyde S-oxide and cyclobutadiene: Competition between the stepwise and concerted routes
Supplemental material, TS_(B-2) for Understanding the mechanism of the 1,3-dipolar cycloaddition reaction between a thioformaldehyde S-oxide and cyclobutadiene: Competition between the stepwise and concerted routes by Bita Mohtat, Seyyed Amir Siadati and Mohammad A Khalilzadeh in Progress in Reaction Kinetics and Mechanism
Supplemental Material
TS_(B-C) – Supplemental material for Understanding the mechanism of the 1,3-dipolar cycloaddition reaction between a thioformaldehyde S-oxide and cyclobutadiene: Competition between the stepwise and concerted routes
Supplemental material, TS_(B-C) for Understanding the mechanism of the 1,3-dipolar cycloaddition reaction between a thioformaldehyde S-oxide and cyclobutadiene: Competition between the stepwise and concerted routes by Bita Mohtat, Seyyed Amir Siadati and Mohammad A Khalilzadeh in Progress in Reaction Kinetics and Mechanism
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
The geometries (MOL files) and the absolute energies (Table) for all of the detected species are listed in the Supplemental material online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.



