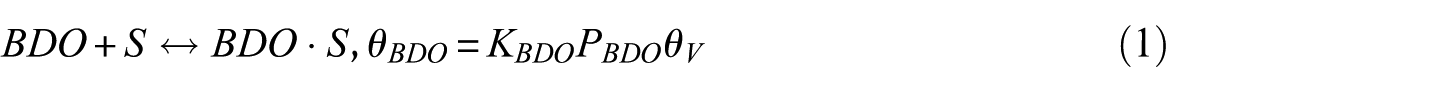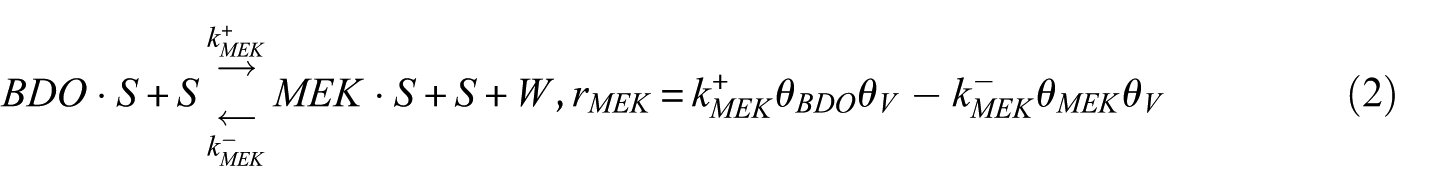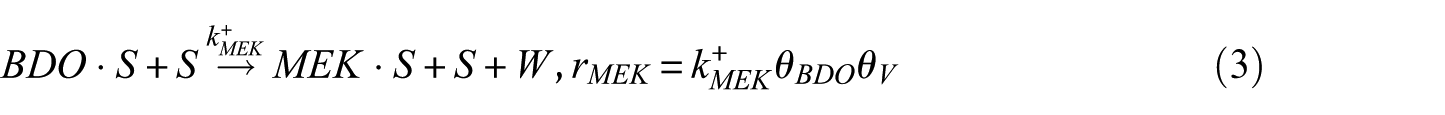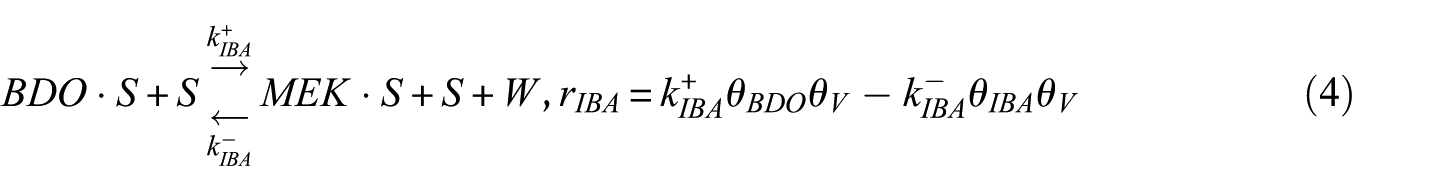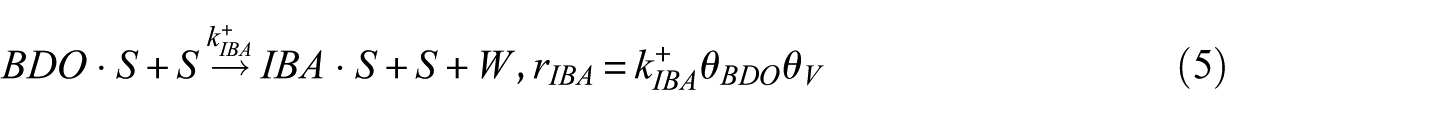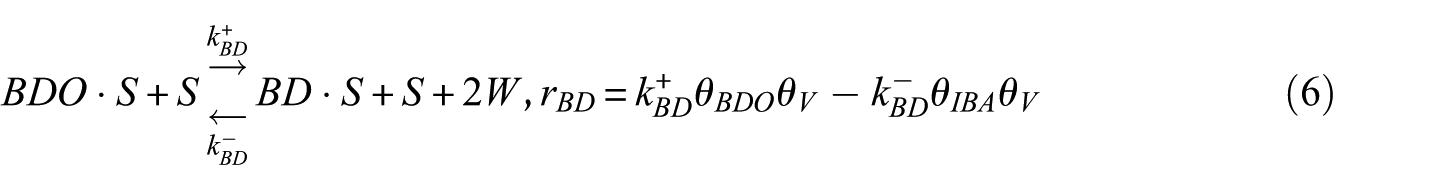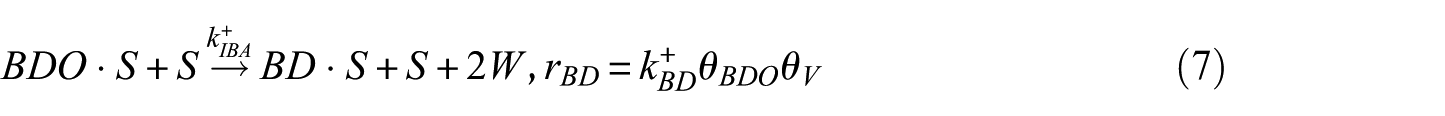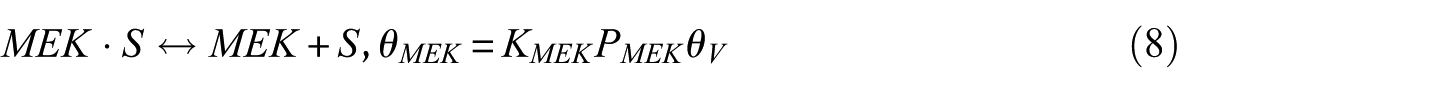Abstract
This study employed Nb2O5.nH2O for the dehydration of butane-2,3-diol, which could be derived from biomass or waste gas using a fermentation process. The experiments were conducted at a temperature ranging from 220 °C to 260 °C and a weight hourly space velocity of 0.01–0.05 min−1. There are three main products that include methyl ethyl ketone, isobutyraldehyde, and butadiene. The yield of products increased with the reaction temperature. Rate data for the dehydration reaction were well represented by Langmuir–Hinshelwood kinetics with adsorption parameters in the rate equations, which assumed the formation of products was reversible with single-site reaction. The apparent activation energies for the dehydration reaction of methyl ethyl ketone, isobutyraldehyde, and butadiene obtained from the Arrhenius plot data were 19.5, 24.0, and 23.7 kJ mol−1, respectively. The adsorption energies for butane-2,3-diol, methyl ethyl ketone, isobutyraldehyde, and butadiene were −182.4, −142.1, −136.1, and −105.6 kJ mol−1, respectively.
Introduction
Butane-2,3-diol (BDO) is produced by a variety of micro-organisms in a process known as butanediol fermentation. 1 BDO production via fermentation started from the Second World War for the production of rubber, plastics, lubricants, latex, and other products. However, the lack of economic competitiveness led to its unsuccessful application in industry. Recently, microbial BDO production has gained global attention as renewable biomass has emerged as a promising route for the development of a low carbon economy. BDO production without concern for the isomer product can produce BDO at a concentration of 150 g L−1. 2 Lately, LanzaTech has developed novel technology that converts local, highly abundant waste and low-cost resources into sustainable, valuable commodities. The patented process uses an acetogenic microbe to convert waste gas containing carbon dioxide (from industrial sources like steel mills and processing plants), or syngas generated from any biomass resources (e.g. municipal solid waste, organic industrial waste, or agricultural waste), into fuels and chemicals including BDO. 3 The catalytic dehydration of BDO has been investigated since the 1940s; however, the known methods for conversation of BDO suffer from several disadvantages, including corrosive reagents, radioactive catalysts, and undesirable products. Dehydration of BDO can be proceeded by different mechanisms depending upon the catalysts used, 4 including Brønsted acid (alumina), 5 silica-supported sodium phosphates, 6 γ-Al2O3, 7 and ceria. 8 As used herein, dehydration refers to a reaction that removes H2O from an alcohol to form an alkene. 9 A diol, such as BDO, may be partially dehydrated by removing one molecule of water, or completely dehydrated by removing two molecules of water. 10 The biologically fermented butane-1,3-diol is usually present in an aqueous solution, so that water-tolerant solid acid catalysts are better for BDO dehydration. Niobic acid (hydrated niobium oxide, Nb2O5.nH2O) has been reported to possess high acid strength 11 and to be a water-tolerant Lewis acid catalyst. 12 In addition, Nb2O5.nH2O is inexpensive, thermally stable, and qualified as an industrial catalyst. The main purpose of this research was to acquire more insight into the dehydration of BDO over Nb2O5.nH2O and the kinetics of BDO dehydration. A series of dehydration experiments was systematically carried out and used to formulate micro-kinetic models. The kinetic constants obtained from this research would be very useful to simulate bio-BDO dehydration on an industrial level.
Experimental
Chemicals
BDO (mixture of stereoisomers) was purchased from Tokyo Chemical Industry Co., Ltd. (Japan) Niobium oxide (HY-340) was provided by Companhia Brasileira de Metalurgia e Mineração (CBMM).
Blank experiment
Nb2O5.nH2O (1.0 g) was loaded into the reactor, which was set at 523 K and purged with nitrogen at a rate of 100 mL min−1. There were no chemicals detected by the gas chromatograph (GC). This indicated that interference from the reactor for these catalytic reactions could be ignored.
BDO dehydration experiments
The catalytic reactions were performed in a conventional continuous flow fixed-bed reactor made of stainless steel (ID = 8 mm) under atmospheric pressure. Prior to reaction, the catalyst sample (weight = 1.0 g) was treated in the reactor under N2 flow (flow rate of 100 mL min−1) at 300 °C for 2 h. The N2 flow was controlled with mass-flow controllers. BDO was fed into the reactor via a micro pump at 3 mL h−1 together with the N2 flow. The reactor temperature was set between 220 °C and 260 °C (Figure 1).
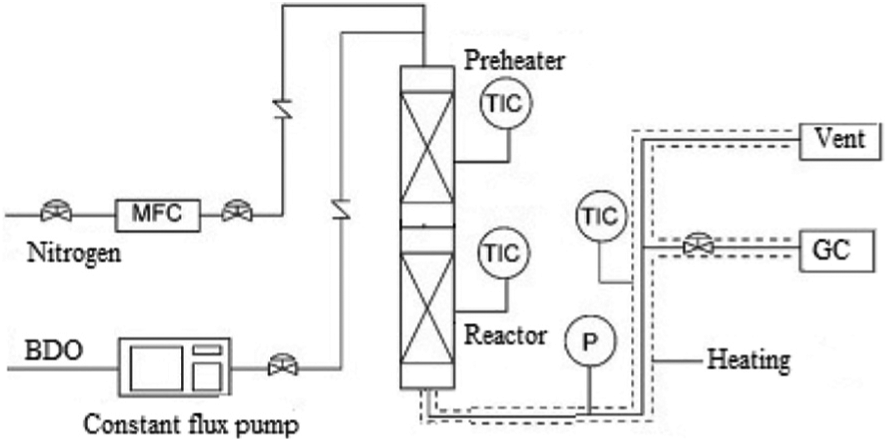
Experimental setup.
Product compositions were analyzed by an on-line GC (SRI 8610C) equipped with an MXT-1 column (non-polar phase, 60 m, ID 0.25 mm, film thickness 0.25 µm), equipped with a thermal conductivity detector (TCD), and flame ionization detector (FID) for the analysis of hydrocarbons and oxygenated chemicals, and quantified by injecting calibration standards to the GC system. The temperature of the tubing from the bottom of the reactor to the inlet of the GC was maintained at 230 °C to avoid the condensation of liquid products. The products were injected through the sample loop, which was controlled by a high-temperature 10-port valve. The oven was kept at 40 °C for 5 min, then raised to 120 °C at a ramp rate of 40 °C min−1, and finally, raised to 250 °C at a rate of 20 °C min−1 and held at this temperature for 10 min. To ensure the identification of products, gas chromatography–mass spectrometer (GC–MS) analyses were also carried out using an Agilent 7890A GC system equipped with an Agilent 5975C MS detector (Agilent, Santa Clara, California, USA) and HP-1 capillary column. The carbon selectivity and conversion were calculated by area normalization methods. Two repeat runs were performed under each reaction condition, and any two trials were generally within 5% of each other. The carbon balances closed with above 90% for all runs in this study.
Results and discussion
Effects of BDO concentration and reaction time
Nb2O5.nH2O used in this study was commercially available. Nb2O5.nH2O was found to exhibit a high acid strength (H0 = −5.6), corresponding to the acid strength of 70% H2SO4, when calcined at relatively low temperatures (373–573 K). Nb2O5.nH2O showed high acid strength on the surface in spite of containing water in an unusual solid acid. To test the selectivity of Nb2O5.nH2O during the reaction, a different set of BDO concentration tests were carried out under the same conditions as before, except for the temperature, which was held at 250 °C.
Nb2O5.nH2O showed excellent stability as a catalyst for BDO dehydration over 60 hours of study. The change in concentration of BDO did not affect the composition of the product (Figures 2 and 3). The selectivity of methyl ethyl ketone (MEK) was always maintained at 80%, and isobutyraldehyde (IBA) and butadiene (BD) were maintained around 10%.
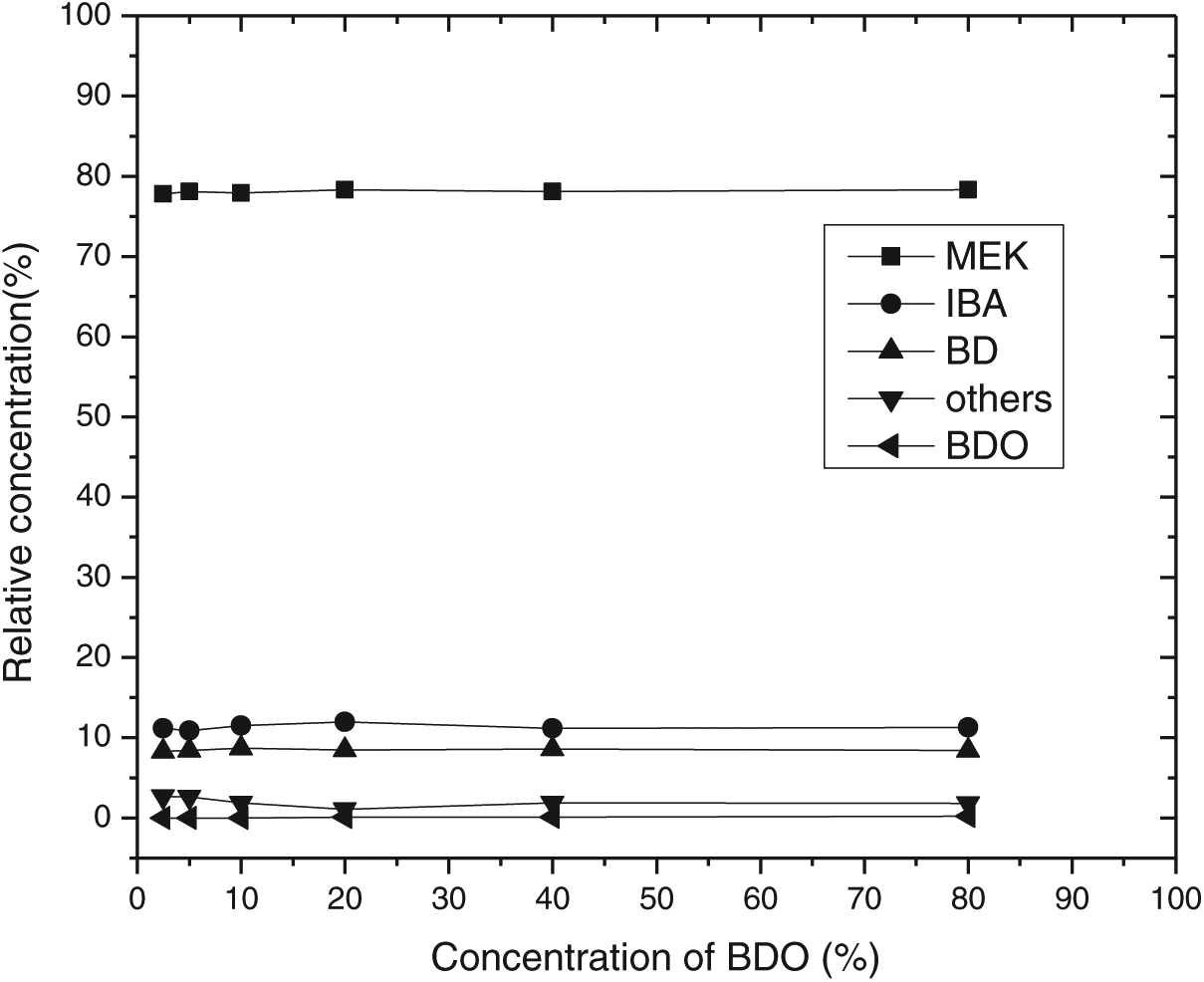
Effect of BDO concentration on selectivity.
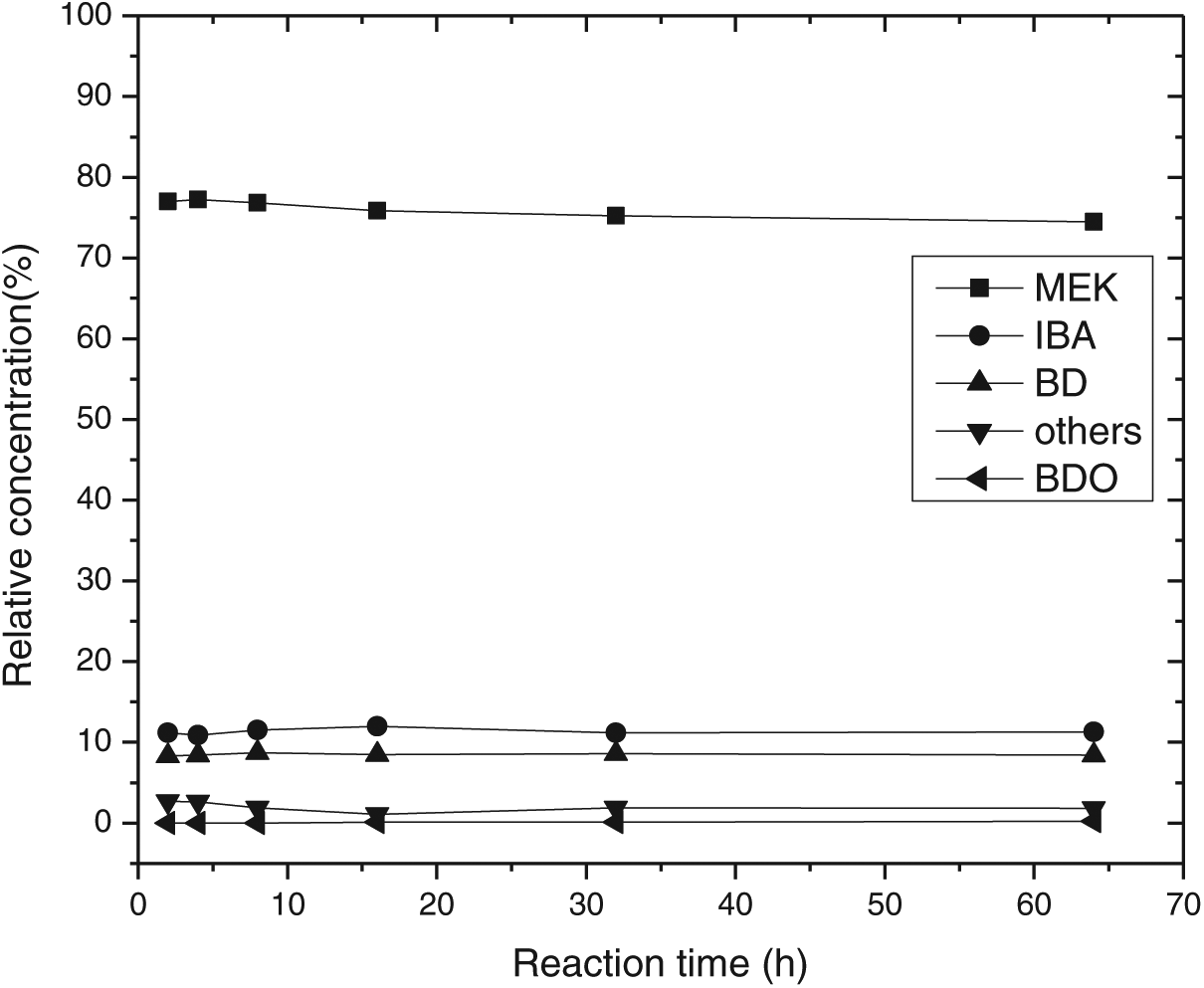
Effect of reaction time on selectivity.
Mechanism of BDO dehydration on Nb2O5.nH2O
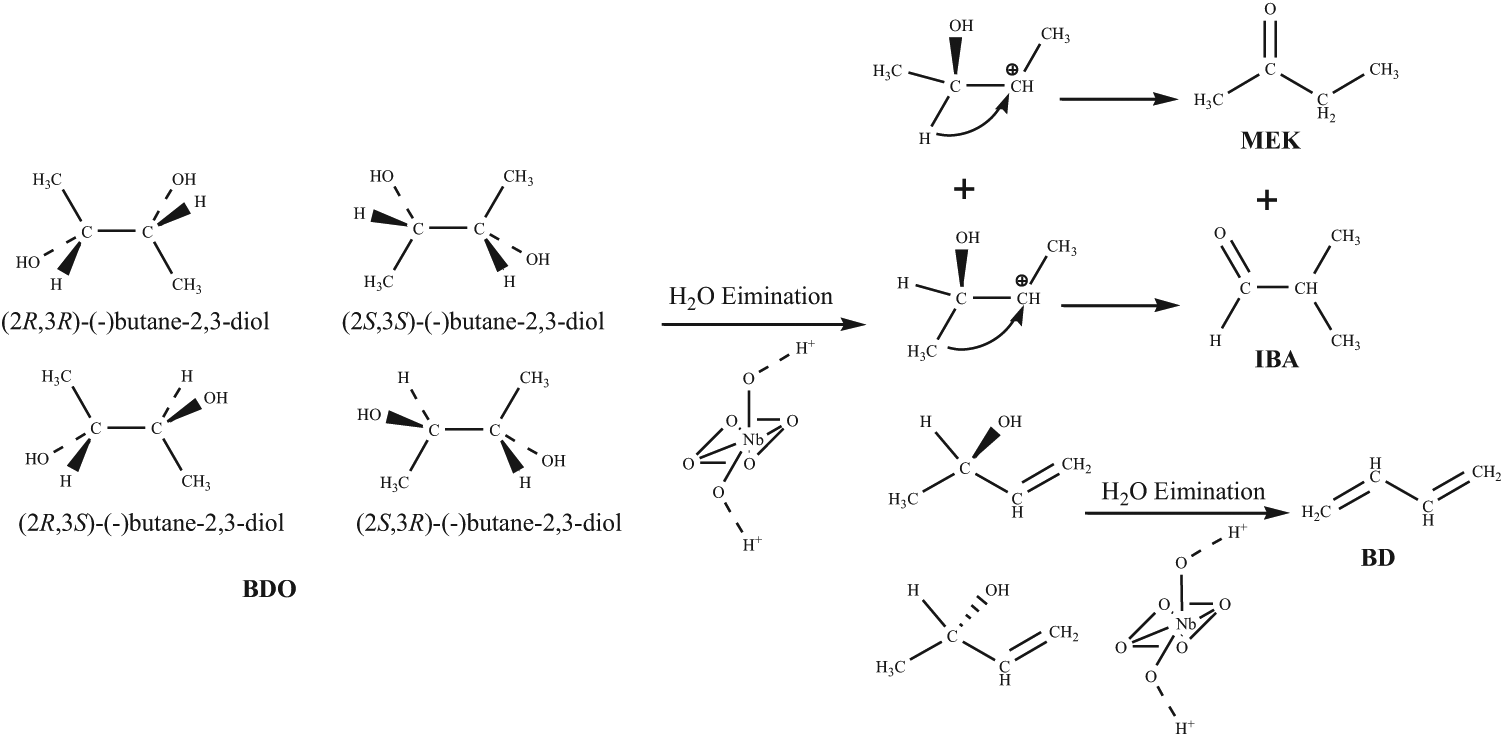
The dehydration reaction of BDO on Nb2O5.nH2O is a very complex process. When Nb2O5.nH2O is taken as the catalyst, there can be mainly three products.
The reaction routes to the products are summarized in Scheme 1. The reaction products over Nb2O5.nH2O catalyst have not been discussed before. It could be observed that MEK and IBA were the primary products from the dehydration reaction of BDO. Acid sites on the surface of the Nb2O5.nH2O 13 could act as the active sites for converting BDO to IBA through a pinacol rearrangement.14,15 3-Buten-2-ol was the dehydration product from the first step and this could be followed by further dehydration to the final product BD at high reaction temperature.
Mechanism of BDO dehydration on Nb2O5.nH2O.
Effects of internal and external diffusion
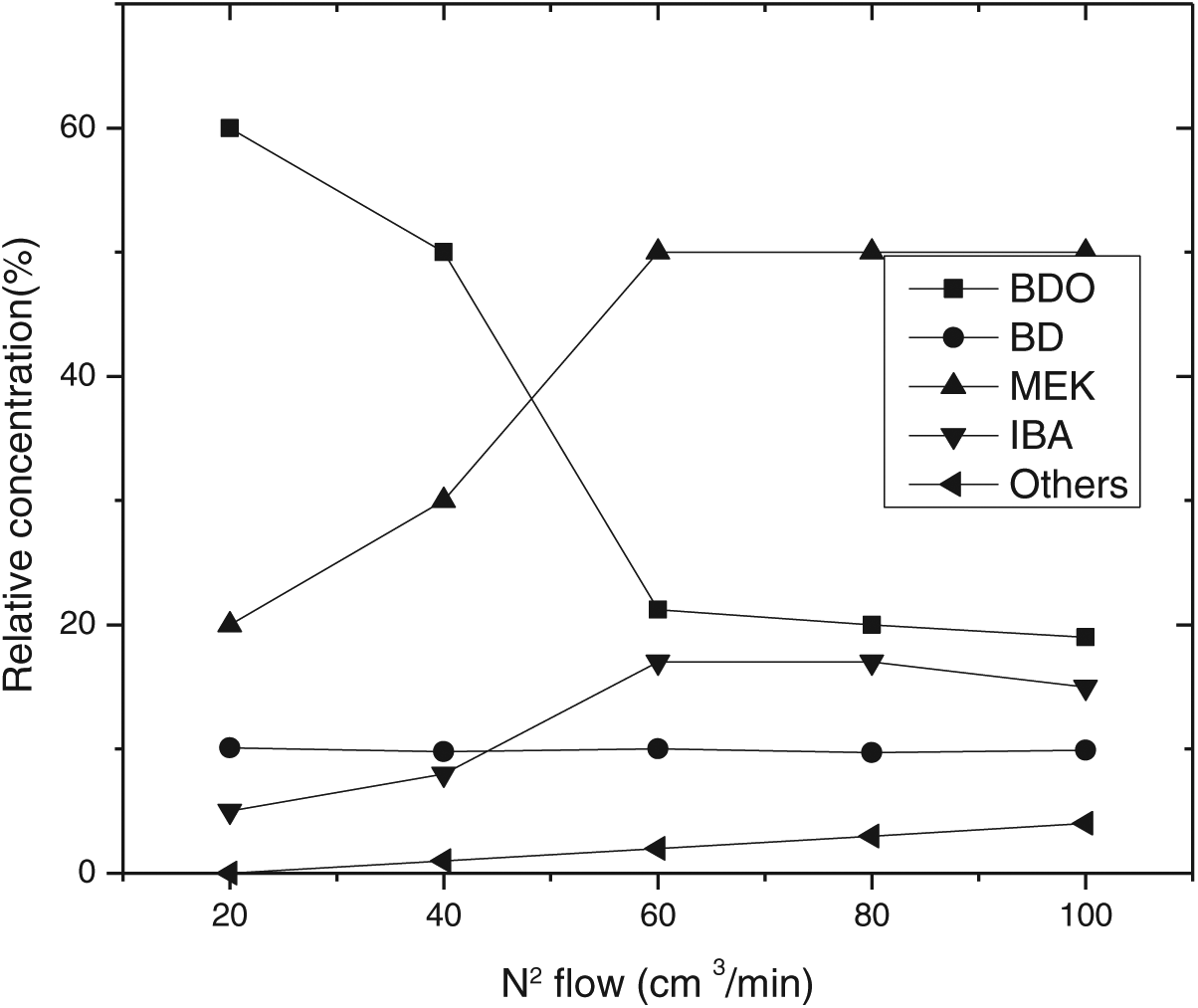
Nb2O5.nH2O used in this reaction was as porous solid particles. In order to induce the reaction, the reacting species were required to be diffused on the active surface of the catalyst. For this reason, the effect of the transport process became more and more significant. External diffusion could be excluded by changing the space velocity of the mixture at nitrogen flows of 20, 40, 60, 80, and 100 mL min−1. The BDO vapor fraction in the feed was 0.02 mL min−1, and the carrier flow was maximized (100 mL min−1) in order to guarantee minimum external resistances. The results, presented in Figure 4, showed that the component of products did not depend on nitrogen flow when it was above 60 mL min−1 and so external diffusion could be neglected.
Effect of N2 carrier flow on selectivity.
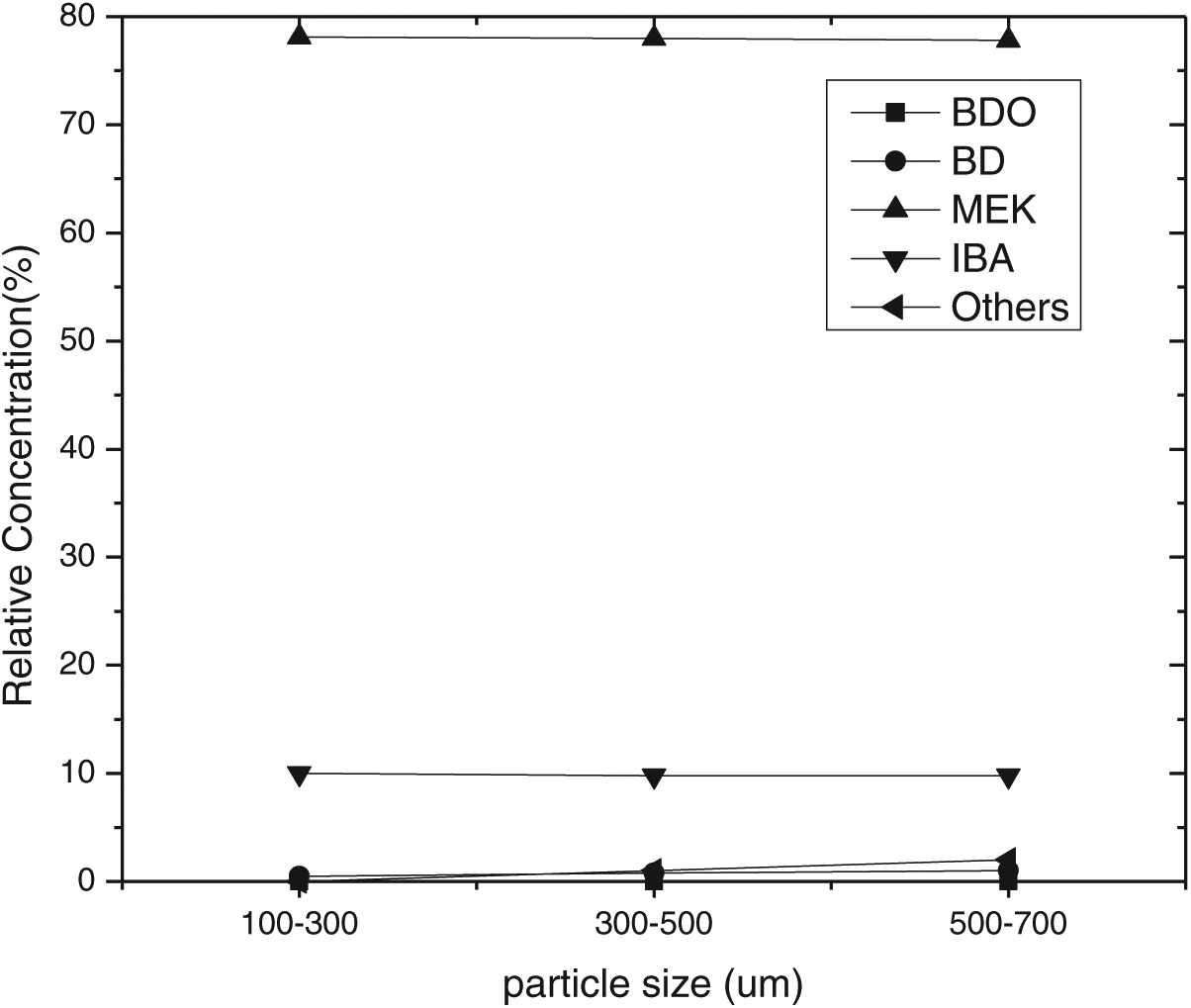
The internal mass transfer resistances were investigated by performing reactions with a 1.0 g load of catalyst at three particle size ranges: namely, 100–300, 300–500, and 500–700 μm. As suggested in Figure 5, the moderate particle size of 300–500 μm was chosen for the remainder of the work.
Effect of particle size on selectivity.
Flow–temperature profiles
Concentration–time profiles on Nb2O5.nH2O catalysts at different temperatures and initial substrate concentrations were obtained to capture the temporal evolution of various intermediate products and to establish acceptable carbon balance closure (Figure 6). Such knowledge is essential to the development of detailed and reliable kinetic models. To confirm the absence of external and intraparticle mass transfer limitations, components of the products were utilized.
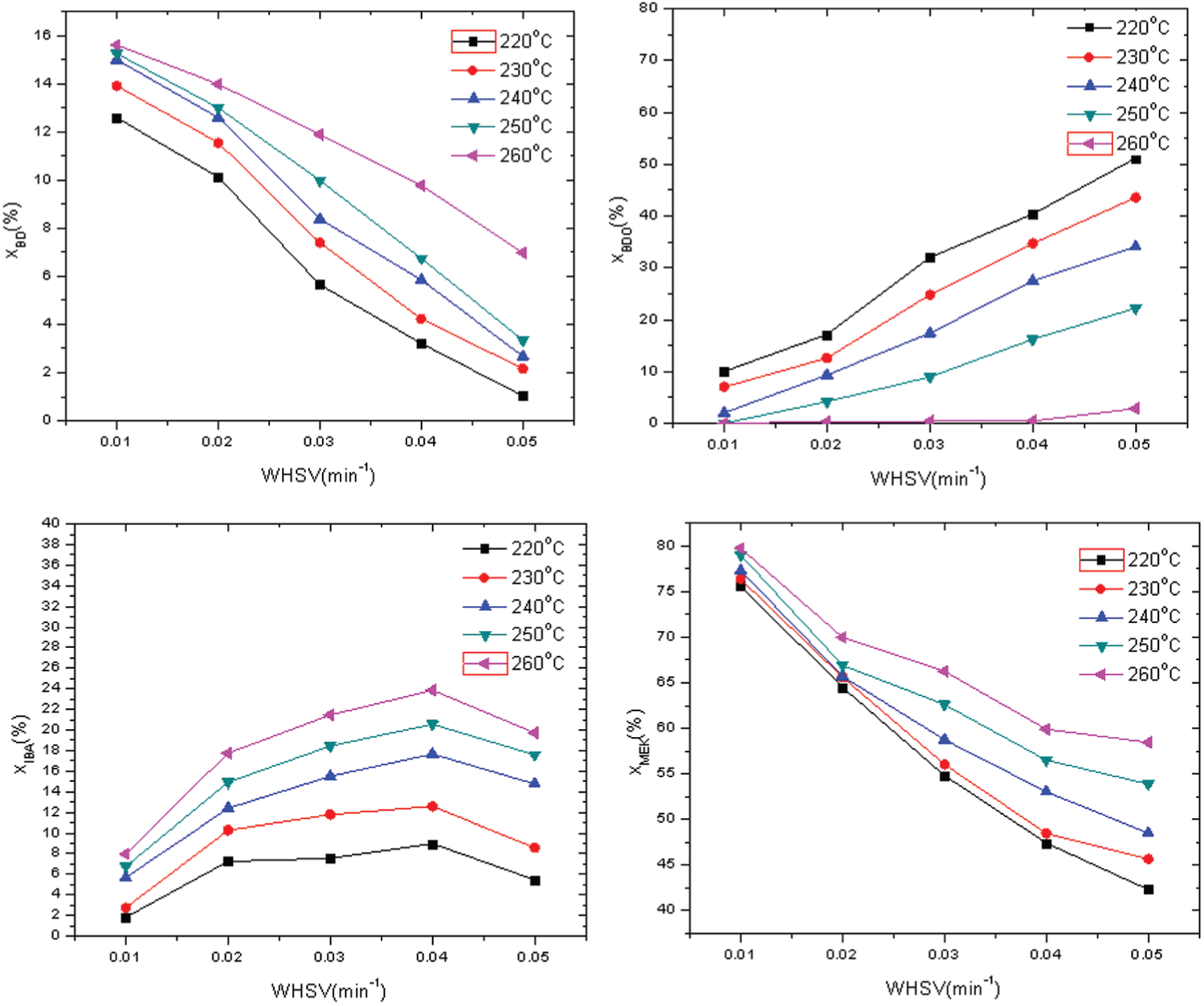
Catalytic results for the conversion of BDO over different temperatures and weight hourly space velocity.
Kinetic model
In this study, the Langmuir–Hinshelwood (LH) model was applied to derive the reaction rate equations. The reaction mechanism followed a classical scheme of reactant–adsorption/surface–reaction/product–desorption, and all the kinetic models included the effects of reactant and product concentrations. This involved adsorption of BDO, reaction between adsorbed reactant (BDO) and the catalyst’s active site (S), and finally, desorption of BD, IBA, MEK, and water from the catalyst surface. Hence, the surface reaction was assumed to be the rate-determining step. The surface reaction mechanism was described by the following equations
The value of the partial pressure of each species was calculated using
Polymath 6.0
16
was used to fit the experimental data. The main function of the Levenberg–Marquardt algorithm was to minimize the sum of square errors
Furthermore, the standard error (SE) was calculated by dividing the standard deviation
To construct an accurate kinetic model, the first step was to make reasonable assumptions for the proposed reactions (Table 1). There were two alternative assumptions for BDO dehydration. The first assumption, as shown in equations (3), (5), and (7), anticipated that the reaction was irreversible. The second assumption, as shown in equations (2), (4), and (6), supposed that the reaction was reversible. The objective of this research was to find out which assumption could generate the most suitable model for BDO dehydration. The results of the regression analysis using the Polymath program are illustrated in Tables 2 and 3.
Brief description of kinetic models.
Rate constants and activation energies of the surface reactions based on Model I.
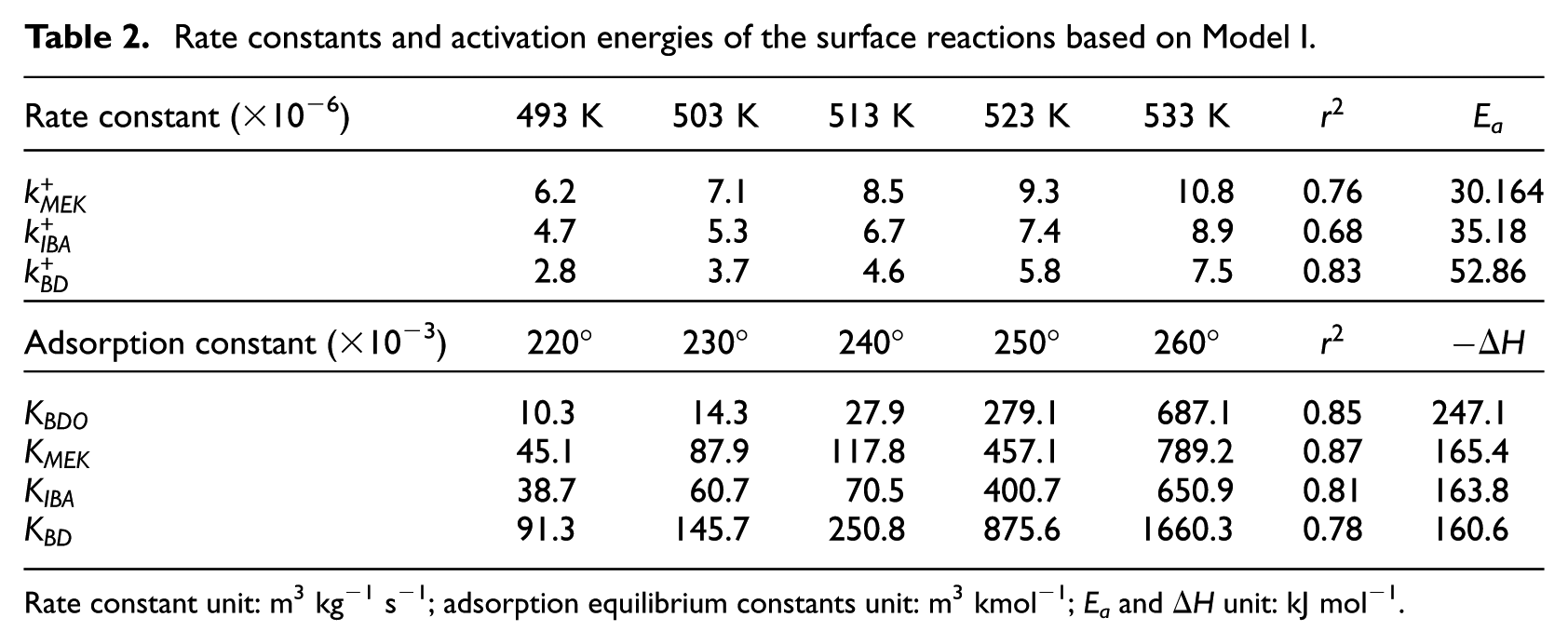
Rate constant unit: m3 kg−1 s−1; adsorption equilibrium constants unit: m3 kmol−1; Ea and
Rate constants and activation energies of the surface reactions based on Model II.
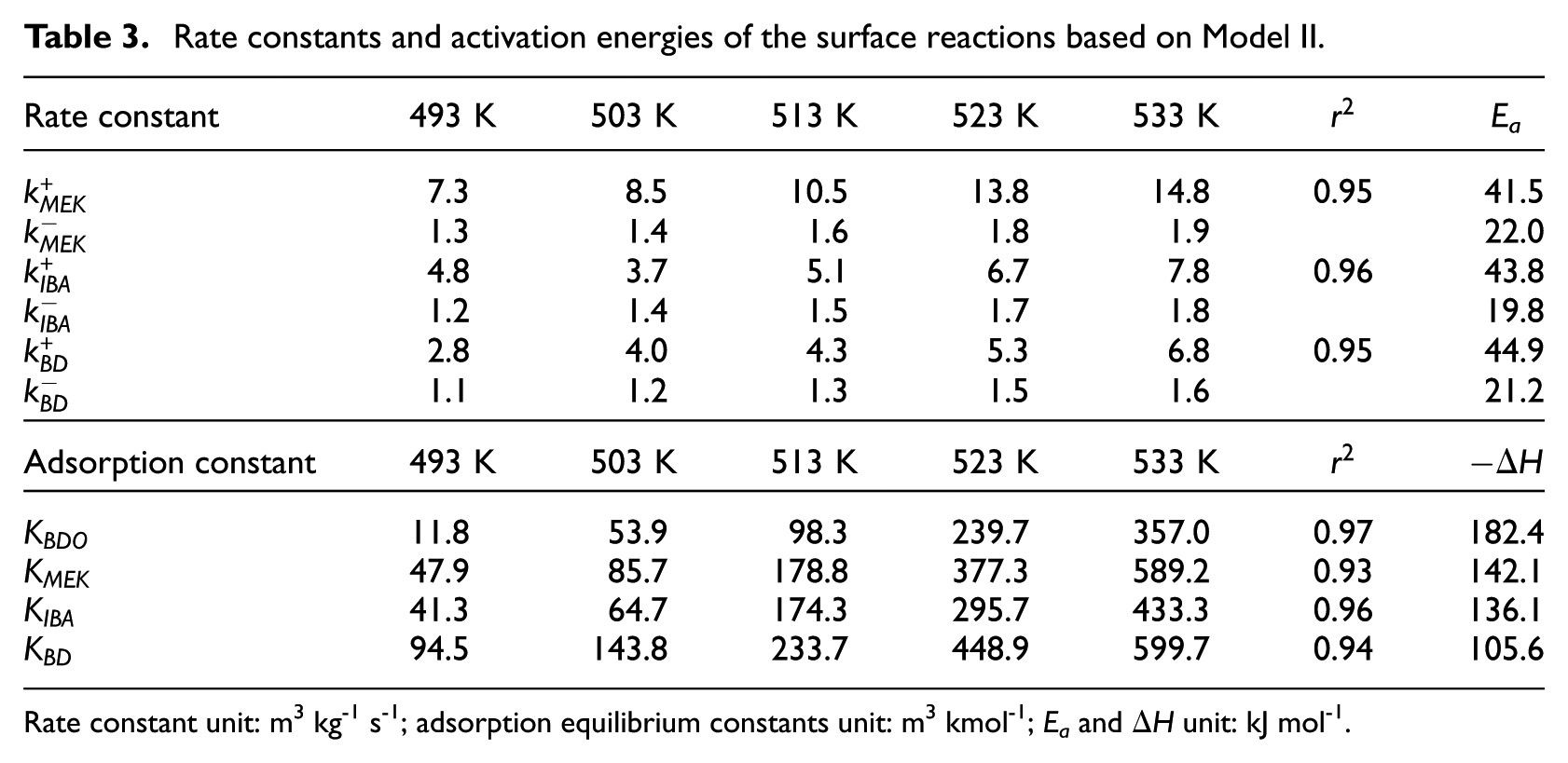
Rate constant unit: m3 kg-1 s-1; adsorption equilibrium constants unit: m3 kmol-1; Ea and ΔH unit: kJ mol-1.
The rate constants for each reaction at various temperatures and the activation energies, and adsorption enthalpies were calculated based on the Arrhenius and van’t Hoff equations. The relative quality of the fit of Models I and II, specifically the sum of squares of residuals, is compared in Tables 2 and 3. It is observed that, among the two models, Model II best represents the experimental data and trends. The estimated parameters of Model II, except r 2 , exhibit a good fit for the concentration of all other species. The kinetic parameters obtained from fitting this model to experimental data are presented in Table 3. The Ea for BDO dehydration to form MEK is approximately 19.5 kJ mol−1, which is smaller than the value for dehydration conversion of BDO on ZSM-5, which is 32.3 kJ mol−1. 17 Ea for BDO dehydration to form IBA and BD are 24.0 and 23.7 kJ mol−1, respectively. Both of these values are larger than the activation energy to form MEK. Furthermore, while the adsorption energies are comparable, and it can be shown that the adsorption energy of BDO is the largest, due to the presence of two hydroxyl groups in the structure of BDO. The adsorption energy of BD is 105.6 kJ mol−1. It is necessary to point out that while Model II describes a reversible reaction on the catalyst surface, diffusion in the catalyst pellets is also important.
Conclusion
Nb2O5.nH2O is an active and selective catalyst for dehydration of BDO and therefore can be used to dehydrate BDO produced through fermentation. More than 80% selectivity of BDO dehydration was produced on the catalyst Nb2O5.nH2O, with BD and two other products detected. Mechanistic modeling of BDO conversion over Nb2O5.nH2O catalyst at 220 °C–260 °C under an inert atmosphere revealed mechanistic insights into the dehydration of BDO. Detailed kinetic analysis illustrated that a model assuming a reversible reaction on the catalyst surface fitted temporal concentration–time data well. The apparent activation energies for the dehydration reaction of MEK, IBA, and BD obtained from the Arrhenius plot data were 19.5, 24.0, and 23.7 kJ mol−1, respectively. The adsorption energies for BDO, MEK, IBA, and BD were −182.4, −142.1, −136.1, and −105.6 kJ mol−1, respectively. These insights should be useful to influence the rational design of catalysts and processes for the selective BDO dehydration in general.
Footnotes
Appendix 1
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grant no. 21602206) and Zhejiang Provincial Natural Science Foundation of China (grant no. LY15B060008).