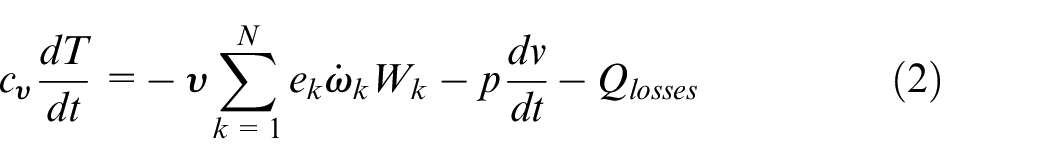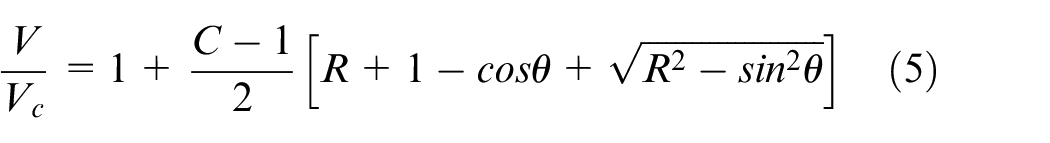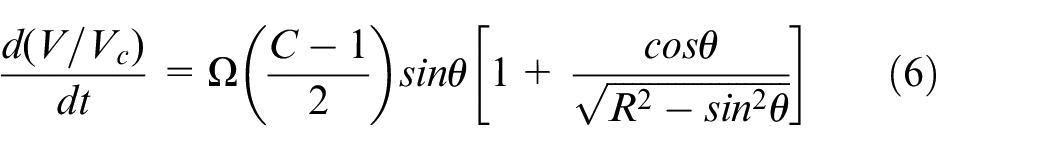Abstract
The potential use of hydrogen peroxide as an ignition promoter to enable the use of ammonia in compression ignition engines is explored in the current study. A simplified numerical HCCI engine model within the Chemkin Pro suite is employed. The numerical investigation reveals that the proposed use of hydrogen peroxide is significantly more advantageous against the more conventional method of preheating the intake charge to achieve ignition, whilst using a glow plug. In particular, the IMEP, power and torque exhibit an increase greater than 65% along with a spectacular decrease of NOx emissions reaching in certain cases a 9-fold decrease. The thermal efficiency exhibits a more moderate, yet non-negligible increase, around 5%. Generally, the incremental increase of hydrogen peroxide leads to the increase of the IMEP, power and torque as well as the maximum temperature and, hence, NOx emissions. These increases are largely linear with the hydrogen peroxide addition. Finally, the introduction of hydrogen peroxide leads to a two-stage ignition process, where the first ignition stage was found to be instrumental to the control of the ignition process, and, therefore, the system’s efficiency. Further research is required to substantiate further the feasibility and the the limitations of the proposed technology which can enable the rapid decarbonization of heavy duty applications, such as marine ships and trucks.
Introduction
The existing climate crisis is a product of human-induced actions over the past centuries. With a rapid increase in human population and demand in various sectors, an exponential growth in greenhouse gases has been observed. The transportation sector plays a significant role in the increase of carbon emissions, with 23% of all emissions generated in Europe arising due to heavy reliance on fossil fuels used by vehicles. 1 This have motivated policy makers, particularly in the west, to introduce mandates that will phase out petrol and diesel cars in the next 10–15 years.2–4 For instance, the UK, Sweden and the Netherlands have decided to end the sales of new petrol and diesel cars by 2030 while Norway has pledged a more ambitious plan, advancing the ban date to 2025.5–8 An important detail to note, is that all the strict and ambitious regulations concern mainly cars, where electrification has made significant technological advancements in the last decade. Hence, electric vehicles (EVs) have been promoted by policy makers and the industry as a viable alternative to petrol and diesel cars and the cost reduction that has been achieved at the same time, have also made EVs a potentially affordable solution.9,10 However, electrification is far from an attractive solution for heavy duty transport applications, such as trucks and marine ships, both predominantly powered by compression ignition (CI) engines due to the requirement for high efficiency.11,12
As a result, the research on decarbonization technologies applicable to heavy-duty transport applications has intensified over the recent years. One of the alternative fuels that have attracted significant interest is ammonia. Ammonia is considered by many as an ideal candidate to replace fossil fuels in ICEs not only for propulsion but also for power generation purposes.13–16 In principle, ammonia is a hydrogen energy carrier with one of the highest gravimetric and volumetric hydrogen densities. 14 The energy density of liquid ammonia is 15.6 MJ/l, that is, much higher than liquid hydrogen (9.1 MJ/l) and even more higher than compressed hydrogen (5.6 MJ/l at 70 MPa). This translates to a driving range of 756 km with a 60.6 l fuel tank of ammonia, which is almost twice the range provided by a liquid hydrogen tank (417 km) holding the same volume and almost three times the range yielded by a compressed hydrogen tank (254 km) again, holding the same volume. 14 When compared to hydrogen, ammonia features two additional important advantages; its storage and transportation is much easier due to an established and reliable infrastructure already being in place.13–16 Therefore, it is not surprising that the use of ammonia in heat engines is not new and dates decades back. 17 However, one of ammonia’s unique features that renders its use in heat engines extremely challenging is the very high auto-ignition temperature, more than 130 K higher than hydrogen. This favors the use of spark ignition (SI) engine mode, where ignition is initiated by a spark plug. As a result, most research on ammonia use in heat engines has been performed in the context of SI engines.13,18–24 On the other hand, the thermodynamic conditions achieved in the engine cylinder in conventional CI engines are such that prohibit the use of ammonia. To tackle ammonia’s high resistance to auto-ignition in CI engines, three strategies have been proposed.17,25 The first is the use of high compression ratios (>35:1).18,26 The second is the use of a glow plug to properly preheat the charge, thereby facilitating ignition, combined with increased compression ratios. 27 The available literature on both these strategies has generally been quite scarce and practically stopped decades back because of the great difficulty in enabling a smooth engine operation as a consequence of ammonia’s low reactivity, high auto-ignition temperature and low flame speed.17,25 Ultimately, using such high compression ratios – in context of vehicular applications, is seen as impractical as it would require extensive engine modifications and also result in higher in-cylinder temperatures. Furthermore, the use of high compression ratios can lead to issues such as ringing, which occurs when the intensity of a combustion cycle is too high. This ultimately leads to an expansion of gas at dangerous speeds causing oscillation within the cylinder. 28
An alternative and more attractive approach is the use of a more reactive fuel along with ammonia, which acts as an ignition promoter to counteract ammonia’s low reactivity and low flame speed. Diesel has been by far the most popular choice to this regard, enabling the use of ammonia to as much as 95% of the fuel blend.29,30 Other fuels that have been tested in a dual fuel CI engine mode are DME,31,32 kerosene,33,34 and hydrogen with proper charge preheating.28,35,36 Of all the aforementioned fuel choices, only hydrogen is non carbon-based. But the preheating of the charge in the case of hydrogen has a detrimental effect on the engine performance characteristics.
28
Therefore, the ignition promoter should ideally be: (i) sufficiently strong to enable ignition without requiring any charge preheating, and (ii) non-carbon based, to avoid the production of any
A potential candidate to serve as an ignition promoter for ammonia’s combustion in CI engines is hydrogen peroxide (
The available scientific literature on the use of ammonia and hydrogen peroxide blends is extremely scarce. Khalil et al.
48
used batch reactor simulations to demonstrate the strong effect that hydrogen peroxide can have on the ignition delay time promotion of ammonia. They showed that a 2% addition of
All the aforementioned studies significantly increased our understanding on ammonia/hydrogen peroxide combustion. The reported promising results clearly demonstrate the necessity for further research that will showcase the feasibility of the proposed technology. Therefore, the aim of the current work is to build on the available knowledge and explore the feasibility of using ammonia/hydrogen peroxide blends in the context of a simplified HCCI engine model, in view of engine performance characteristics, combustion phasing and NOx emissions. The simplified numerical model available in the Chemkin Pro 52 suite is selected because it will allow for a wide exploration of the parametric space with a small computational cost. The employed computational model is a simplified one, therefore has some inherent weaknesses due to the underlying assumptions made to reach a simple mathematical formulation. Yet, it can still provide some valuable insight if carefully used, which justifies its wide use by the research community.35,53,54 Subsequently, the results produced by such a simplified model will need to be treated with care, and the emphasis should be placed on exploring the qualitative aspects rather than the quantitative ones. In addition, the obtained results will be used to identify trends that can credibly inform subsequent analyses of the fuel blend in either high-fidelity simulations or real-world engine testing.
Material and methods
In the current work a single zone zero-dimensional HCCI engine model was employed, available within the Chemkin Pro suite. The employed model is fully closed (no mass exchange) and solves the species mass fractions and energy equations, along with an additional equation for the volume change. In principle, the model considers the system to be fully homogeneous, with the effect of the transport processes being negligible. Since the HCCI combustion is a chemical kinetic combustion process controlled by the thermodynamic conditions of the in-cylinder charge and not by the flame propagation these approximations are reasonable. 55 Yet, the results should be treated with care because the preparation of a fully homogeneous charge with no spatial compositional and thermal inhomogeneities may not always be used (sometimes intentionally) and in these cases mixed combustion modes (deflagration and spontaneous ignition) can co-exist, thereby affecting the overall engine performance.56–58 For a detailed description of the model the reader is referred to the Chemkin Pro manual. 52 Here, only a brief description will be provided.
Considering a system of
where
where
where
where
where
where
where
where
The chemistry in the simulations was modeled by the detailed chemical kinetic mechanism of Zhang et al. 59 which includes 38 species and 262 chemical reactions and has been extensively validated. 59 The engine’s parameters are summarized in Table 1. In all simulations, the compression ratio was maintained at 17:1, whilst the bore and stroke were 100 and 105 mm, respectively. Unless otherwise stated, the intake temperature and pressure remained constant at 320 K and 1.065 atm. Since hydrogen peroxide is in liquid phase at these conditions, a fuel vaporizer would need to be used, like those typically used for diesel operation in HCCI mode.60–62 The study focused on two engine speeds, 750 and 1150 rpm. Finally, all simulations started at −180 crank angle degrees after top dead center (CAD aTDC) and ended at +180 CAD aTDC.
Engine parameters and operating conditions.

Addition of H2O2 was done on a mole fraction basis of NH3. For example, a 10% addition of H2O2 specifies the mole fraction of H2O2 added on a 10% basis of the mole fraction of NH3. However, as a result of hydrogen peroxide acting as an oxidiser (and a fuel), the traditional method of calculating the equivalence ratio:
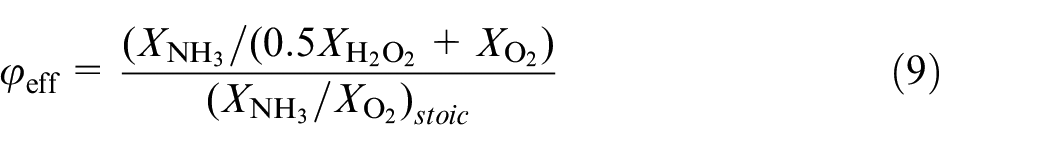
Equation (9) is derived on the assumption that 1 mol of H2O2 generates 1 mol of H2O and 0.5 mol of O2. The notation stoic indicates that stoichiometric conditions must be considered. Indicative values of the initial mole fractions used for the examined compositions are shown in Table 2.
Indicative values of the initial mole fraction values of the charge at various equivalence ratios.
On the basis of the indicated work being Wind, and the engine speed denoted by N, the indicated power can be defined as:

Subsequently, torque can be derived using

whilst the swept volume (V disp ) can be used to define the indicated mean effective pressure,

using

where sfc indicates the specific fuel consumption (sfc = (mfN)/(2Pind)),
Results and discussion
Comparison of the hydrogen peroxide use strategy against the glow plug approach
The first part of the investigation is focused on comparing the proposed technology (i.e. the use of hydrogen peroxide as an ignition promoter) against the more conventional use of a glow plug which preheats sufficiently the charge to enable its ignition close to the TDC. We firstly consider adiabatic conditions (i.e.
Engine performance results for different engine speeds (750 and 1150 rpm) and effective equivalence ratios (
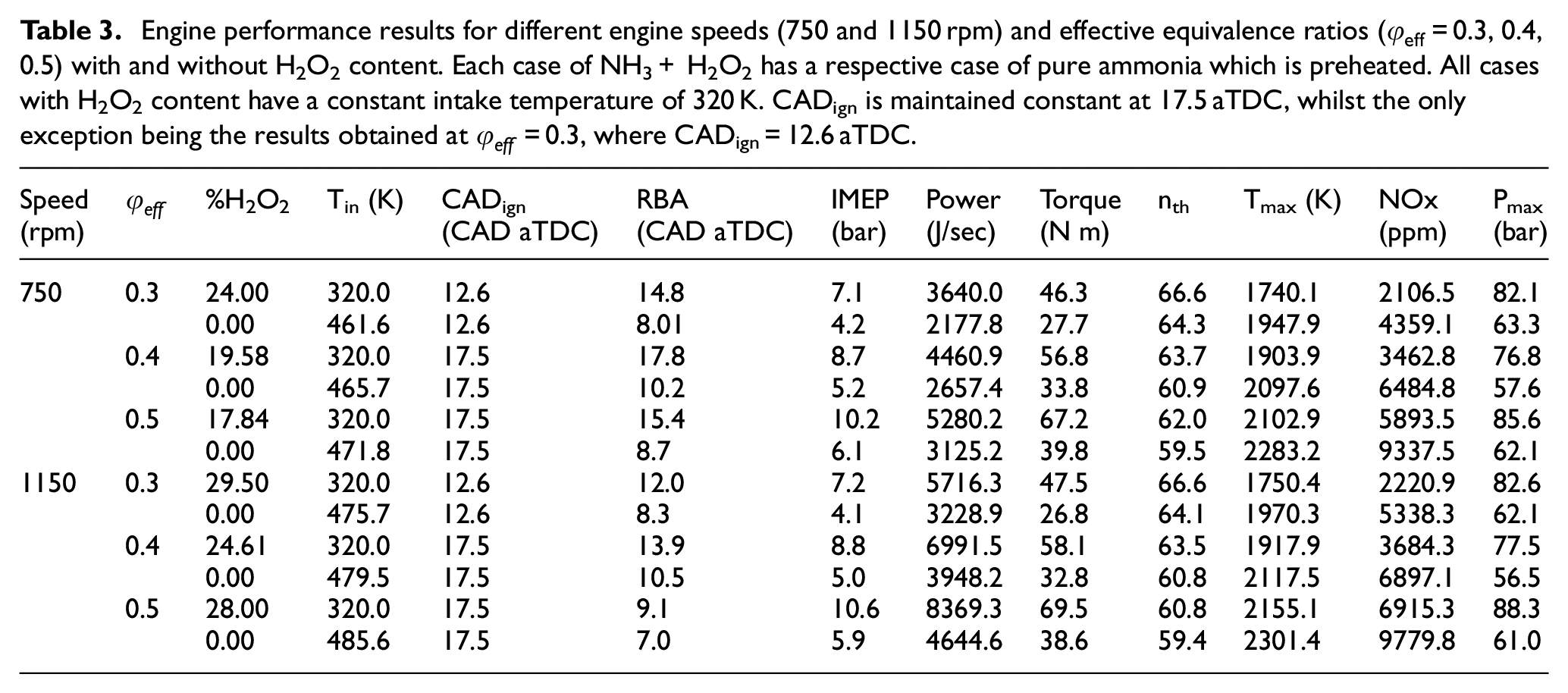
In particular, the results at an effective equivalence ratio of
Increasing the engine speed from 750 to 1150 rpm while maintaining the effective equivalence ratio and the
Moving onto
At
In summary, the following conclusions can be drawn for the hydrogen peroxide addition approach when compared to the inlet preheating method:
IMEP, power and torque all increase significantly (>65%), with their increases remaining practically unaffected by the increase of the effective equivalence ratio but becoming more pronounced at higher engine speeds. This will be discussed further in the following chapters.
the thermal efficiency exhibits a small increase, regardless the effective equivalence ratio and the engine speed.
the maximum temperature is notably decreased by 6%–11%. This decrease has a significant effect on the production of NOx, leading to a decrease of 29%−52%. The decreases of
the maximum pressure increases significantly by 30%–45%. Its increase is favored by the increase of the effective equivalence ratio and/or the increase of the engine speed.
the RBA increases significantly by 30%−85%, with its increase becoming less pronounced as the effective equivalence ratio and/or the engine speed increase.
In order to understand what causes the higher engine performance with the use of hydrogen peroxide, we have to remember that the introduction of hydrogen peroxide leads to the increase of the mixture’s energy density. Considering that the introduction of hydrogen peroxide occurs on a volumetric basis and that hydrogen peroxide has significantly higher volumetric energy density than ammonia at the examined conditions (4.428 MJ/l for
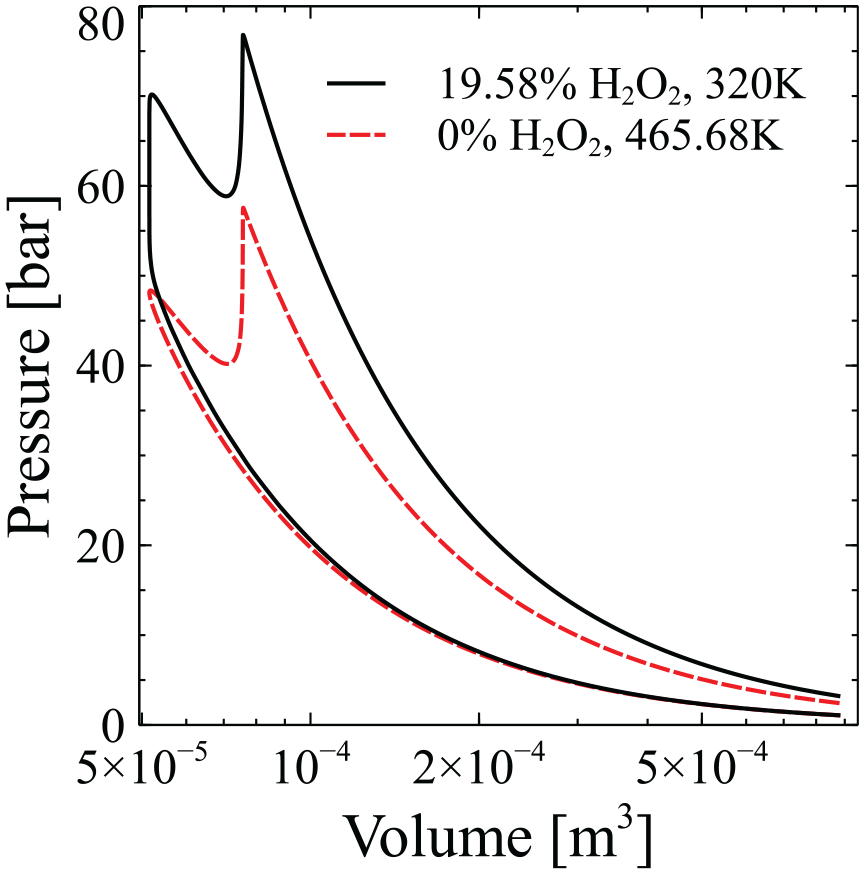
The P-V diagram for the cases of 19.58%
It is worth noting that for the cases considered in Table 3, the inlet temperature in the absence of any hydrogen peroxide had to be increased by 44%–52%. At the same time, the hydrogen peroxide approach required hydrogen peroxide addition ranging between 24% and 28%. A closer investigation of the inlet temperature and hydrogen peroxide addition values reveals something unexpected. In the cases where no hydrogen peroxide is added to the mixture, the inlet temperature has to be increased with the increase of the equivalence ratio. This is not surprising since the reactivity of ammonia decreases as the mixture becomes richer, that is, the ignition delay time increases when moving from fuel lean to stoichiometric mixtures.66–68 The unexpected finding is the variation of the required amount of hydrogen peroxide at 1150 rpm; from fuel lean to fuel rich conditions, the required percentage of hydrogen peroxide initially decreases and then increases. This anomaly is observed at 1150 rpm and not 750 rpm where the hydrogen peroxide percentage decreases with the increase of the effective equivalence ratio.
In order to investigate further this unexpected finding, in Figure 2 we present the variation of
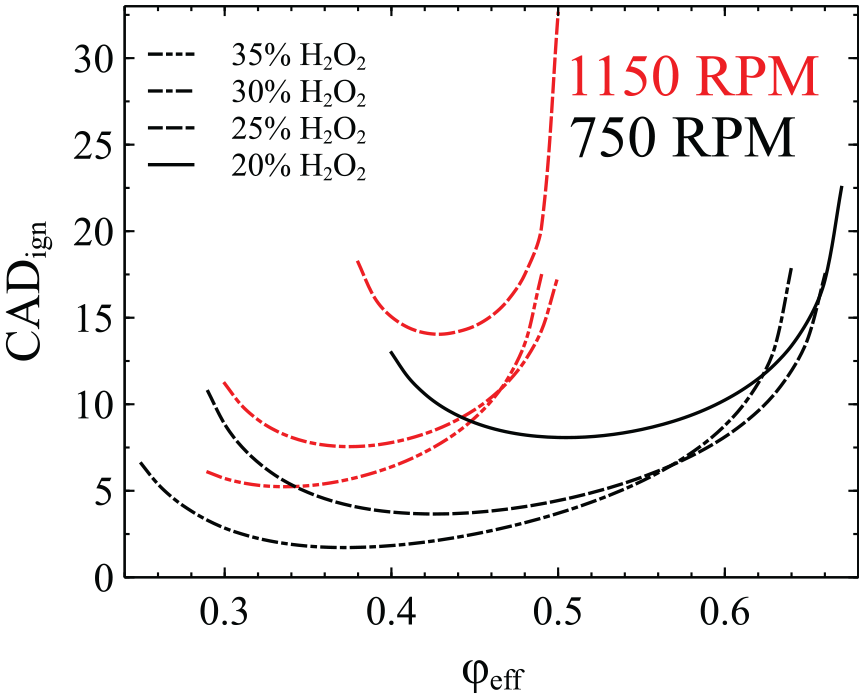
The variation of
In order to understand what causes the non-monotonic response in
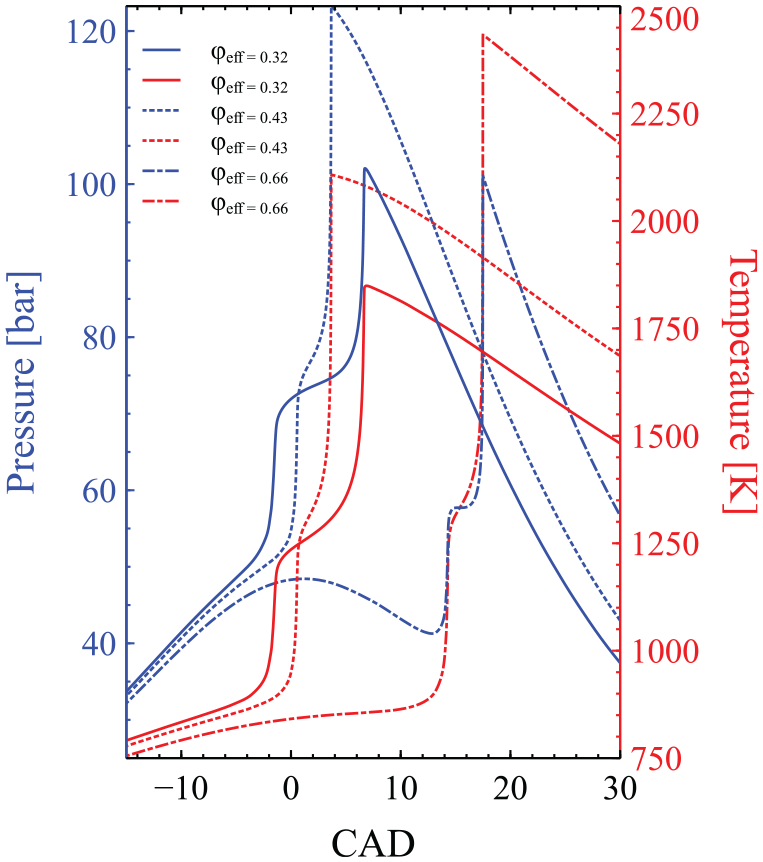
The evolution of pressure (bar) and temperature (K) against CAD for three cases of
To substantiate further the critical role of the first stage ignition on the non-monotonic response of
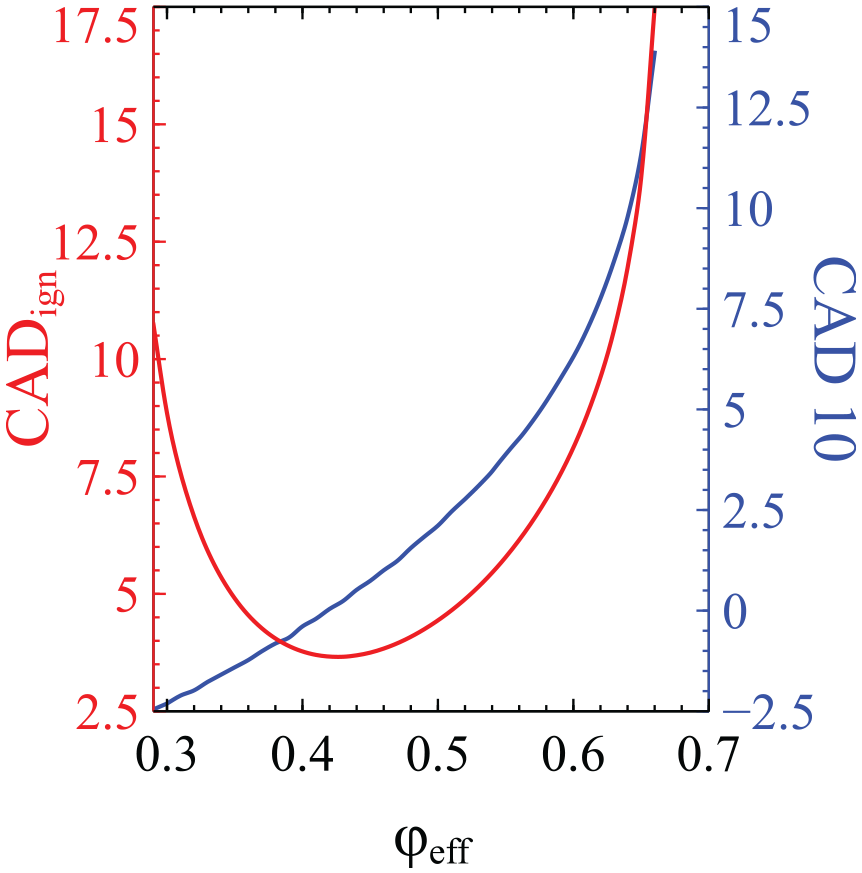
The variation of
The effect of the incremental increase of the initial amount of hydrogen peroxide on the engine performance characteristics at constant equivalence ration (
)
The next analysis takes into consideration the incremental addition of H2O2 at
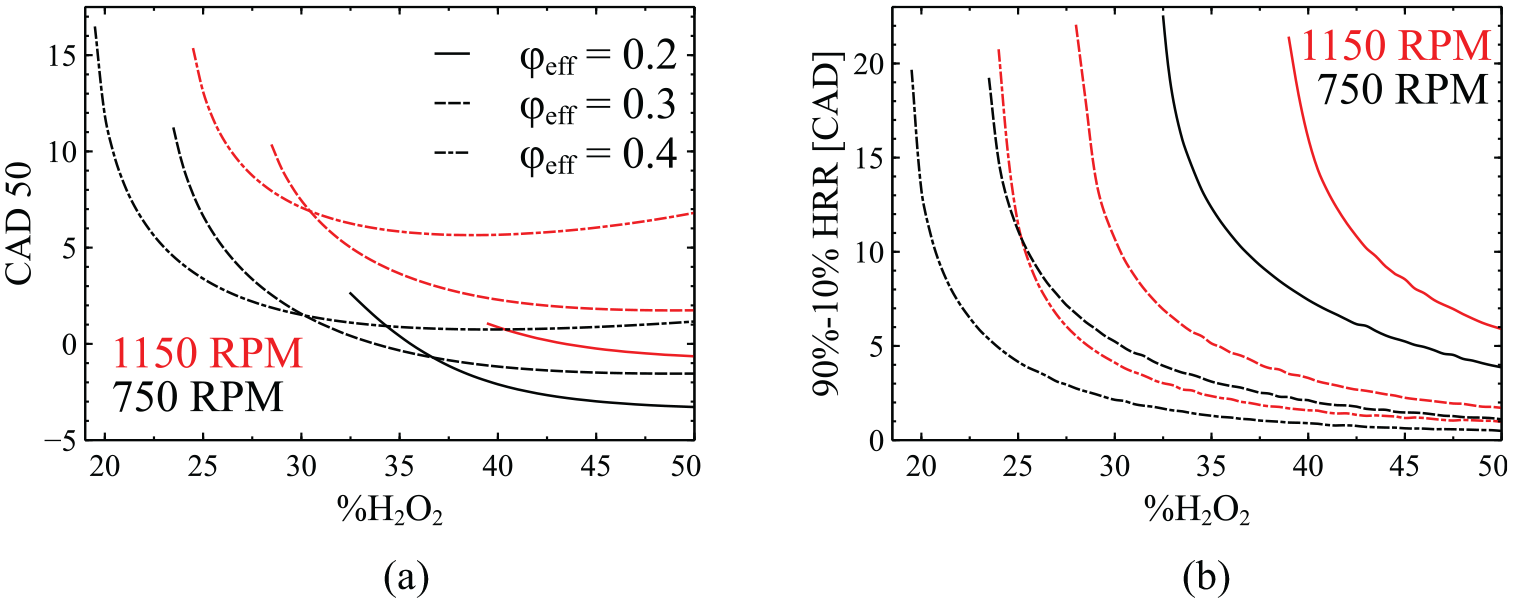
Variation of CAD50 (a) and the RBA (b) as a function of H2O2 addition for three different cases of
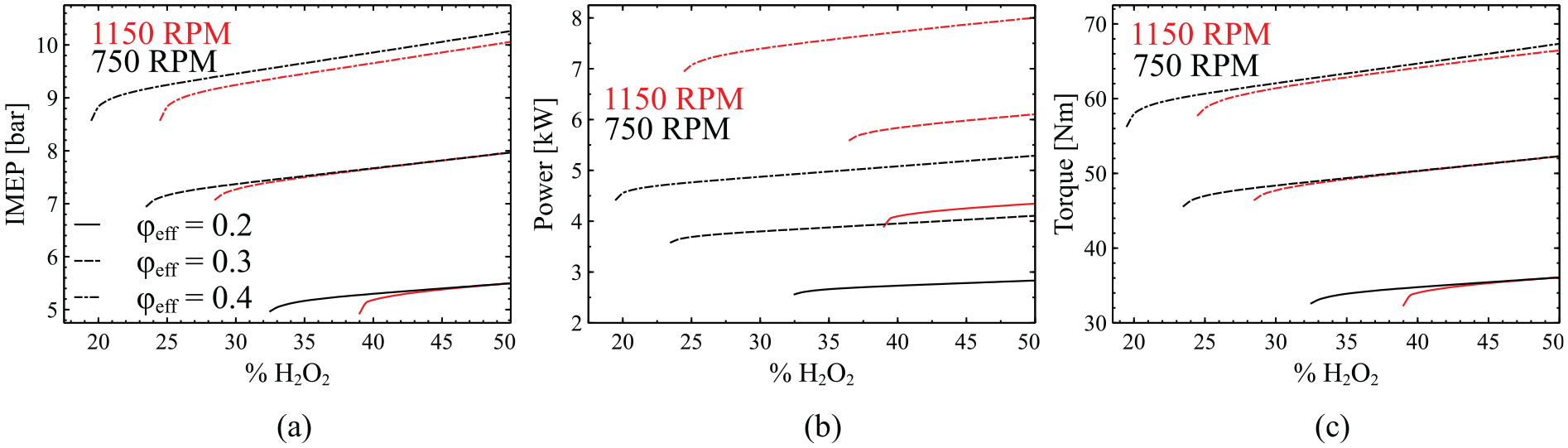
Variation of IMEP (a), power (b), and torque (c) as a function of H2O2 addition for three different cases of
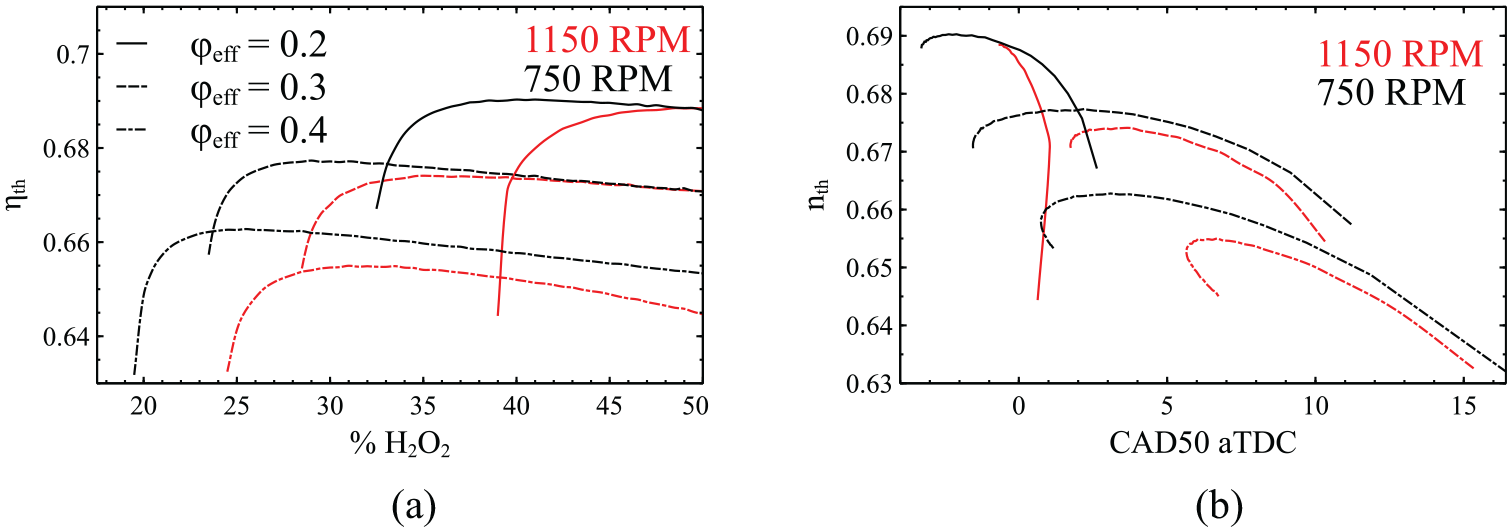
Variation of the thermal efficiency as a function of H2O2 addition (a) and CAD50 (b) for three different cases of
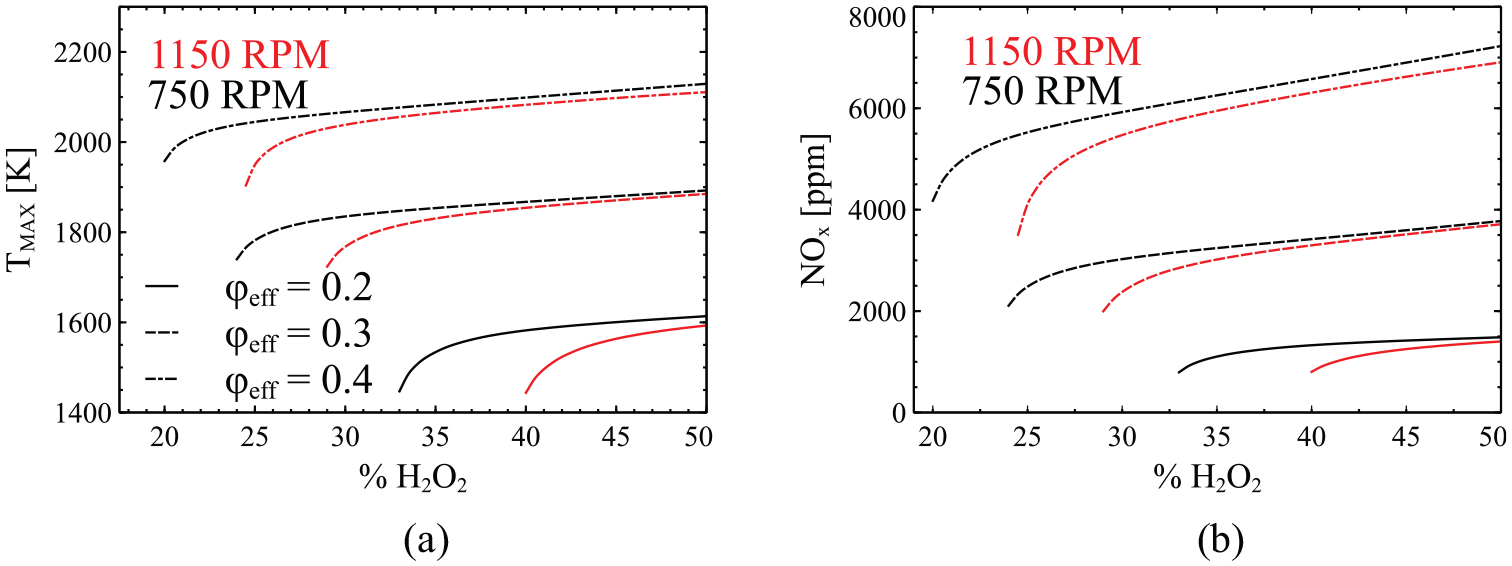
Variation of exhaust NOx emissions (a) and maximum temperature (b) as a function of H2O2 addition for three different cases of
In Figure 5(a) it is shown that the addition of hydrogen peroxide initially has a strong effect on the CAD50 advancement but shortly afterward levels off. For instance, at
As the engine speed increases, it becomes more difficult for the system to reach negative CAD50 values. In fact, the advancement of CAD50 to negative degrees is barely achieved for extremely lean mixtures of
The significant influence of the
The analysis will now focus on the effect of incremental hydrogen peroxide addition on IMEP, power and torque, as illustrated in Figure 6. Firstly, it can be easily observed that all three performance outputs exhibit in principle a linear response to the hydrogen peroxide addition. Exploiting this feature, a linear regression analysis was performed to determine how the response of IMEP, torque and power is affected by the change in
Figure 6 also shows that at 750 rpm and 35% H2O2: (i) at
The effect of H2O2 addition on the thermal efficiency is also displayed in Figure 7. The first observation that can be made is that the thermal efficiency generally drops with the increase of
To obtain further insight on the relation between the thermal efficiency and the combustion process, in Figure 7(b) the thermal efficiency is plotted against CAD50. The percentage of the added
The last part of this analysis focuses on NOx emissions. Figure 8 displays the variation of NOx emissions, on a basis of ppm units and maximum in-cylinder temperature, as a function of H2O2 addition at engine speeds of 750 (a,c) and 1150 (b,d) rpm and at
The effect of the incremental increase of the initial amount of hydrogen peroxide on the engine performance characteristics at constant engine load conditions
The next part of analysis considers the effect of H2O2 addition on NOx emissions, CAD50 and thermal efficiency whilst keeping the engine load (IMEP, indicated power, and torque) constant across an
In Figure 9 we firstly observe that the required amount of hydrogen peroxide addition to achieve the desired load outcome reaches 100% when the mixture becomes very lean (
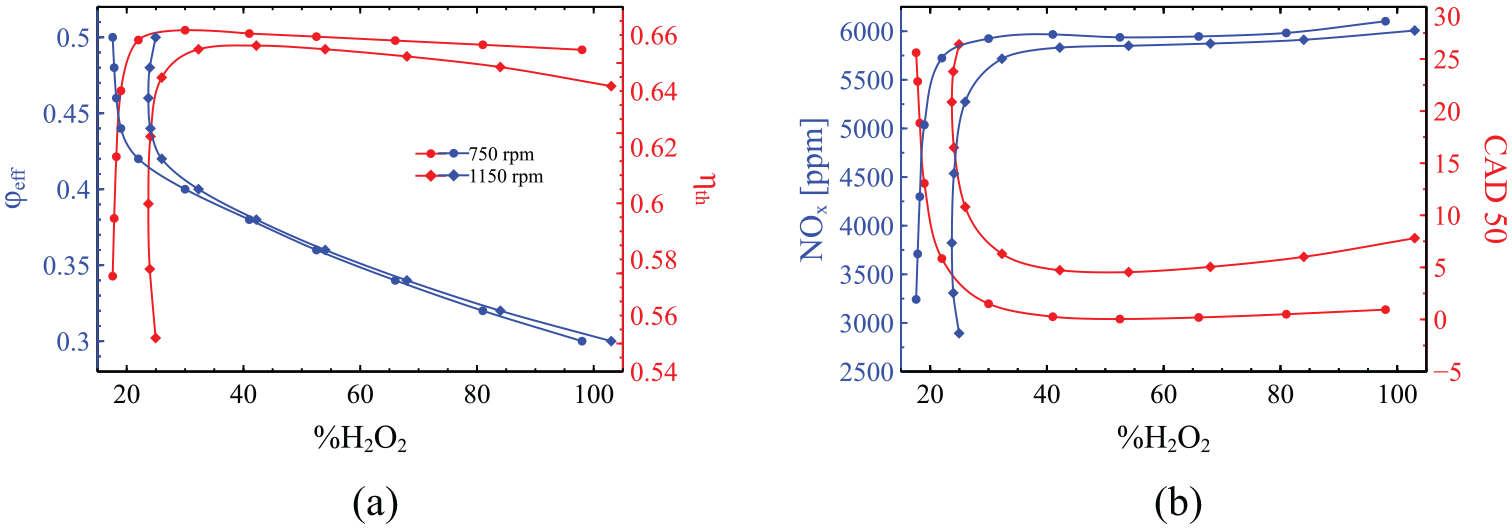
The effect of H2O2 addition on the thermal efficiency, the NOx emissions and CAD50 as the effective equivalence ratio is incrementally increased whilst adjusting the
In the last part of the analysis, the aim was to highlight the benefits of H2O2 addition on the engine performance characteristics whilst providing a direct comparison with the conventional method of pre-heating ammonia, but opposite to the analysis so far, now heat losses are considered. The heat losses are the cause for some notable differences in the results compared to the respective ones reported in Figures 5 to 8 for the adiabatic cases. Initially, H2O2 was incrementally added on a mole fraction basis of NH3 in steps of 1% from 27% to 35%. The load output obtained from this exercise was then reproduced by removing the hydrogen peroxide and increasing properly the inlet temperature. If an increase in temperature did not suffice that is, the same power was not achieved using the glow-plug approach as for the case containing H2O2, the
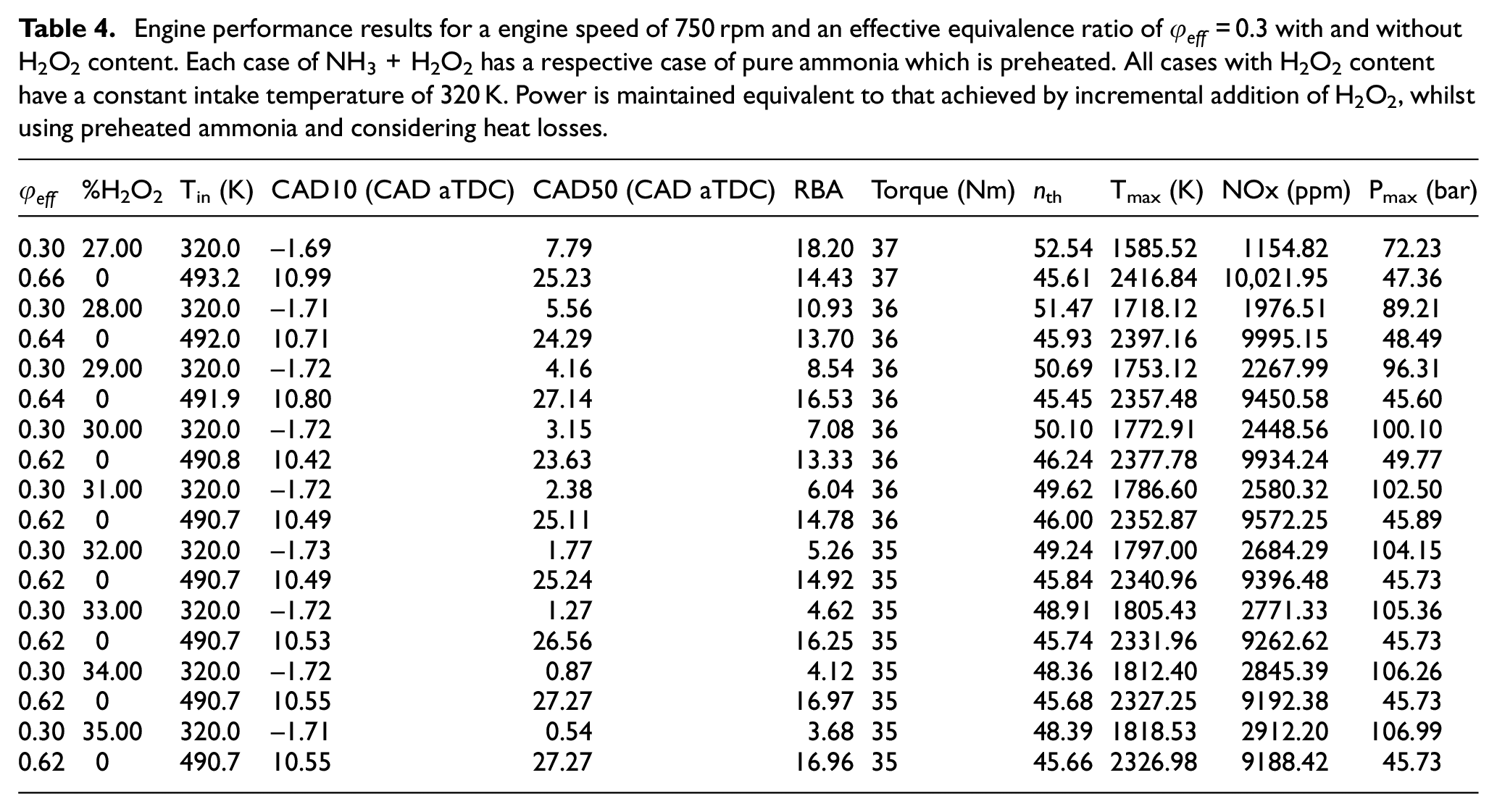
The results concerning this analysis are shown in Table 4. Firstly, the glow plug approach generally requires that the equivalence ratio is doubled and the inlet temperature is increased by more than 50% for all examined cases, while with the hydrogen peroxide approach the effective equivalence ratio and the inlet temperature are both kept fixed at 0.3 and 320 K, respectively. With regard to combustion phasing, CAD10 is consistently maintained to negative values for the hydrogen peroxide approach while in the glow plug approach it is maintained at ∼11 CAD aTDC. The difference of the CAD10 values between the two approaches is in the neighborhood of 12 degrees for all cases under consideration. On the other hand, the difference in the CAD50 values between the two approaches becomes more pronounced ranging between 17 and 28 degrees. This occurs because the increase of the hydrogen peroxide addition leads to the gradual advancement of CAD50 and at the same time the increase of the equivalence ratio to reach the desired load outcome in the glow plug approach leads to the gradual retarding of CAD50. This phenomenon also explains why the difference in the RBA values between the two approaches increases gradually with the increase in the hydrogen peroxide addition. In addition, unlike to what was reported earlier in the discussion on Table 3, the difference in the thermal efficiency between the two approaches becomes more notable. The thermal efficiency with the hydrogen peroxide addition approach varies in the range of 48%–53%, which is consistently higher than the respective thermal efficiency with the glow plug approach, by 3%–7%, depending the conditions. This is a reasonable finding since most of the heat release in the hydrogen peroxide addition approach occurs close to the TDC, much closer compared to the glow plug approach. More spectacular though is the difference of the maximum reached temperature. The glow plug approach leads consistently to much higher maximum temperatures, with this difference ranging between 28% and 52% against the hydrogen peroxide addition approach. The higher maximum temperatures reached in the glow plug approach are the result of the higher employed equivalence ratios and inlet temperatures. These higher temperatures directly affect the production of NOx emissions which increase by a factor of 3 to a factor of 9 (almost an order of magnitude) compared to the hydrogen peroxide addition approach. Finally, in agreement with what was reported earlier in the discussion on Table 3, the only caveat with the use of hydrogen peroxide is the increased maximum pressures, which when compared to the glow plug approach exhibit increases which range between 50% and 130%.
Engine performance results for a engine speed of 750 rpm and an effective equivalence ratio of
Conclusions
Ammonia is one of few main candidates for future use in heavy duty applications, however its unrealistic ignition delay times due to its high auto-ignition temperature has made it an unattractive solution for the decarbonization of heavy duty applications. In the current study, hydrogen peroxide was considered as an ignition promoter of an ammonia-fulled HCCI engine. The numerical work was conducted on the basis of a single zone simplified model available in the Chemkin Pro suite, using a detailed chemical kinetic model. 54 The objective of the work was to provide numerical evidence for the feasibility of the proposed technology, while also identifying potential challenges in its implementation. The employed numerical model, albeit simplified, enabled the qualitative analysis of the proposed technology and the identification of trends that will inform future experiments and/or computationally expensive high fidelity numerical simulations. The employed numerical model despite its inherent weaknesses provided valuable insight about the feasibility of the new technology, yet the produced results and the reached conclusions should be interpreted with care, due to lack of proper validation with engine experiments.
The conclusions can be summarized as follows:
hydrogen peroxide addition was proven to be advantageous against the more conventional approach of preheating the ammonia/air charge, typically done with the use of a glow plug. In particular, the hydrogen peroxide addition led to significantly higher IMEP, power and torque, mainly due to the increase of the mixture’s energy density. The thermal efficiency exhibited much lower, yet non-negligible, increase. NOx emissions were also found to decrease tremendously. Under constant load conditions, the hydrogen peroxide addition approach led to a 9-fold decrease in NOx. Yet the obtained NOx emission values were still high, thereby suggesting the necessity for the use of an additional technology for their treatment.
the introduction of hydrogen peroxide leads to a two-stage ignition process. The first ignition stage is instrumental in the control of the whole ignition process since it was found to be retarded with the increase of the hydrogen peroxide addition while the second ignition stage was advanced. This led to: (i) a rapid decrease of the RBA and (ii) a non-monotonic response of the ignition CAD to the hydrogen peroxide addition.
hydrogen peroxide addition generally leads to higher IMEP, power, torque, and NOx emissions values. In fact these variables largely exhibit linear response to the hydrogen peroxide addition. Linear regression analysis revealed that hydrogen peroxide tends to increase IMEP, power and torque and this tendency is favored as the effective equivalence ratio increases. As the engine speed increases the response of IMEP, power and torque are characterized by a higher rate of change but this phenomenon becomes attenuated as the effective equivalence ratio increases. Additionally, hydrogen peroxide addition leads to increase of NOx but this increase becomes more pronounced as the effective equivalence ratio and/or the engine speed increase.
the incremental addition of hydrogen peroxide leads initially to a rapid increase of the thermal efficiency which at some point reaches a maximum value and after that point further increase of the hydrogen peroxide addition leads to a gradual decrease of the thermal efficiency. The thermal efficiency is achieved when CAD50 gets closest to the TDC. As the mixture becomes leaner the CAD50 associated with the maximum thermal efficiency is advanced and in the case of sufficiently lean coditions it even becomes negative.
The results presented herein for the feasibility of the proposed use of hydrogen peroxide as an ignition promoter for ammonia use in CI engines, are promising. Yet, further research is required. Few of the issues that we believe merit further investigation, as highlighted by the current works, are:
the chemical mechanism that is responsible for the control of the two ignition stages, particularly the first stage ignition which was found to be key to the control of the ignition process;
the implementation of different strategies for NOx mitigation such as the use of exhaust gas recirculation (EGR) or steam dilution;
the limitations in terms of the pressure rise rate and the combustion efficiency.
Footnotes
Appendix
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: OS received financial support from Edinburgh Napier University through the Starter Grant scheme (N452-000).