Abstract
In dual-fuel compression-ignition engines, replacing common fuels such as methane with renewable and widely available fuels such as methanol is desirable. However, a fine-grained understanding of diesel/methanol ignition compared to diesel/methane is lacking. Here, large-eddy simulation (LES) coupled with finite rate chemistry is utilized to study diesel spray-assisted ignition of methane and methanol. A diesel surrogate fuel (n-dodecane) spray is injected into ambient methane-air or methanol-air mixtures at a fixed lean equivalence ratio
Introduction
Diesel engines have still a dominant role in large scale power production and marine engines. 1 While the technology is relatively mature and robust, the major concern is to limit its pollution contribution. 1 This concern has led to promising technologies over the past decade, including dual-fuel (DF) combustion, which feature low NOx and soot emission. 2 In particular, the main research focus of DF combustion has been on extending the operational range of DF compression ignition (CI) engines while gaining higher efficiency and lower emissions.2,3 In the context of renewable fuels, one of the common challenges is to ensure smooth and robust control over the DF ignition process for different fuel combinations and operational ranges.4,5
Ignition process in multi-fuel combustion systems is an intricate process due to the interlinked length and timescales of chemistry and mixing phenomena. 6 Of particular importance is to acquire knowledge on the mixing process, low temperature chemistry (LTC), high temperature chemistry (HTC), and ignition delay time (IDT), which helps to develop and implement the technology in practice. Despite the recognized importance of DF combustion implementation, specifically in heavy-duty engines,2,7 a fine-grained understanding of the ignition phenomenon in such stratified reactivity systems, particularly for renewable fuels such as methanol, is still lacking. In the present study, three-dimensional (3d) large-eddy simulations (LES) are employed to investigate mixing and ignition phenomena in DF diesel/methane and diesel/methanol cases corresponding to the Spray A baseline conditions of the engine combustion network (ECN) (https://ecn.sandia.gov). Such an LES investigation helps in understanding the intertwined physics and chemistry processes in reacting DF sprays. 8
In a typical CI DF combustion, a low-reactivity fuel (LRF) with a relatively long IDT is the background primary fuel. The LRF is interactively burned with small amounts of a high-reactivity fuel (HRF) with a relatively shorter IDT, such as diesel. In this configuration, the liquid HRF is injected into the lean LRF/oxidizer mixture in the cylinder and IDT stratification is created. The role of HRF is to facilitate the ignition process, whereas the LRF moderates the system reactivity and delivers the main energy release. 3 Recently, there has been substantial progress in the characterization of ignition mechanism in DF diesel/methane under CI engine relevant conditions. In an LES study of DF ignition in diesel-surrogate/methane under the ECN Spray A injection conditions, Kahila et al. 6 specified the role of ambient methane on prolonging the ignition delay time by a factor of two. They explained this effect by detailed LTC and HTC analyses. In a following study, 9 they demonstrated the impact of diesel pilot injection duration on spray overleaning and consequently, the IDT. The findings were supported by engine experiments. More recently, in the studies by Tekgül et al. 10 and Kannan et al. 11 , effects of ambient temperature on IDT with different chemical mechanisms were investigated. The present study is a continuation of these works to study the ambient methanol influence on the ignition process, as compared to methane, under various ambient temperatures.
Recently, a few experimental studies on DF combustion have been carried out with methane as the LRF,12–15 which mainly focus on the performance, efficiency, emissions, and combustion instability. Nonetheless, several insights have been obtained on ignition characteristics. For instance, the previous studies indicate that mixing methane with oxidizer retards both low- and high-temperature ignition stages. These findings match well with the findings in previous numerical studies.6,9–11,16–20 Moreover, direct numerical simulations (DNS) have provided numerical evidence on the importance of LTC and HTC in the DF ignition process.21,22 Using renewable fuels such as methanol in DF CI engines have been studied both experimentally23–26 and numerically.27–34 Those studies predominantly focus on the engine geometry and performance,23,28,30–32 fuel reactivity,24,27,28 combustion instability, 25 and emissions characteristics.26,27,29–31,33
Using methanol over methane, natural gas, and gasoline as the LRF has attracted the interest of DF engines manufacturers over the past few years due to several reasons. 35 First, methanol is a renewable fuel, and it is widely available. 4 Second, its high octane number can mitigate knock tendency in engines. 36 Third, since there is an oxygen atom in the methanol formula and it is free of aromatics, soot formation is mitigated. Fourth, methanol has a higher heat of vaporization compared to methane, natural gas, and gasoline, which leads to cooling effects during combustion and consequently, lower charge peak combustion temperature leading to smaller NOx emissions. 4 With relevance to this study, methanol ignition is a particularly sensitive process and several challenges on the smooth ignition of methanol in the DF engines context have been reported in the literature, where for example the crucial role of intake temperature control has been acknowledged in the works by Zou et al. 31 , Wang et al. 37 , Hu et al. 38 and Wang et al. 39 Recently, Xu et al. 34 investigated effects of increasing the initial ambient methanol-air mixture fraction on ignition at a constant temperature of 900 K. They provided evidence that adding methanol to the ambient may delay the ignition of n-heptane and prolong the transition from low to high-temperature ignition. More recently, we have investigated tri-reactivity ignition of methane/hydrogen 40 and methanol/hydrogen 41 at fixed temperature of 900 K by diesel spray. However, in those studies we did not thoroughly discuss the dual-reactivity, that is dual-fuel case of diesel/methanol versus diesel/methane at various ambient temperatures. Here, we carry out a systematic analysis to fill the research gap with regard to the effect of ambient temperature on diesel spray assisted ignition of methane and methanol.
The overarching goal of this study is to investigate utilization of methanol, as a renewable fuel, in the DF CI context as compared to methane through spray LES, following the recent research framework developed by Kahila et al.6,9 This is the first study comparing n-dodecane/methanol and n-dodecane/methane DF spray ignition for different ambient temperatures using LES and finite rate chemistry. Accordingly, the main objectives of this study are to (1) understand DF ignition at different ambient temperatures when methane is replaced by methanol, (2) compare the mixing and ignition characteristics of DF spray systems including n-dodecane/methanol and n-dodecane/methane, (3) identify ignition modes and the relative importance of LTC/HTC, (4) provide a further insight into chemical reactions yielding different ignition delay times for n-dodecane/methane and n-dodecane/methanol using reactions sensitivity analysis, and (5) provide and test a simple approach to estimate spray ignition delay time using zero-dimensional (0d) homogeneous reactor calculations and non-reacting spray LES data. We note that the main goal of this work is to study differences of diesel spray-assisted ignition of methane and methanol. However, these results do not represent the overall combustion in an engine cycle since the effect of volumetric compression is not included. Finally, a list of all abbreviations used in this work is provided in Table 1 to improve the readability of the paper.
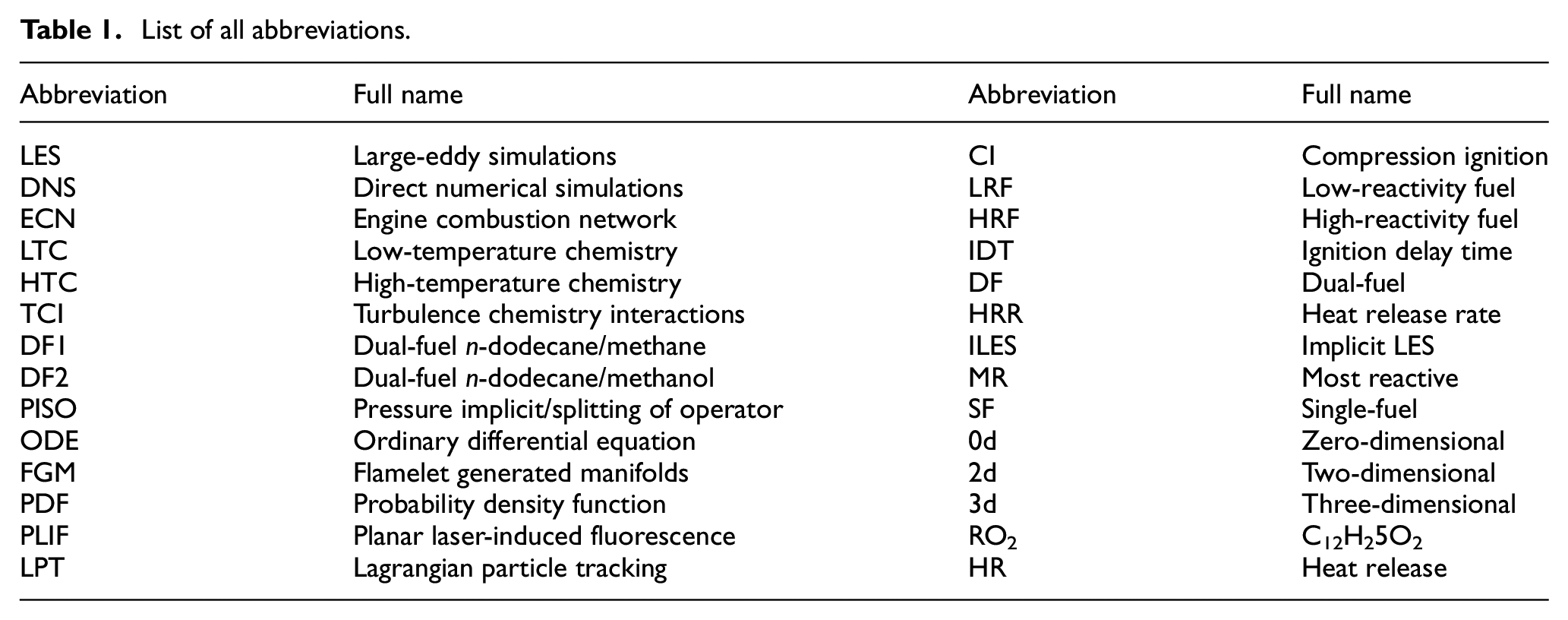
List of all abbreviations.
Methodology
Case setup
The simulation configuration is based on the modified ECN Spray A baseline conditions for DF ignition.6,9 As in the ECN Spray A setup, liquid n-dodecane (diesel-surrogate) at T = 363 K is injected from a 90
Ambient premixed gas initial conditions in spray
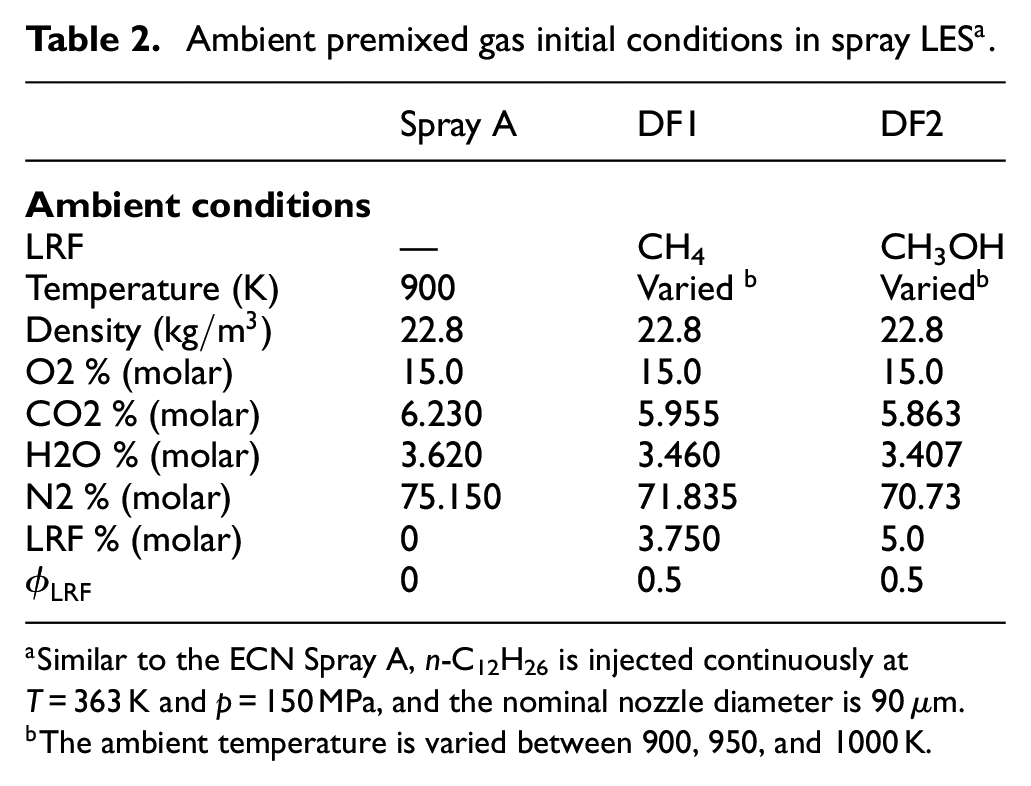
Similar to the ECN Spray A, n-C12H26 is injected continuously at T = 363 K and p = 150 MPa, and the nominal nozzle diameter is 90
The ambient temperature is varied between 900, 950, and 1000 K.
Numerical methods
Reacting LES is carried out within the OpenFOAM finite-volume framework 42 integrated with our open-source finite-rate chemistry solver.43,44 Modeling assumptions and discretization methods are similar to our previous LES studies.6,9,10 Nonetheless, assumptions and methods are briefly mentioned here. For further details, the reader is referred to the work by Kahila et al. 6
LES formulation of the governing equations, that is continuity, momentum, species concentration, and enthalpy, with Favre-filtering is the same as those by Kahila et al.,6,9 and Tekgül et al. 10 which are provided as:

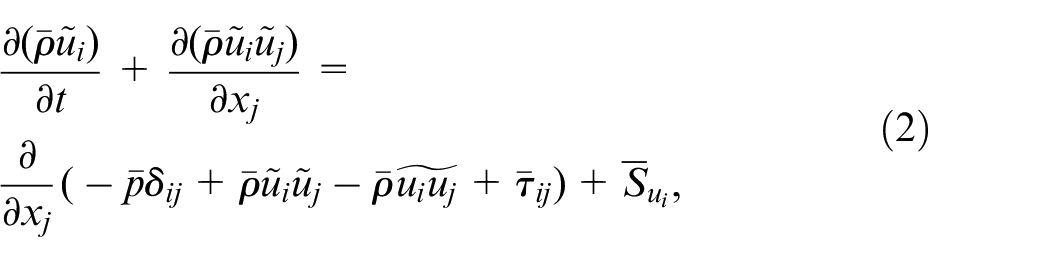
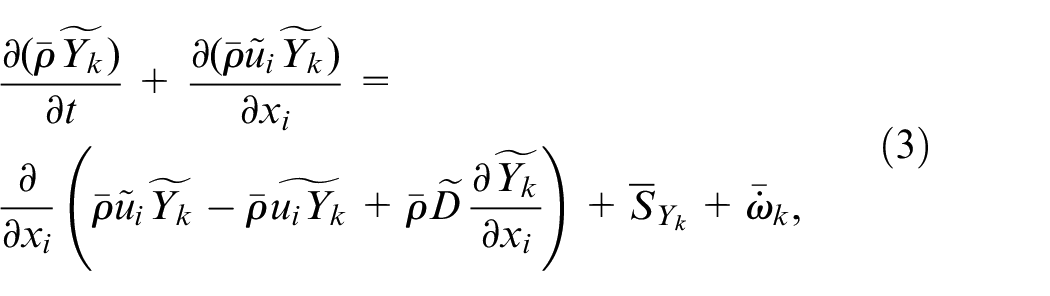
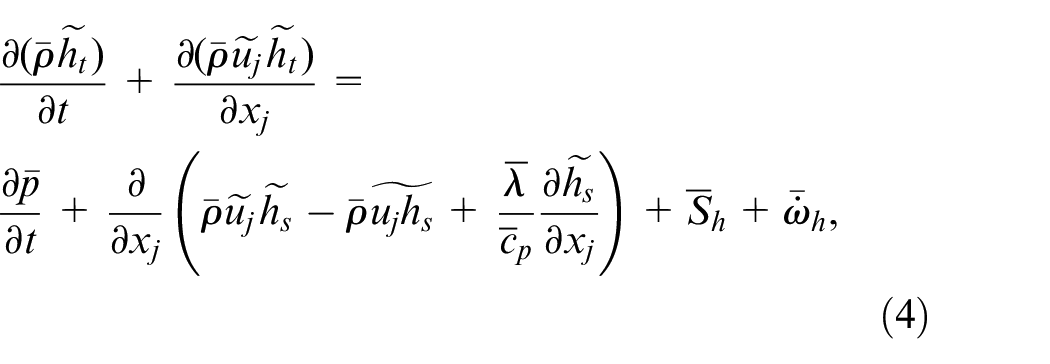
where
In the finite volume solver, an implicit, three time-level, second-order scheme is used for time integration. A second-order central scheme discretization for diffusion terms along with a non-linear flux limiter for convective flux treatment 45 are utilized as in the authors’ previous spray studies.46,47 The compressible solver utilizes the standard PISO (pressure implicit with splitting of operator) pressure-correction approach. For the injected liquid phase modeling, the Lagrangian particle tracking (LPT) method is applied using the cylindrical injection volume approach, as discussed by Gadalla et al., 48 which extends the standard disk injection model of OpenFOAM to 3d. The standard correlations of Ranz and Marshall 49 and Frossling 50 are used for modeling heat and mass transfer between the liquid and gas phases.
It is noteworthy that the utilized Implicit LES (ILES) approach has been previously demonstrated to be a reliable assumption as compared to other SGS models, c.f. Grinstein et al., 51 Grinstein and Fureby, 52 Aspden et al., 53 and it has been applied to several different problems for example in studies on free shear flows, 54 supersonic jets, 55 and sprays.6,9,46,47,56 Here, the same implicit LES/LPT approach as in the previous studies by the authors is used wherein extensive validation studies on reacting6,9–11 and non-reacting48,56 single-fuel (SF) and DF ECN Spray A are provided. Furthermore in this work, the no breakup model approach for the droplet secondary atomization is utilized with ILES following our recent model sensitivity study by Gadalla et al. 48
The utilized reacting flow solver is based on the recent work by Kahila et al.
6
In the solver, the open source library pyJac
57
is coupled with OpenFOAM providing the analytical Jacobian matrix formulation required by the ordinary differential equation (ODE) system solver. The operator-splitting strategy is utilized to separate the chemical source terms
Mesh structure and sensitivity analysis
Figure 1 provides the physical dimensions and structure of the mesh in the computational domain. The mesh consists of three refinement regions similar to our previous studies, for example Kahila et al.
6
Here, a standard mesh resolution, 125
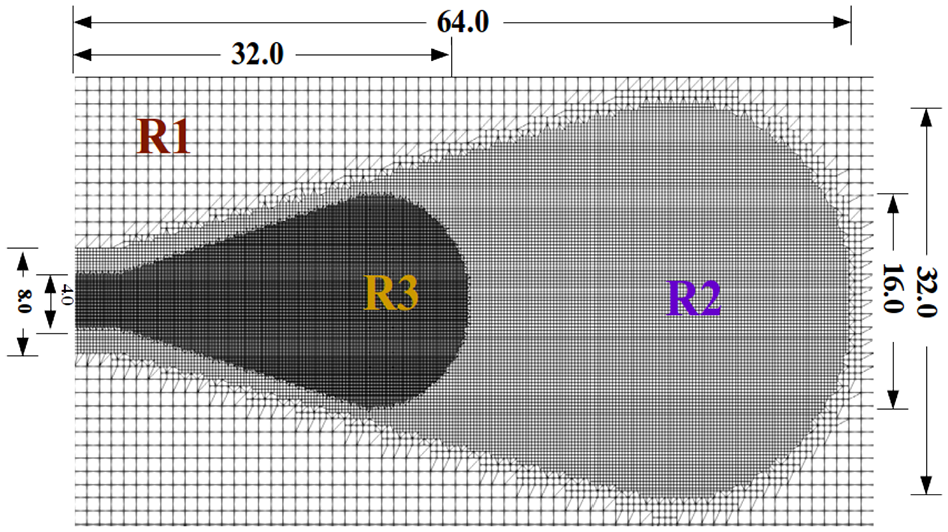
The computational domain and mesh structure:
For completeness, a mesh sensitivity analysis is provided in Appendix A for the DF1 case at 1000 K ambient temperature with two different grid resolutions: a very fine 62
Chemical kinetics mechanism
A reduced chemical kinetics mechanism developed by Frassoldati et al., 59 called Polimi-reduced hereafter, is used. The mechanism has been developed for n-dodecane combustion under engine relevant conditions. Polimi-reduced as well as another chemical mechanism, Yao et al. 60 mechanism, were earlier validated and used for similar DF ECN Spray A studies with methane at 900 K ambient temperature by Kahila et al. 9 We note that as the Yao mechanism does not include methanol, it is not considered in this study. According to Kahila et al. 6 and Tekgül et al., 10 Polimi-reduced presents a longer IDT compared to Yao mechanism for DF n-dodecane/methane. However, numerical tests carried out by the authors have shown that the relative ratio of the first- and second-stage ignition time remains approximately constant between the two mechanisms for DF n-dodecane/methane. In addition, Polimi-reduced mechanism has been recently validated for methanol combustion under engine relevant conditions by Karimkashi et al. 61 and it is used in numerical simulations for DF n-dodecane/methanol by Karimkashi et al. 62 This mechanism has been also compared against other mechanisms in the DF context by the authors 17 and a sensitivity analysis of the chemical mechanism is provided in our previous publication under dual-fuel conditions. 6 Finally, we note that all the modeling assumptions related to the present results are of numerical character. In particular, the considered chemical mechanism is only validated against SF experimental data for n-dodecane, methane, and methanol. Therefore, it should be noted that the conclusions of this work have direct relevance with the selected chemical mechanism and further validation is not currently possible due to the lack of experimental data for DF ignition.
Results and discussion
In this section, DF ignition properties of n-dodecane/methane (DF1) and n-dodecane/methanol (DF2) are presented and discussed at three different ambient temperatures: 900, 950, and 1000 K. First, insight into DF1 and DF2 ignition characteristics is given using 0d homogeneous reactor simulations. Second, high-fidelity LES of n-dodecane spray-assisted ignition of methane and methanol at different ambient temperatures are provided along with discussions on the first- and second-stage ignition. Different ignition types are identified for DF2 compared to the robust DF1 ignition at various ambient temperatures. Next, heat release map analysis is provided to give further details on the significance of LTC versus HTC in DF1 and DF2 ignition. Moreover, a reaction sensitivity analysis on the second-stage ignition time, reveals the key reactions causing a longer IDT in DF2 compared to DF1. Finally, a simple approach based on the non-reacting LES and 0d homogeneous reactor IDT data is proposed, which can estimate the second-stage ignition (
Insight into DF ignition properties using 0d simulations
Polimi-reduced is used in constant pressure 0d homogeneous reactors using Cantera
63
to acquire knowledge on the ignition properties of the three fuels used in the present LES. Hereafter, IDT refers to the second-stage ignition time defined as when the highest gradient of the maximum temperature within the domain is reached. Figure 2(a) depicts premixed ambient IDT (
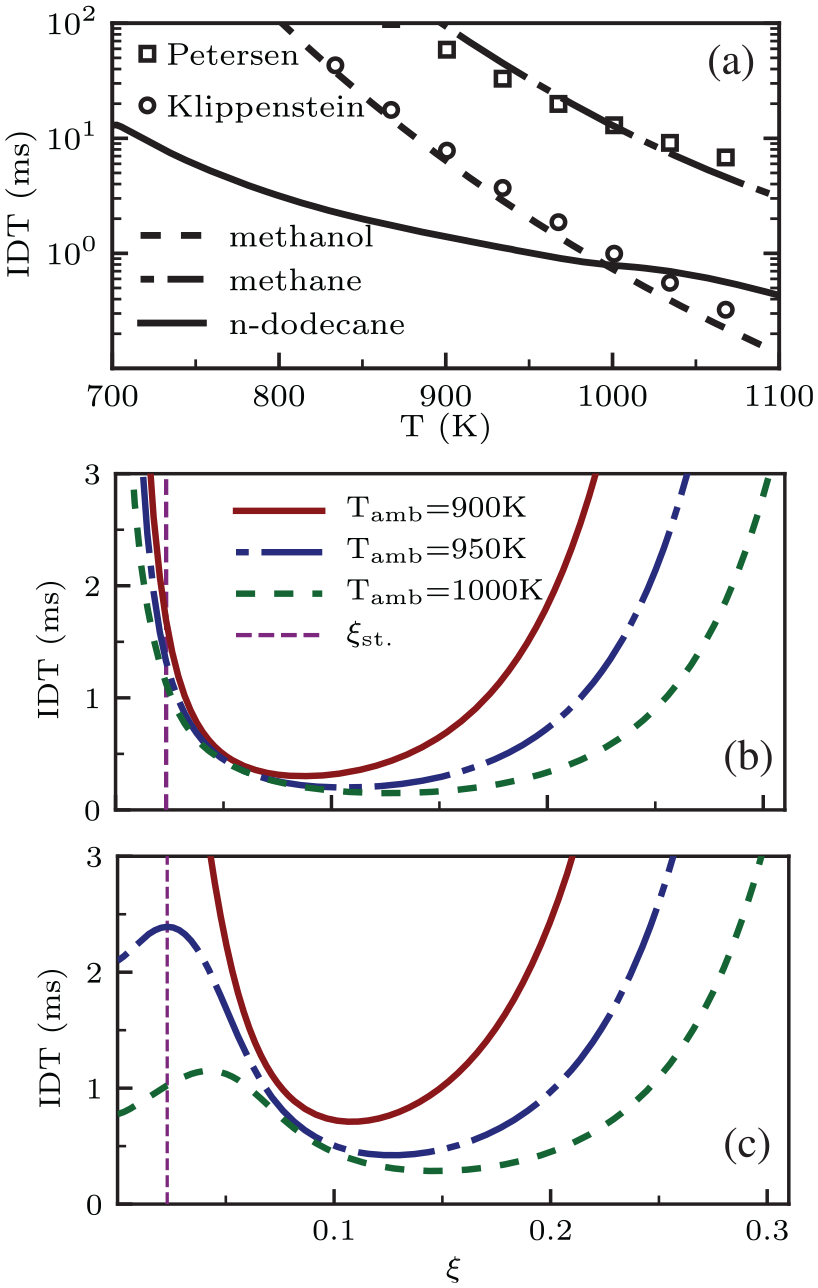
0d homogeneous reactor calculations using Polimi-reduced mechanism (lines): (a) IDT versus T for single-fuel methane, methanol, and n-dodecane (
In this work, the premixing ratio is defined based on the mixture fraction,
Results of IDT against mixture fraction according to the described mixing line concept are presented in Figure 2(b) and (c) for DF1 and DF2, respectively. It has been previously observed in the literature that adding methane to the ambient retards ignition compared to the ECN Spray A conditions.6,10,17 According to Figure 2(b) and (c), adding methanol to the ambient delays IDT even further compared to methane, consistent with the observations by Karimkashi et al.
62
For instance, IDT at the most reactive mixture fraction (the point with the shortest IDT within the considered range of mixture fraction) is observed to be longer in DF2 compared to DF1 at similar
The following observations in Figure 2 motivate the temperature sweep in the present study for DF1 and DF2. At
We note that as long as a validated chemical mechanism for DF n-dodecane/methanol ignition at engine-relevant conditions is not developed, the validity of the observed IDT trend and the inflection effect may pose uncertainties. However, the authors have performed similar 0d tests with DF n-heptane (another common diesel surrogate) and methanol. In those simulations we used a chemical mechanism provided by Lu et al.,
69
which is validated and used for DF n-heptane/methanol by Hu et al.
38
Similar IDT trends and the inflection effect at
DF1 and DF2 LES
Figure 3 depicts volume rendering of DF1 (n-dodecane/methane) and DF2 (n-dodecane/methanol) at
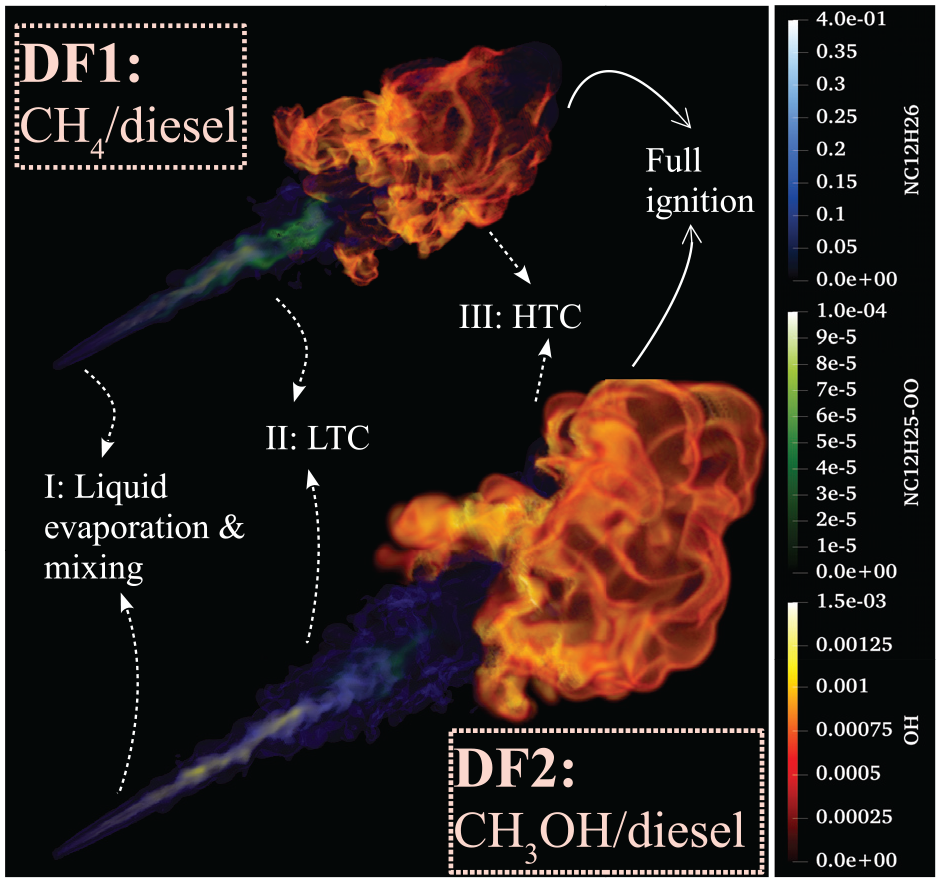
N-dodecane surrogate spray in ambient methane (DF1) and methanol (DF2) with the marked regions of evaporation (I), LTC (II), and HTC (III). Here, n-dodecane, RO2, and OH radicals are rendered for liquid evaporation and mixing, LTC, and HTC regions, respectively.
Within the LTC regime, low-temperature reactions lead to the early decomposition of n-dodecane and intermediate radicals such as RO2 and H2O2 are formed.
70
According to Westbrook,
71
the dodecyl peroxy radical (RO2) is among the first species formed after n-dodecane decomposition and therefore, it is considered as an appropriate marker for LTC activity and the first-stage ignition. Here, the first-stage ignition time (
Temporal evolution of the maximum RO2 mass fraction (
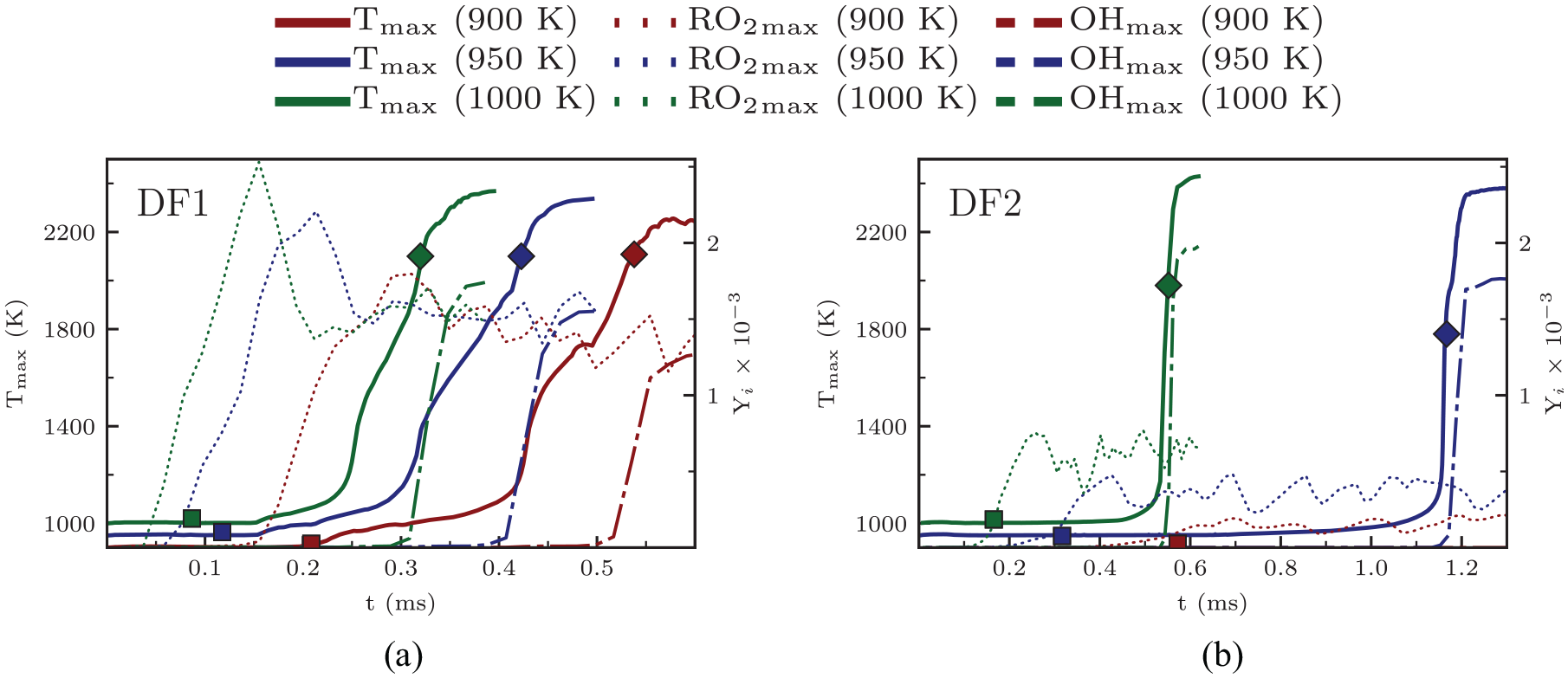
Temporal evolution of
Three observations are made according to Figure 4. First, higher ambient temperature advances the first-stage ignition and consequently, the second-stage ignition in both DF1 and DF2. A similar trend for DF1 was previously reported by Tekgül et al.
10
Second,
The observed trend of advanced
Ignition time calculations from 0d homogeneous ignition at
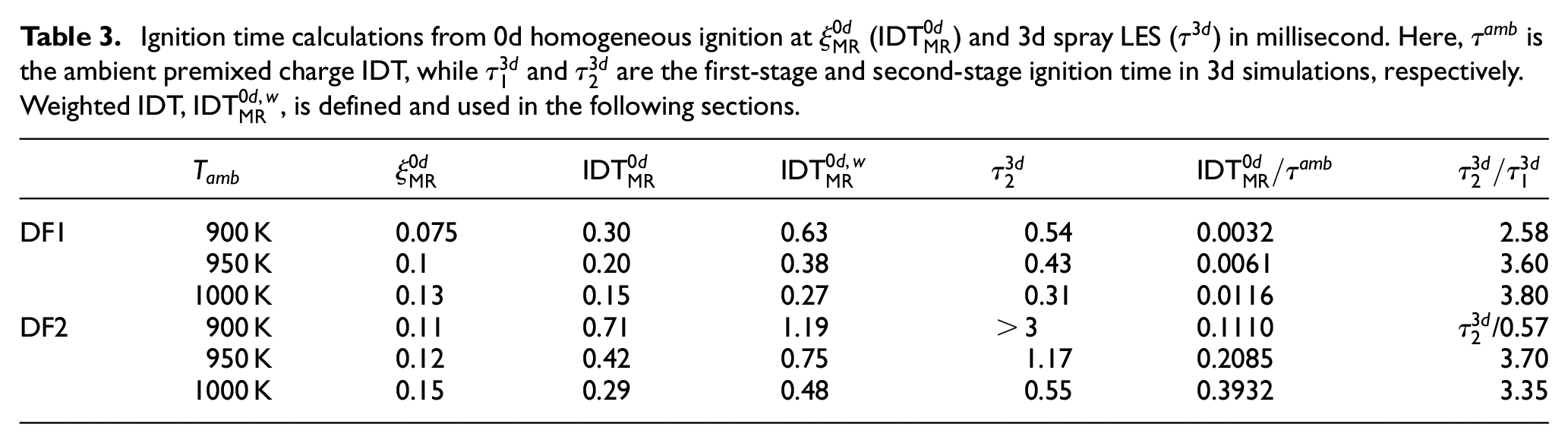
Figure 5 depicts 2d cut-planes for spatial distribution of temperature at the time 1.2
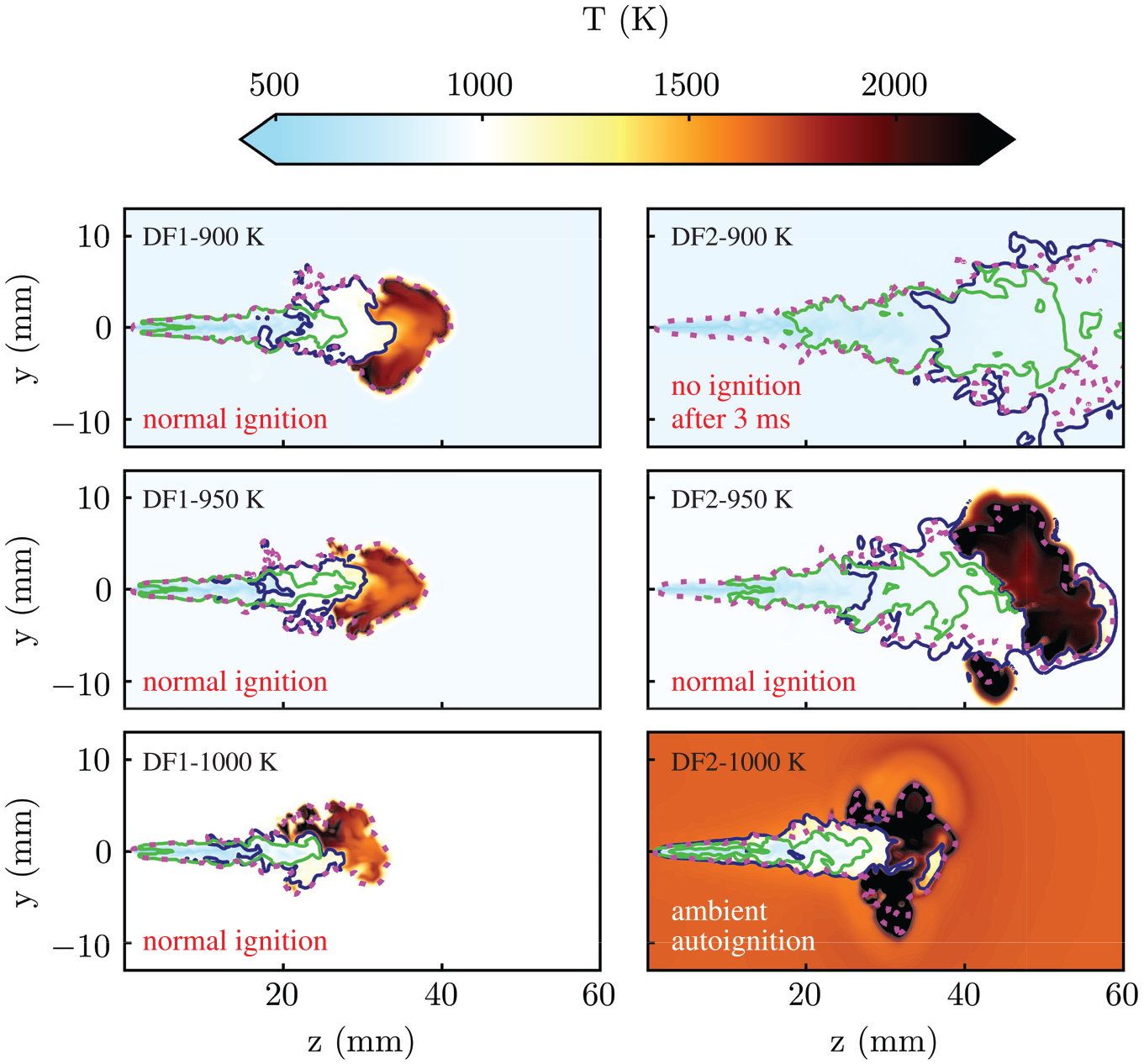
Spatial distribution of temperature at 1.2
Contour-lines of
Figure 6 demonstrates scatter plots of temperature against mixture fraction for LES cases at their respective 1.2
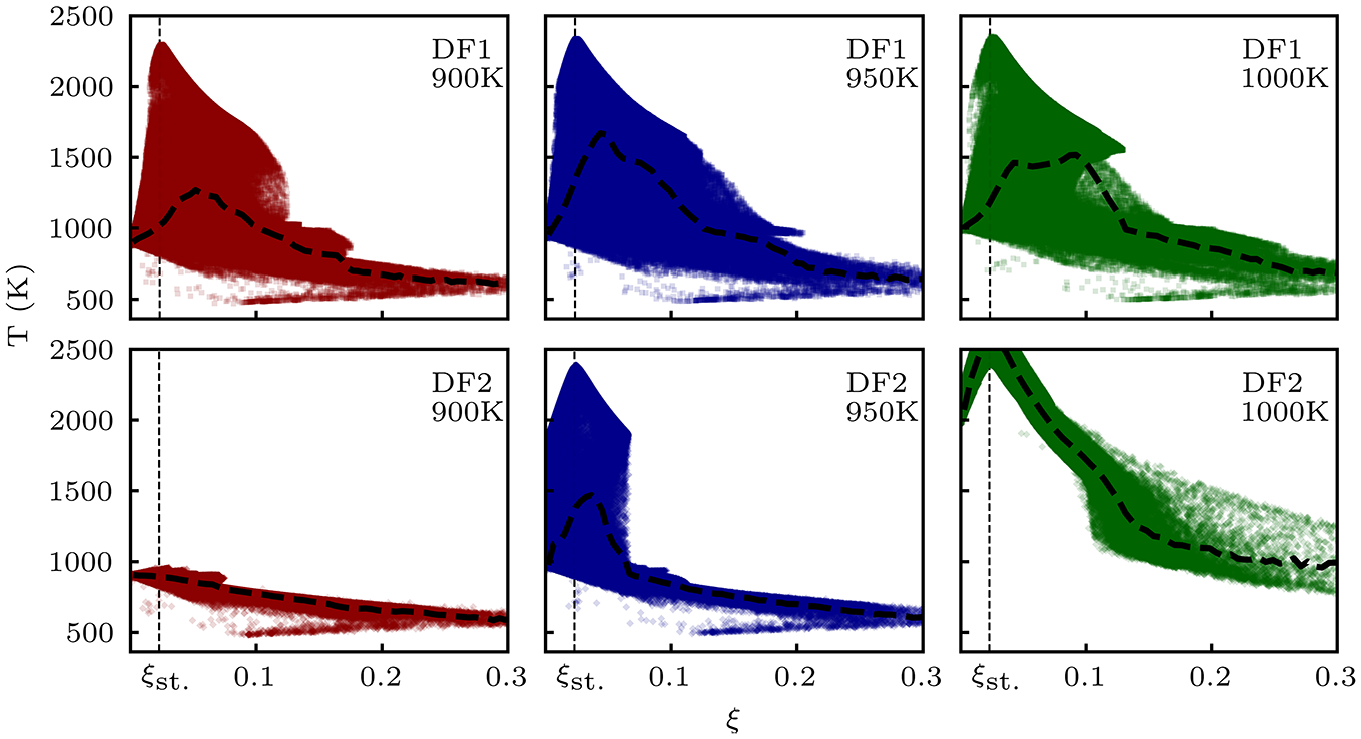
Scatter plots of temperature versus mixture fraction from LES simulations at their respective 1.2
Heat release map analysis
Our observations in the former section indicated significant differences in the ignition process of n-dodecane/methane (DF1) and n-dodecane/methanol (DF2) sprays at various
Criteria for HR maps a.
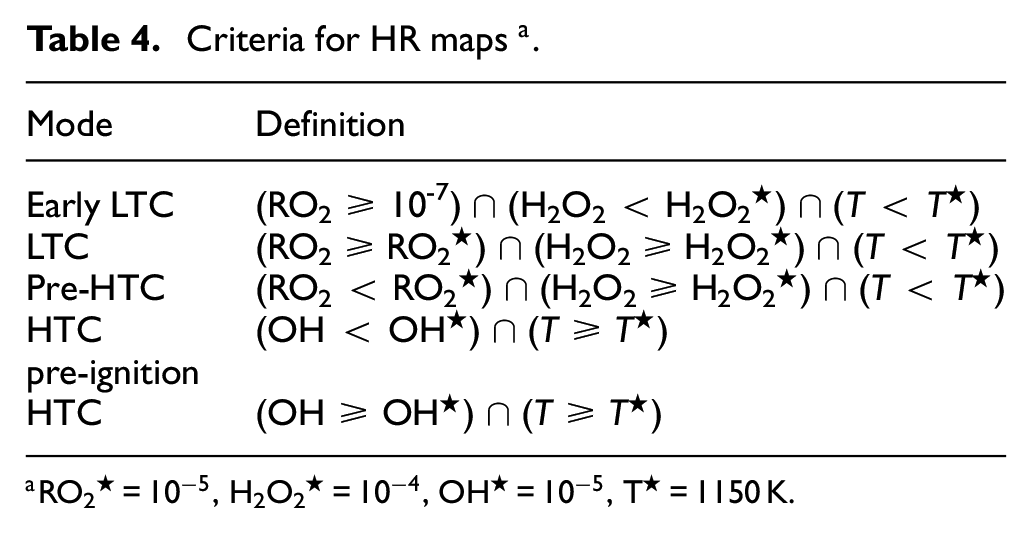
RO2★ =
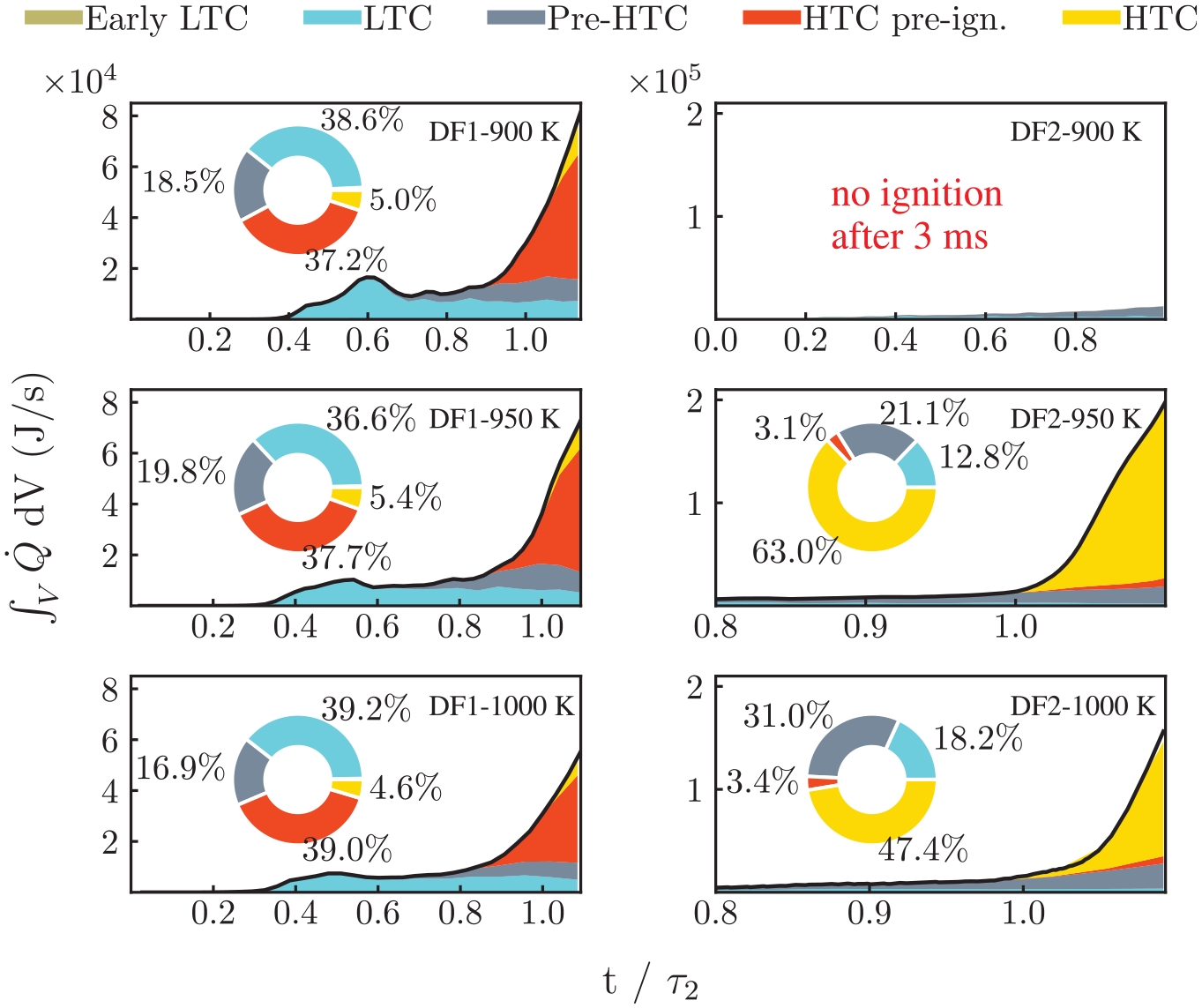
Modal decomposition of the HR maps within the spray region (
In DF1, changing
Reaction sensitivity analysis
For completeness, we carry out a reaction sensitivity analysis in order to understand why the second-stage ignition time is longer in DF2 compared to DF1 at similar
According to Yao et al.,
60
we define a sensitivity indicator,

wherein

We note that according to our tests, using different
The relevant reactions of Polimi-reduced mechanism which have larger
List of top priority reactions from Polimi-reduced mechanism for SF, DF1, and DF2 ignition at the considered conditions of this study.
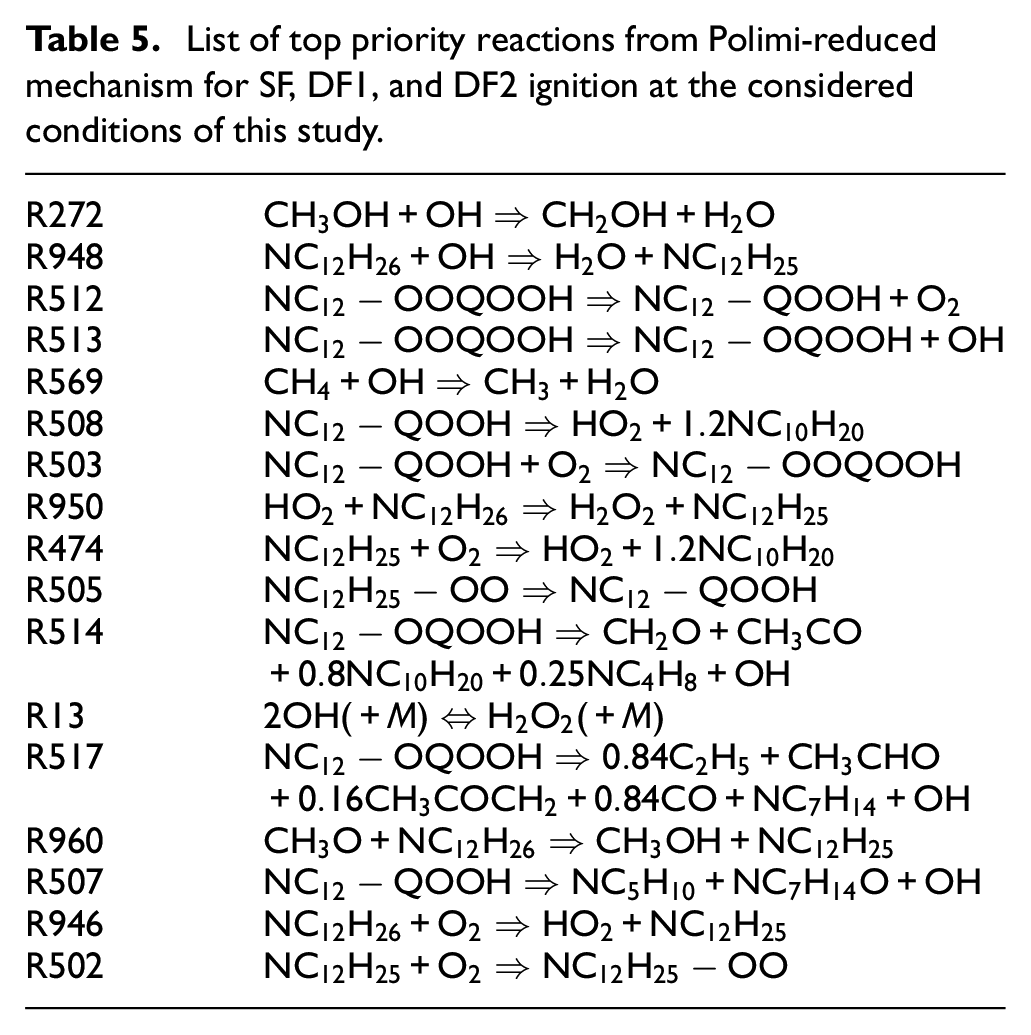
In the considered DF cases, all of the above-mentioned reactions are still among the top sensitive ones according to their
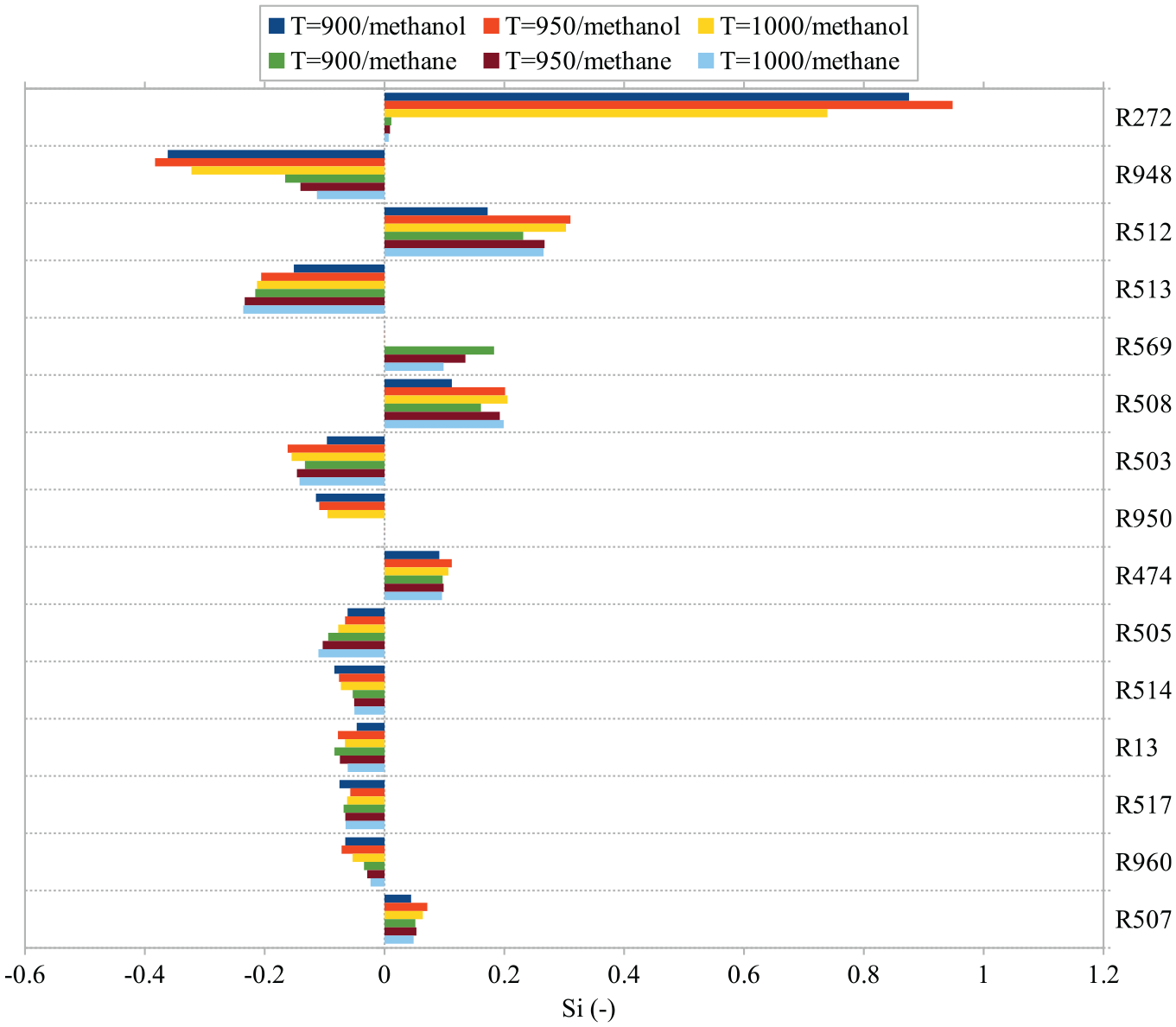
Reaction sensitivity analysis for DF2 and DF1 ignition at
According to Figure 8, variation of temperature at the corresponding
The acquired knowledge from reaction sensitivity analysis along with the 3d spray results provide insight into the reason of the observed narrow ignition window in DF2. From the reaction sensitivity analysis we know that R272 in DF2 dramatically prolongs ignition of n-dodecane spray compared to that in DF1. This prolongation leads to further dilution before ignition in DF2 cases compared to DF1 cases. The over-dilution itself leads to further delay of the spray ignition, which typically starts from richer pockets on the tip of the spray. Accordingly, in n-dodecane/methanol cases, different ignition behaviors are observed depending on the ambient temperature: at lower
Mixing and residence time analysis
Considering the high computational cost of LES with finite rate chemistry, for example in DF2 with longer IDT leading to longer spray penetration, it would be beneficial to predict the DF spray ignition delay time based on 0d homogeneous reactor calculations and computationally less expensive non-reacting LES. Here, we outline a simple approach for such a purpose as follows. It is discussed by Kahila et al. 6 and shown in Table 3 of the current study that the calculated IDT from 0d homogeneous reactor calculations is typically two to three times shorter than the ignition time in LES spray simulations. This difference mainly corresponds to the mixing time in spray simulations. The idea here is to estimate the mixing time using non-reacting 3d spray simulations and contribute it to 0d IDT to provide a better estimation of the spray ignition timing.
For estimating mixing time, a residence time,
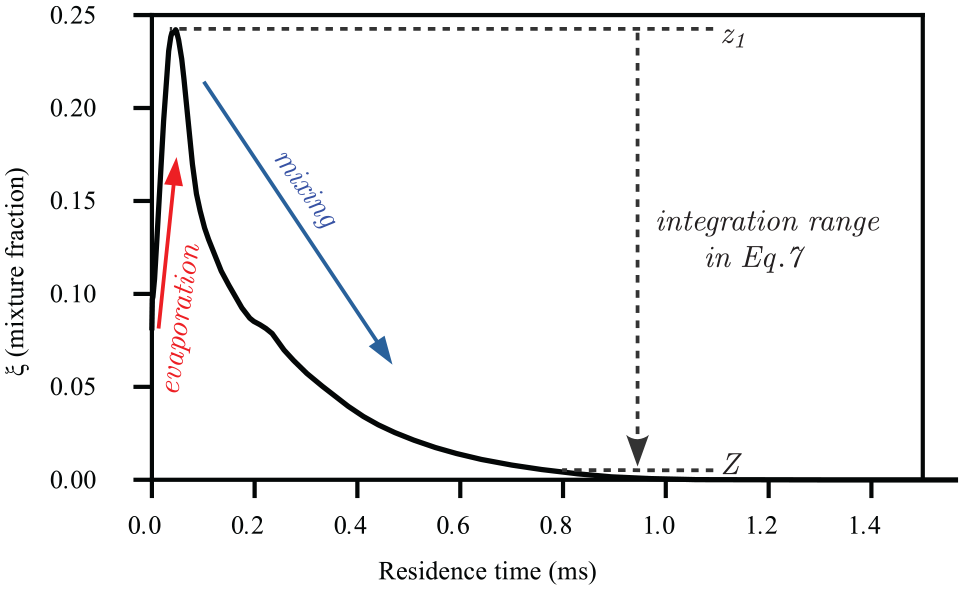
Calculated residence time along the mixture fraction using non-reacting LES for DF1-900 K case. Along the spray axial direction (
We estimate ignition time in 3d spray simulations by weighting the 0d IDT data, c.f. Figure 2, with
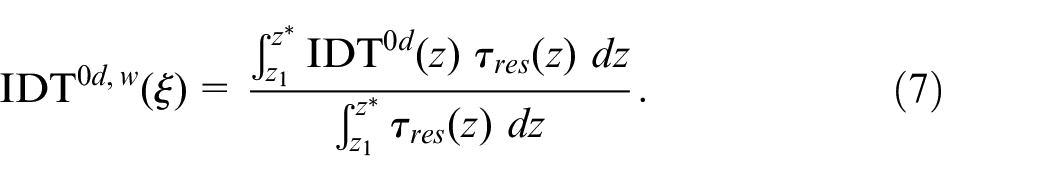
Here, we neglect the evaporation phase and we only integrate along the descending branch of the profile in Figure 9, in which
Figure 10(a) displays weighted and unweighted IDT profiles against mixture fraction for DF1-900 K. It is observed that the most reactive IDT is further delayed and shifted toward leaner mixture fractions. Figure 10(b) and (c) compare the most reactive weighted IDTs (
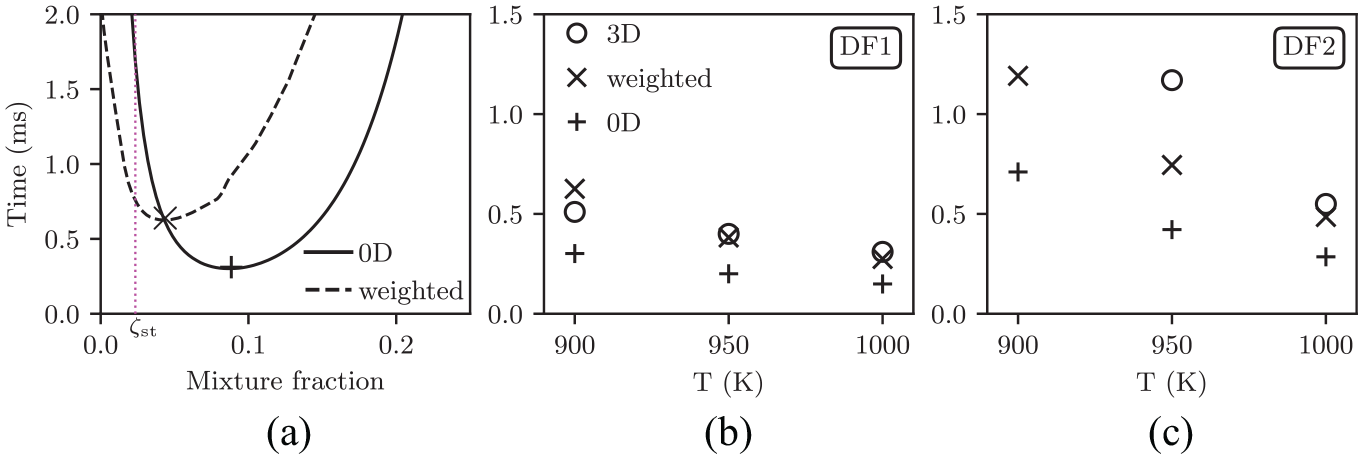
(a) Unweighted and weighted IDT from 0d data for DF1-900 K case. Comparison of the most reactive 0d IDT (
In summary, we note that the proposed weighted IDT concept provides a simple correction to the DF1 cases with reasonably good IDT prediction. However, the concept was shown to be less accurate, although improved, for the more sensitive DF2 case.
Conclusions
Ignition characteristics of diesel surrogate (n-dodecane) injection into hot and lean (
Replacing methane with methanol in the ambient lean mixture of DF sprays leads to inhibited ignition almost by a factor of two. Moreover, higher
With methanol, different ignition types are observed for different
With methanol, the
Weighting 0d IDT data using residence time from non-reacting LES is shown to offer a correction factor for estimation of spray IDT with substantially lower computational expenses compared to 3d reacting spray LES. Although the concept is more reliable for methane DF cases, it provides improvements even for more sensitive methanol DF cases. The method is proposed as a diagnostic tool to assess spray ignition delay time.
Heat release maps indicate the small contribution of LTC in ignition for methanol cases compared to methane cases. LTC becomes weaker with methanol and an extended LTC region is observed, mainly due to the longer IDT.
According to the reaction sensitivity analysis, R272 (
Considering the reported limitations in robust ignition of methanol in DF conditions in this work, our recent research results suggest that adding H2 to methanol/air ambient may favorably impact ignition. 62 In our recently published studies, tri-reactivity ignition of methane/hydrogen and methanol/hydrogen by a diesel spray at a fixed ambient temperature (900 K for methane/hydrogen and 950 K for methanol/hydrogen) are investigated.40,41 As a closing remark, we note that the conclusions of this study are dependent on the selected chemical mechanism for DF mixtures of n-dodecane/methane and n-dodecane/methanol. Due to the lack of experimental data for validation of the utilized chemical mechanism under the considered conditions, the presented conclusions need to be further assessed using experimental engine tests, in specific for n-dodecane/methanol DF combustion.
Footnotes
Appendix A: Mesh details and sensitivity analysis
The computational geometry, mesh structure, and mesh sensitivity analysis of the studied LES are presented here. The volume of the computational domain is the same as the experimental combustion vessel for the ECN Spray A. The computational domain and the sizes (in mm) are displayed in Figure A1. The standard mesh (the upper half) – which is used for the studied LES – is compared to a finer mesh (the bottom half) – which is used for the sensitivity test. The only difference between the two meshes is the higher resolution in the spray envelope region of the finer mesh. Four regions are marked, which specify resolutions: R1: 1000, R2: 250, R3: 125, and R4: 62.5
The finer mesh is used in an LES test for validation of our results in one of the studied cases, DF1-1000 K. The result of this sensitivity test is presented in Figure A2, which shows temporal evolution of the maximum temperature (
Acknowledgements
We acknowledge CSC (Finnish IT Center for Science) and Aalto University for providing the computational resources.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is financially supported by the Academy of Finland (grant numbers 332784 and 318024).
