Abstract
Introduction:
Patient and public involvement and engagement (PPIE) in research is an essential component of high-quality research. Patients and the public can identify which research topics are most relevant to them, contribute to study design, and interpretation and dissemination of findings. While inclusion of PPIE is widely adopted in medical research, awareness within the dental research community is more limited.
Aim:
To examine patient and public involvement and engagement in orthodontic research activity.
Design:
Identification and appraisal of use of PPIE in orthodontic research reporting and funding applications using a systematic approach.
Methods:
Three sources of information were examined: (1) research articles published between September 2018 and September 2019 in four major orthodontic journals. Articles were examined for reported PPIE; (2) common funding bodies for orthodontic research were assessed to establish whether PPIE was mandated (National Institute for Health Research, Medical Research Council, Wellcome Trust, Chief Scientist Office (Scotland), Health and Care Research Wales, British Orthodontic Society Foundation, Royal College of Surgeons and CLEFT); and (3) publication guidance for authors in these journals was examined to identify whether reporting of PPIE was included.
Results:
Of the 363 research articles, 2 (0.6%) mention patient/public involvement. None of the 363 research articles mention patient/public engagement. Of nine funding bodies, 2 (22%) request evidence of patient/public involvement as a condition of receiving funding with one (11%) expecting evidence of public engagement to be provided as a condition of receiving funding. None of the four major orthodontic journals include patient/public involvement and/or engagement in their guidance for authors.
Conclusion:
There is currently: (1) a notable lack of reporting of PPIE in orthodontic research; (2) variability in the requirements of funding bodies for researchers to include PPIE in funding applications and throughout the research process; and (3) no stipulation in journals’ instructions for authors.
Introduction
Patient and public involvement (PPI) in research refers to ‘research being carried out “with” or “by” the public rather than “to” or “about” or “for” them’ (National Institute for Health Research [NIHR] Central Commissioning Facility [CCF], 2019). Patient and public engagement (PaPE) describes activities that encourage the findings and benefits of research to be shared with the patients/the public (National Co-ordinating Centre for Public Engagement [NCCPE], 2019). Patient and public involvement and engagement (PPIE) in research is an essential component of high-quality research; patients and the public can contribute in a number of ways (Table 1) to help identify which research topics are most relevant, to improve study design and delivery, and to identify effective methods for disseminating research to the public (Staley, 2009, 2015). PPIE aims to create a mutually beneficial relationship between researchers, patients and the public.
Examples of patient and public involvement and engagement activities in research (NCCPE, 2019; NIHR CCF, 2019; Staley, 2015, 2019).

PPIE in the UK is increasingly important to justify funding and maximise the value and impact of research. The two-way conversation that public engagement facilitates is one which builds trust between researchers and the public and helps to build understanding and appreciation of the research which is conducted. It also helps researchers to shape their projects to meet the needs and expectations of the public (NCCPE, 2019; NIHR CCF, 2019). The evidence base for PPIE has grown in recent years and its inclusion is becoming increasingly prevalent in medical research (Boivin et al., 2018; Regan de Bere et al., 2016). However, awareness and uptake in the dental research community appears slower (Needleman, 2014). This study idea arose from discussions about PPIE with Mr Neil Hillyard, an orthodontic patient who was a founding member of British Orthodontic Society patient panel and is the author of the ukadultbraces.co.uk blog.
Aim
The aim of the present study was to examine patient and public involvement and engagement in orthodontic research.
Design
Orthodontic literature and funders of orthodontic research were appraised for use of PPIE using a systematic approach. A patient contributor (NH) advised on the purpose and analysis of this study.
Methods
Three sources of information were examined:
Research articles published between September 2018 and September 2019 in four orthodontic journals (Journal of Orthodontics, American Journal of Orthodontics and Dentofacial Orthopaedics, The Angle Orthodontist and European Journal of Orthodontics). These were identified by assessing each article in each journal electronically to see whether it fulfilled the eligibility criteria. The eligibility criteria included any original research articles and systematic reviews that had a structure of background, methods, results, conclusion and discussion or similar. There was exclusion of case reports, case series, expert opinion papers, audits, service evaluation, letters to the editor and other obvious non-research articles. Each article was independently judged against the criteria by two authors (JS and VAP) and any disagreements were resolved by a third author (SB). All research articles fulfilling the eligibility criteria were included for analysis. Corresponding authors of the papers included in the analysis were emailed to investigate whether they used PPIE but did not report it in the final publication. A single email was sent and authors asked to respond within two weeks.
The websites of common funding bodies for orthodontic research were assessed to establish whether PPIE was mandated—NIHR, Medical Research Council (MRC), Wellcome Trust, Chief Scientist Office (Scotland) (CSO), Health and Care Research Wales (HCRW), British Orthodontic Society Foundation (BOSF), Royal College of Surgeons (RCS) and CLEFT.
Publication guidance for authors in these major orthodontic journals was examined to identify whether reporting of PPIE was included.
Judgement of sources:
Sources were judged against pre-defined criteria (Table 2) by two independent reviewers (VAP and JS). Any disagreements were discussed with a third reviewer (SB). Where the criteria were scored as ‘Yes’, details were recorded to allow further analysis.
Where PPI was reported in a paper, the quality of PPI reporting was evaluated using the Guidance for Reporting Involvement of Patients and Public (GRIPP2) checklist (Staniszewska et al., 2017).
Criteria used to judge requirements for, and reporting of, patient and public involvement and engagement (PPIE; this was devised by the authors for the purpose of the study).
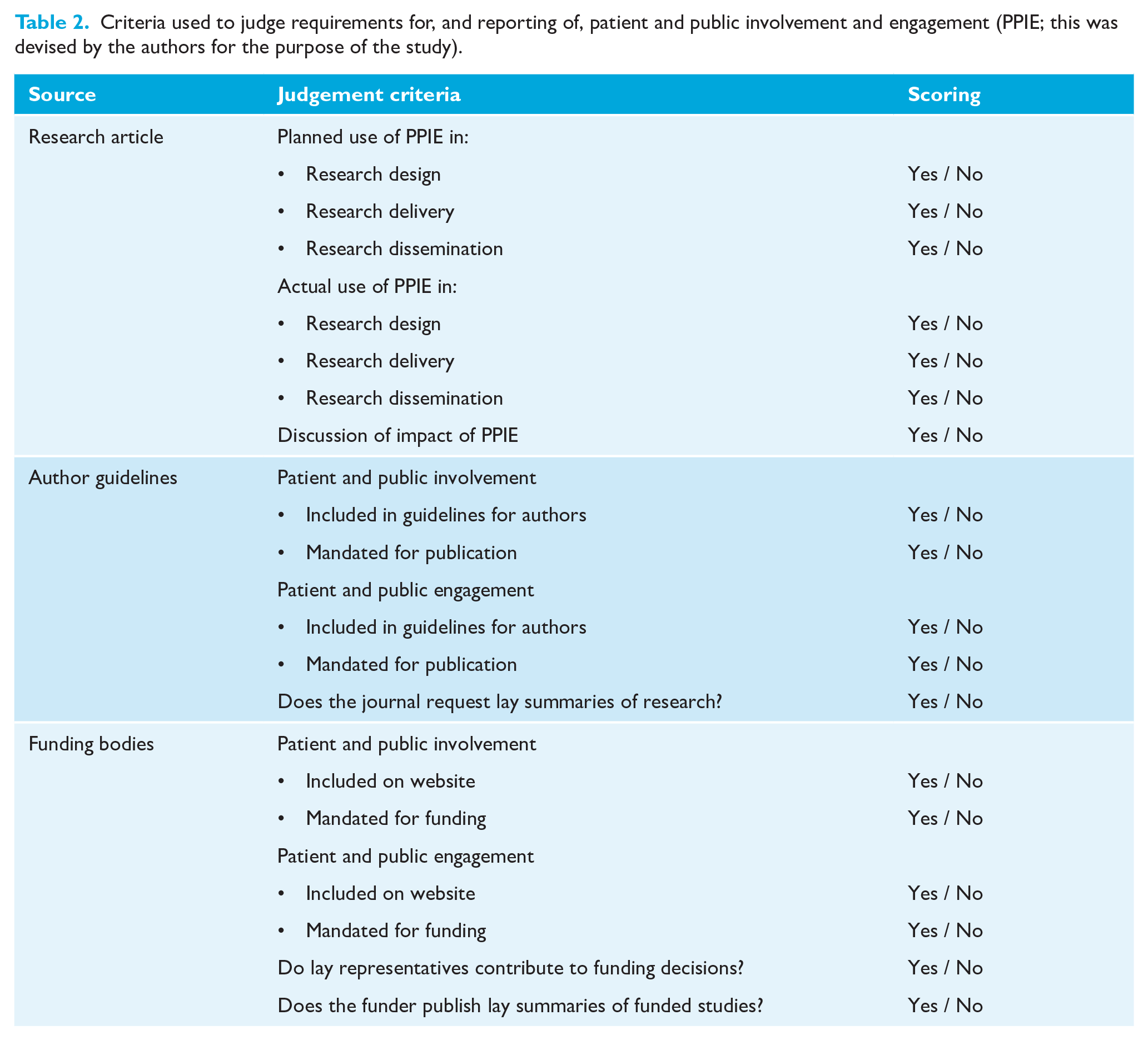
Results
Reporting of PPIE in published research
From the four journals examined, 707 articles were identified, of which 363 were eligible for inclusion (Figure 1). Of the 363 research articles, only 2 (0.6%) report PPI and both were in the design of patient-facing materials (Ahn et al., 2019; Chambers and Zitterkopf, 2019). None of the 363 research articles reported PPI in the other aspects of research design and delivery. Neither of the papers that did utilise PPI, reported their use of PPI against the GRIPP2 checklist. Since PPI was not the main focus of either of the two studies, we have used the GRIPP2 short form to analyse the two papers against the criteria (Table 3).
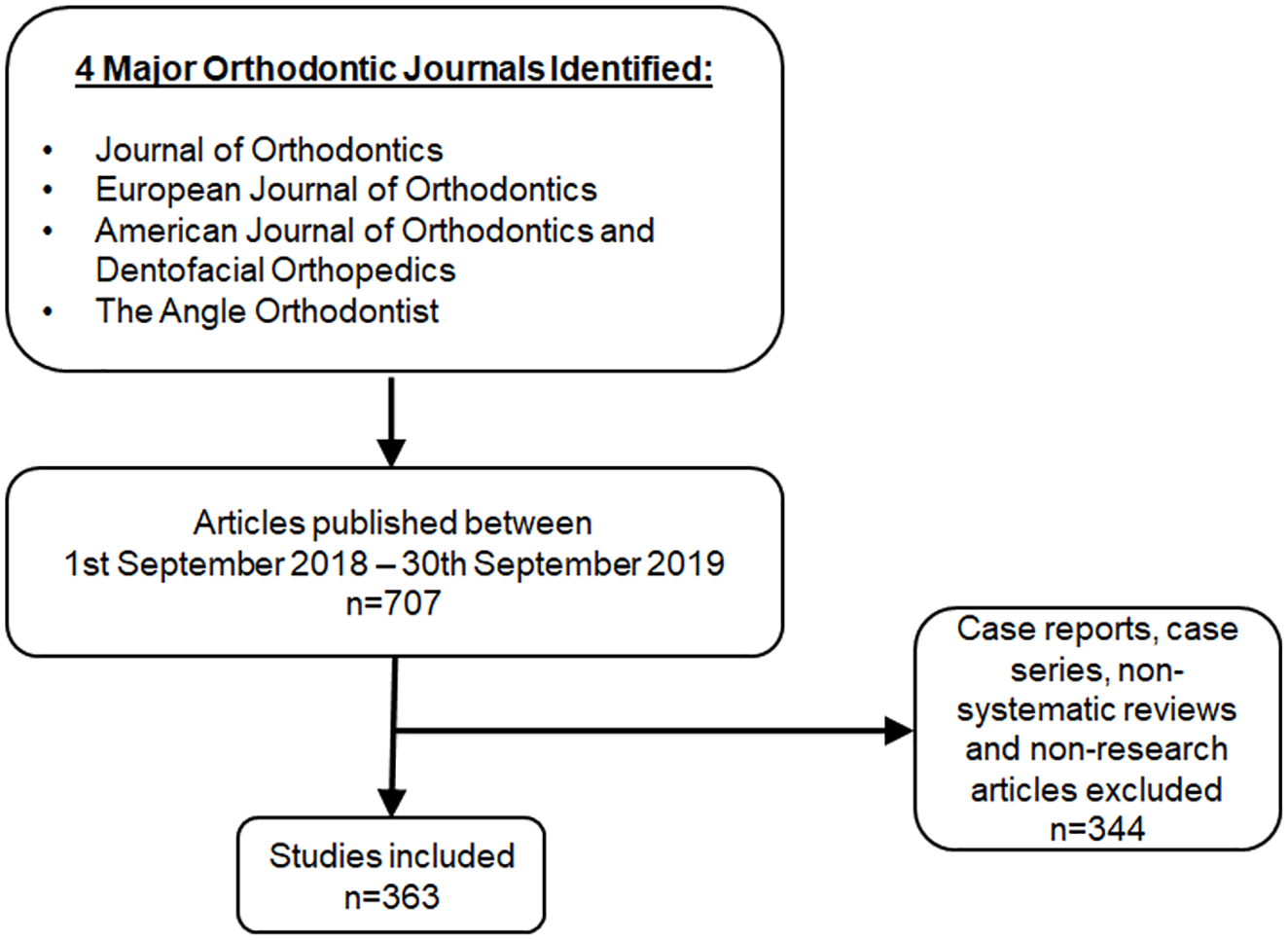
Summary of the methods for identifying studies to include in the analysis.
GRIPP2 short form applied to both papers which mention patient and public involvement (PPI).

Authors for 52 (14%) of the 363 studies replied to our email request regarding the use of PPIE and, of these, only 2 (4%) reported use of PPI. One of these authors has previously been identified, as the use of PPI was reported in their paper (Chambers and Zitterkopf, 2019). The second reply was from a co-author of the present study (SB), who had used PPI in another study published in the Journal of Orthodontics but had not reported it (Barber et al., 2019). The author provided a summary of their use and experience of PPI (Box 1).
Co-author SB’s experience of using patient and public involvement (PPI) in research.

None of the 363 research articles reported PaPE in disseminating the research findings to the public. Corresponding authors of 5 (9.6%) of the 52 studies for which we received replies confirmed their use of PaPE. All five authors exclusively reported posting their research on social media platforms but of these, only 2 (40%) stated that they provided a plain English summary to accompany the post.
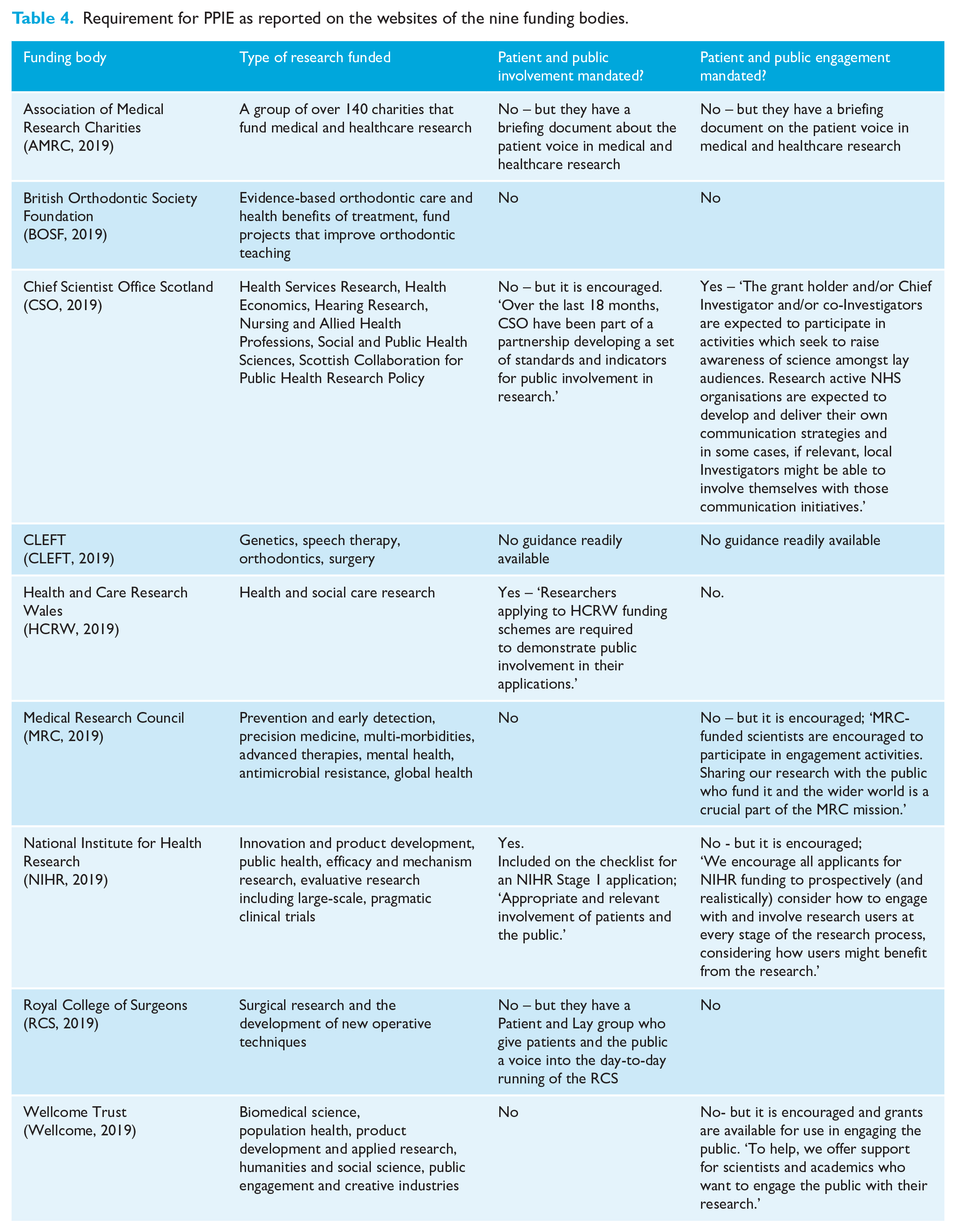
PPIE requirements of funders
PPIE requirements of the funders is summarised in Table 4. Two (22%) of the nine funders (NIHR, 2019; HCRW, 2019) request evidence of PPI as a condition of receiving funding, while one funder (11%) (CSO, 2019) expects evidence of PaPE as a condition of receiving funding. Three funders (33%) (CSO, 2019; MRC, 2019; NIHR, 2019) clearly state a requirement for lay or plain English summaries to be provided in the publication of research.
Requirement for PPIE as reported on the websites of the nine funding bodies.
PPIE requirements of journals
None of the four orthodontic journals or their publishers include PPIE in their guidance for authors (American Journal of Orthodontics and Dentofacial Orthopedics, n.d.; The Angle Orthodontist, n.d.; Elsevier, n.d.; European Journal of Orthodontics, n.d.; Journal of Orthodontics, n.d.; Oxford University Press, n.d.; SAGE Journals, n.d.).
Discussion
The results of this study suggest a lack of reporting of PPIE in orthodontic research, an absence of prompting in guidance from journals and publishers, and inconsistent requirements from funding bodies regarding the need for, and reporting of, PPIE. Our results substantiate findings from a systematic review of the quality of reporting PPI in surgical specialties, which found that only a limited number of studies reported use of PPI (Jones et al., 2015). We were not able to find any studies examining the reported use of public engagement in health research.
There are many potential detrimental consequences of not employing PPIE in research. Chalmers and Glasziou (2009) estimated that 85% of all clinical research is wasted, even when huge investments and public funding have been utilised. The authors also deduced that there are four research practices which could result in such waste: (1) prioritising research questions which are not relevant to the public (as well as clinicians); (2) conducting unnecessary or inappropriate studies or study designs; (3) failing to publish research findings; and (4) selective reporting of research findings. NH provided a patient’s perspective on these findings (Box 2), which highlights both the need and drivers for PPIE, but also potential challenges.
Patient perspective (NH) of the review findings.

Alongside impacting on research quality, a lack of PPIE reporting means that others cannot learn from the other researcher’s experience and it is difficult to evaluate the effectiveness of different approaches to PPIE. Improved reporting of PPIE will lead to a more expansive and high-quality evidence base for PPIE, allowing us to better understand its uses and effectiveness (Staniszewska et al., 2017). The EQUATOR network (EQUATOR, 2019), which is an international initiative to promote transparent and accurate reporting of health research, includes the GRIPP2. However, no guidelines on the reporting of PaPE could be identified and the International Committee of Medical Journal Editors (ICMJE, 2019) recommendations do not provide guidance on the reporting of PPI or PaPE. GRIPP2 reporting guidelines were published in 2017 and these are the first evidence-based, international, consensus community-informed guideline of its kind in health and social care research. The guidance includes two checklists: one is a short version that can be used to report PPI in any study; and the other is a long form which should be used where PPI is the focus of the study and can be used to structure the entire paper. The forms can be used prospectively to guide the use of PPI and retrospectively to help structure the reporting of PPI (Staniszewska et al., 2017). Perhaps a requirement to report use of any PPIE could also be included in existing checklists for reporting research papers such as CONSORT, PRISMA and ROBINS-I.
It may be the case that many researchers are using PPI or PaPE in their research but are not reporting it. Indeed, one of the research papers reviewed in this study was undertaken by one of the authors of the present article and although the examined study (Barber et al., 2019) was funded by the NIHR and used extensive PPI, this is not reported in the paper. Of the 363 studies analysed, 161 were funded and this included funding from the NIHR, suggesting there will be other studies that used PPI but did not report it explicitly. However, in this study, although only 14% of authors replied to an email regarding use of PPIE, the low rates of PPIE utilisation identified within this sample correspond with the lack of reporting. The British Medical Journal described PPI reporting before and after a journal policy was introduced. The policy required authors to write if and how PPI was used, and this concluded that sporadic reporting of PPI was not only due to a lack of reporting but also due a lack of actual PPI activity and/or unwillingness to report unsuccessful PPI (Price et al., 2018).
Recommendations for increasing the use and reporting of PPIE are given in Box 3. A number of concerns may be raised in response to these recommendations: certain types of studies, such as laboratory studies, are too conceptual for laypeople to understand; if publishers and funding bodies make PPI or PaPE a compulsory requirement, this may limit the research that can be undertaken by those who may not have funding and support available to them; PPI may become tokenistic and lose any value (Domecq et al., 2014). The counter to this is that research with an immediate or eventual clinical impact should be able to be translated into a format that is accessible to a lay audience, and if patients and the public do not believe that a project is worth pursuing, then perhaps the proposed benefits are not adequately explained, or more importantly, are not valued. Capturing patient or public perspective may mean less money is wasted and resources are channelled into research that is deemed to be more useful (Boote et al., 2014).
Recommendations for improving the use and reporting of patient and public involvement and engagement (PPIE) in orthodontic research.

INVOLVE is a national advisory board funded by the NIHR, which aims to support the use of public involvement in the NHS and health and social care research. INVOLVE provide freely accessible resources to support researchers and this is recommended as a starting point for researchers interested in PPIE. Furthermore, journals dedicated to PPIE, such as BioMed Central’s Research Involvement and Engagement and the National Coordinating Centre for Public Engagement’s Research for All may be a useful source for inspiration and shared learning for dental researchers. ‘Critical appraisal guidelines for assessing the quality and impact of user involvement in research’ (Wright et al., 2010) is also a useful resource to refer to when planning to use PPI.
However, PPI can be as simple as asking patients to contribute as authors to papers by providing a layperson’s perspective as demonstrated in this paper, and PaPE could be a social media post of a research paper accompanied by a plain language summary. Currently, to our knowledge, no orthodontic journals published in English provide plain language summaries of research articles for sharing via their website or other platforms, such as social media. A plain language summary example has been provided for this paper (Box 4). During the preparation of this paper the Editor of the Journal of Orthodontics was contacted to discuss whether plain language summaries may be considered in the future. The Editor expressed interest in this, and it is now being explored further.
Plain language summary for this paper.

Requirements for using and reporting PPIE by research bodies was found to be variable and while a number of funders request evidence of PPI in applications for funding, it is unclear how PPIE activity throughout the research is monitored. An example of guidance for PPI in research funding applications from HCRW is provided below.
‘Applicants should state in 2000 words:
How they have involved and engaged public partners in the development of the application and the benefits this is expected to yield.
Provide descriptions of the experience or area of activity of the public.
Outline the activities in which they have been involved.
The reasons for taking the approach.
Explain how this involvement has/has not influenced or changed the research application.
If the public were not involved in identifying the research topic and preparing the application, or if there are no plans to for active involvement, then there must be an explanation given for why this is not thought necessary.’ (Research for Patient and Public Benefit Wales [RfPPB], 2019).
This is useful for researchers for PPI during planning research but does not necessarily promote ongoing PPI throughout the research delivery and dissemination.
One challenge, highlighted by NH, is how to effectively recruit, train, support and retain PPI contributors. A review by Domecq et al. (2014) found most studies reported the positive effects of PPI but some patients involved in research became frustrated at the time required for training, attendance and transportation. However, with the advent of online video-conferencing platforms such as Zoom and Microsoft Teams, the cost of training individuals who reside in different parts of the country and travel may become less of an issue. It may be argued that patients and the public who contribute to PPI may not be representative of the population the research is being designed to benefit. They may have particular motivations for being involved with a research team and thus have an influence on steering the research in a particular direction. Researchers should take this into account when employing the use PPI. Similarly, researchers should think of ways to engage patients from a variety of different backgrounds rather than simply those who may be easiest to target when planning PaPE.
It is acknowledged that this study was limited to four orthodontic-specific journals, so orthodontic research published elsewhere was not considered and may provide different results. The lack of replies from authors regarding whether they actually employed the use of PPIE and simply did not report it may have been due to a number of factors. These include outdated contact details, language barriers, a lack of interest in PPIE and lack of use of PPIE, meaning they are less motivated to respond or the fact that this is a busy and difficult time period with regards to the COVID-19 pandemic.
Conclusions
This study found that there is currently:
A lack of reporting of PPIE in orthodontic research.
Variability in the requirements of funding bodies for researchers to include PPIE in funding applications and throughout the research process.
No stipulation in journals’ instructions for authors for reporting PPIE or provision of a plain language summary.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SKB is an Associate Editor for the Journal of Orthodontics.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
