Abstract
To reduce the hydrophilicity of polyvinyl alcohol (PVA), various methods are employed. Acid cross-linking is one technique, but it decreases the polymer's strength. To address this, different reinforcements are utilized to enhance the polymer's strength while mitigating the side effects of acid cross-linking. The present research intends to improve the physical, mechanical, and thermal properties of PVA by cross-linking it with fumaric acid and reinforcing it with modified graphite particles. The particles were prepared through oxidative acidic treatment and added in different weight proportions (0.5, 1, 1.5, and 2 wt.%) to the PVA matrix. Water absorption (WA) tests were conducted to affirm the formation of cross-linked bonds, and Fourier transform-infrared spectroscopy was employed to confirm the oxidation of the graphite particles with acid. The composites were examined using scanning electron microscopy, which revealed a robust interfacial adhesion between the modified graphite and cross-linked PVA, resulting in better mechanical characteristics. The highest ultimate tensile strength was observed when using 1.5 wt.% of modified graphite particle reinforcement, resulting in a 31% increase in comparison to pure cross-linked PVA. Moreover, the thermal stability increased from 358°C (PVA alone) to 375°C (composite with 2 wt.% treated graphite particles). Dynamic mechanical analysis revealed an increase in glass transition temperature from 68.2°C to 72.9°C, and activation energy from 604.84 to 1028.21 kJ mol−1 (neat PVA to composite with 1.5 wt.% modified particles). The damping coefficient of the cross-linked composite was 0.257, making it suitable for acoustic damping applications like speakers.
Keywords
Introduction
Biodegradable polymers and composites are becoming increasingly popular in various industries because of their environmentally friendly nature and low cost of production. These materials can degrade naturally over time, reducing the impact on the environment compared to traditional nondegradable plastics.1–4 Polyvinyl alcohol (PVA) is indeed a biodegradable polymer that has gained popularity in recent years due to its versatility and biodegradable properties.5–7 PVA is a widely used polymer because of its biocompatibility and lack of toxicity. Hermann and Haehnel first synthesized it in 1924 by hydrolyzing poly (vinyl acetate) in ethanol with potassium hydroxide. 8 PVA is commonly used in composites such as PVA–fiber composites, PVA–nanoparticle composites, PVA–ceramic composites, etc. for its strong interfacial bonding with reinforcing materials, improving mechanical strength and toughness. 9
The reactivity of hydroxyl (-OH) groups of PVA with water's H+ ions make it readily soluble in water, posing a significant challenge. However, the complete solubility of PVA in water limits its application in moisture-exposed settings. To overcome these limitations, researchers have successfully employed acid cross-linking techniques using various inorganic and organic acids such as HCl, boric acid, citric acid, and others. This crosslinking approach pre-engages the hydroxyl (-OH) groups of PVA with acid, effectively reducing the hydrophilicity of PVA and enhancing its compatibility with moisture-sensitive applications.10–15 Crosslinking agents like fumaric acid (FA), oxalic acid, and tartaric acid are used to crosslink PVA, which is suitable for food packaging along with drug-delivery systems. FA is a dicarboxylic acid that reacts with hydroxyl groups of PVA to form an ester, increasing its strength, stability, and heat resistance. It is low-cost, reactive, and commonly used in industrial applications, including composites manufacturing. FA is also environmentally friendly, biodegradable, and nontoxic. 16
The incorporation of carbon-family reinforcements, such as graphene, carbon fibers, carbon nanotubes, etc. enhances the mechanical and physical properties of polymer composites, improving fatigue resistance, fracture toughness, and overall performance.17–19 The use of graphite as a reinforcing material in polymer composites has allowed researchers the development of high-performance materials with a range of desirable properties, making it a valuable material in many industries and applications.20–22 Graphite is a naturally occurring form of carbon that consists of stacks of graphene sheets.23–34 The graphene sheets are composed of carbon atoms that are bonded in a hexagonal pattern and arranged in a two-dimensional plane.35–40 Each carbon atom in graphene is sp2 hybridized, meaning it forms three strong, covalent bonds with other carbon atoms and one weak π bond.36,37 Inert graphite particles face challenges of poor dispersion and weak bonding, but surface modification improves interfacial interactions in PVA composites, enhancing mechanical properties. Modified graphite in composites has attracted significant attention in recent years due to its high specific strength, low density, and good thermal conductivity. These properties make modified graphite a promising reinforcement material for a wide range of composites, including polymer composites, ceramic composites, and metal composites.35,38–41 The modification of graphite is a process of modifying its surface for improved performance in composites. This can be achieved through oxidative acid treatment, which chemically activates the graphite by functionalizing it with acids. 42
Khoramishad et al. 43 investigated the effect of 0.3 wt.% graphene oxide nano-platelets (GONPs) on fracture behavior in epoxy adhesive-bonded joints. They achieved a 69% improvement in fracture energy. Numerical analysis revealed that GONPs influenced the fracture process zone length. In the optimal case, incorporating 0.3 wt.% GONPs increased cohesive strength by 33% and failure separation by 27%, indicating an extended fracture process zone length. Ashofteh et al. 44 investigated the impact of incorporating 0.1 wt.% GONPs on the creep behavior of adhesively bonded joints. The inclusion of 0.1 wt.% GONPs resulted in reductions of elastic shear strains by 21%, 31%, and 34%, and reductions of creep shear strains by 24%, 31%, and 37% at temperatures of 30°C, 40°C, and 50°C, respectively. Khoramishad et al. 45 explored the impact of GONPs on nanocomposite adhesive joint strength at elevated temperatures. The inclusion of GONPs altered the joint strength, with the improvement diminishing as temperature increased. Above a critical temperature, GONPs negatively affected joint strength. The critical temperature depended on the weight percentage of GONPs: 60°C for 0.1 wt.% and reduced to 40°C for 0.3 wt.%. Ashofteh et al. 46 studied the hygrothermal aging effects on the creep behavior of multiwalled carbon nanotubes (MWCNT) and GONP-reinforced adhesive joints. Results showed that elastic shear strain increased by 14%, and creep shear strain by 25% from 30°C to 50°C. 0.1 wt.% MWCNTs maximally reinforced, reducing elastic and creep strains by 56% and 33% compared to neat joints. GONPs reduced elastic and creep strains by 45% and 20% in nanocomposite adhesive joints. Tareq et al. 17 investigated the three-point flexural fatigue behavior and mode I interlaminar fracture toughness of carbon fiber-reinforced polymer composites modified with graphene nanoplatelets (GnP). Incorporating 0.1% GnP improved mean and predicted fatigue life by 155% and 190% (0.5 failure probability), respectively. GnP-reinforced composites showed up to 40% higher mode I interlaminar fracture toughness. NajiMehr et al. 18 evaluated the shear strength of steel-bonded lap joints reinforced by MWCNT and GNP after limited fatigue loading. The maximum static shear strength improvements were associated with 1.0 wt.% MWCNT and 0.5 wt.% GNP contents. Zamani et al. 19 examined the influence of hybrid insertion of GNP and silica nanoparticle (SNP) additives on aluminum-to-GFRP bonded joints. The study includes different GNP + SNP mixing ratios: 30%GNP + 70%SNP, 50%GNP + 70%SNP, and 70%GNP + 30%SNP. The findings highlight specific mixing ratios that resulted in maximum average static failure load, highest static strength, and longer crack initiation life.
Dalmis et al. 47 examined the nanoparticulate reinforcement effects on the physical and mechanical properties of ZA27 alloy composites. Density decreased with increasing graphite content, as the porosity ratio of nano graphite-added nanocomposites (GANs) increased. Higher graphite rates also led to decreased ultimate tensile strength (UTS) and Brinell hardness values. Hammood et al. 48 investigated Nickel-5 vol.% Zirconium dioxide composites with varying graphite particle contents. Powder metallurgy was used for fabrication, followed by optical microscope characterization. Their results showed that increasing graphite content improved micro-hardness, diametral compressive strength, and wear resistance up to 4%. Higher loads reduced wear resistance. Hybrid composites with 4% graphite particles demonstrated excellent mechanical and wear properties, suitable for corrosion-resistant applications. Irhayyim et al. 49 examined aluminum-5 wt.% graphite self-lubricating composites with MgO nanoparticles. Powder metallurgy was employed for fabrication, and scanning electron microscopy (SEM) and X-Ray Diffraction were used for characterization. Increasing MgO content up to 2.5 wt.% enhanced micro-hardness and diametral compressive strength, while reducing wear rate. Hybrid composites with 2.5 wt.% MgO nanoparticles demonstrated superior mechanical and wear properties.
Anstey et al. 39 modified the biocarbon structure using oxidative acidic treatment. Biochar derived from Miscanthus biomass was refluxed with nitric acid, sulfuric acid, and their mixture. The treatment increased sulfur and oxygen content and introduced carboxylic acid and nitro groups. HNO3-H2SO4 treatment showed the most effective functionalization for enhanced polymer matrix interaction. Sezer et al. 41 investigated the oxidative acid treatment of MWCNTs by refluxing them with varying molar concentrations of HNO3 and HNO3/H2SO4. MWCNTs treated with 15 M HNO3 showed 5.15% oxygen content, higher than 10 M HNO3 (3.2%) and 10 M H2SO4/HNO3 (2.2%). Furthermore, weight loss was 17%, 6.8%, and 5% for 10 M H2SO4/HNO3, 15 M HNO3, and 10 M HNO3, respectively. Thus, 15 M HNO3 treatment resulted in the highest dispersion stability.
Despite an extensive literature review, no prior studies have utilized FA as a cross-linking agent to enhance the mechanical properties and reduce the wettability of PVA. The bio-friendly nature of FA makes it a promising candidate for food packaging and biomedical materials. Furthermore, the novel use of sulfuric acid (H2SO4) for surface modification of graphite particles embedded in the PVA matrix, leveraging its strong oxidative properties, remains unexplored. This study introduces a unique approach to developing cross-linked PVA composites that have the potential to replace conventional plastics.The study involved the preparation of optimized cross-linked PVA composite film samples with varying concentrations of functionalized graphite particles. Tensile tests were conducted to evaluate mechanical properties, including UTS, percentage elongation, and modulus of elasticity. Water absorption (WA) experiments examined the influence of graphite particles on water uptake, while thermo-gravimetric analysis (TGA) experiments analyzed thermal stability. The dynamic mechanical analysis (DMA) determined the viscoelastic behavior and the glass transition temperature (Tg). The hypothesis posits that different concentrations of functionalized graphite particles will affect the mechanical and thermal properties of the cross-linked PVA composite films. The analysis aimed to identify the optimal concentration of graphite particles to achieve the desired mechanical and thermal characteristics.
Materials and methods
Materials
For the current study, PVA hot (C2H2O4) n molecular weight 14,000, degree of hydrolysis (mole %) 87–89%, and with the viscosity of 11.6–15.4 cp was procured from Research-Lab Fine Chem Industries, Mumbai for experimentation. To cross-link with the PVA, FA (C4H4O4—molecular weight 116.07) was purchased from the Research-Lab Fine Chem Industries (Mumbai, India). Additionally, graphite powder with a particle size of 0.25 mm and a molecular mass of 12.01 (purchased from Alpha Chemika in Mumbai, India) was added to the matrix material as a reinforcement. The mixture was dried in an oven using a Petri dish with a 20 cm diameter as the mold. 50
Modification of graphite powder
The current study aims to enhance the reactivity of graphite particles with PVA by modifying them through an oxidative acidic treatment. The process involved mixing graphite powder with a 5 M H2SO4 solution (112 mL H2SO4 and 288 mL distilled water) and refluxing the solution at 90°C for 24 h with magnetic stirring. Consequently, hydroxide and carbonyl groups became attached to the surfaces of the graphite particles, creating a negatively charged and hydrophilic surface. The separated particles were then dried in a vacuum oven for 24 h and confirmed to contain hydroxide and carbonyl functional groups through Fourier transform-infrared (FT-IR) spectroscopy. Figure 1 provides a comprehensive visualization of the process flowchart, depicting the sequential steps involved, alongside highlighting the oxidative acidic treatment of graphite particles employed in the study.

Flowchart of the process (left) and oxidative acidic treatment of graphite particles (right).
Fabrication of composite film
The cross-linked reinforced films were prepared using the solution casting method. To initiate the process, 5 g of PVA was dissolved in 150 mL of distilled water. The solution was then stirred for 60 min at 85°C and 300 RPM. FA at 8 wt.% and modified graphite particles are then added as cross-linker and reinforcing material, respectively. The solution is stirred for another 60 min at 85°C and 300 RPM before being cast into a glass petri dish and dried in a vacuum oven at 70°C for 24 h. The resulting films are then peeled off and subjected to various tests, and further heated at 120°C for 60 min to prepare thermally cross-linked (TC) films. Table 1 shows the nomenclature of the different cross-linked films.
Nomenclature of cross-linked and reinforced PVA composite films.

To ensure optimal results, a temperature of 85°C and a 60 min duration were selected to achieve a clear solution of PVA and FA and facilitate efficient cross-linking. Stirring the solution at 300 RPM effectively disperses graphite particles within the crosslinked solution. Lower stirrer speeds risk agglomeration of graphite particles in the PVA matrix, compromising load transfer and resulting in inferior mechanical properties.51–53
Cross-linked reinforced films were fabricated utilizing the solution casting method, meticulously adhering to the prescribed sequence of steps. Figure 2 showcases specific images associated with the aforementioned procedure.
5 g of PVA was dissolved in 150 mL of distilled water, and the solution was stirred for 60 min at 85°C and 300 RPM to ensure thorough mixing and dissolution. FA at a concentration of 8 wt.% was added as a cross-linker, promoting the formation of cross-links within the PVA matrix. Modified graphite particles were incorporated as a reinforcing material into the solution to enhance the mechanical properties of the resulting films. The solution was stirred for an additional 60 min at 85°C and 300 RPM to ensure proper dispersion of the graphite particles throughout the solution. The well-mixed solution was transferred into a glass petri dish, ensuring uniform distribution. The solution-filled petri dish was dried in a vacuum oven at 70°C for 24 h, facilitating the removal of excess water and allowing the formation of a solid cross-linked film. After the drying process, the resulting film was ready for further characterization or application.

Casting of PVA composite, vacuum oven, and petri dish (from left to right).
This step-by-step process ensured the proper preparation of cross-linked reinforced films, optimizing the dispersion of graphite particles, and the formation of cross-links within the PVA matrix.
Specification of the prepared specimen
The tensile testing procedure adheres to the widely accepted ASTM D882-02 standard, specifically designed for evaluating the tensile properties of thin plastic sheets. Conforming to ASTM D882-02, specimens were precisely cut to a width of 10 mm and a length of 60 mm. Additionally, the film thickness was meticulously measured at three distinct locations utilizing a dial vernier callipre, yielding an average thickness value of 0.25 mm. These tests were conducted under ambient temperature conditions, with each test condition being assessed using five individual samples. Refer to Figure 3 for a visual representation of the specimens prepared for the tensile testing process.

Images of tensile tested specimens of graphite particles reinforced composite films.
Sample characterization
Scanning electron microscopy
In this research, SEM was utilized to investigate the structure and dispersion of modified graphite in PVA resin composites. Fractured surfaces of tensile test samples were examined via SEM images to evaluate interfacial adhesion, phase segregation, blending, internal cracking, and casting level. The pictures were obtained through microscopic inquiry at 1000× magnification with the Jeol JSM-6610LV model (from Tokyo, Japan) using an accelerated voltage of 5 kV.
FT-IR spectroscopy
In the current examination, the material (sulfuric acid-treated graphite particles) was subjected to FT-IR spectroscopy utilizing a Bruker alpha Eco-ATRIR furnished with a ZnSe crystal of German origin. The instrument's scope extends from 4000 to 400 cm−1 with a spectral precision of 0.2 cm−1. To initiate the FTIR analysis, individual graphite particles were meticulously placed in a designated location within the FT-IR spectroscope. This placement ensures that the infrared rays pass through the material accurately, enabling the acquisition of spectroscopic data. The purpose of the infrared spectroscopy was to verify whether or not the hydroxide and carbonyl functional groups on the graphite particles after modification had been connected.
WA test
As PVA-based composites are prone to WA, a WA test is necessary to evaluate their behavior under dynamic environmental and loading conditions. The samples of varying compositions, with a size of 25 mm × 25 mm, were first dried for 2 h at 80°C using a microwave oven. The initial weight (W1) was recorded before immersing the samples in 100 ml of water at room temperature. After 20 min, the sample was gently wiped with tissue paper and re-weighed (W2). The percentage (%) of WA was calculated using equation (1) every 20 min for up to 2 h. Additionally, the WA test was performed under controlled environmental conditions using a humidity cabinet (Scientech, India) at 30°C and 90 RH for 4 days, and the percentage of WA was calculated using equation (1) as well.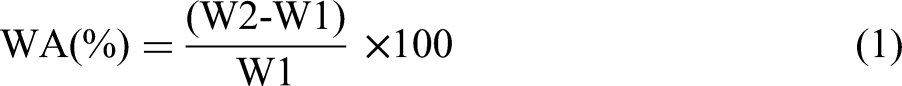
Tensile test
Using a universal testing machine (model AMT-SC-01521), the mechanical properties of modified graphite particle-reinforced PVA-based cross-linked films were determined. The UTS, strain, Young's modulus, and toughness were evaluated through tensile testing conducted following ASTM D882-02, the Standard Test Method for Tensile Properties of Thin Plastic Sheeting (Superseded). 54 The test method is suitable for composites with a thickness of less than 1 mm. Samples were cut to a width of 10 mm and a length of 60 mm, and the thickness of the films was measured at three different locations using a dial vernier calliper. The average thickness value was used for the tests. The tests were performed at ambient temperature, and each test condition was evaluated with five samples. The machine load cell's least count was 0.5 N, and the jaw separation rate of UTM was set to 1 mm min−1 for the test speed.
Thermal analysis test
To evaluate the thermal stability of the material, a thermal analysis was conducted. TGA was performed to assess the reduction of material concerning temperature under a consistent heating speed. The derivative TGA (DTGA) was utilized to obtain data on the mass loss rate. The STA 7300 HITACHI machine was used for this test, and the N2 supply rate was set to 150–200 mL min−1. 55 A platinum crucible was used, and a finely chopped sample of mass 3–6 μg was placed in it. The temperature range was set between 30°C and 500°C, and the heating rate was maintained at 10°C min−1.
DMA test
To adequately characterize a material under dynamic loading conditions, traditional elasticity, and plasticity measurements may not be sufficient. Instead, the viscoelastic response of the material becomes more apparent. To identify the viscoelastic properties of the material, DMA tests were conducted using a DMA analyzer (7100 HITACHI) under sinusoidal tensile mode. Samples, which were approximately 20 mm × 10 mm in size, were tested in a temperature range of 30°C–200°C at a constant heating rate of 2°C min−1. The measurements were taken at a frequency of 10 Hz with a 5 µm amplitude. The DMA test provided data on the storage modulus, loss modulus, tan δ, and Tg.
Biodegradability test
Due to biological factors like the presence of microorganisms in the soil, the weight of a material is reduced in a process known as biodegradability. The biodegradability of materials based on high molecular weight polymers is affected by the glass transition temperature and degree of crystallinity, where a higher degree of either results in reduced biodegradability and vice versa.
56
In this study, samples of 25 mm × 25 mm were taken for each composition and dried for 1 h in a microwave oven at 80°C, with their corresponding weight recorded as Wb. These samples were then buried in garden soil for 60 days. After they were removed, rinsed with water, and dried again in a microwave oven at 80°C for one hour before being weighed as Wa. Using equation (2), the amount of weight loss that represents biodegradability was calculated.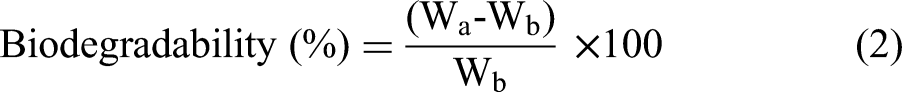
Results and discussion
FT-IR test
Figure 4 represents the FT-IR spectrum of modified and unmodified graphite particles. Two spectrums show the clear-cut changes in graphite particle structure after the treatment with sulfuric acid. There is a medium sharp peak at 3742.06 cm−1, which shows the stretching of the hydroxyl (-OH) group in modified graphite. Again, a deep sharp peak at 1548.09 cm−1 is attributed to C=O and C=C stretching vibrations.
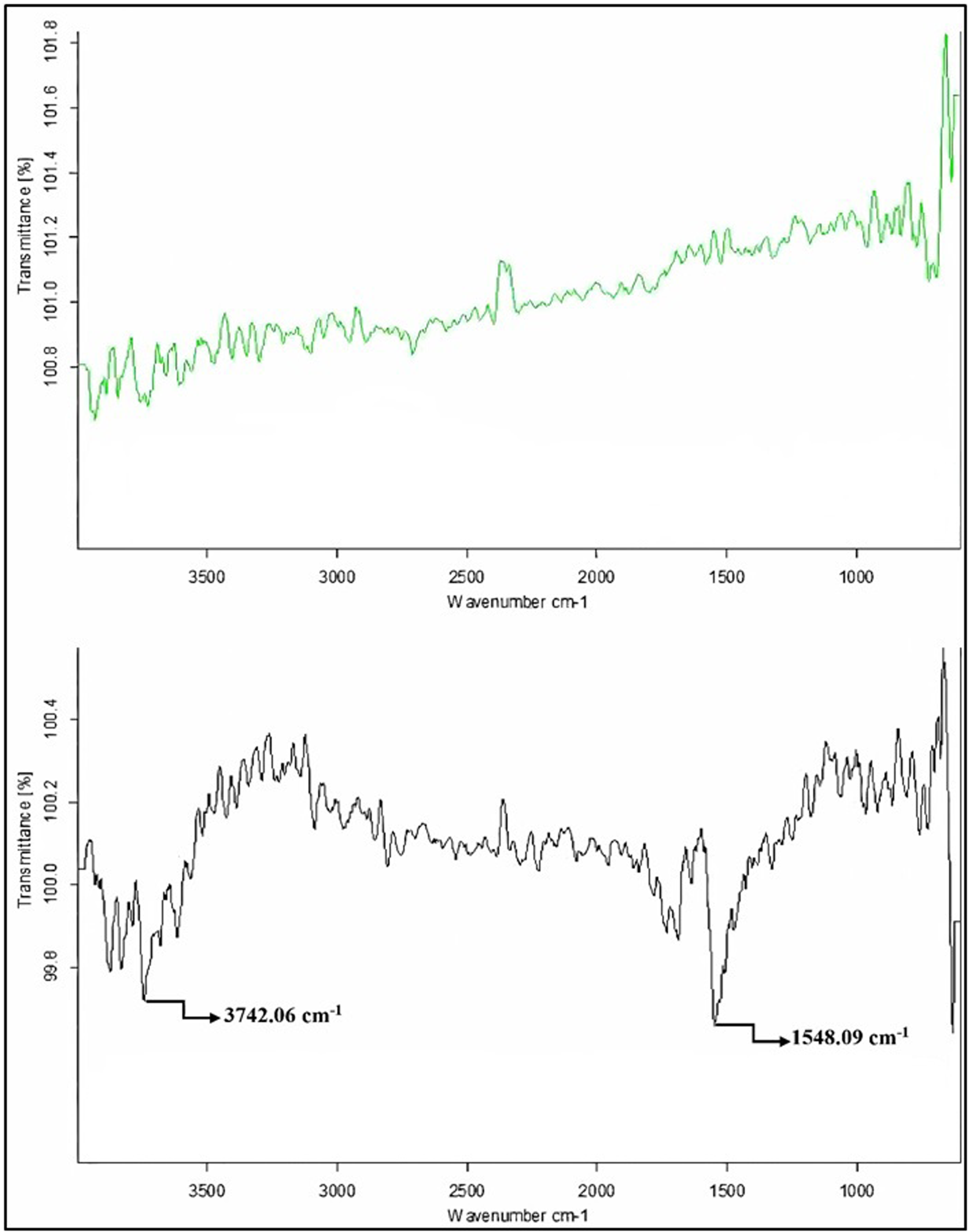
Comparative FT-IR spectroscopies of nonfunctionalized (upper figure) and functionalized graphite (lower figure) particles.
Above results showed that oxygen atoms are attached in the form of hydroxyl (-OH) group and C=O ketone group to the inert graphite after modification with sulfuric acid, which is known as an oxidative acidic treatment. As a result, modified graphite is more prone to attach to PVA composite matrix. The attachment of hydroxyl (-OH) groups via oxygen enhances water affinity in the composite to a small extent, while significantly increasing mechanical strength, which is the primary objective of incorporating graphite particles.
WA test
Due to the hydroxyl group in its side chain, PVA is a hydrophilic polymer that absorbs water and swells in moist environments. The water uptake capacity of PVA can be reduced by adding FA, which reacts with the hydroxyl group of PVA via its carboxylic group.16,50 Graphite particles, which are typically inert toward the water, can increase WA when added to PVA depending on their size.
Figure 5 shows the WA characteristics of the composite. As shown in Figure 5(a), in the first 20 min, the WA rate of PVA reaches a maximum of around 120% due to its hydrophilic nature and voids in the film. Over the next hour, it decreases until it becomes almost constant after 80–120 min. The water absorptivity increases from 120% to 133% with an increase in graphite weight percentage. Figure 5(b) shows a similar pattern when H2SO4-treated graphite particles are reinforced in cross-linked PVA. However, in this case, the WA increases from 120% to 155% because the modified particles have hydroxyl (-OH) bonds, which absorb more water than the untreated particles.
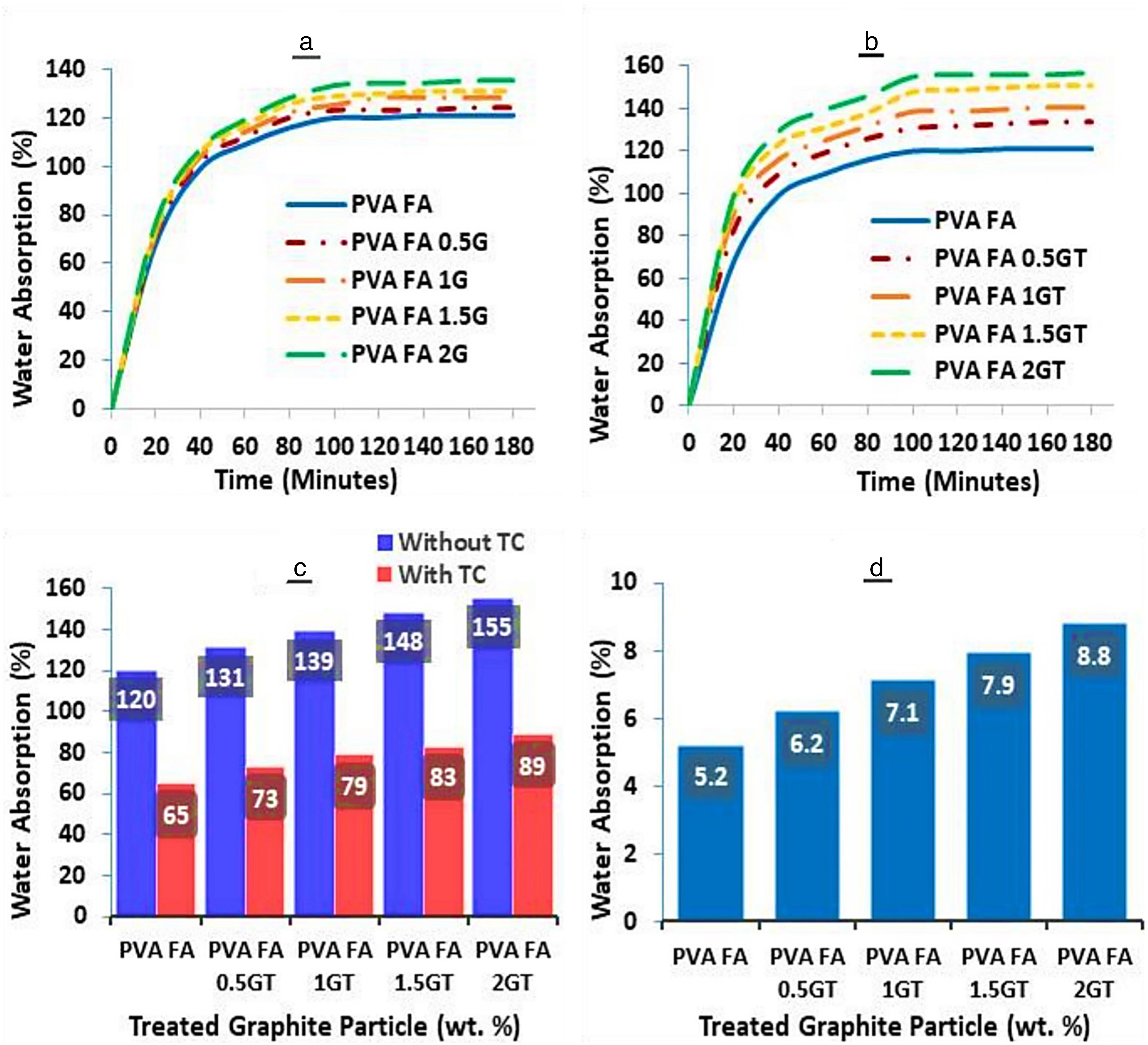
(a and b) Variation of WA with time for different wt.% of untreated and treated graphite particles reinforced composites, (c) effect of thermal crosslinking on WA of treated particles reinforced composites, and (d) variation of WA with different wt.% of treated graphite particles reinforced composites under a controlled environment (90 RH at 30°C).
Figure 5(c) shows the effect of thermal cross-linking on modified graphite particle-reinforced films. After thermal cross-linking at 120° C for 60 min, the films show decreased WA due to the collapse of voids. For FA cross-linked PVA, WA is 65%, but adding treated graphite particles up to 2 (wt./wt.%) increases it to a maximum of 89%.
Under controlled environmental conditions (90% relative humidity at 30°C) for four days, the samples showed very little gain in weight. For PVA-FA cross-linked film, the WA rate was 5.2%. However, in the case of 2 (wt./wt.%) modified graphite particle reinforced composite film, the WA rate was reduced to 8.8%, as shown in Figure 5(d).
Tensile test
Table 2 provides the UTS and percentage elongation results for various weight percentages of PVA composites strengthened with modified graphite.Adding 1.5 wt.% sulfuric acid treated graphite to cross-linked PVA matrix enhanced UTS by 31%, exhibiting optimal weight for maximum UTS compared to neat cross-linked PVA. Simultaneously, the strain was reduced by 35% when incorporating 1.5 wt.% sulfuric acid-treated graphite, indicating increased brittleness of the film.
Tensile and WA properties of graphite particle reinforced PVA composite.
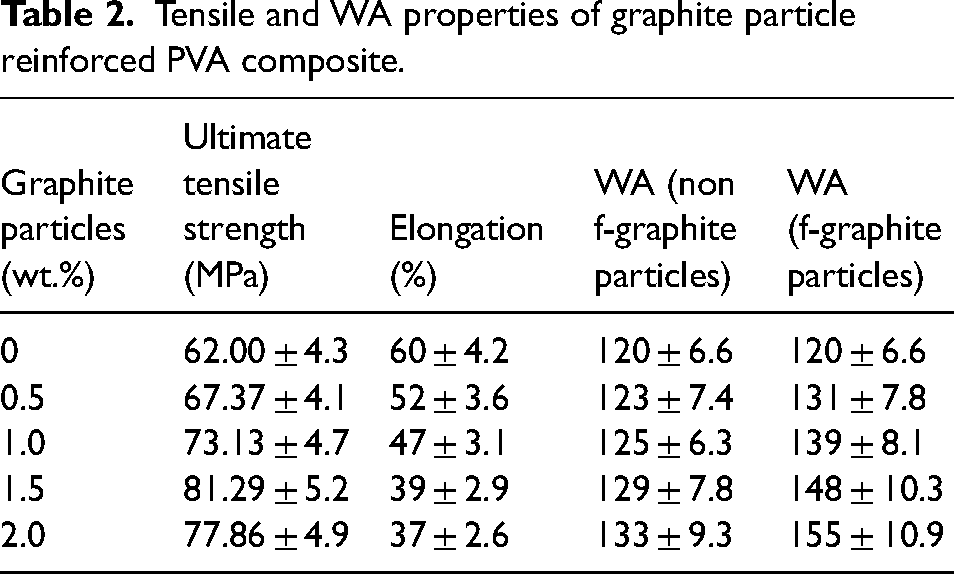
Figure 6 depicts the outcome of the tensile test. In Figure 6(a), the stress-strain curve showed UTS differences. Neat FA cross-linked PVA film had the lowest UTS (62 MPa). The UTS of composite material initially increased with modified graphite weight percentage in PVA matrix up to 1.5 wt.% (81.29 MPa), but decreased with a further increase in weight percentage. This observation can be attributed to the phenomenon of particle agglomeration. When the wt.% of graphite particles exceed the threshold, and agglomeration occurs, resulting in the formation of clusters. These clusters introduce stress concentration points, consequently diminishing the overall strength of the composite material.
(a) Stress–strain curve for cross-linked PVA composite reinforced with various wt.% of modified graphite particles, (b) cross-linked PVA composite reinforced with 1.5 wt.% modified graphite and nonmodified graphite particles, (c) UTS comparison chart for normal and TC composite reinforced with 1.5 wt.% of unmodified and modified graphite particles, (d) Young's Modulus line chart for non-TC composite reinforced with 1.5 wt.% of unmodified and modified graphite particles, and (e) Pearson correlation heatmap of various physical and mechanical properties of fabricated composite concerning modified reinforced particles.
Figure 6(b) compares the tensile strength of 1.5 wt.% modified and nonmodified graphite particle-reinforced composites with virgin cross-linked PVA. Both have higher tensile strength than PVA. The optimal weight percentage is 1.5 wt.%, beyond which the stress transfer rate reduces, causing weak interfacial bonding between particles and matrix and early composite failure. Strain decreases due to increased stiffness and particle-matrix interaction.
Figure 6(c) and (d) is the comparative graphical representation of maximum stress and Young's modulus of 1.5 wt.% unmodified and modified graphite particle-reinforced composites as compared to virgin cross-linked PVA. Young's modulus of the film increased by 20% and 61% in unmodified and modified graphite particle-reinforced composites, respectively, compared to virgin cross-linked PVA due to enhanced stiffness.
Figure 6(e) heatmap that displays the Pearson correlation among various factors of PVA composite reinforced with modified graphite particles. As the weight percentage of graphite particles increases, the WA of the film increases, indicating a positive correlation of 1.0. The weight percentage of graphite particles has a substantial positive impact (0.93) on UTS but a negative impact (−0.99) on elongation. The heatmap confirms the results of the WA and mechanical tensile tests. Please note that these heatmaps validate and reinforce the investigation findings; thus, aiding in the optimization of composite performance by visually representing correlations and patterns among factors, facilitating a deeper understanding of their influence.
SEM test
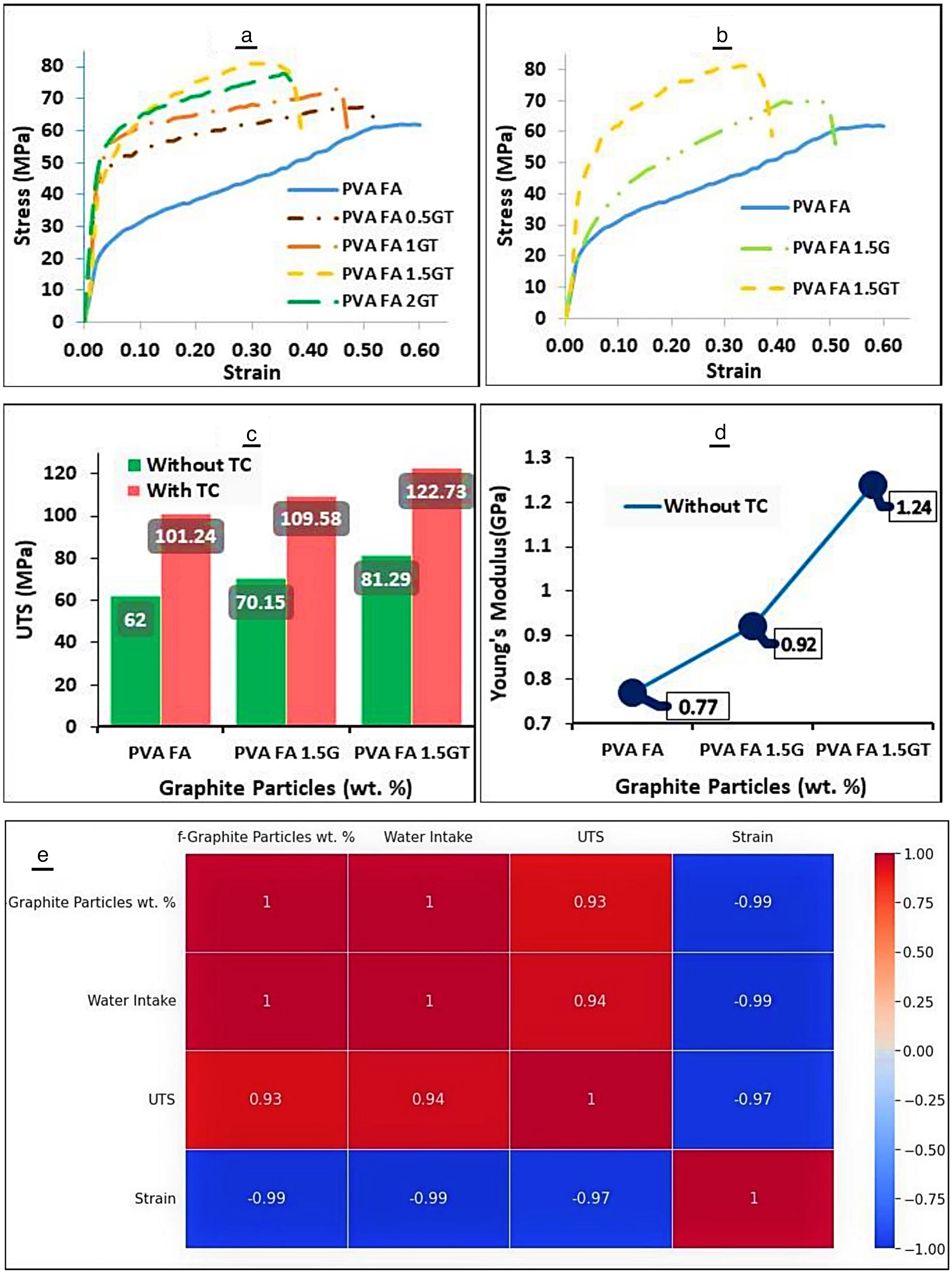
In this study, the surface characteristics of composite materials were analyzed using SEM. Cross-sectional views of composites made from PVA, 8% FA cross-linked PVA, and various modified graphite particle weight percentages are presented in Figure 7. The smooth surface of neat PVA film is shown in Figure 7(a), while Figure 7(c) to (f) showed the microstructure of cross-linked PVA composites with modified graphite particles.
SEM (magnification level of 1800×) of the fractured surface of (a) virgin PVA film, (b) 1.5 wt.% unmodified graphite particles reinforced cross-linked PVA composite film, (c) 0.5 wt.% modified graphite particles reinforced cross-linked PVA composite film, (d) 1 wt.% modified graphite particles reinforced cross-linked PVA composite film, (e) 1.5 wt.% modified graphite particles reinforced cross-linked PVA composite film, and (f) 2 wt.% modified graphite particles reinforced cross-linked PVA composite film.
The fractured surface of untreated and treated graphite-reinforced cross-linked PVA composites are displayed in Figure 7(b) and (e), respectively. The 1.5 wt.% modified graphite-PVA composite system exhibited the highest tensile strength due to the even distribution of particles within the matrix and strong adhesion between the treated graphite particles and polymer. The bonding between functionalized graphite and cross-linked PVA in composites with 0.5 wt.% and 1 wt.% was weaker than that of the 1.5 wt.% composite, as shown in Figure 7(c) and (d), respectively. As the weight percentage of graphite particles increased, the interfacial bonding between the matrix and reinforcement weakened, resulting in inadequate load transfer and agglomeration, as depicted in Figure 7(f).
The study concluded that the optimum weight percentage of modified graphite particles in cross-linked PVA composites was 1.5 wt.% based on the SEM analysis. This weight percentage resulted in proper bonding between the modified graphite particles and cross-linked PVA, leading to the best tensile strength of the composite material. This finding will be confirmed in the mechanical test subsection.
Thermal analysis test
Figure 8 shows the impact of modified graphite particle reinforcement on the thermal behavior of cross-linked PVA composite. The thermal examination was conducted to explore the alterations in the composite's thermal properties after reinforcing the modified graphite particles.
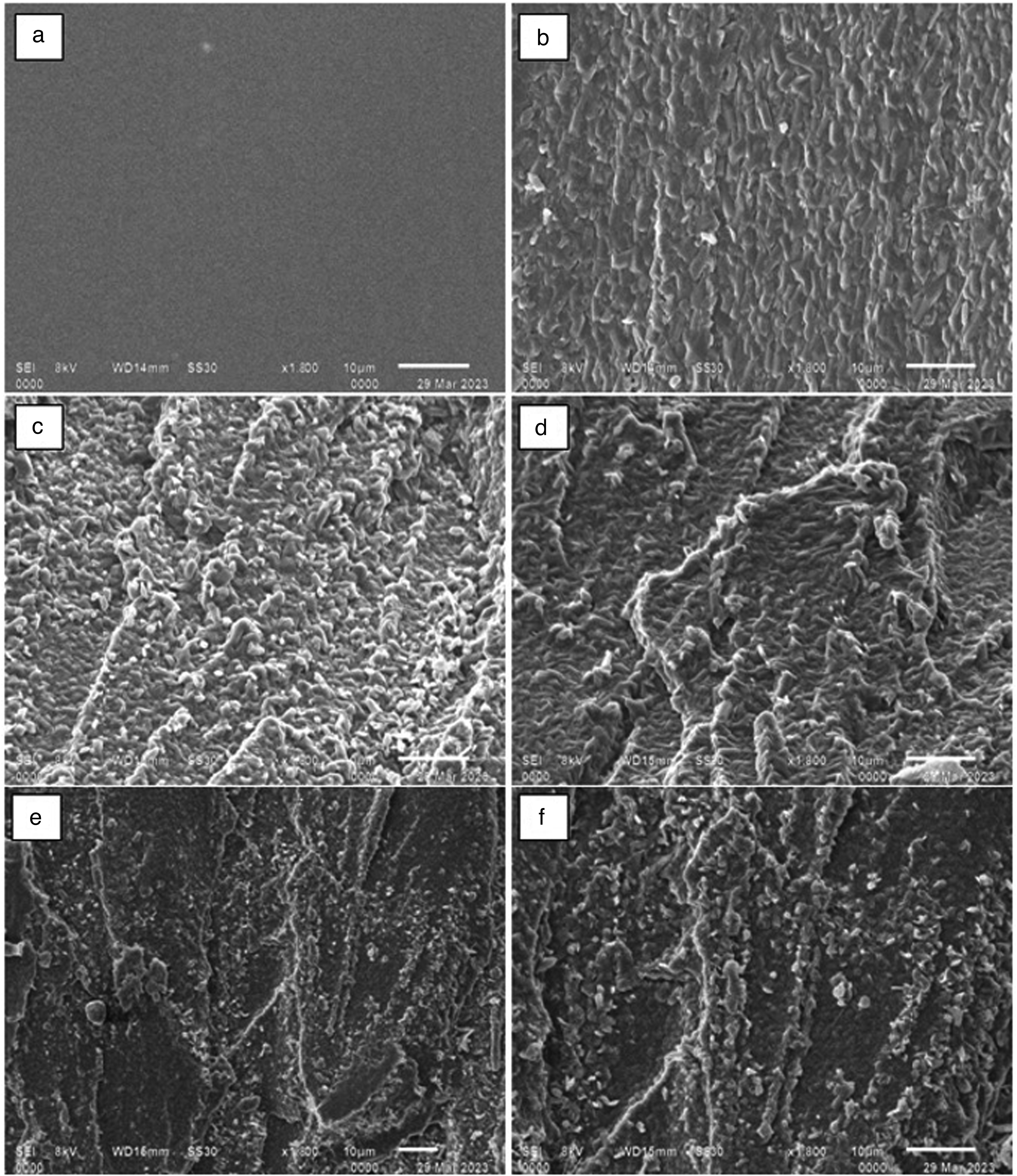
(a) TG graph of cross-linked PVA composite reinforced with varying wt.% of modified graphite particles and (b) DTG graph of cross-linked PVA composite reinforced with varying wt.% of modified graphite particles.
The thermo-gram (TG) in Figure 8(a) indicates that there are three temperature ranges where the decomposition or mass loss of PVA is most significant. 57 The first range is 80–160°C, where the material undergoes dehydration or evaporation. The second range is 225–370°C, where thermal degradation of the PVA backbone chain occurs, leading to the transformation of the composite into ethanol, aldehyde, and ketone, as well as gaseous products such as carbon dioxide and carbon monoxide. The third range is from 400°C to 460°C, where the breakage of bonds results in the formation of carbons and hydrocarbons and subsequent mass loss. 58 Figure 8(a) shows that in the first range, there is a mass loss of 0.8–4% due to evaporation, and in the second range, the mass loss is sharper for simply cross-linked PVA compared to PVA reinforced with treated graphite particles. Acid-modified graphite improves the heat resistance of PVA because modified graphite particles act as a heat sink, resulting in greater thermal stability. The overall stability of the composites increases as the proportion of modified graphite increases.55,59
The addition of treated graphite particles to PVA composites enhances interfacial bonding which restricts the polymer chain's mobility, resulting in increased thermal stability (Figure 8(b)). This is evidenced by a shift in the DTGA curve from 358°C (for PVA alone) to 375°C (for the composite containing 2 wt.% treated graphite particles). The peak shift confirms the composite's superior thermal stability compared to solely FA cross-linked PVA. 59
Dynamic mechanical analysis
In Figure 9 the dynamic mechanical behavior of PVA-based composite film was studied using DMA to determine E’ and tan δ. The stiffness of a material is typically described by its storage modulus. 60 The mobility of PVA is constrained by modified graphite particles, thereby boosting the storage modulus of composites via interfacial adhesion. The graph in Figure 9(a) indicates the storage modulus of cross-linked PVA composites reaches the maximum at 1.5 wt.% of modified graphite particle reinforcement with the highest interfacial strength. However, further, increase in wt.% causes agglomeration and reduces the storage modulus.
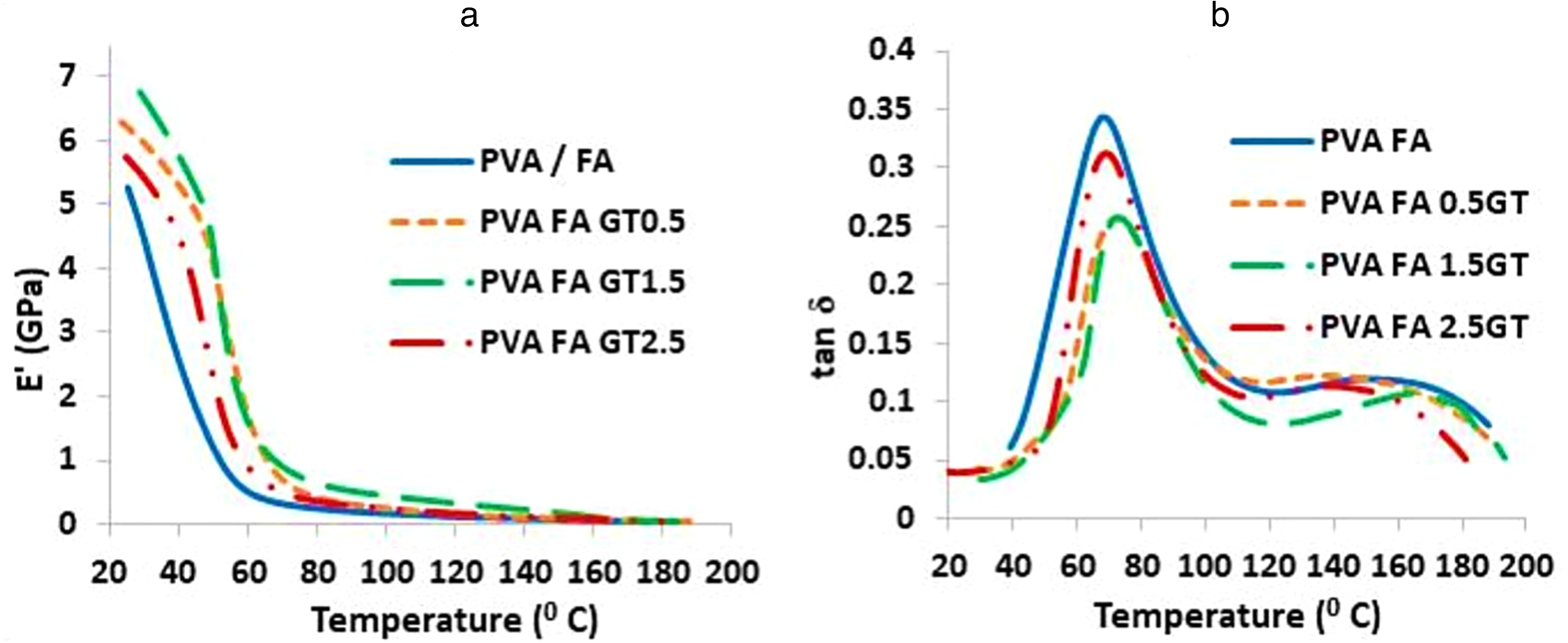
(a) E’ graph of cross-linked PVA composite reinforced for varying wt.% of modified graphite particles and (b) tan δ graph of cross-linked PVA composite reinforced for varying wt.% of modified graphite particles.
As per the basic definition, tan δ represents the quotient of loss modulus and storage modulus. As per Table 3, the damping coefficient (tan δ) was analyzed to assess energy dissipation in PVA composites with different graphite filler percentages. The 0% sample displayed a higher damping coefficient and enhanced energy dissipation. The lower tan δ values for 0.5% and 1.5% samples suggest lower energy dissipation, potentially due to reduced interfacial friction and increased stiffness from filler incorporation. Nonetheless, the 2.5% sample showed a slight improvement in damping coefficient and energy dissipation compared to the 1.5% sample. Figure 9(b) has two peaks in the tan δ curve: the first represents β-relaxation (500–900°C) and the second represents α-relaxation (1100–1700°C). This analysis is similar to that of PVA cross-linked with citric acid by Brick et al.. 57 β-transition is due to the side chain movement, and α-transition is due to main chain movement. Tg is determined by the temperature corresponding to the first peak.
Glass transition temperature, tan δ, biodegradability and activation energy of modified graphite particles reinforced cross-linked PVA composite film.
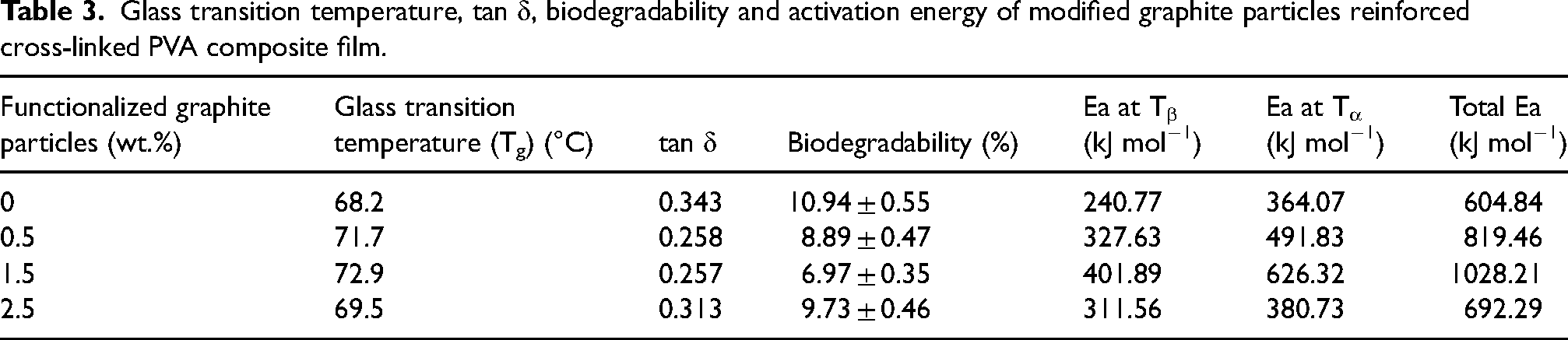
The composite material displays characteristics of both elasticity and viscosity. The incorporation of modified graphite particles improved the composite's elasticity, and reduced tan δ, indicating higher elastic properties. Figure 9(b) showed adding modified graphite particles increased the glass transition temperature, with the highest obtained at 1.5% weight reinforcement (72.9°C) beyond which further increment caused a decrease in glass transition temperature (Table 3).
Biodegradability analysis
Biodegradability refers to a material's ability to break down via soil enzymatic activity. Microbes first target the amorphous, then the crystalline portion, resulting in lower biodegradability with higher crystallinity and glass transition temperatures. Furthermore, lower glass transition temperatures increase the biodegradability. 56
The weight loss percentage of PVA cross-linked composite following 60 days in the soil is displayed in Figure 10(a). As evidenced by Table 3, PVA composites with 1.5% weight of graphite particle reinforcements exhibit inferior biodegradability compared to other compositions due to their elevated glass transition temperature.
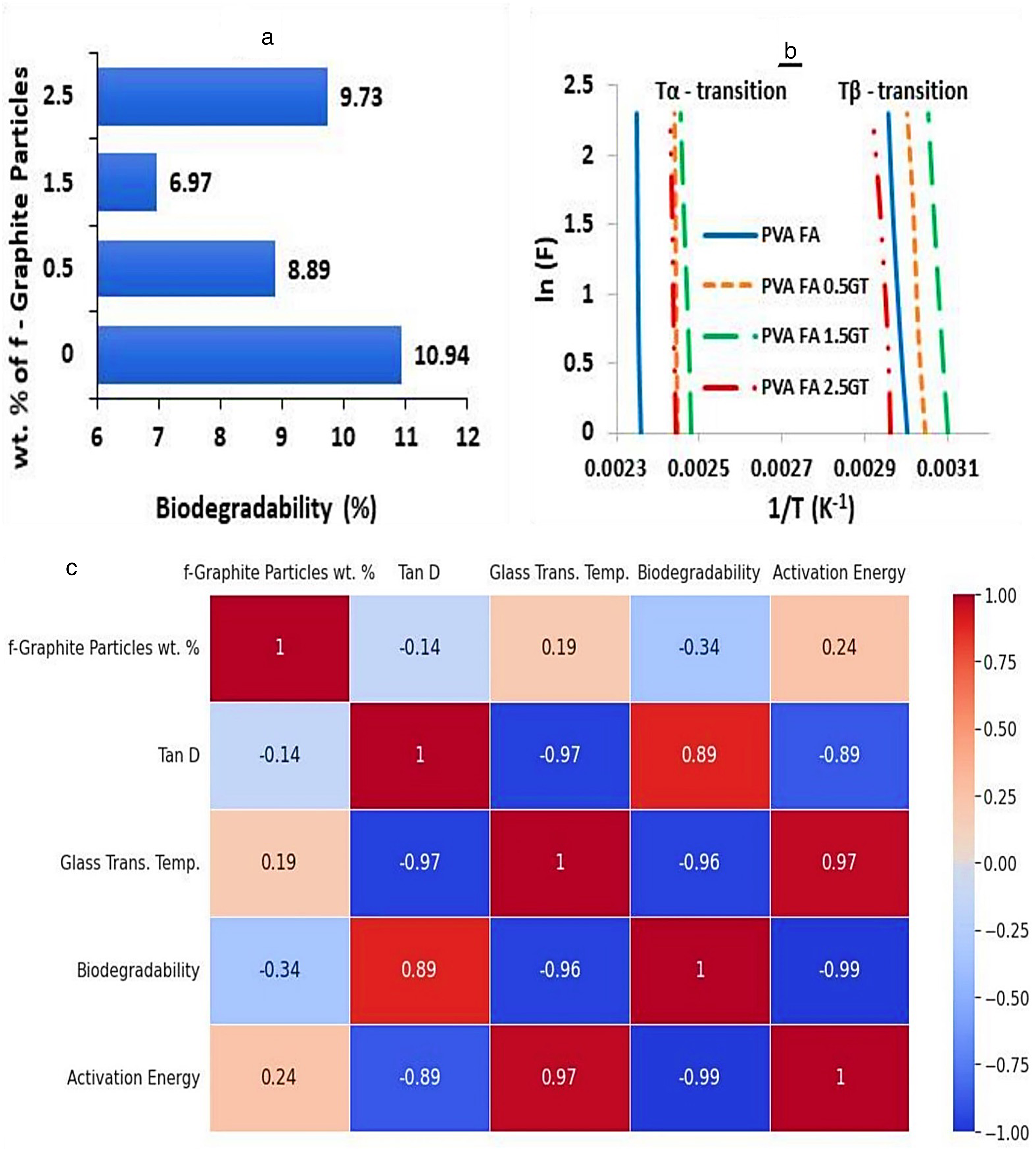
(a) Biodegradability (%) of f-graphite particles reinforced composite after 60 days, (b) Arrhenius plot to determine activation energy, and (c) Pearson correlation heatmap of various properties of fabricated composite concerning modified reinforced particles.
Activation energy
During the breakdown of polymer molecules, the polymer takes in energy at β-relaxation and α-transition when subjected to dynamic stress, referred to as activation energy, which denotes the least energy indispensable for the breakdown of the polymer. The Arrhenius equation 
Table 3 shows activation energy at β-relaxation and α-transition, obtained from Figure 10(b), and the sum of activation energy at Tβ and Tα. Activation energy increases continuously and peaks in the 1.5 wt.% modified graphite particle reinforced PVA composite.
Figure 10(c) showcases the heatmap of Pearson correlation for PVA composite reinforced with f-graphite particles, shows a weak positive correlation (+0.19) between wt.% of graphite particles and glass transition temperature, and a moderate negative correlation (−0.34) with biodegradability. The tan δ correlation (−0.14) indicates a mild increase in stiffness with graphite particle reinforcement, and activation energy shows a moderate positive correlation (+0.24) demonstrating the composite's chemical stability. The results well align with the biodegradability and DMA test outcomes.
Conclusions
This study presents a novel approach to fabricating PVA-modified graphite-reinforced composites via oxidative acid treatment. The methodology involved comprehensive analyses of mechanical properties, SEM imaging for dispersion assessment, thermal analysis using TG & DTG, and DMA measurements. Incorporating 1.5 wt.% sulfuric acid-treated graphite in cross-linked PVA resulted in a significant 31% increase in UTS, representing an optimal weight percentage for maximum UTS compared to neat cross-linked PVA. However, this addition also increased brittleness, reducing strain by 35%. SEM analysis confirmed uniform dispersion of graphite particles without phase separation or internal cracking, contributing to higher UTS. Increasing graphite weight percentage raised water absorptivity from 120% to 133%, while H2SO4-treated graphite exhibited increased WA (120–155%) due to hydroxyl (-OH) bonds. Thermal cross-linking reduced WA, with FA cross-linked PVA at 65% and 2% modified graphite reaching 89% absorptivity. Under controlled conditions (90% RH, 30°C, 4 days), PVA-FA film absorbed water at 5.2%, while 2% modified graphite composite reduced it to 8.8%. Thermal analysis showed improved stability with 2 wt.% treated graphite (358–375°C). The DMA demonstrated enhanced storage modulus and glass transition temperature, particularly at 1.5 wt.% modified graphite with moderate tan δ. Overall, this investigation provides valuable insights into the fabrication and properties of PVA-modified graphite-reinforced composites, offering potential applications in automotive components, consumer goods, and medical devices requiring strength, flexibility, and stiffness. However, high WA may limit certain applications, while its suitability for filtration membranes in water and fluid purification shows promise.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the TEQIP-III grant at College of Technology, Govind Ballabh Pant University of Agriculture and Technology, Pantnagar, India.
