Abstract
In this research, tin was added to the AZ31/2Al2O3 magnesium metal matrix composite to investigate its influence on degradation rate in the presence of simulated body fluid (SBF) and wear behaviour in dry conditions. The AZ31-xSn/2Al2O3 (x = 0, 2, 4, 6, 8, and 10 wt%) composites were manufactured using the bottom pouring stir casting route. The microstructure of the manufactured AZ31/2Al2O3 and AZ31-xSn/2Al2O3 composites revealed that they were composed of the α-Mg solid solution and the intermetallic compound □-Mg17Al12, which was situated near the grain boundaries. When tin was added, the Sn-rich intermetallic compound Mg2Sn was formed, increasing the volume fraction of the Mg2Sn phase. Compared to AZ31/2Al2O3 composite, the wear test revealed that AZ31/2Al2O3 composites containing Sn particles exhibited a higher ability to generate more stable tribo-layers at higher applied loads, which protected the worn surface and reduced the wear rate. The wear resistance of composites was improved primarily by the behaviour of the tribo-layer in the wear process. The degradation rates (mm/year) of the AZ31 composites were carried out for 72 h in SBF. The degradation rate was reduced when the Sn content in the AZ31/2Al2O3 composite was increased to 6 wt% and then increased with further Sn addition. It was observed that when the amount of Sn particles increases up to 6 wt%, the severity of the degradation decreases.
Introduction
One of the most exciting research topics in biomaterials is the study of advanced biodegradable implant metals. Biodegradable implant materials can be progressively dissolved, absorbed, consumed, or excreted in the human body, rejecting the necessity for secondary surgery to remove implants after the surgical regions have healed. Current biodegradable polymer implants 1 have insufficient mechanical strength and thus have limited applications. Magnesium alloys have the potential to be biodegradable materials because of their superior biological performance. 2 Mg2+ is an important element that is the fourth most abundant cation in the human body. A normal adult's daily Mg intake is around 300–400 mg, and redundant Mg cations can be evacuated through urine securely and efficiently. 3 It was recently reported that bone strength and growth are enhanced by the existence of magnesium in the bone system. 4 In addition, the density and modulus of elasticity (1.74–2 g/cm3, 41–45 GPa) of Mg alloys are near those of human bone (1.8–2.1 g/cm3, 3–20 GPa), but conventionally used artificial implant materials such as stainless steels, cobalt–chrome alloys, and titanium alloys, have a density of >4 g/cm3 and a Young's modulus of >110 GPa, which is higher compared to that of human bone. As a result, Mg alloys are mostly better compared to polymer or metallic implants 5 in terms of mechanical and physical properties for orthopaedic and bone repair/replacement purposes, as the difference in modulus of elasticity between an implant and natural bone causes stress shielding effects that cause stress to concentrate at the bone and implant interface, lowering stimulation of new bone growth and reducing durability. 6
Lim et al. 7 have concluded that alumina particles of 1.11 vol.% can considerably develop pure magnesium's hardness, ductility, and wear resistance. Saravanan and Surappa 8 have investigated the characterization of pure magnesium reinforced with 30 vol.% silicon carbide (SiCp) particles composites. It is found that pure Mg reveals a dendritic structure, whereas the composites show a cellular dendritic structure. Up to 400 °C, the composites exhibit higher wear resistance and superior strength than pure magnesium. Muley and Singh 9 studied the volumetric wear loss at a constant speed of 1 m/s with the rise in the sliding distance under various applied loads of 5, 10, and 15 N for AZ91 alloy, AZ91 with 3 wt% silicon, and AZ91 with 5 wt% silicon composites. It has been concluded that the volumetric wear loss rises linearly with the sliding distance, and AZ91–5 wt% silicon composite has better wear resistance for all the loading conditions. Girish et al. 10 studied the effect of AZ91-silicon carbide-graphite particle hybrid reinforcements on the wear characteristics. The wear test in dry conditions uses a pin-on-disc apparatus with parameters such as applied load ranging from 20 N to 80 N and sliding velocities (1.047 to 2.618 m/s). At a load of 40 N and a speed of 1.047 m/s, a 6.3 mm3/km wear rate is observed for the unreinforced alloy, and 3.8 mm3/km is observed for the 3% reinforced composite. Adding 1 to 3 wt% silicon carbide and graphite content to the magnesium alloy results in improved anti-wear ability and a reduced wear rate of the composites. Increasing the reinforcement content from 1% to 3% at an 80 N load and 1.047 m/s speed results in a reduced wear rate from 11.8 to 9.1 mm3/km. Better wear resistance is detected when the sliding speed and normal load increase in the reinforced composites and the unreinforced alloy.
Song et al. 11 used electrochemical impedance spectroscopy tests to investigate the biodegradable performances of AZ31 alloy in simulated body fluid (SBF) by varying the immersion time intervals, followed by a discussion of biodegradable mechanisms using scanning electron microscopy (SEM) micrographs. A protective film layer is observed on the AZ31 surfaces. In addition, the deposition of hydroxyapatite particles on the film layer was observed. Human bone primarily comprises hydroxyapatite, proving the AZ31 magnesium alloy's complete biocompatibility. Liu et al. 12 studied the corrosion and wear properties of the Mg–2Zn–0.2Mn alloy in stimulated body fluid. The selected magnesium alloy's wear resistance is very low when using SBF solution as a medium compared to sliding in dry and deionized water conditions.
Magnesium alloys are widely used in the biomedical industry due to their excellent biodegradability. However, certain magnesium alloys that contain rare earth elements, cadmium (Cd), zirconium (Zr), and heavy metals are not ideal for use in biomaterials. 13 On the other hand, pure magnesium lacks sufficient wear and corrosion resistance. Among different magnesium alloys, the AZ31 alloy with a low amount of aluminium has shown promising wear and corrosion resistance properties, making it suitable as a biodegradable material. However, the degradation rate of the AZ31 alloy is different than expected. This research investigates how adding tin (Sn) to the AZ31/2Al2O3 magnesium metal matrix composite affects its degradation rate in SBF and its wear behaviour in dry conditions. The study seeks to understand the impact of various concentrations of tin on the degradation and wear resistance of the composite material. Analysing the microstructure and adjusting the tin content aim to enhance the composite's wear resistance and reduce its degradation rate.
Experimental details
Materials preparation
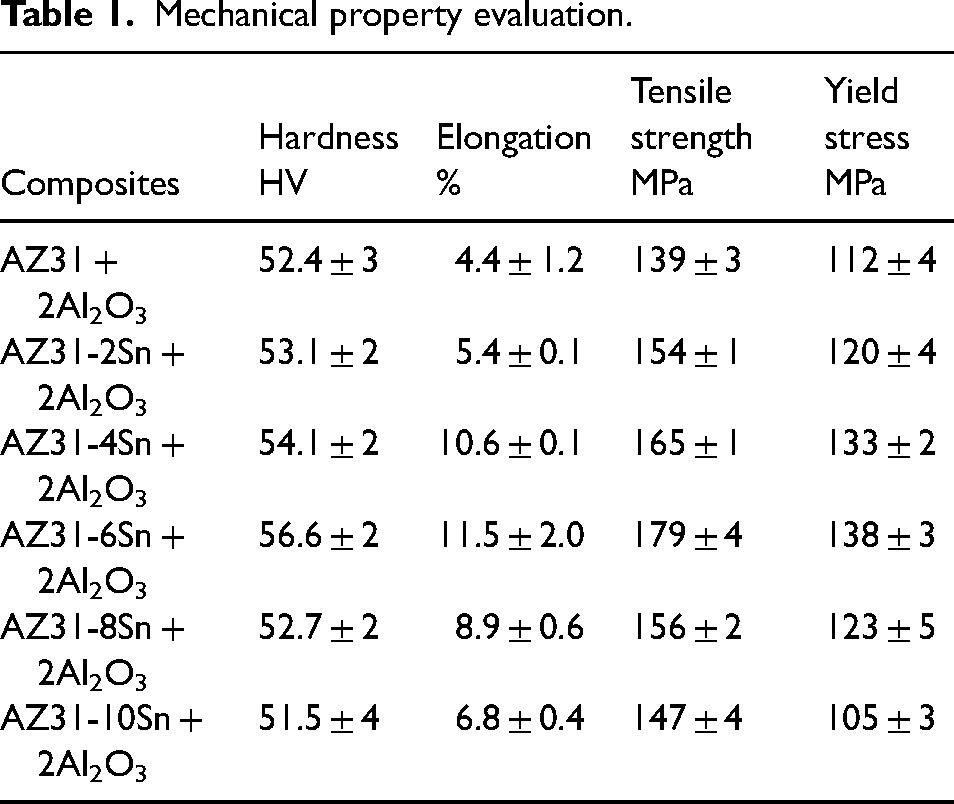
In this experiment, the AZ31 alloy is strengthened by incorporating nano-sized aluminium dioxide particles (40 nm in size) and micro-sized tin powder (44 µm in size) with a purity of 99.9%. Through the stir casting route, first, the AZ31 alloy rods are melted in the furnace (650 °C), followed by the addition and stirring of preheated reinforcements such as Al2O3 and Sn powders. This molten mix is cast as a cylindrical rod (210 mm × Ø 25 mm) and a square plate (150 mm × 150 mm × 25 mm). The samples are taken from the steel die, followed by cooling in the air. After fabrication, the microstructure and phase compositions of the fabricated composites are examined using an optical microscope and X-ray diffraction (XRD). The mechanical parameters such as microhardness, yield stress, elongation (%), and ultimate tensile strength are tested, and the findings are discussed elsewhere 14 and displayed in Table 1.
Mechanical property evaluation.
In vitro degradation test
The disc samples are cut from the fabricated composites with a diameter of 12 mm and a thickness of 2 mm, ground with waterproof abrasive papers of 800, 1200, 1500, 2000, and 2500 grit to achieve uniform surface roughness, and cleaned ultrasonically in acetone and alcohol in sequence. The current study used the SBF proposed by Kokubo et al.
15
as a degradation medium. The pH level of SBF was kept at 7.4, and 37 ± 0.5 °C temperature was maintained. The ion content of the SBF is shown in Table 2. Figure 1 displays the experimental setup of the immersion test, which is done for 72 h according to the G31-72 ASTM standard.
16
The deteriorating medium volume to specimen surface area ratio is set to 50 ml/cm2 under ambient conditions, and the solution is changed every 24 h. As a result, the pH of SBF can stay constant. After completion of the immersion test for 72 h, the immersed samples are cleaned with a 5 g K2Cr2O7 + 10 ml H2O + 90 ml H2SO4 mixed solution, rinsed with ethanol, and finally, air dried to eliminate corrosion products before being weighed for weight loss measurement. The following equation (1) is used to compute the deterioration rate of the immersed samples.
18

Immersion setup.
The ion content of the simulated body fluid (SBF) and human blood plasma. 15
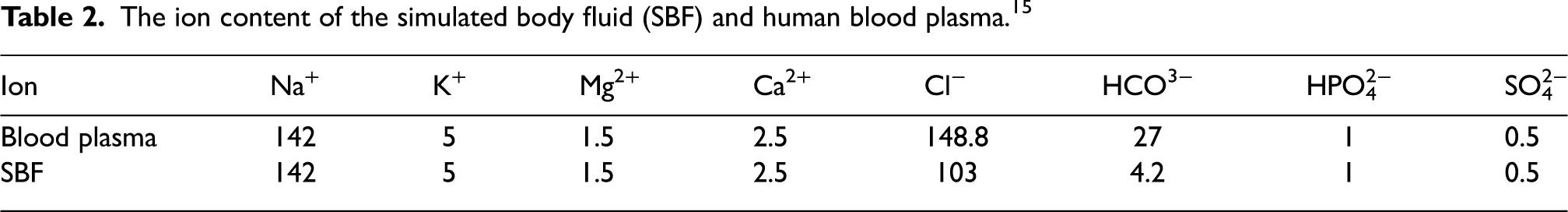
where K is a constant (8.76 × 104), T is the exposure time (hours), W is the mass loss (g), A represents the exposed area in cm2, and D represents the actual density of the samples in g/cm3. Furthermore, after 72 h of immersion, the chemical composition of the fabricated composite samples and the corroded morphology were examined using a field emission scanning electron microscopy equipped with energy-dispersive X-ray spectroscopy (EDS).
Wear analysis
At room temperature, dry sliding wear tests are performed using a DUCOM pin-on-disc tribometer (Model: TR-20LE-PHM400-CHM600) containing an EN-31 steel disc with 60 HRC hardness per the ASTM G99 standard. The spherical-headed cylindrical pin specimen used in the wear test experiment has a specification of 32 mm height and 8 mm diameter. The test is executed with a fixed sliding distance of 1000 m, varying sliding velocities of 1 and 2 m/s, and normal loads of 10, 15, 30, and 40 N. The specimen's sliding disc and end were ground using silicon carbide metallographic paper and then cleaned with acetone. An electronic weighing machine with an accuracy of 0.001 g is used to weigh the pin before and after the wear test. Worn surfaces, wear debris, and wear mechanisms are characterized using the SEM.
The research involved analysing the microstructure and phase composition of the fabricated composites using optical microscopy and XRD. In vitro degradation tests are conducted by immersing disc specimens in SBF for 72 h, with the degradation rate determined through weight loss measurement and examination of corroded morphology using SEM with an equipped EDS. Wear tests under dry sliding conditions are performed using a pin-on-disc apparatus, with analysis of worn surfaces and wear mechanisms conducted using SEM.
Results and discussion
Microstructure behaviour
Figure 2 depicts the microstructure images of the AZ31/2Al2O3 and AZ31–Sn/2Al2O3 composites made by stir casting. The AZ31/2Al2O3 composite exhibits large grains, as shown in Figure 2(a), with grain sizes ranging from 100 to 150 µm. It is seen that the grain size got reduced with the increment in the amount of Sn particles in the AZ31/2Al2O3 composite. It can be observed that as the amount of Sn particles in the AZ31/2Al2O3 composite increased, the grain size reduced. The microstructures of the composite AZ31–10Sn/2Al2O3 are depicted in Figure 2(f). The grains are refined, with an average particle size of 50 to 80 µm. The composites comprise α-Mg solid solution and the intermetallic compound β-Mg17Al12, situated near the grain boundaries. Figure 2 also depicts the distribution of “α-Mg + β-Mg17Al12” regions between the intermetallic compound Mg17Al12 and the Mg-solid solution, as mentioned in Figure 2(a) and (b). Various investigations showed similar microstructure images along grain boundaries with the β-Mg17Al12 phase.17–19 The existence of Al in the Mg alloy affects the volume fraction of the Mg17Al12 phase, which increases as Al content increases. 19
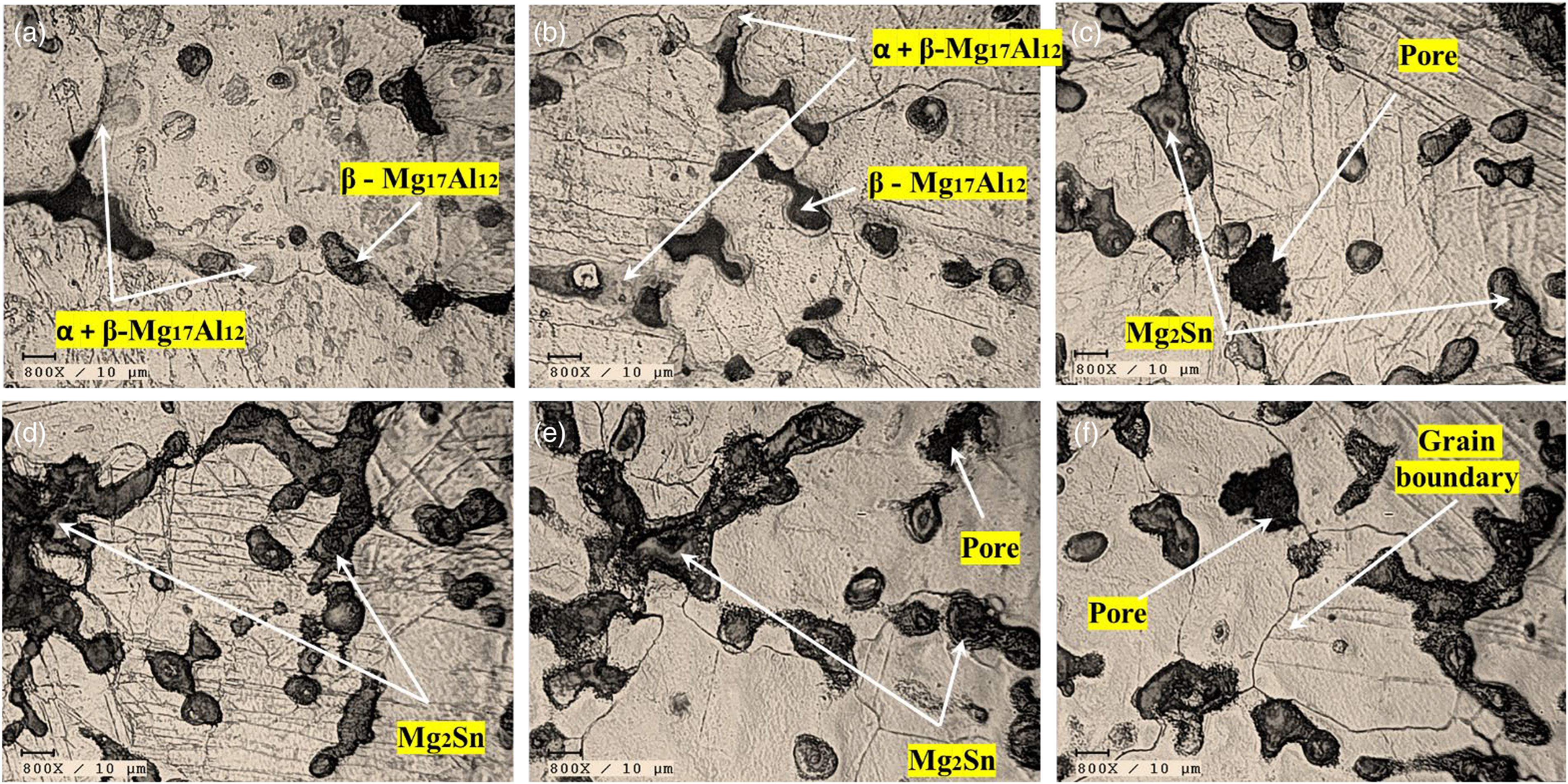
Optical micrographs of fabricated composites: (a) AZ31 + 2Al2O3, (b) AZ31-2Sn + 2Al2O3, (c) AZ31-4Sn + 2Al2O3, (d) AZ31-6Sn + 2Al2O3, (e) AZ31-8Sn + 2Al2O3, and (f) AZ31-10Sn + 2Al2O3.
Figure 2(c) to (f) illustrates the Sn-rich intermetallic compound Mg2Sn formation when Sn reinforcement is introduced. It is observed that adding Sn powder improved the volume fraction of the Mg2Sn phase. Most Sn is dissolved into the matrix during solidification when the Sn addition is <2 wt%. The remaining Sn that did not dissolve in the matrix when the amount of Sn exceeded its solubility limit formed the Mg2Sn phase.20–22 The hard Mg2Sn phase is a strong barrier against grain boundary slippage and deformation, and it is also important to the precipitation-strengthening process. 21 An EDS mapping investigation revealed information about the AZ31/2Al2O3 composites’ chemical composition and elemental distribution. An EDS mapping finding is shown in Figure 3, indicating the presence of evenly distributed Mg, Al, and O components. This demonstrates the existence of alumina (Al2O3) particles in the composite material, which form clusters that are equally dispersed across the sample. Figure 4 also shows the EDS mapping results for the AZ31–10Sn/2Al2O3 composite. The mapping shows that the elements Mg, Al, O, and Sn are distributed uniformly throughout the composite. This indicates that the Sn particles are efficiently incorporated into the composite matrix, resulting in a homogeneous distribution.

The energy-dispersive X-ray spectroscopy (EDS) mapping of the AZ31/2Al2O3 composites.

The energy-dispersive X-ray spectroscopy (EDS) mapping of the AZ31-10Sn/2Al2O3 composite.
Figure 5(a) and (b) displays the XRD graphs of the AZ31–10Sn/2Al2O3 nano-composites and (b) AZ31/2Al2O3. Figure 5(a) confirms the existence of the Mg2Sn secondary phase in the AZ31–10Sn/2Al2O3 composite. Figure 5(b) explains the presence of MgO with Mg and Al2O3 peaks.
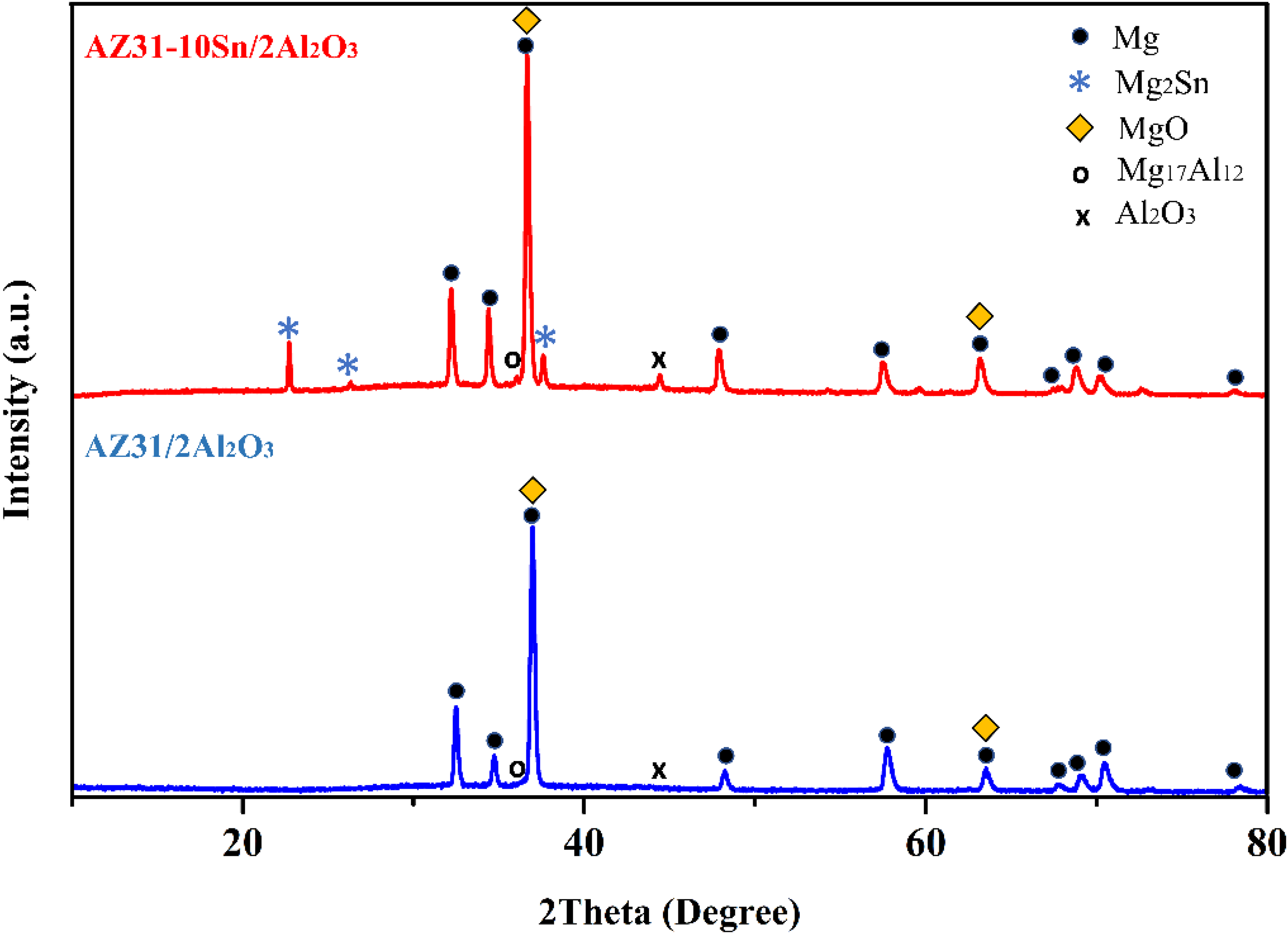
X-ray diffraction (XRD) graphs of magnesium composite: (a) AZ31–10Sn/2Al2O3 and (b) AZ31/2Al2O3..
Wear study
Three independent wear measurements were performed for each condition during dry sliding wear tests, and average values were considered. Figure 6 shows that higher loads increase wear rates due to elevated contact pressure, potential dislodging of reinforcement particles, and elevated temperature, which accelerate wear and damage. In addition, it is also observed that an increment in tin powder content results in a reduced wear rate, which shows that the existence of the ceramic phase Mg2Sn is acting as resistance against wear on the contact surface. A wear rate of 0.025 g/km is observed in the case of AZ31/2% Al2O3 composites, whereas that of 0.015 g/km is found in the case of AZ31–10% Sn/2% Al2O3 for a load of 10 N, a sliding velocity of 1 m/s, and a sliding distance of 1000 m. For different applied loads and sliding distances, the wear resistance for Sn powder-reinforced composites is higher when compared to AZ31/2% Al2O3 composites due to the absence of Sn powder on the contact surface in the softer matrix phase. The AZ31/2% Al2O3 composite wear rate is 0.025, 0.026, 0.028, and 0.038 g/km, whereas in the case of the 10 wt% Sn composite wear rate, it is found to be 0.015, 0.02, 0.024, and 0.029 g/km for the sliding distance of 1000 m, sliding distance of 1 m/s, and applied load of 10, 15, 20, and 40 N, respectively. From Figure 7, it can be seen that the average coefficient of friction (COF) value exhibited specific trends based on the normal load and Sn powder content. As the normal load increases, there is an observed increase in the average COF value. This can be attributed to the increase in contact pressure between the composite and the counter-surface, resulting in higher resistance to motion and an overall increase in the frictional force. It is observed that the average COF value decreased slightly with the addition of Sn particles. In the case of AZ31/2% Al2O3 composites, an average COF value of 0.28 is observed, while for AZ31–10% Sn/2% Al2O3, a lower average COF value of 0.19 is found. These values are obtained under specific conditions, including a load of 10 N, a sliding velocity of 1 m/s, and a sliding distance of 1000 m. Furthermore, when the normal load is increased to 20 N, the average COF of AZ31/2% Al2O3 composites remained at 0.28, while the average COF of AZ31–10% Sn/2% Al2O3 increased significantly from 0.19 to 0.25. At the higher applied load of 40 N, the average COF value of the AZ31/2% Al2O3 composite increased to 0.3, whereas the AZ31–10% Sn/2% Al2O3 composite exhibited a lower average COF value of 0.26. These findings suggest that including Sn powder in the AZ31–10% Sn/2% Al2O3 composite contributes to a lower COF, indicating reduced friction and potentially improved sliding behaviour compared to the AZ31/2% Al2O3 composite.
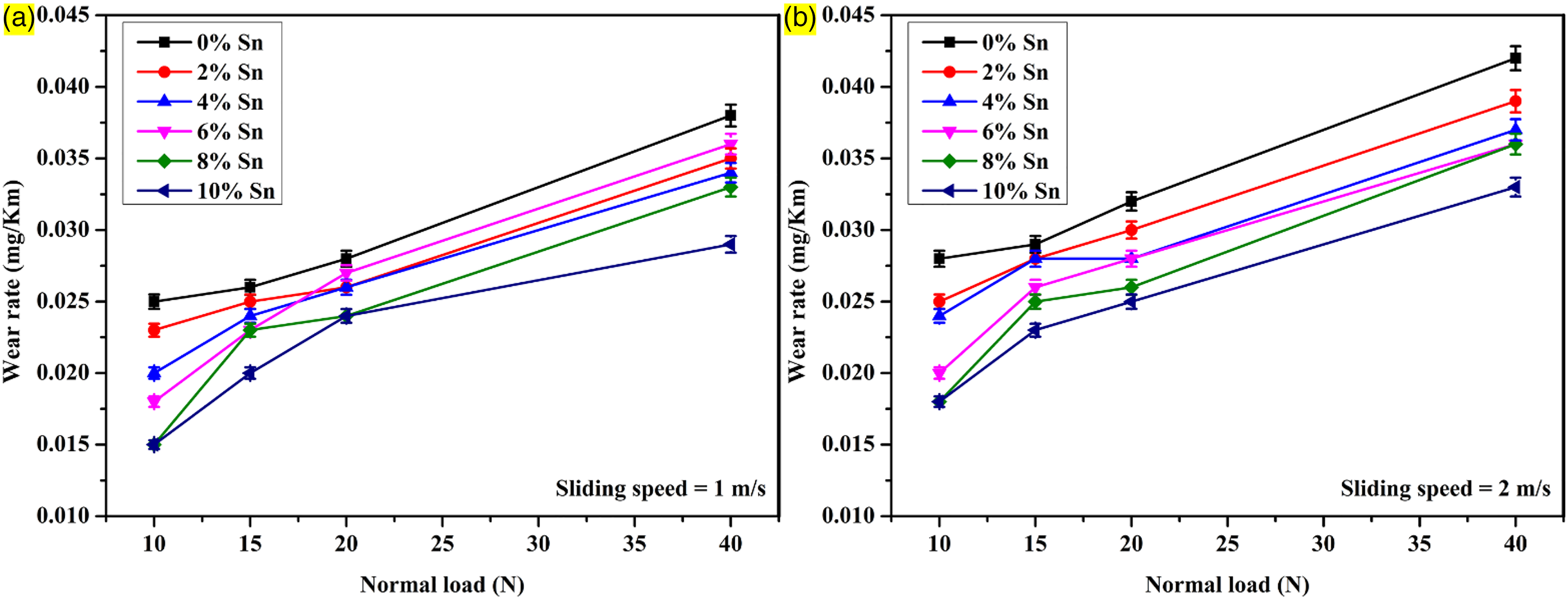
Influence of load against sliding velocity and reinforcement on wear rate.
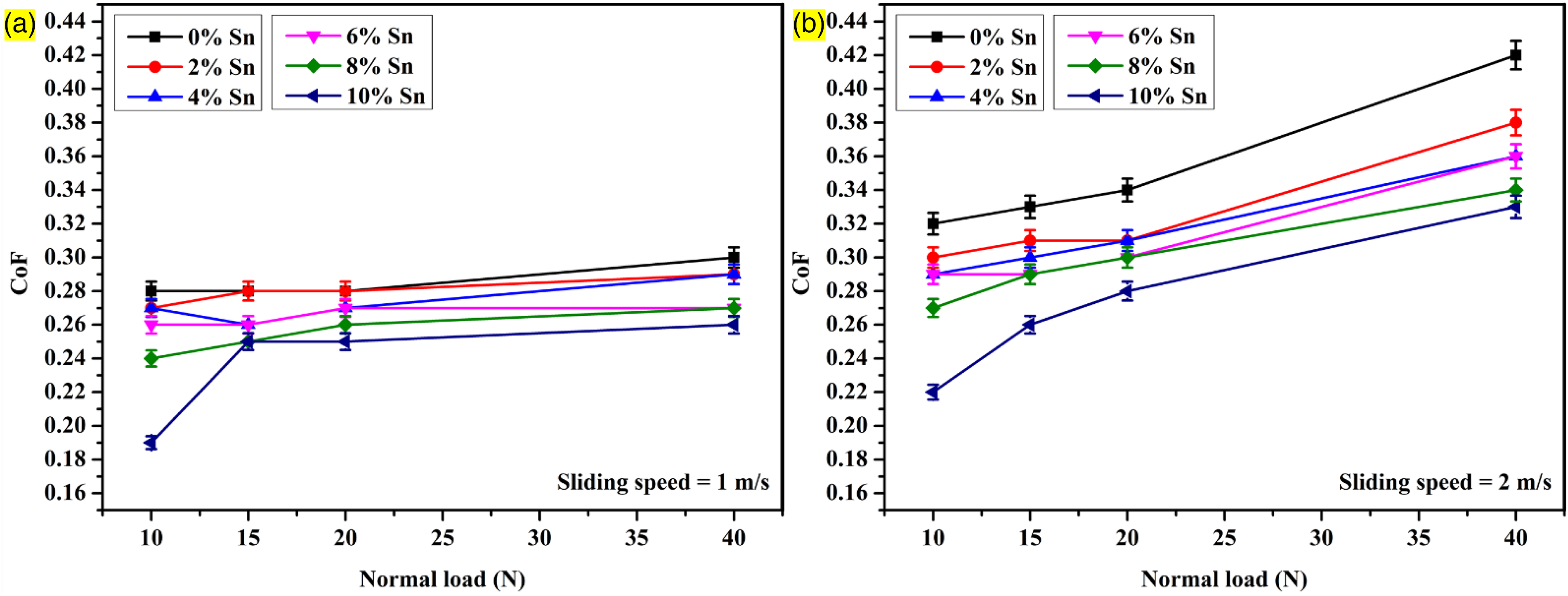
Influence of load against sliding velocity and reinforcement on coefficient of friction (COF).
A SEM study of the worn surfaces of AZ31/2% Al2O3 and AZ31–10% Sn/2% Al2O3 composites is conducted based on the wear rate and COF results for various loads (10 and 40 N) and sliding velocities (1 and 2 m/s). The samples are reduced to fit within the parameters of the SEM apparatus. For all sliding conditions, Figure 8(a) to (d) shows deep grooves parallel to the sliding direction on the worn surface of the AZ31/2Al2O3 composite. This describes abrasive wear brought on by the existence of tightly bound Al2O3 particles that cut the counterface. At 1 m/s sliding velocity under all loading conditions, the micro-cutting wear mechanism material is removed by abrasion and plastic deformation. When the sliding velocity is raised to 2 m/s, the main wear mechanisms under a 10 N load are abrasive wear and plastic deformation brought on by micro-cutting and micro-ploughing. It has been noted that at 30 and 40 N loads, oxides are formed on the worn surface of the pin, which leads to debris build-up in the AZ31/2% Al2O3 composite. Delamination wear traces are observed on the worn-out surface of the AZ31/2% Al2O3 composite with a high load of 40 N and a speed of 2 m/s, as illustrated in Figure 8(d). Under higher loads, the process of debris formation in the AZ31/2Al2O3 composite results in delamination on the surface of the pin. Loading and deformation during sliding could cause microcracks to expand and propagate, leading to the delamination of a thin sheet. Under all testing conditions, the removed wear debris is observable on the worn surface. The material loss caused by severe plastic deformation explains the higher wear rate and COF.
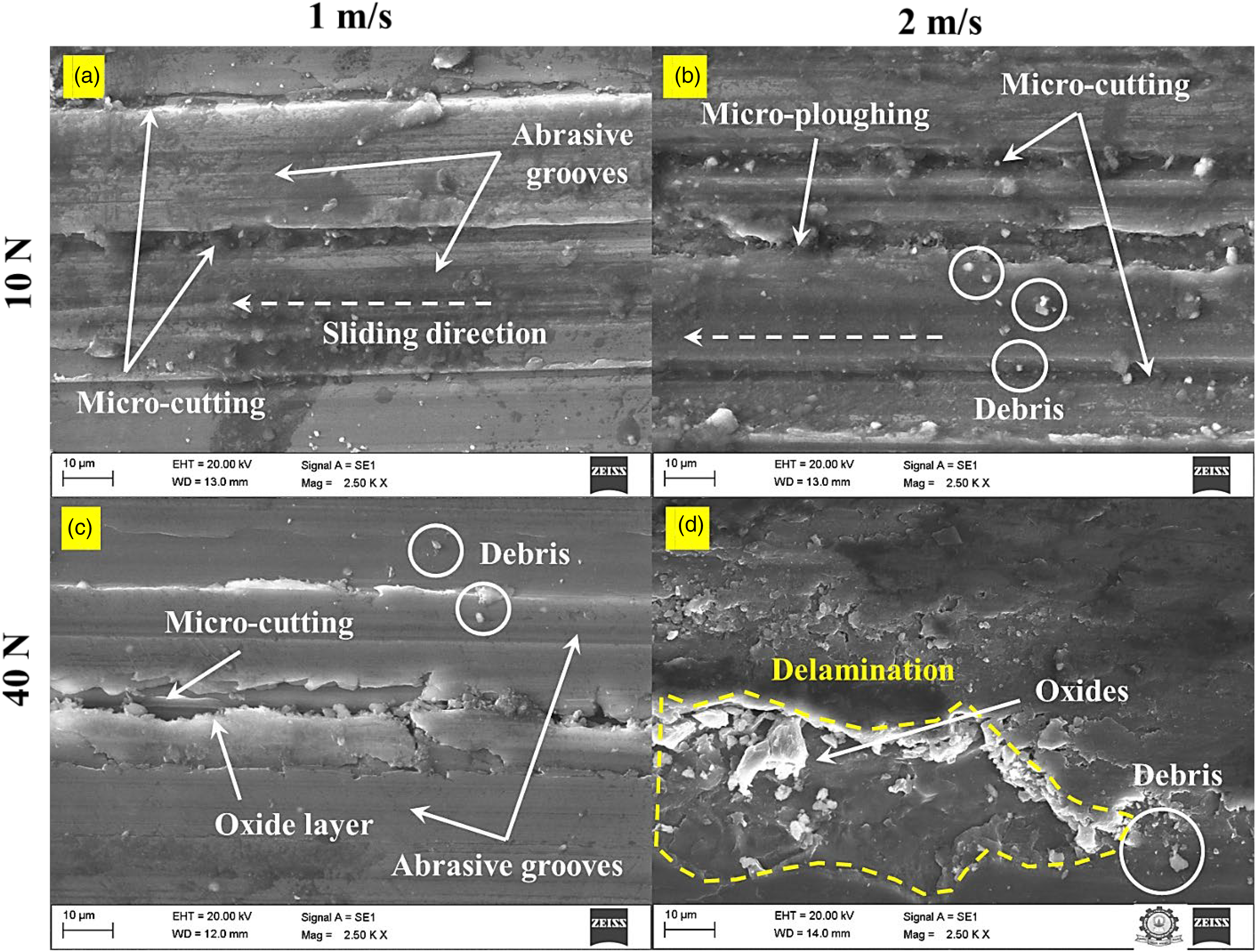
Scanning electron microscopy (SEM) micrographs on the worn surface of the AZ31/2Al2O3 composite: (a) 10 N, 1 m/s, (b) 10 N, 2 m/s, (c) 40 N, 1 m/s, and (d) 40 N, 2 m/s.
The micrograph images of the worn-out surfaces of the AZ31–10Sn/2Al2O3 composite are shown in Figure 9(a) to (d). Figure 9(a) and (b) shows the occurrence of shallow abrasive grooves on the surface of an AZ31–10Sn/2Al2O3 composite subjected to 10 N and 20 N normal loads, respectively. Also, it is observed that the development of a stable tribo-layer on the worn surface. And the more visible layers are observed on the worn surface at higher loading conditions (30 and 40 N), as shown in Figure 9(c) and (d). As the sliding process proceeds, the particles adhered to the worn surface undergo a mix of compacting and sintering to form a load-bearing tribo-layer.23,24 Compared to AZ31/2Al2O3 composite, AZ31/2Al2O3 composites containing Sn particles exhibited a higher ability to generate more stable tribo-layers at higher applied loads, which protected the worn surface and reduced the wear rate. The wear resistance of composites is improved primarily by the behaviour of the tribo-layer in the wear process. Similar results were obtained by various studies.25,26 In conclusion, the AZ31/2% Al2O3 composite exhibited abrasive wear with deep grooves, while the AZ31–10% Sn/2% Al2O3 composite showed shallower abrasive grooves and the formation of a protective tribo-layer. The tribo-layer in the AZ31–10% Sn/2% Al2O3 composite improved wear resistance, emphasizing the significance of particle composition and tribo-layer formation in determining wear behaviour and performance.

Micrograph images of the worn surfaces of the AZ31–10Sn/2Al2O3 composite: (a) 10 N, 1 m/s, (b) 10 N, 2 m/s, (c) 40 N, 1 m/s, and (d) 40 N, 2 m/s.
Effect of reinforcement on the degradation behaviour
All samples possess visible signs of degradation on the surface. Figure 10 shows photographs of the samples before and after corrosive product removal after 72 h of immersion in SBF. Also, it has been observed that when the quantity of Sn particles increases up to 6 wt%, the severity of the degradation decreases. Figure 11 shows the AZ31 composite degradation rate (mm/year) after 72 h in SBF. After 72 h of immersion, the AZ31/2Al2O3 composite degradation rate was 18.5 mm/year, whereas it had decreased by 17.8% for the AZ31–2Sn/2Al2O3 composite. The degradation rate was reduced when the Sn content of the AZ31/2Al2O3 composite was increased to 6 wt% and then increased with additional Sn inclusion. After 72 h of immersion in SBF, the AZ31–6Sn/2Al2O3 composite degraded at an incredibly low rate (7.6 mm/year). The corrosion morphologies of the AZ31/2Al2O3 composite and the AZ31–6Sn/2Al2O3 composite after 72 h of immersion in SBF are shown in Figure 12 at lower magnification (500×, Figure 12(a) and (c)) and higher magnification (1000×, Figure 12(b) and (d)). Figure 12(a) and (b) shows a substantial amount of corroded area on the AZ31/2Al2O3 composite surface, whereas Figure 12(c) and (d) shows that the percentage of the corroded area has dropped greatly with the inclusion of Sn (up to 6 wt%). Corroded surfaces are covered by comparatively enormous corrosion products with vast clusters of flake-like crystals (Figure 12(a) and (c)). Kubásek et al.,
27
Siti Nur Hazwani Mohamadet al.,
28
and Bakhsheshi-Rad et al.
31
reported similar flake-like structures on corroded surfaces. Surface cracks were detected in the AZ31/2Al2O3 composite (Figure 12(a) and (b)), indicating dehydration of the corrosion products and consequent differential shrinkage.
29
Through these cracks, the SBF solution comes into direct contact with the matrix material, accelerating corrosion.
30
The severity of the crack and pit formation is reduced with further Sn particle addition. The EDS examination results of the composites AZ31/2Al2O3 and AZ31–6Sn/2Al2O3 (Figures 13 and 14) indicate the presence of Mg, O, and a minor atomic fraction of Ca and P, indicating the formation of a hydroxyapatite layer.28,31 Hydroxyapatite is formed when phosphate and calcium ions combine with OH– in a solution.
31
According to the EDS result, the higher oxygen concentration certainly suggests composites’ surface layers include Mg(OH)2 and MgO.28,30 The degradation of AZ31 composites in the SBF followed the following equation
32
: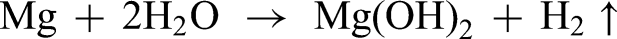

Photographs of the specimens immersed in simulated body fluid (SBF).
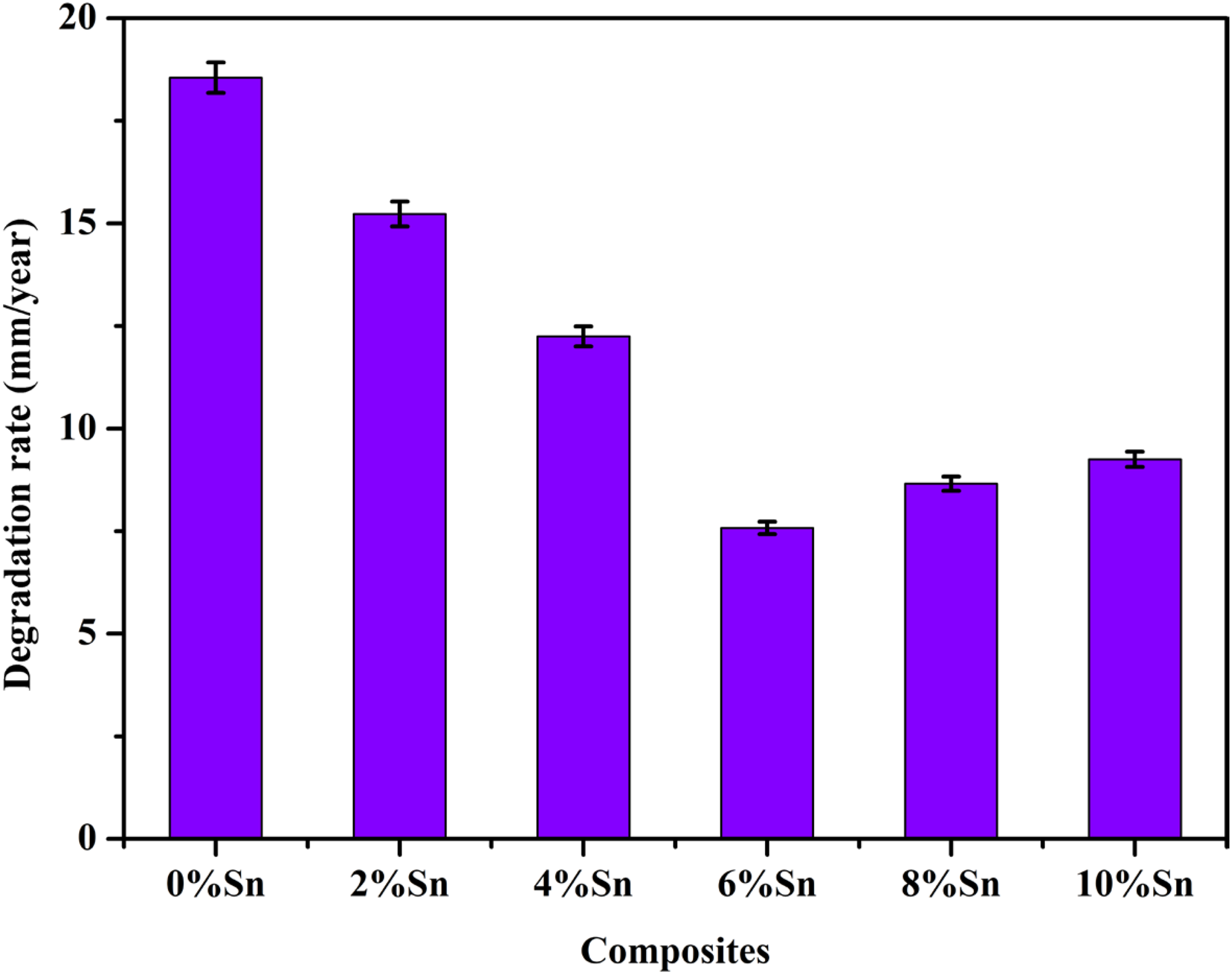
Degradation rate after 72 h immersion in simulated body fluid (SBF) solution.

Corrosion morphologies of (a, b) the AZ31/2Al2O3 composite and (c, d) the AZ31-6Sn/2Al2O3 composite after 72 h of immersion in simulated body fluid (SBF).
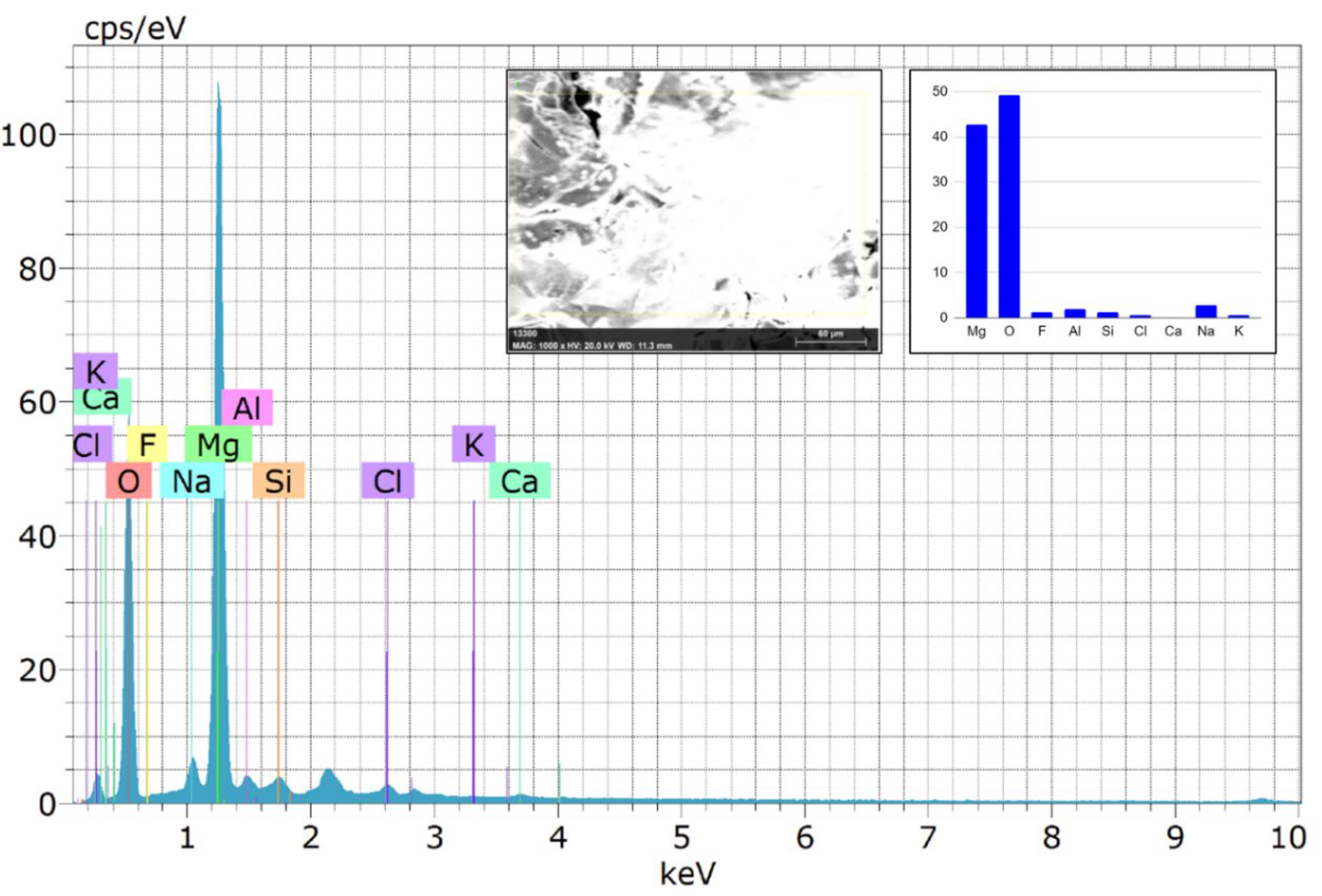
Energy-dispersive X-ray spectroscopy (EDS) spectrum of the AZ31/2Al2O3 composite.
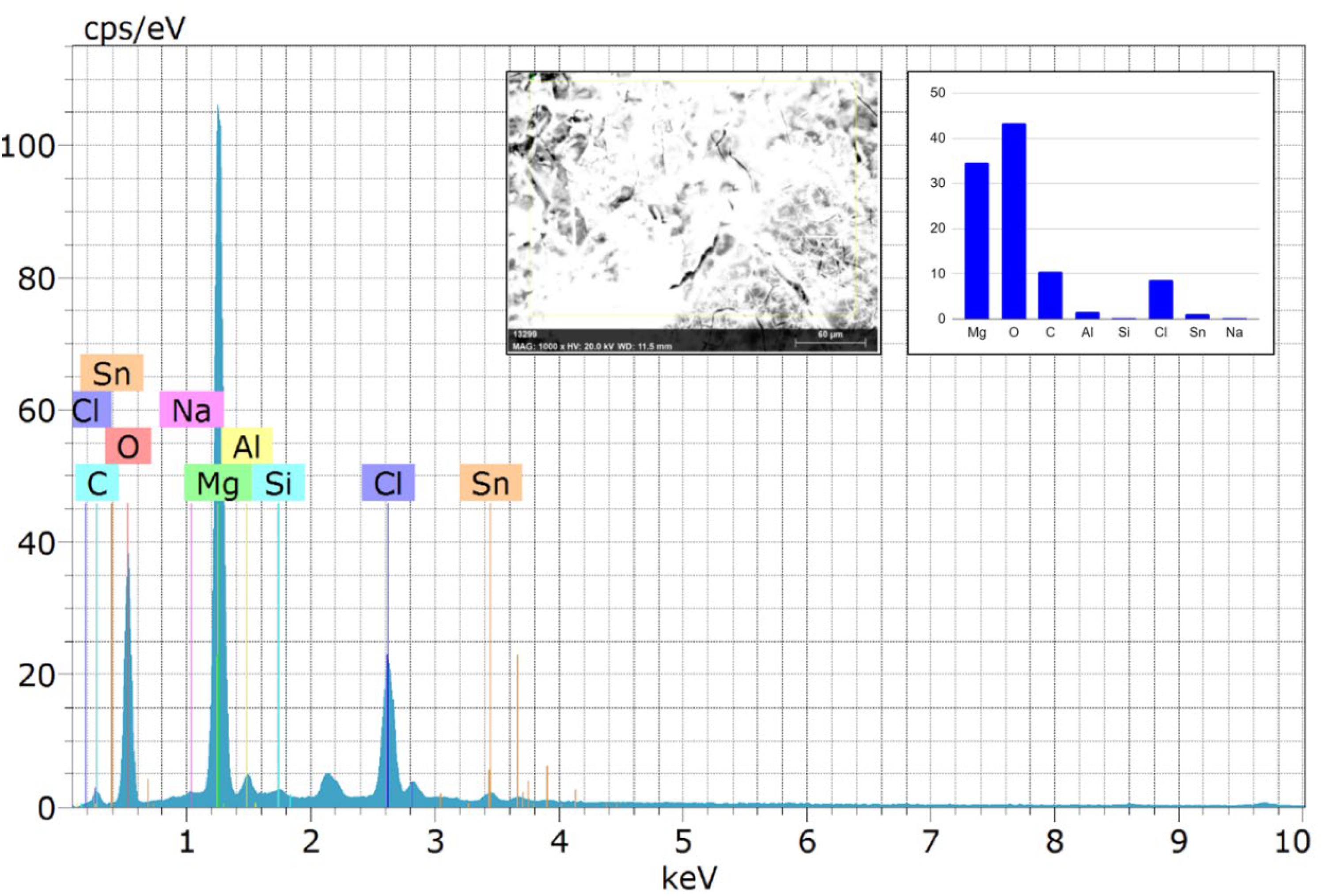
Energy-dispersive X-ray spectroscopy (EDS) spectrum of the AZ31-6Sn/2Al2O3 composite.
On the other hand, the existence of chlorine ions (Cl−) in the SBF solution instigated Mg(OH)2 to convert into the soluble magnesium chloride (MgCl2). 31 The presence of Cl in the EDS reports of the composites (Figures 13 and 14) confirms the formation of MgCl2. Microstructure analyses showed that the amount of secondary phase Mg2Sn increased as the Sn concentration increased. After 72 h of immersion, composites with more than 6% Sn degraded more rapidly. This could be attributed to an increase in the second-phase Mg2Sn, which increases corrosion. 33
Conclusion
In this current work, AZ31/2Al2O3 and AZ31-Sn/2Al2O3 composites were manufactured by the stir casting process. The influence of Sn reinforcement addition on wear behaviour and in vitro degradation rate was evaluated. The following insights are drawn from the study:
As Sn reinforcement content increases, the wear rate gets reduced due to the ceramic phase Mg2Sn acting as the resistance to wear at the contact surface. Due to the lack of Sn particles on the contact surface in the softer matrix phase, the wear rate for Sn particle-reinforced composites was lower under different applied loads and sliding distances compared to AZ31/2% Al2O3 composites. The presence of shallow abrasive grooves on the surface of an AZ31–10Sn/2Al2O3 composite subjected to 10 and 20 N normal loads was observed. In addition, it is observed that the formation of a stable tribo-layer on the worn surface. And the more visible layers are noted on the worn-out surface at higher loading conditions (30 and 40 N). Compared to AZ31/2Al2O3 composite, AZ31/2Al2O3 composites containing Sn particles exhibited a higher ability to generate more stable tribo-layers at higher applied loads, which protected the worn surface and reduced the wear rate. The wear resistance of composites is improved primarily by the behaviour of the tribo-layer in the wear process. The AZ31 composite degradation rate (mm/year) is carried out for 72 h in SBF. After 72 h of immersion, the degradation rate of the AZ31/2Al2O3 composite was 18.5 mm/year, whereas it decreased by 17.8% for the AZ31–2Sn/2Al2O3 composite. The degradation rate is reduced when the Sn content of the AZ31/2Al2O3 composite is increased to 6 wt% and then increased with additional Sn inclusion. It is observed that when the amount of Sn particles increases up to 6 wt%, the severity of the degradation decreases. After 72 h of immersion in SBF, the AZ31–6Sn/2Al2O3 composite degraded at an incredibly low rate (7.6 mm/year).
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
