Abstract
There is an underutilization of smoking cessation treatment among patients with chronic conditions, which indicates a need for new strategies to engage them. Web-based smoking cessation decision-making tools can be beneficial. This study assessed the effectiveness of the Pare de Fumar Conosco software at increasing engagement to smoking cessation counseling groups among Brazilian smokers with chronic conditions. Clinical trial participants were randomized to Pare de Fumar Conosco or standard of care. Engagement in the smoking cessation counseling group was the primary outcome. Secondary outcomes included motivation level, cessation rate, completion, and treatment adherence. The t-tests and Chi-square analyzed baseline differences between groups and the Poisson models, the effects of the intervention. Engagement rates were higher in the Pare de Fumar Conosco (IR=2.22; 95% CI: 1.06 - 4.63) concerning the standard of care. Pare de Fumar Conosco group was more adherent to treatment over time (6.4% versus 4.2% in the final week). Cessation rate was 26.3% in standard care and 23.1% in Pare de Fumar Conosco. Both interventions maintained significant motivation levels to quit smoking. The Pare de Fumar Conosco intervention effectively increases the engagement in the smoking cessation counseling groups. There is a need to improve the smoking cessation rate.
Introduction
Tobacco use remains the leading cause of reversible morbimortality worldwide. 1 By 2030, tobacco will be responsible for approximately 8000 deaths per year. Of these, more than 80% will be in low- and middle-income countries. 2 Offering smoking cessation treatment in various modalities is one-actions indicated to minimize this projected impact. 3
In Brazil, population-based surveys show a decrease in the prevalence of tobacco use in the past 30 years. 4 However, Brazil still has more than 20 million tobacco users and is listed among the ten countries with the highest number of smokers.5,6 Brazilian clinical practice guidelines recommend the combination of counseling and pharmacotherapy for smoking cessation. 7 As part of Brazil’s universal healthcare system, group counseling is offered to all smokers at no cost. Group counseling covers coping methods, social skills training, contingency management, self-control, and cognitive-behavioral strategies.8,9 The Brazilian universal health care system also covers pharmacotherapy (nicotine replacement therapy, bupropion, and varenicline). However, the uptake of these resources remains low. 7 This underutilization of smoking cessation treatment is even more evident among patients with chronic conditions (e.g. hypertension, diabetes, kidney disease, and obesity). Studies demonstrate the limited engagement and low cessation rate in smokers with multiple conditions.10,11
There is an urgent need to develop new strategies to engage smokers with chronic conditions in smoking cessation treatment. 12 Web-based interventions are a promising strategy for smoking cessation, 13 particularly in developing countries. 14 There are several types of web-based smoking cessation interventions available (e.g. standardized or tailored information, interactive features, multimedia resources, etc.).15–20 Web-based interventions stimulate the desire to quit smoking and guide smokers to achieve this goal. 21 Moreover, these interventions are a credible source of information and mitigate barriers to smoking cessation. 22 Previous studies showed that web-based smoking cessation interventions increase motivation to quit and pharmacotherapy use, which caused remarkable abstinence rates.21,22,23,24
Pare de Fumar Conosco, a web-based decision-making tool for smoking cessation, has significantly increased interest in pharmacotherapy and counseling among Brazilian smokers with chronic conditions. 25 However, additional testing as a formal randomized controlled trial is needed to test its effectiveness. This study aimed to assess the effectiveness of Pare de Fumar Conosco at increasing engagement to smoking cessation counseling groups among Brazilian smokers with chronic conditions.
Methods
Trial design
The study hypothesis was: Among Brazilian smokers with chronic conditions, the Pare de Fumar Conosco software increases engagement in the smoking cessation counseling groups. This engagement in cessation demonstrated by attendance to the first in-person smoking cessation counseling group was the primary outcome. Motivation level, cessation rate, completion, and treatment adherence were secondary outcomes. Study procedures were approved and monitored by the Research Ethics Committee of the Hospital University of UFJF (24883413.5.0000.5133). The study' report follows the recommendation of the CONSORT Declaration 26.
Participants
Brazilian smokers with chronic conditions were recruited in the waiting rooms of two secondary care clinics by a research assistant. Eligible participants were ≥18 years of age, self-identified as current smokers, were treated at healthcare centers for chronic conditions, and were willing to sign an informed consent form. We obtained written informed consent was from each participant and did not reimburse them for their participation in the study.
Interventions
We submitted participants in the intervention group to the Pare de Fumar Conosco software and the control group to the standard of care. Both interventions and group counseling for smoking cessation are specified below.
Pare de Fumar Conosco
The one-arm feasibility study of the Pare de Fumar Conosco software describes in detail this intervention.
25
In summary, a panel of Brazilian healthcare professionals adapted Pare de Fumar Conosco from an existing web-based decision-making tool for smoking cessation developed in English and Spanish for use among Latinos in the United States
21
and Mexico.27–29 They adapted the content to reflect the resources, perceptions, and sociocultural characteristics of Brazilian smokers. Pare de Fumar Conosco follows the Social Cognitive Theory, which comprises motivational messages, behavioral change support, and smoking cessation pharmacotherapy use. The decision-making tool included videos featuring a professional actor guiding smokers through the program, as well as a testimonial from an ex-smoker. The software also collected sociodemographic information and smoking behaviors. It used this information to develop a personalized quit plan: pharmacotherapy selection and an invitation to a counseling group. Finally, the software asked participants to set up a smoking quit date. The engagement in cessation demonstrated by the attendance to the first in-person smoking cessation counseling group is the main functionality of the software. The use of Pare de Fumar Conosco took approximately 15–25 minutes per participant to complete. Figure 1 shows some aspects of Pare de Fumar Conosco. Pare de Fumar Conosco software in its original language (Portuguese).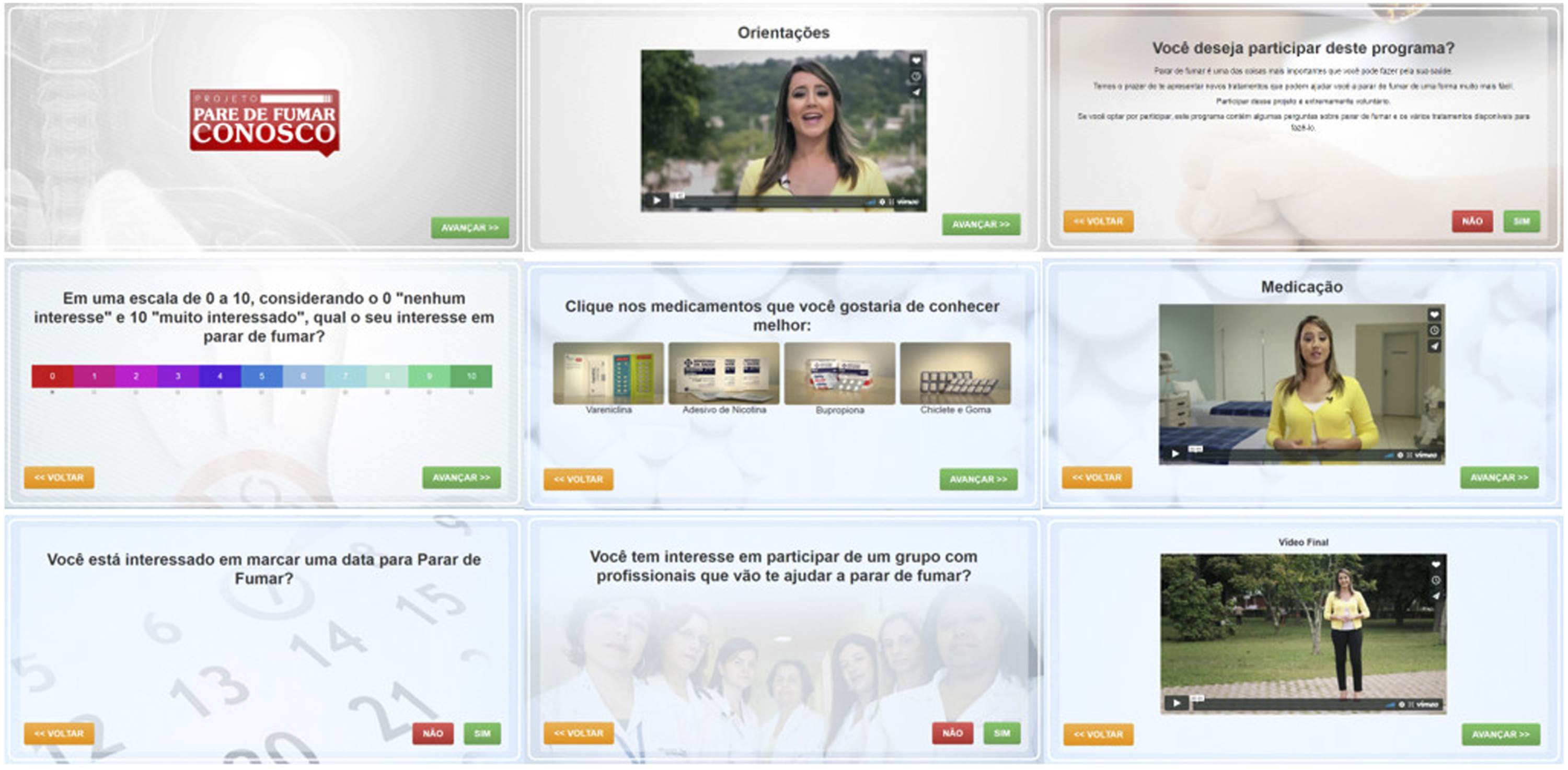
Standard of care
Following Brazil’s National Cancer Institute (INCA, based on its acronym in Portuguese), standard of care, basic approach for smoking cessation treatment, was substantiated by the 5 A’s approach: Ask, Advise, Assess, Assist, and Arrange. 30 The procedures consisted of asking participants about their smoking behaviors, advising participants to quit smoking, assessing their willingness to make a quit attempt, assisting participants with a quit date selection, and inviting them to participate in the smoking cessation counseling groups. These procedures took approximately 15-25 minutes per participant to complete. Lastly, we gave participants printed educational material with motivational content and the contact information for the counseling groups.
Smoking cessation counseling group
We invited all participants to the in-person group-based counseling program. This program consisted of intense cognitive-behavioral therapy, which included behavioral skills training and pharmacological intervention if indicated. The program was structured in four 90-minute weekly sessions. Subsequently, these meetings were held every fifteen days until the conclusion of the twelve-week follow-up. The smoking cessation counselor conducting the sessions was not aware of the participants’ allocation group.
Outcomes
Research staff conducted an in-person assessment at baseline and follow-up (Week 12). The baseline survey collected sociodemographic information (e.g. age, gender, and educational level). Smoking-related variables included two questions from the Fagerström Test for Nicotine Dependence 31 (number of cigarettes smoked per day and time of smoking the first cigarette). Moreover, we assessed if participants made a previous quit attempt. The Patient Health Questionnaire (PHQ-2) 32 screened depression.
The engagement rate in cessation (percentage of participants who attended the first in-person smoking cessation counseling group) was the primary outcome. As part of the counseling program, we assessed: motivation level, evaluated by an analog scale of 0 to 10 (10 being the maximum motivation level), self-reported smoking abstinence (7-day point prevalence abstinence), completion rate (percentage of participants who attended all the sessions in the counseling program), and treatment adherence (percentage of participants who attended in each session in the counseling program). We analyzed smoking abstinence, completion rate, and treatment adherence using intention to treat analysis.
Participants in the intervention group were contacted eight weeks after utilization of the software to assess satisfaction. This over-the-phone survey consisted of questions about the degree of satisfaction with the software, its most valuable aspects, and suggestions for improvement.
Sample size
Data from a study conducted in one of the secondary healthcare centers that reported 11.9% of the patients as current smokers 33 supported the sample size calculation performed in the OpenEpi software. The primary outcome was the engagement in the smoking cessation counseling group. We expected a 25% 34 and 10% (assumed effect size) engagement rate in the Pare de Fumar Conosco group and standard of care group, respectively. The effect size in terms of relative risk was 2.5 in favor of the intervention group. We set the significance level and statistical power at 5% and 80%, respectively. As a result, the estimated sample size was 100 subjects per group. With an assumed loss of 10%, the adequate size of eligible participants should total at least 110 subjects per group, or 220 in total.
Randomization
After the baseline assessment completion, participants were randomized to one of two conditions: (1) Pare de Fumar Conosco or (2) standard of care. For allocation of the participants, the statistical program R created a computer-generated list of random numbers. This allocation was open and strictly followed the random number list to avoid cross-contamination between groups. Researchers and participants were not blinded. Figure 2 shows the summary of the entire clinical trial design. Clinical trial design.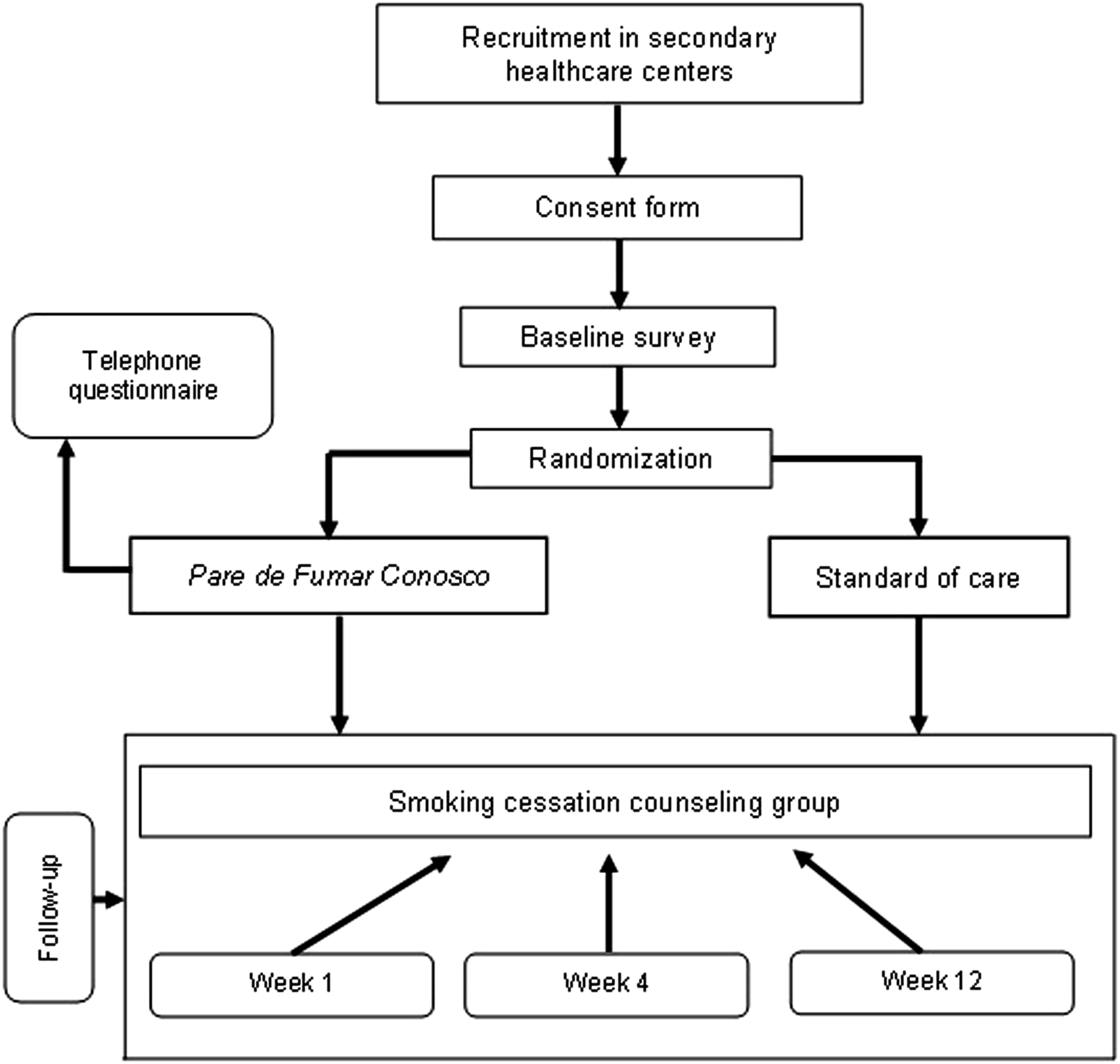
We continuously trained the research team to standardize both interventions – standard of care and Pare de Fumar Conosco. Standard of care is part of the regular training of health professionals, with at least two training per year. Training on the implementation of Pare de Fumar Conosco followed the same periodicity during the study time.
Statistical methods
Based upon the data stored in REDCap 35 (Research Electronic Data Capture), we performed descriptive and inferential statistical analyses with the statistical package R version 3.4.4 and STATA 13.0. The confidence level adopted was 95%, and we interpreted the p-values and confidence intervals following the literature recommendation.36,37
We reported the categorical variables in absolute (n) and relative (%) frequencies and continuous variables in mean, median, and standard deviation. Differences in baseline between groups were analyzed using Chi-square and t-tests.
To analyze the effects of the intervention, we used regression models appropriate to the outcome type and always adjusted or stratified for potentially confounding variables. In estimating treatment effects, we used Generalized Linear Models, 38 a class of statistical models that allows for highly flexible probability distributions, according to the type of response variable. In the case of dichotomous variables (e.g. engagement in the smoking cessation counseling group), we used Poisson models with a log-link function to provide the incidence ratio as a measure of association.
Results
During the recruitment process, 281 participants were eligible for the study. However, it was not possible to contact 38 individuals. Hence, the final sample comprised was 243 participants, with 124 (51%) in the Pare de Fumar Conosco group (intervention group) and 119 (49%) in the standard of care (control group) (Figure 3). Participant recruitment process and follow-up.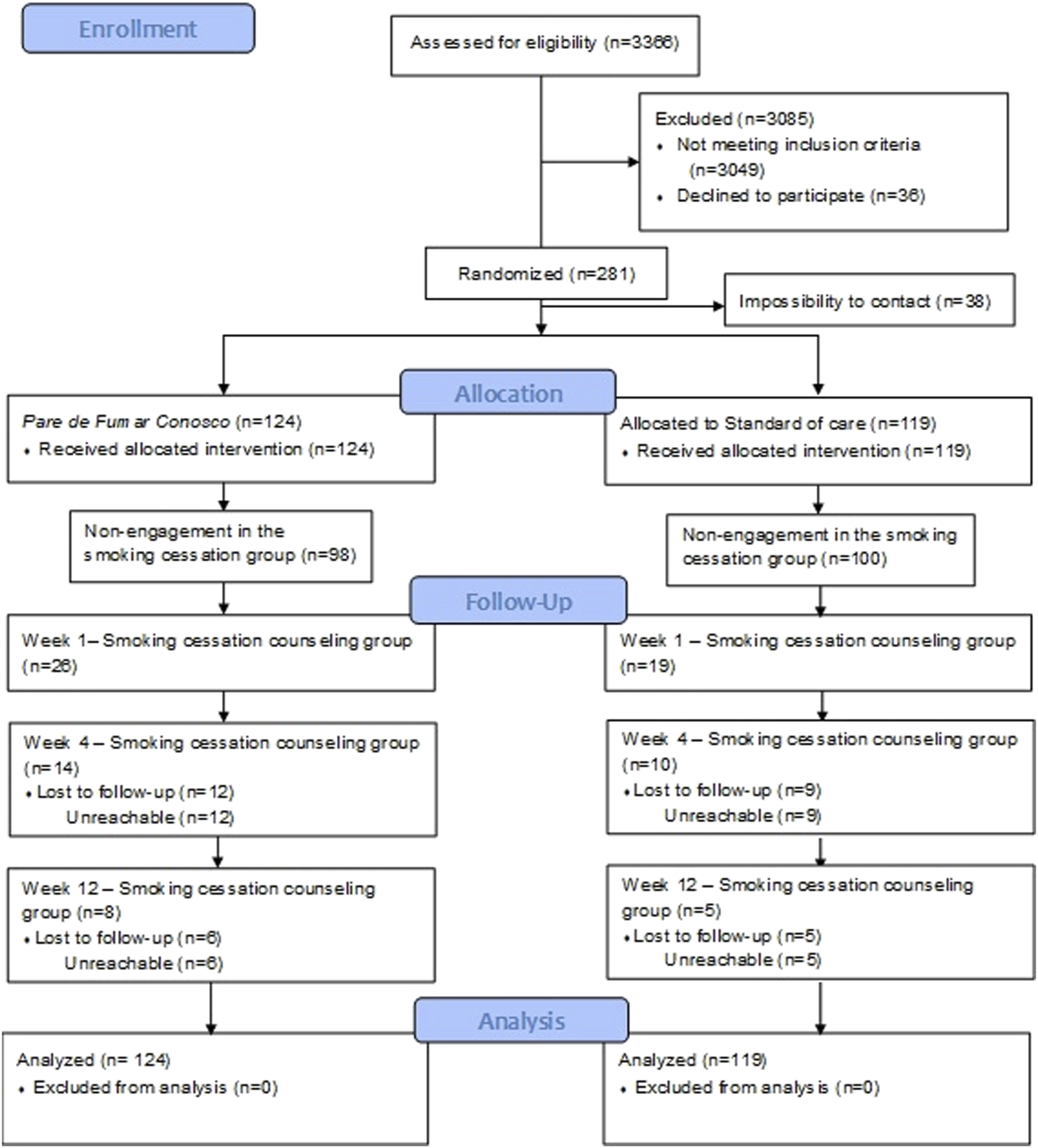
Participants were primarily middle-aged women. Most of them were daily smokers, had screened negative for depression, and had incomplete or no elementary education. Almost half of the participants (44%) consumed a maximum of ten cigarettes a day, and 41% smoked their first cigarette five minutes after waking up.
Baseline characteristics of study participants.
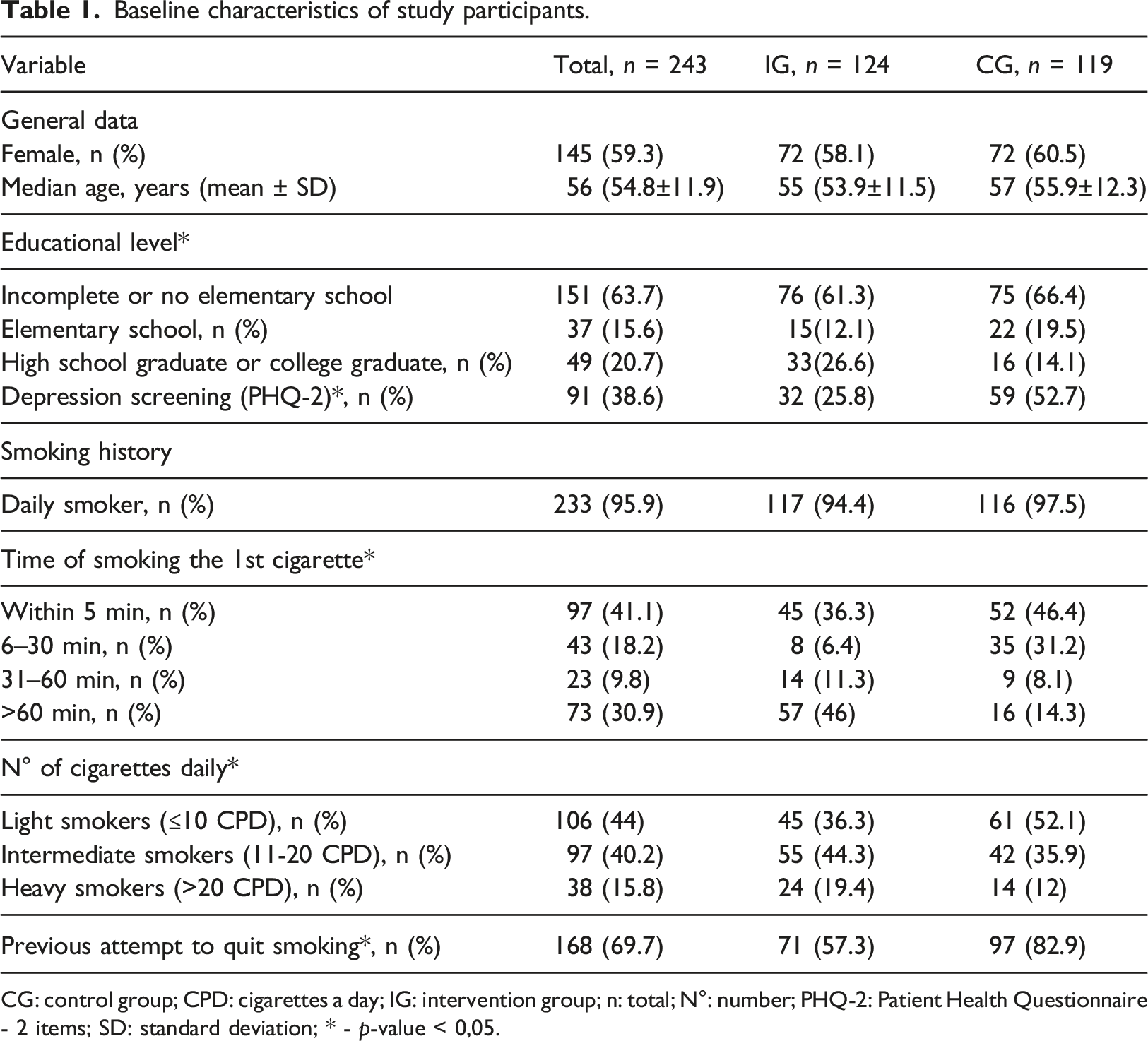
CG: control group; CPD: cigarettes a day; IG: intervention group; n: total; N°: number; PHQ-2: Patient Health Questionnaire - 2 items; SD: standard deviation; * - p-value < 0,05.
Comparison of outcomes between study groups.
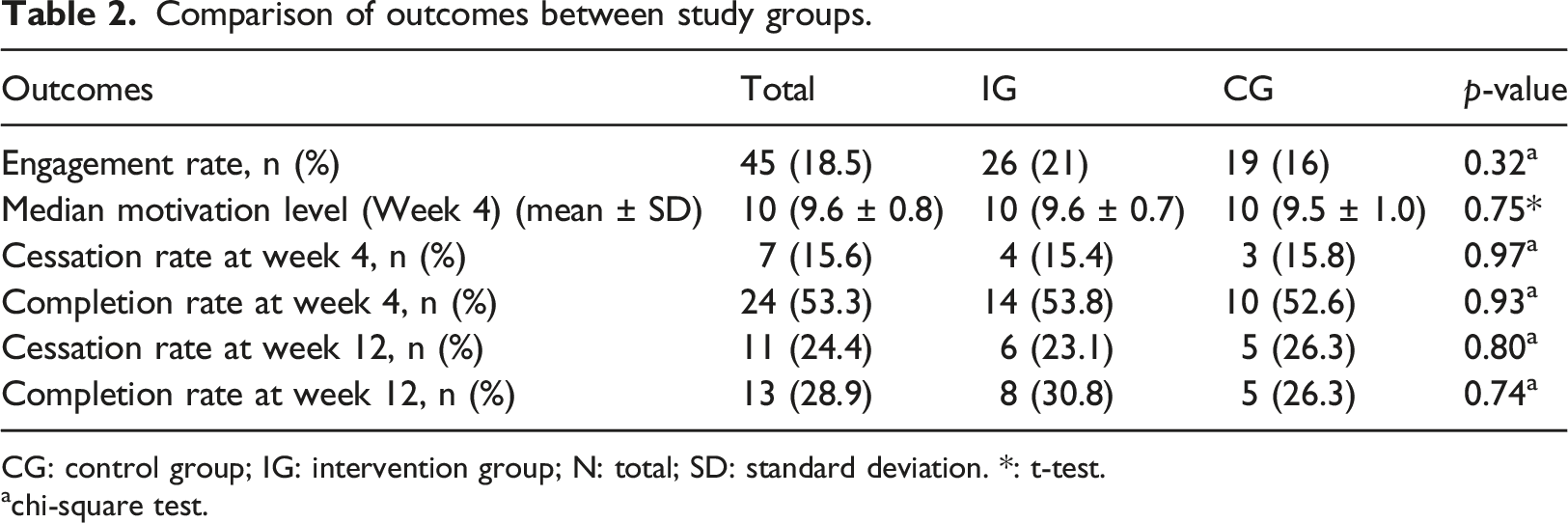
CG: control group; IG: intervention group; N: total; SD: standard deviation. *: t-test.
achi-square test.
At Week 4, eleven participants used smoking cessation medications (43% in the intervention group and 50% in the control group). Sessions adherence of the in-person smoking cessation counseling group decreased over time (Figure 4). At week 4, adherence treatment was 53% (24/45). At Week 12, 6% of participants in the intervention group attended the counseling group session, compared to 4% in the control group. Treatment adherence (intention to treat).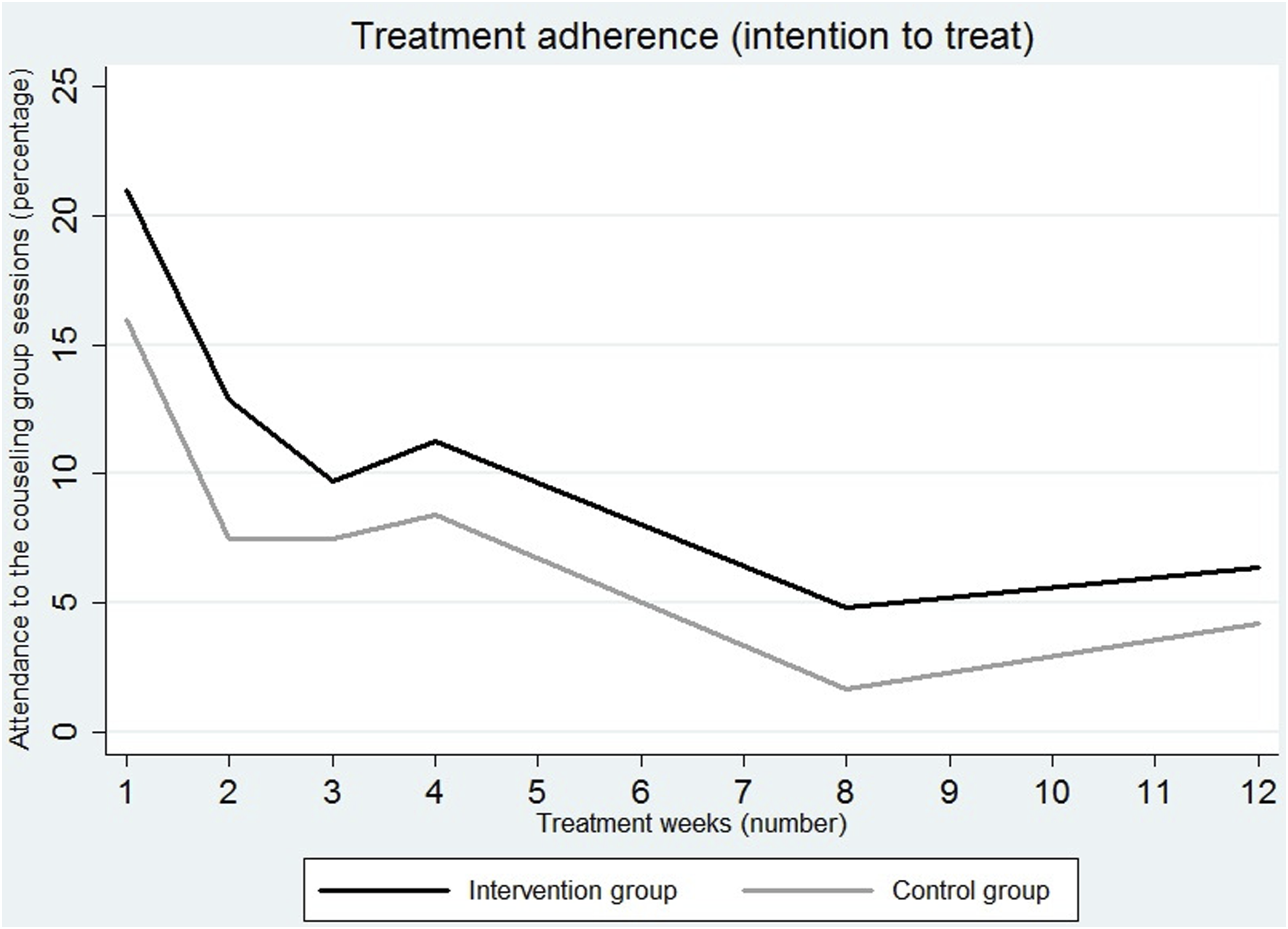
Multivariate Poisson regression models for primary and secondary outcomes.
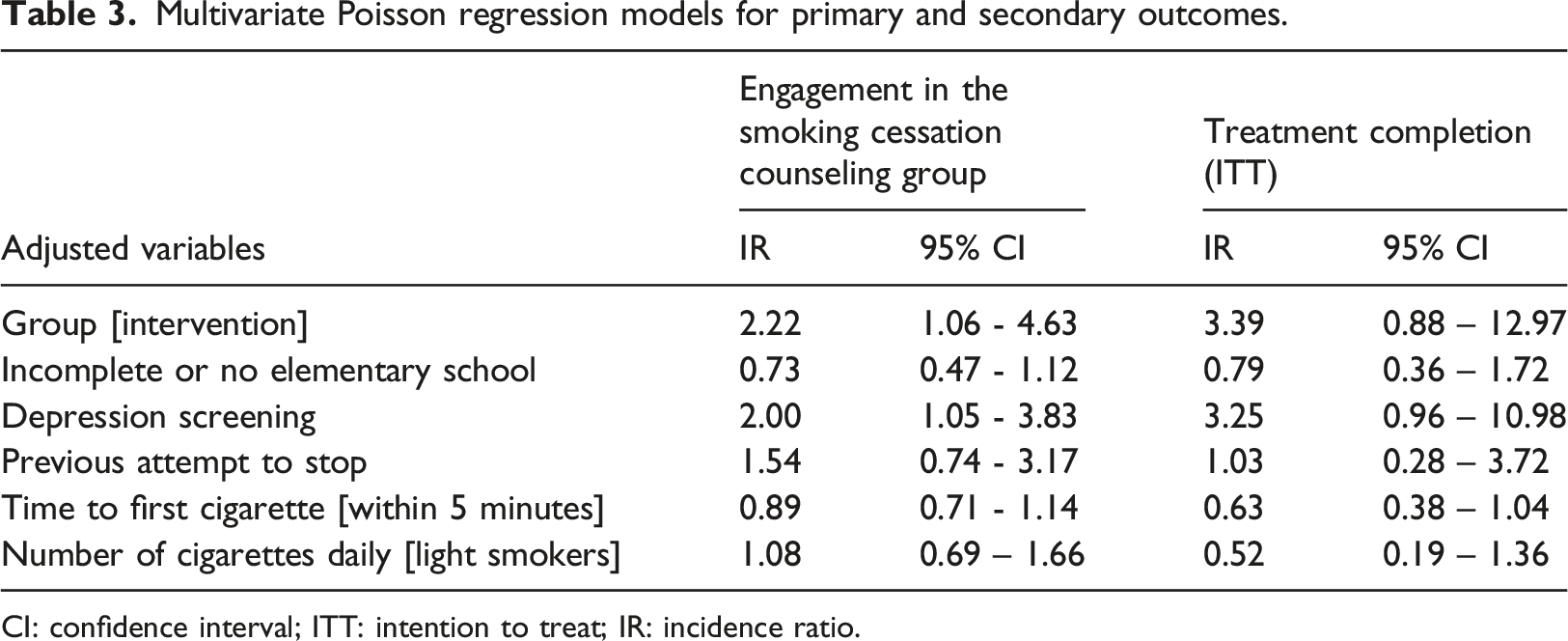
CI: confidence interval; ITT: intention to treat; IR: incidence ratio.
Eighteen participants in the intervention group completed the over-the-phone survey at Week 8. Almost all participants (94.4%) reported high satisfaction with the technology and the setting of utilization (e.g. waiting areas). The most valuable aspects of the interventions were information about smoking cessation medications (27%), testimonials (20%), interest in smoking cessation (20%), the importance of quitting smoking (13.3%), and technology (20%). Two participants suggested enhancing the psychological aspects of smoking cessation and developing more innovative technological features. A total of 83% of participants reported that they cut down the number of cigarettes in the past seven days, with a median (mean ± standard deviation) of consuming six (9.2 ± 6.3) cigarettes per day.
Discussion
Participants using Pare de Fumar Conosco reported a similar engagement rate (21%) to smokers in Mexico using a similar web-based smoking cessation decision-making tool (19.7%). 27 The cessation rate described in this study (4.5%, 11/243) is consistent with the rate found in population-based surveys.39-41 However, this result was lower than that obtained by the web-based smoking cessation decision-making tools in the United States 21 and Mexico 27 (11.8% in the United States and 19.1% in Mexico).
A systematic review and network meta-analysis showed that cessation, overall, is higher among participants who use smoking cessation electronic tools compared to participants who do not receive an intervention or use generic self-help resources (RR=1.14, 95% CI 1.07 to 1.22). 42 The same effect occurs when comparing electronic tools with computer-generated and tailored printed materials concerning no intervention, standard of care, or untailored printed self-help materials, with or without concurrent co-interventions (RR=1.51. 95% CI 1.15 to 1.97). 42 In the Social-Local-Mobile study, the combination of psychopharmacological therapy with a digital mobile health solution favored smoking cessation by per-protocol analysis (adjusted OR = 3.45, p = 0.01) and by intention-to-treat analysis (adjusted OR = 3.13, p = 0.002). 43
In our study, cessation rates in the control group were higher than in the Pare de Fumar Conosco group, especially in Week 12. One aspect that could explain this difference is that of the total number of individuals in the intervention group who had quit smoking in Week 4, 50% abandoned the treatment before the end of the 12 weeks. Cessation rates may differ depending on the length of treatment. 44 Studies reported higher success rates in early periods than in later treatment periods.45–47 In pooled analyzes of the effects of the web- and computer-based smoking cessation programs, short-term (3 months), midterm (6–10 months), and long-term (12 months) follow-up resulted in a decline in smoking cessation rates over time of treatment. 48 It is worth mentioning the need to improve the effectiveness of the cessation of a decision-support tool. Web-based interventions have room for improvement.49,50 Its combination with different types of interventions can enhance smoking cessation.42,51-53
Even with the tobacco cessation treatment (combination of counseling and pharmacotherapy)54–59 available at no cost, treatment adherence decreased over time, especially in the control group. This finding corroborates with those described in the literature. Similar to our study, in Brazilian patients with chronic conditions, the adherence rate to the 4-week treatment of the smoking cessation counseling group was 57%. 60 Despite lack of standardized research in adherence to smoking treatment, the counseling sessions adherence is positively associated with cessation.55,61-63
Audio-visual communication can eliminate the barrier of low literacy.64,65 Accessibility via the Internet or internal networks permits a single production to be used several times with a minimal recurring cost. 66 Unlike standardized health, electronic tools have a personalized focus on the choices and results of users, guiding them to decision-making according to their preferences, values, and beliefs. 67 Multimedia interventions can standardize counseling for decision-making, saving the time healthcare providers spending to educating their patients. 68 The reduction of health system costs and the potential informed decision support has on patient health are policymakers' objectives for improving health, equity, and patient satisfaction. 69 The SmokeFreeBrain project, funded by the European Commission H2020 program, 70 analyzed the success rate of various smoking cessation interventions in terms of health economics. 71
The opportunity to help smokers, who mostly live in low- and middle-income countries, may lie within their public health systems. 72 Incorporating smoking cessation into the assistance programs of health centers facilitates the successful integration of approaches. 72 Undoubtedly, technology-enabled healthcare has a high potential to help improve outcomes in key-risk patients. 73 Computer-tailored interventions have proven to be effective in smoking cessation.74–76 By achieving an increase in cessation rates, these approaches can contribute to reducing the incidence of diseases directly associated with smoking and those resulting from exposure to secondhand smoke. 77
Pare de Fumar Conosco should be linked, in the future, with counseling resources such as telephone calls and or text messages. Text messaging is a powerful resource in health education. 78 There is evidence that the potential for text messages to deliver smoking cessation treatment may be even more among hard-to-reach and socioeconomically disadvantaged populations, 79 like the one assessed in this study. Cruvinel et al. showed the feasibility of a hybrid telephone and text messaging counseling intervention among post-discharge hospitalized smokers in Brazil. 80 This hybrid intervention was well accepted by participants and resulted in noteworthy retention and cessation rates at 12 weeks.
The results can be generalized with caveats. We cannot eliminate differences in the implementation of the intervention. Standard of care and smoking cessation counseling groups follow Brazilian guidelines. Differences between what guidelines recommend and what healthcare centers implement can occur. Moreover, groups were not well balanced at baseline. Important variables such as depression screening, educational level, time to first cigarette, previous attempts to quit smoking, and the number of cigarettes daily were significantly different between groups. The sample size and type of block randomization generated by a statistical program can explain these differences. There is the possibility of favorable or unfavorable results because of the unbalanced allocation. 81 Randomization did not eliminate but reduced biases in potential. By being performed by a list generated on the computer, the investigator, even unconsciously, would not influence the participants’ allocation in the treatment groups. Randomization enabled a valid static inference. 81
Some factors may still have biased the study’s outcomes after multivariate adjustments. The lower education level (incomplete or no elementary school) may have caused difficulty understanding the information provided related to smoking cessation.82,83 Smoking within the first five minutes after waking up, an indicator of high nicotine dependence, may interfere with the decision-making process and treatment uptake.84,85
Depression could negatively affect the study results. This condition is related to heavier smoking, relapses, and difficulties for smoking cessation. 86 Pare de Fumar Conosco has obtained a level of user motivation, which we reiterated due to the engagement in the smoking cessation counseling group being two times higher for the group intervention.
This study had several limitations. First, we were not able to contact 13.5% (38/281) of eligible subjects. There is a need to further evaluate the barriers and facilitators in clinic settings to reach and recruit Brazilian smokers into cessation studies. Second, we did not assess nicotine dependence. So it remains unknown whether these results vary by nicotine dependence level. Third, follow-up was limited to 12 Weeks. Fourth, we did not use biomarkers to verify smoking status. Fifth, at the end of the treatment, we did not measure the motivation level. Sixth, we did not present the results of the Poisson model for smoking cessation in the 12th week. The number of participants who reached this outcome was small, making meta-regression difficult. Seventh, this study was conducted at two clinics with wireless internet capacity; many healthcare clinics in Brazil still lack high-speed internet connections, especially in rural areas.
Conclusions
The Pare de Fumar Conosco intervention effectively increases the engagement in the smoking cessation counseling groups among Brazilian smokers with chronic conditions. However, more research is needed to find innovative ways to enhance adherence to smoking cessation treatment and improve the cessation rate. Future studies should evaluate the integration of Pare de Fumar Conosco as part of routine care and its cost-effectiveness.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Foundation of the State of Minas Gerais [grant number FAPEMIG - CBB APQ 03603-13]; the Fogarty International Center at the National Institutes of Health [grant number R03TW008723]; and the Mineiro Researcher Program [grant number PPM-00129-18].
Trial registry
Brazil’s Registry of Clinical Trials; number RBR-6tjmw3 (https://ensaiosclinicos.gov.br/rg/RBR-6tjmw3).
Data accessibility
The clinical trial participants’ data used to support this study’s findings are available from the corresponding author upon request.
