Abstract

Clinical scenario
A paramedic crew attends a patient who has fallen from a third-storey balcony. No other pre-hospital teams are available to assist. The patient has a deformed femur and apparent pelvic asymmetry. He is alert with a heart rate of 114 bpm and blood pressure of 115/80 mmHg. Adequate analgesia is achieved with inhaled methoxyflurane to facilitate the application of a pelvic binder and Kendrick Traction Device. The crew wish to give tranexamic acid (TXA) but are unable to gain intravenous (IV) access. The patient declines intraosseous (IO) access and is conveyed to the nearest major trauma centre where a cannula is placed under ultrasound guidance and IV TXA is administered. The crew subsequently discuss the case with colleagues, who inform them that the latest update to the JRCALC guidelines 1 allows intramuscular (IM) administration of TXA as an alternative to IV/IO. They wonder whether the patient may have benefitted from this.
Three-part question
In [adults at risk of significant haemorrhage], does [pre-hospital IM administration of TXA vs IV administration in hospital] improve [morbidity and mortality]?
Search strategy
On 6th November 2022, the MEDLINE, Embase and Cochrane Library databases were searched.
Title and abstract: Tranexamic AND (intramuscular OR prehospital OR pre-hospital).
The search was limited to papers published after 2010.
Search outcome
Results were identified for title and abstract screening (MEDLINE: 154, Embase: 570 and Cochrane Library: 69).
No systematic review, clinical trial or observational study was found comparing the effectiveness of IM TXA vs IV TXA or IM TXA vs placebo. Three systematic reviews examining the effect of pre-hospital IV TXA were identified. The primary studies included in these reviews overlapped and the findings were similar. The review with the most recent evidence and largest number of patients is included. One further observational study, published after that systematic review, was identified and included. One observational study examining the effect of pre-hospital TXA on the timing of administration was identified. One pharmacokinetic study of IM TXA in human trauma patients was identified. The included studies are summarised in Table 1.
Comment
A subgroup analysis of the CRASH-2 trial 6 showed that the beneficial effect of TXA in reducing deaths due to bleeding was only seen when the first dose was administered within 3 h of injury and the effect was probably greater still when it was given within 1 h. Li et al. 7 performed a subgroup analysis of the STAAMP trial 8 and found that the survival of shocked trauma patients was significantly improved if pre-hospital IV TXA was administered within one hour of injury but not if it was delayed. Finally, Gayet-Ageron et al. 9 combined the results of the CRASH-2 trial and the WOMAN trial, which examined the effect of TXA on survival in post-partum haemorrhage. They found ‘Immediate [IV] TXA improved survival by more than 70% (OR 1·72, 95% CI 1·42–2·10; p < 0.0001). Thereafter, the survival benefit decreased by 10% for every 15 min of treatment delay until 3 h, after which there was no benefit.’
NICE guideline NG39 10 recommends ‘Use IV TXA as soon as possible in patients with major trauma and active or suspected active bleeding. Do not use IV TXA more than 3 h after injury in patients with major trauma unless there is evidence of hyperfibrinolysis.’
Pre-hospital TXA availability appears to reduce the time to administration following injury and reduce mortality by a similar amount as would be expected from the CRASH-2 data, which is based on patients in hospital. A further randomised placebo-controlled trial is currently underway with a target recruitment of 1316 patients at risk of traumatic coagulopathy based on a Coagulopathy in Severe Trauma score ≥3. 11 Like the STAAMP trial, it seeks to compare the effectiveness of IV TXA initiated pre-hospital vs placebo.
IM TXA administration is associated with rapid absorption even in shocked trauma patients and has minimal side effects. IM TXA is now recognised as an alternative to IV/IO administration for patients at risk of haemorrhage in the most recent update to the JRCALC guidelines1 for pre-hospital practitioners.
Given its increased effectiveness, avenues should be explored to facilitate patients receiving TXA as soon as possible after injury. This has led some to propose that the potential use of IM autoinjectors should be investigated in certain settings.12,13 Even if IM TXA were less effective than TXA via the IV route, this may be more than compensated for by the potential benefit of earlier administration in situations where IV/IO access and administration may be delayed.
Clinical trials to investigate the potential for IM TXA to reduce morbidity and mortality could include a randomised placebo-controlled trial or a non-inferiority trial vs IV/IO administration.
Clinical bottom line
Current guidelines support early pre-hospital administration of TXA in trauma patients. If there is likely to be a significant delay in IV/IO administration, then patients may benefit from IM administration. This should be guided by clinician judgement.
Summary of included studies.
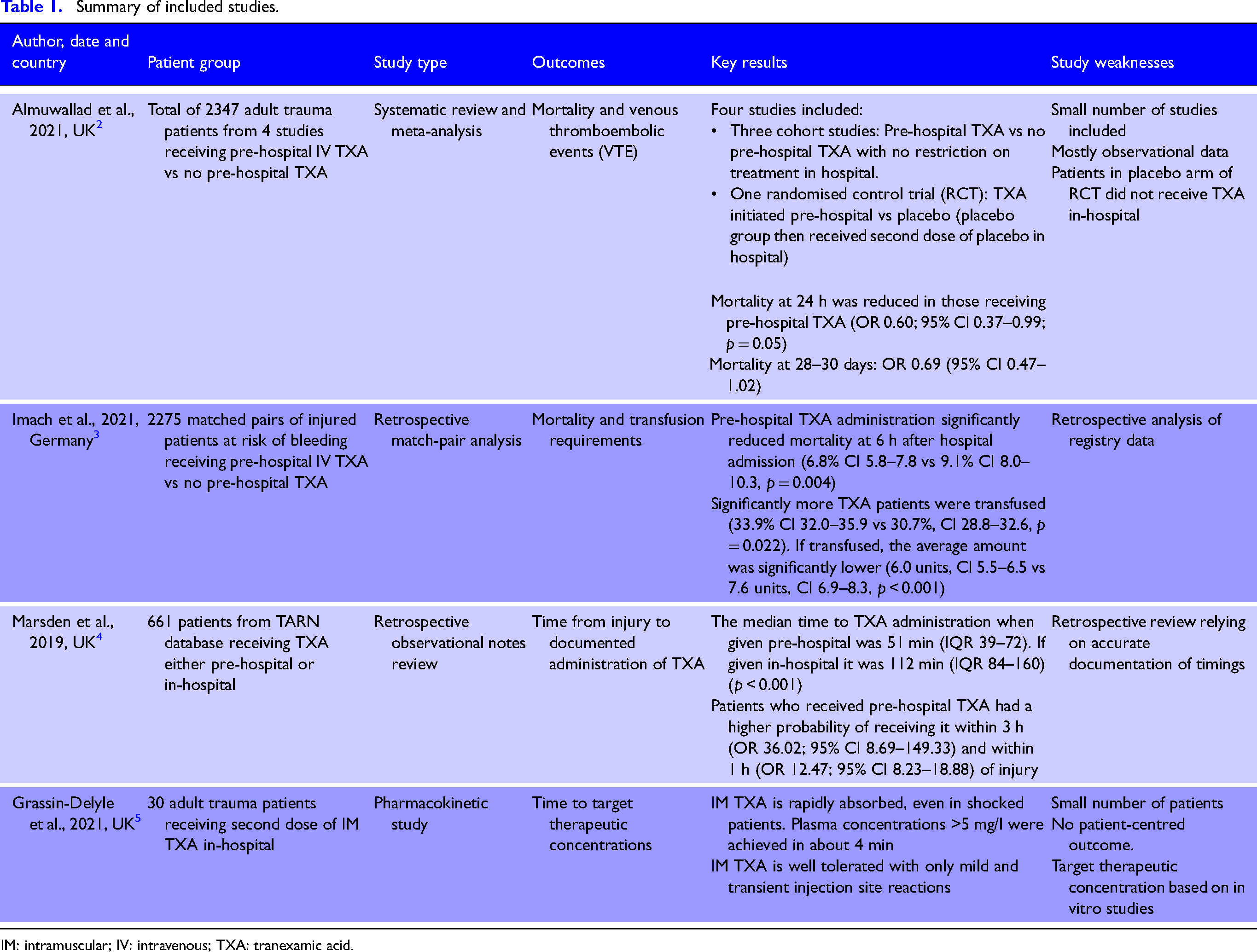
IM: intramuscular; IV: intravenous; TXA: tranexamic acid.
Footnotes
Contributorship
FW: Study conception; FW: Protocol development; FW and LW: Literature search and article review; FW and LW: Analysis; FW: Writing - draft; FW and LW: Writing - review and editing.
Acknowledgements
The authors wish to thank Jason Smith, Harvey Pynn and Phil Cowburn for offering their thoughts on early versions of this manuscript.
Correction (February 2024):
This article has been updated to remove the Abstract since its original publication
