Abstract
Background
The Clinical Randomization of an Antifibrinolytic in Significant Hemorrhage-2 (CRASH-2) trial proved that tranexamic acid (TXA) is a time-dependent drug, having a better outcome if given within 1-hour of injury. In order to test this theory, studies have been conducted to examine the effect of TXA in the pre-hospital setting. We conducted a systematic search and meta-analysis to evaluate the role of TXA administration in the civilian pre-hospital setting on patient outcomes.
Methods
Embase, Medline, CINAHL and Cochrane were searched for randomized control trials (RCTs), retrospective, and prospective studies that examined the effect of TXA on patients in the pre-hospital setting versus a control group. Outcome measures were overall mortality rate and thromboembolic events. Two authors extracted the data independently. To appraise the included studies, we used the NIH quality assessment tool for cohort and cross-sectional studies. Results are presented as Risk Ratio (RR), a random-effect model was implemented, and the I2 test was used to assess heterogeneity.
Results
The search identified 1886 papers, but only five retrospective studies met the inclusion/exclusion criteria and were selected for further analysis. A meta-analysis confirmed that TXA reduced the overall mortality rate (pooled risk ratio of 0.74 (95% CI 0.45, 1.25)) and thromboembolic events (risk ratio of 0.71 (95% CI 0.35, 1.44)).
Conclusion
The pooled effects for both outcome measures favour the administration of TXA in the pre-hospital setting, although none of the findings reported a significant effect. Our study highlights the need for additional high-quality evidence to validate the significance of these findings.
Level of evidence
Level III, therapeutic study.
Background
Recent statistics reported by the World Health Organization (WHO) indicate that trauma accounts for 9% of worldwide deaths, which is double the number of deaths resulting from HIV/AIDs, tuberculosis and malaria combined. 1 It is worth mentioning that around 65% of the trauma mortality is the result of secondary complications such as, acute coagulopathy and hemorrhagic shock as a result of excessive bleeding.2–4 Early treatment of these secondary complications is seen as a way to significantly reduce mortality and complications after trauma5,6 and many hospitals have implemented protocols which aim to reduce the effect of secondary complications resulting from excessive bleeding, one of which is the administration of Tranexamic acid (TXA).
TXA is a cost-effective antifibrinolytic agent proven to significantly decrease the mortality rates and complications associated with excessive bleeding if used within 3-hours of injury.7,8 Before the implementation of TXA as a treatment for traumatic bleeding, it was used on patients who were undergoing surgery or patients with heavy menstrual bleeding. Due to the similarity between how the human body reaches homeostasis when bleeding has occurred because of surgery or menstrual bleeding compared to when bleeding has occurred because of trauma, Roberts and colleagues initiated a large, double-blinded, randomized control trial (RCT) to test the efficacy of TXA on patients with traumatic bleeding. 7
The Clinical Randomization of an Antifibrinolytic in Significant Hemorrhage-2 (CRASH-2) trial is the largest study to date to examine the effect of TXA on approximately 20,000 trauma patients 7 in 40 countries and confirmed that TXA is associated with a decrease in overall mortality, without any increased side effects. Importantly the effect of TXA was time-dependent, being associated with better outcomes only if given early (<1 hour from injury). However, the limitations of the trial, which did not report injury severity of the recruited patients, have called into question the integrity and generalizability of these findings. Hence, following CRASH-2, to test the time-dependent effect of TXA, a considerable number of studies have been carried out to examine the safety and efficacy of TXA in the pre-hospital setting. Almost all of these studies followed the same inclusion/exclusion and intervention protocols as the CRASH-2 trial and investigated mortality rate and the risk of venous thromboembolism (VTE) as a side effect of TXA.
This study systematically reviewed published data regarding the efficacy of administering TXA in the pre-hospital setting, where the <3 hours from time of injury to TXA treatment can be observed. The aim is to examine whether early administration of TXA in the pre-hospital setting has a better outcome in terms of overall mortality rate and the risk of VTE compared to a control group of patients who did not receive TXA, in civilian populations.
Methods
Literature search
The search strategies recommended for the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) reporting guidelines were used. Two reviewers (AMA and ZA) systematically searched for studies from inception to 25 February 2020 that showed the efficacy of TXA in the pre-hospital setting in trauma patients, using four data bases: Embase, Medline, CINAHL and Cochrane. We used the PICO strategy to identify keywords and help with the search; for population we used the keywords “adult”, “in-hospital”, “prehospital”, “prehospital care”, “TBI”, “traumatic brain injury”, “trauma”, “severe trauma”, “coagulopathy”, “trauma-induced coagulopathy”, “bleeding”, “hemorrhagic shock”, “ambulance”, “hospital” all in title and abstract. The medical subject headings (MeSH) terms used were pre-hospital AND coagulopathy AND bleeding AND trauma AND traumatic brain injury. For intervention, we used TXA OR Tranexamic acid in the title and abstract and the MeSH term used was tranexamic acid. Finally, for outcome, we used “mortality rate” and “thromboembolic events” OR “VTE” in the title and abstract and the MeSH term was thromboembolic events. The referencing list of the reviewed articles were also screened for relevant studies.
Inclusion and exclusion criteria
The inclusion criteria were: the study design was RCT, prospective or retrospective series the study took place in the pre-hospital setting (either in ground ambulance or flight ambulance) adult civilian trauma patients treated with TXA compared with control group of patients who did not receive TXA inclusion of one or both of overall mortality or VTE the study used the CRASH-2 trial guideline for TXA administration (adult patients aged between 16 and 80 years, signs of hemorrhagic shock and within 3-hours of injury) the study was published in English.
The following were excluded: studies without a full-text, conference abstracts, reviews and quality improvement studies. In addition, studies where TXA was administered for reasons other than trauma (i.e. surgery, menstrual bleeding etc.) were also excluded as were studies that did not follow the CRASH-2 trial guidelines (i.e. TXA was administered later than three hours after injury). Military patients were excluded since we were interested in the use of TXA in a civilian pre-hospital setting; the majority of military patients suffer from penetrating injuries whilst the majority of civilian patients suffer blunt force trauma and hence the comparisons may not be valid for these different types of injuries.7–9
Data extraction
After title, abstract and full-text screening of the articles, the relevant studies were selected. Article screening was conducted by two reviewers (AMA and ZA) and any differences were resolved by discussion after which, data extraction was performed. The extracted data included, study characteristics (first author, year, title, design, setting, country, intervention protocol, inclusion and exclusion criteria and outcome measures), population characteristics (sample size, age, Injury Severity Score (ISS), mechanism of injury (penetrating and blunt), gender (male and female), Glasgow Coma Scale (GCS)) and outcome measures (mortality rate and thromboembolic events).
Data synthesis and statistical analysis
The data was synthesised using both a narrative synthesis as well as meta-analysis. As the outcome is dichotomous (patients’ mortality: dead or alive and thromboembolic events: events or no events), and there are sufficient number of events in each group, a risk ratio (RR) was chosen as the method to quantify the risk between the two groups. A random-effects approach was utilized for the meta-analysis and any significant heterogeneity explored via a subgroup analysis if possible. Between-study heterogeneity was assessed by the I2 value, and an estimate of the between-study variance (Tau 2 ), in a random-effects meta-analysis, is also be presented. The analysis was carried out in R version 3.6.2 using the ‘meta’ package version 4.13.
Results
Study selection
The search identified 1884 abstracts plus 2 papers from searching bibliographies of related articles; after removal of duplicates there were 1475 abstracts and after screening the abstracts for inclusion and exclusion criteria, 19 articles were included for full text review. After full text screening, five articles were included in this review and 14 articles were excluded (Figure 1).
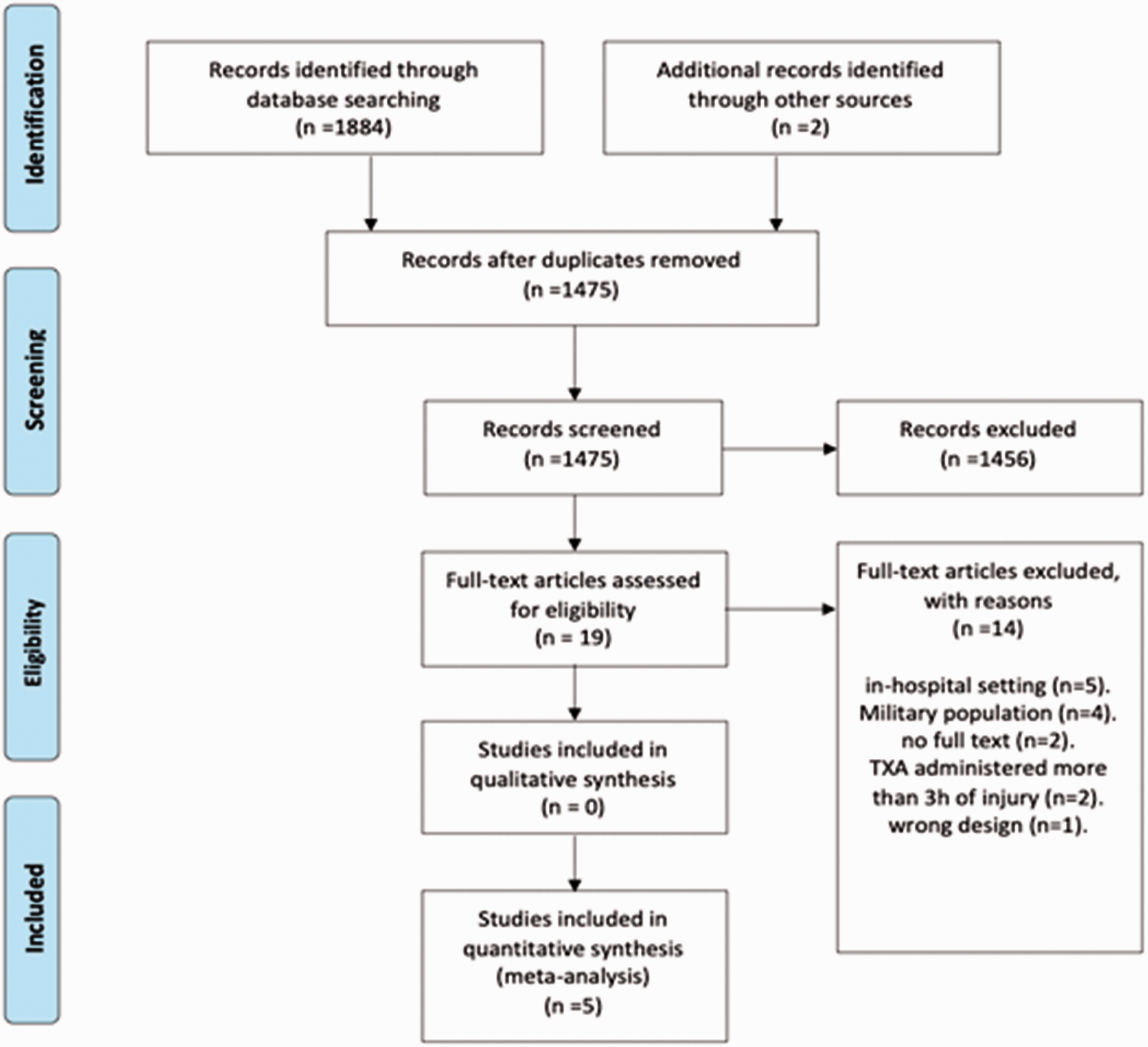
PRISMA flow chart.
Study characteristics
The total number of patients from the included studies was 2077 patients.10–14 Of those, 1021 of patients had received their first dose of TXA in the pre-hospital setting, compared to 1056 in the control group. A propensity score matching analysis was conducted in four of the included studies,10,12–14 while one study used manual matching. 11 The matching yielded two similar groups between the TXA and control group with no significant differences in four studies,11–14 while in the remaining study the matching was not identical. 10 All of the included studies were retrospective analysis studies conducted in the United States,10,12 Qatar, 11 Japan 13 and Germany. 14 One study used 16 years of age as the minimum age 14 and another study did not report using age as an inclusion criterion. 14 The intervention protocol consists of two doses: the first dose is 1 g of TXA diluted in 100 mL of Normal Saline (NS) over 10 minutes administered intravenously or intraosseous, and the second dose is 1 g of TXA diluted in 100 mL of NS over 8 hours. Table 1 summarises the study characteristics of all the included studies.
Study characteristics and outcomes of included studies.
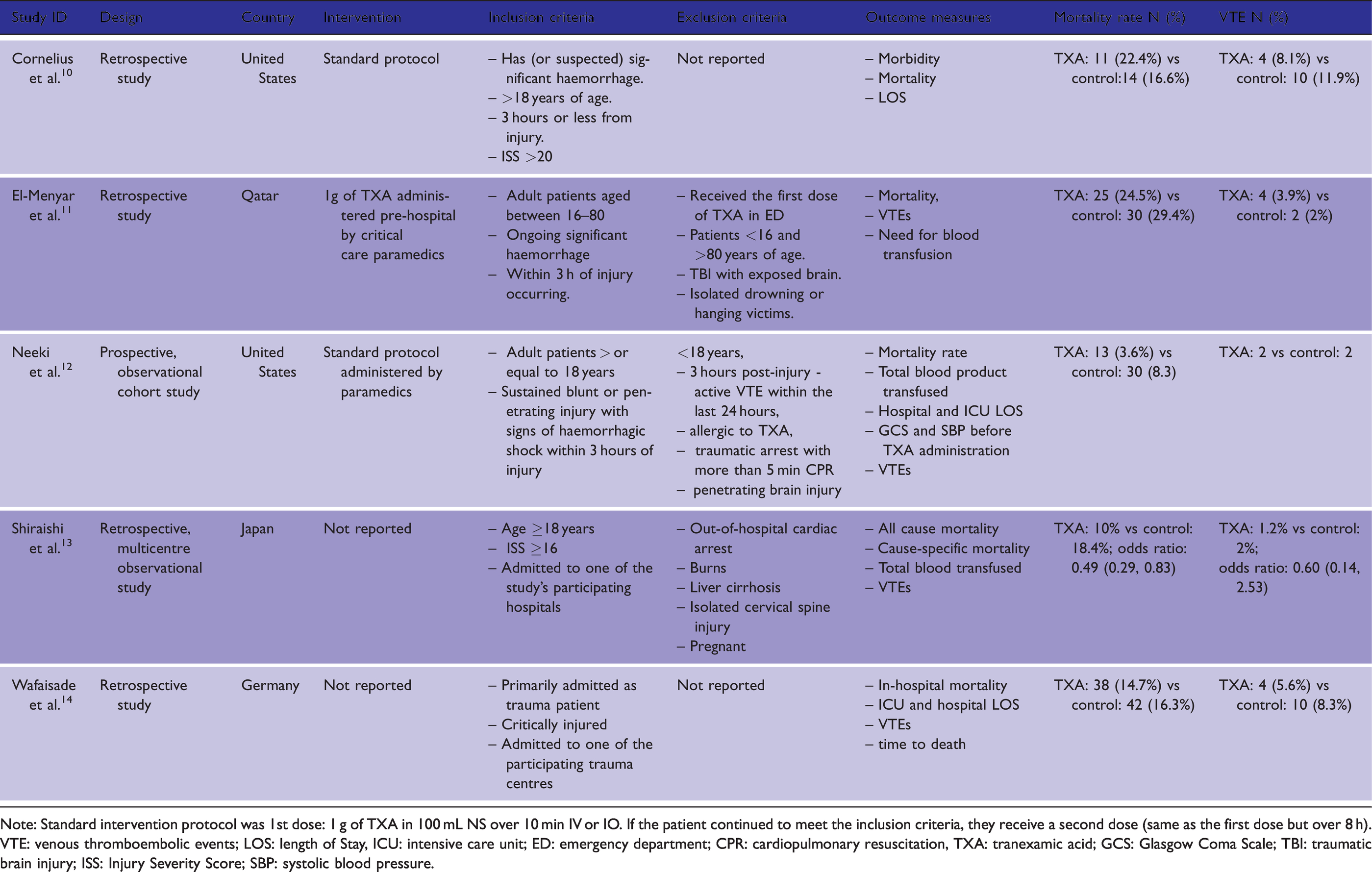
Note: Standard intervention protocol was 1st dose: 1 g of TXA in 100 mL NS over 10 min IV or IO. If the patient continued to meet the inclusion criteria, they receive a second dose (same as the first dose but over 8 h).
VTE: venous thromboembolic events; LOS: length of Stay, ICU: intensive care unit; ED: emergency department; CPR: cardiopulmonary resuscitation, TXA: tranexamic acid; GCS: Glasgow Coma Scale; TBI: traumatic brain injury; ISS: Injury Severity Score; SBP: systolic blood pressure.
Quality assessment
Since all of the studies included were retrospective studies, a pre-designed quality assessment tool was used to assess the included studies, the NIH quality assessment tool for observational cohort and cross-sectional studies. 15 Four out of the five studies were rated as fair quality because they did not report using statistical power and did not measure for potential confounding factors between exposures and outcomes. This is problematic as confounding factors increases the risk of bias and either exaggerate or dilute any potential difference between arms. The following domains were not assessed as they were not applicable to the design of the included studies: measurement of exposure overtime, blinding of outcome assessment and loss to follow-up overtime. Only one study was determined to be of good quality. 12
Patient characteristics
All of the included studies reported similar patients’ baseline characteristics including age, gender, ISS, mechanism of injury, systolic blood pressure (SBP) and GCS (Table 2). Four studies reported mean age with associated standard deviation,10,12,14 and one reported the median age with inter quartile range 13 ; only four studies included the gender of the recruited patients,11–14 and most were male in all of the studies within both the TXA and the control groups. As with age, four studies reported ISS as mean value10–12,14 and one study as the median. 13 Mean ISS values ranged from 16.08 12 to 31 10 and the median was 25 in the TXA groups. Mean ISS value in the control groups ranged from 17.15 12 to 29 10 and the median was again 25. 13 Blunt mechanism of injury was reported to be higher in four studies,10,12–14 while one study 12 had more patients with penetrating injuries. Four studies represented the neurological status of their patients using GCS.11–14 Two studies represented a moderate neurological status11,14 with mean values of (8.8 and 10.5 vs. 9.6 and 10.2, TXA vs. non-TXA, respectively), while the other two studies12,13 had mild neurological status (12.78 and 13 vs. 13 and 13, TXA vs. non-TXA, respectively).
Patients baseline characteristics of included studies.
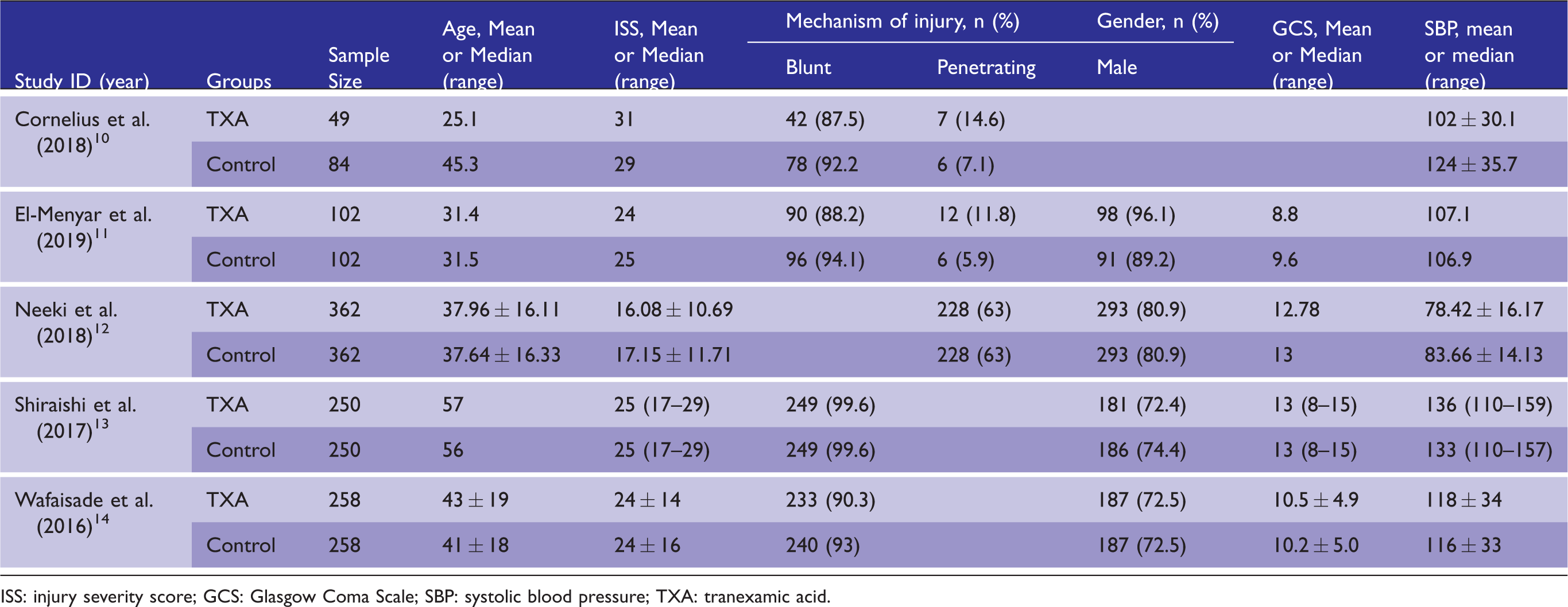
ISS: injury severity score; GCS: Glasgow Coma Scale; SBP: systolic blood pressure; TXA: tranexamic acid.
All included studies reported SBP at the scene before TXA administration. One study’s 10 TXA group had worse SBP than its control group (102 vs. 124). In two other studies,11,12 both groups had low SBP on scene (107.1 and 78.42 vs. 106.9 and 83.66, TXA vs. control group, respectively), while in the final two studies13,14 both groups had normal SBP on scene.
Narrative analysis
Mortality rate
All the included studies reported mortality rate; four reported decreased mortality rate among their TXA groups while one TXA group had a higher mortality rate. 10 In two studies that reported decreased mortality, the decrease did reach statistical significance12,13 and in two it did not;11,14 the mortality rate also did not reach statistical significance in the fifth study that reported a higher mortality rate in patients who received TXA. 10 Multivariable logistic regression analysis was performed in a single study to predict mortality after adjustment for age, sex, head AIS, ISS, and pre-hospital heart rate and SBP 11 ; predictors of mortality were massive transfusion, serum lactate on arrival to hospital and GCS in ED.
Thromboembolic events
The rate of VTE was reported by all the included studies. In three studies, the rate of VTE was lower in the TXA groups,10,13,14 in one there was a higher number of VTE among its TXA group, 11 while the other reported equal numbers of events in both its TXA group and control group 12 – none of these events were reported to be fatal.
Meta-analysis
Mortality rate
A total of 112 patients died in the TXA group, while 162 patients died in the control group. The overall mortality rate is lower for the TXA group compared to the control group with the pooled RR estimate of 0.74 with 95% confidence interval of [0.45; 1.25]; there is a moderate heterogeneity (I2 = 53%) between the studies. The pooled results of the overall mortality rate are presented in Figure 2.
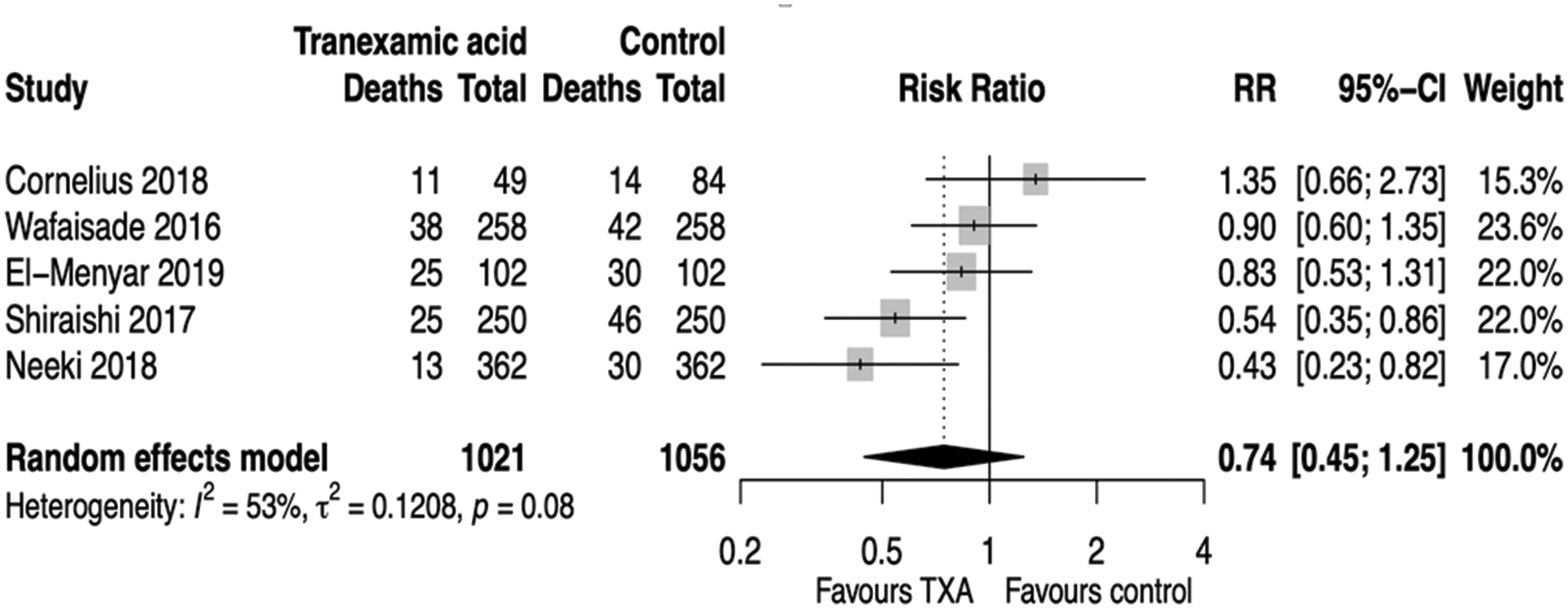
Forest plot of mortality rate.
Thromboembolic events
Overall, 17 patients developed thromboembolic events in the TXA group and 29 patients in the control groups.10–14 Thromboembolic events were lower in the TXA group compared to the control group; the pooled RR point estimate is 0.71 with 95% confidence interval of [0.35; 1.44]. There was no heterogeneity (I2 = 0%) between the studies. The pooled results of thromboembolic events are presented in Figure 3. Both outcome measures were not statistically significant.
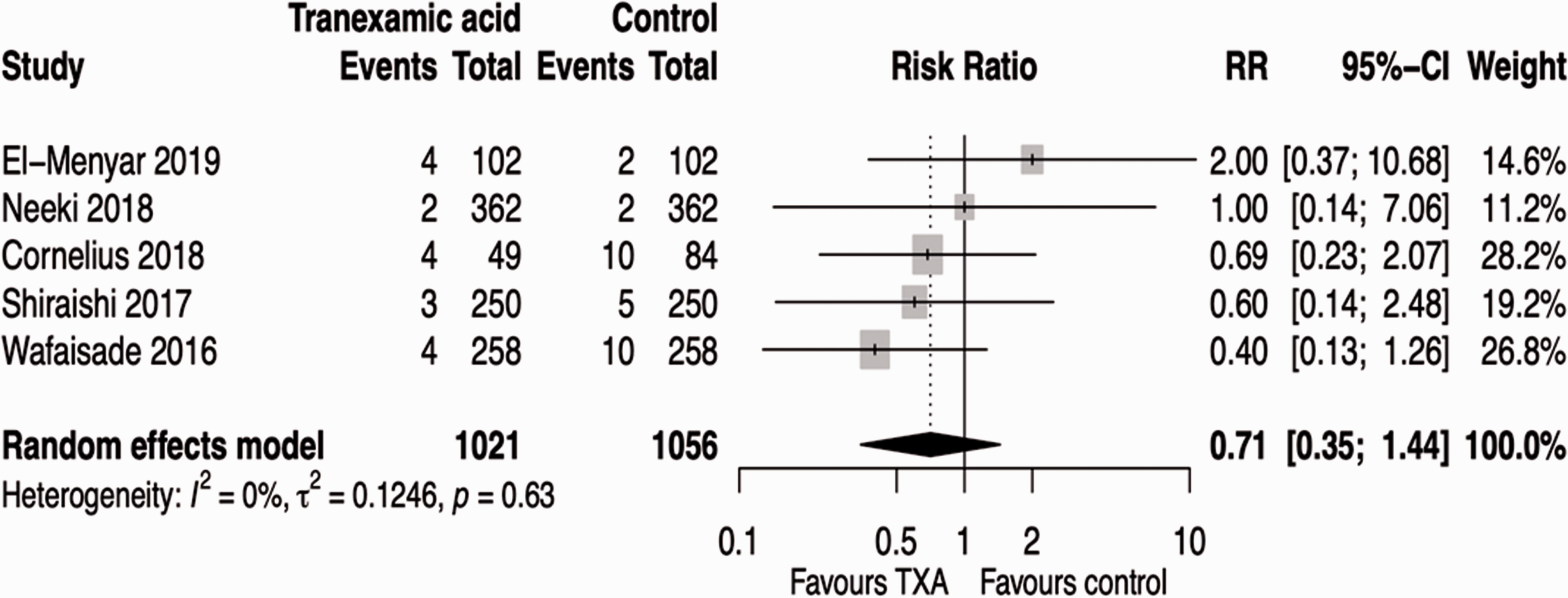
Forest plot of thromboembolic events.
Discussion
This study conducted a systematic review and meta-analysis of the pre-hospital use of TXA in trauma patients and their associated outcomes. It found that TXA is associated with lower overall mortality compared to patients who did not receive TXA (RR 0.74; 95% CI [0.45, 1.25]) and the TXA group had lower thromboembolic events compared to the control group (RR 0.71; 95% CI [0.35, 1.44]; however, neither of these findings reached statistical significance. The main merit of this study is that it included all of the clinical experiences published to date making it the most comprehensive study to evaluate the role of TXA in trauma patients and indicates a potential benefit in this patient group. However, further studies are needed to validate the potential use of TXA in the pre-hospital setting for trauma patients.
An important point to consider when using TXA in trauma patients is timing of the intervention. If TXA is administered within 3 hours of injury, all-cause mortality rates are reduced, with an increased effect if TXA is given within 1 hour of injury.7,16 Hence, it was hypothesized that if TXA was given in the pre-hospital setting it would result in better outcomes than if it was given later in the hospital emergency department.17,18 In one study based in a rural setting, it was found that most trauma patients arrived at the hospital outside the 3-hour time window for TXA administration and suggested TXA to be given by helicopter emergency medical services staff. 19 It is now generally accepted that TXA must be given within 3 hours to have a positive effect on trauma patients.20–23
The study by Cornelius et al. 10 found that TXA is associated with higher mortality, however, that study had some serious limitations. The matching between the TXA group and control group was not identical as the TXA group had higher ISS, which affected the results of the study. On the other hand, Neeki et al. 12 concluded that patients with higher ISS would benefit the most from TXA administration. Both studies were underpowered, hence, their findings were inconclusive and the effect of TXA on the patient’s severity remains unclear. The retrospective nature of the included studies may have also limited the integrity of the findings. As reported by Wafaisade et al. 14 patient’s files were incomplete, impacting the results of their study, whilst Shiriashi et al. 13 discussed that the retrospective analysis prevented the adjustment for some confounders that were unmeasured, but would likely have been associated with different findings. Moreover, and perhaps most importantly, four out of the five included studies had insufficient sample sizes.10–13 These limitations and weaknesses may have biased our findings. Unfortunately, there are no published RCTs that examine the role of TXA in the pre-hospital setting, but there are number of ongoing clinical trials.24,25
A previous meta-analysis by El-Menyar et al. 26 aimed to assess the early effect of TXA by evaluating 24-hour mortality rate and the late effect by reviewing 30-day mortality rate, along with thromboembolic events. The pooled results of their meta-analysis showed a significant decrease in early mortality (24-hour), however, the pooled number of patients were underpowered for 30-day mortality rate and thromboembolic events. In order to examine the overall effect of TXA and its complications with an appropriately powered sample size, and since there are studies that were published after the El-Menyar et al., 26 we aimed to investigate the overall mortality rate and thromboembolic events in all of these studies.
Our meta-analysis showed a 26% decrease in the overall mortality rate for the TXA group compared to the control group, although the confidence interval of 0.45-1.25, means that the risk of mortality may range from 55% lower to 25% higher in the TXA group compared to the control group. The other finding of this study is that the TXA group had a 29% lower chance of developing thromboembolic events compared to the control group with a confidence interval of 0.35-1.44; as both intervals span the null value of 1, hence, there is little evidence to show a true difference. This limitation in findings might be due to the retrospective nature of the included studies, as well as the fact that most of the studies included in this review had insufficient sample sizes. The data regarding the overall effect of TXA administration in the pre-hospital setting is still scarce and this field needs a high-quality RCT study with a sufficiently powered sample size.
Some ongoing RCTs conducted in the pre-hospital setting were also observed24,25 which will fill some gaps regarding pre-hospital TXA administration, and hopefully these trials will also have sufficient sample sizes to reach statistical significance in terms of mortality rate and thromboembolic events. One is investigating patients with traumatic brain injuries (TBI) who received TXA in the pre-hospital setting 24 and also trialling different doses of TXA (a 2 g pre-hospital bolus) and as such is the first study to use different drug doses that CRASH-2.
Limitations
The generalizability of these results is subject to certain limitations. The search strategy did not include non-English language articles nor grey literature making this study subject to publication bias. All the included studies were retrospective studies and had incomplete data accrual which might have changed the results of this analysis. The generalizability is also setting-dependent as for example, in the UK, pre-hospital guidelines are robust and recommend that all personnel have minimum in-hospital training before embarking into the pre-hospital setting and further competencies are recommended prior to undertaking sole practice in the UK hospital setting. The benefits of such training are self-evident from previous studies, which showed high level competencies among paramedics and critical care paramedics (CCPs). 27 Increasingly in the UK however, the use of pre-hospital critical care physicians (PHCC physicians) has been utilized along with increased access to training programmes for CCPs, which have significantly improved pre-hospital care.28,29 Like the UK, pre-hospital care in Europe is also most often provided by experienced consultant level physicians working with experienced paramedics, whilst the pre-hospital setting has been a sub-specialty for many years with well-defined training programmes. 29 In other countries such as the US, pre-hospital care is usually provided by paramedics and hence could account for some variations in mortality and thromboembolic events.
Conclusions
This study set out to systematically review the effect of TXA administration on trauma patients in the pre-hospital setting compared to a control group with overall mortality rate and thromboembolic events as the outcomes of interest. This study has identified that early administration of TXA in the pre-hospital setting is associated with lower mortality and lower risks of developing thromboembolism compared to controls but these findings do not reach statistical significance and may simply reflect training competencies of the pre-hospital care provider. There remains need for definitive high quality RCTs that are appropriately powered to assess the using of TXA in the pre-hospital setting. Future studies should compare mortality rates and the frequency of thromboembolic events in pre-hospital trauma patients in the UK, where robust training of paramedics, physician-led care and national guidelines have been in place for several years.
Footnotes
Declaration of conflicting interest
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval was not sought for this article because it was not required for this study as per advice from the NHS Health Research Authority, since it is a systematic review of published literature.
Informed consent
Informed consent was not sought because no patients or members of the public were involved.
Guarantor
ZA.
Contributorship
AMA and ZA conceived the study and developed the protocol with ASA and BK. AMA and ZA completed the data synthesis and wrote the first draft of the manuscript. All authors contributed to data analysis, reviewed and edited the manuscript and approved the final version.
Provenance and peer review
Not commissioned, externally peer reviewed.
Acknowledgements
None.
