Abstract
Background and aims:
The anti-inflammatory effects of dexamethasone may reduce the inflammatory response after pancreatoduodenectomy. The aim of this retrospective observational study was to evaluate the association between intraoperative dexamethasone and postoperative complications in patients undergoing pancreatoduodenectomy with a special focus on patients with pancreatic ductal adenocarcinoma (PDAC).
Methods:
All eligible patients undergoing pancreatoduodenectomy in our hospital between January 2018 and December 2021 (n = 319) were included comparing the postoperative outcomes in patients who received intraoperative dexamethasone (n = 178) to patients not given any intraoperative glucocorticoids (n = 142).
Results:
PDAC was the most common diagnosis (n = 166) and of these patients 92 received intraoperative dexamethasone and 74 no glucocorticoids. Patients with PDAC who received dexamethasone experienced fewer severe Clavien–Dindo complications than those not receiving glucocorticoids (n = 13/92 (14.1%) vs n = 21/74 (28.4%), P = 0.033). Multivariable analyses confirmed that a single dose of dexamethasone was associated with a reduced risk of severe complications in this patient group (odds ratio (OR) 0.40, 95% confidence interval [CI] 0.18–0.91, P = 0.030). When considering all pancreatoduodenectomy patients, no statistically significant differences in postoperative complications were observed. The incidence of postoperative infections was similar between the groups, although postoperative C-reactive protein (CRP) levels were lower in pancreatoduodenectomy patients who received dexamethasone (CRP on the second postoperative day: 102 (69–146) vs 159 (112–208) mg/l, P < 0.001). Patients who received dexamethasone experienced postoperative fever less frequently than those not given an intraoperative glucocorticoid (n = 68/178 (38.4%) vs n = 73/141 (51.8%), P = 0.023). Dexamethasone had no statistically significant influence on overall survival of PDAC patients.
Conclusion:
A single dose of dexamethasone was not associated with decreased postoperative complications across all pancreatoduodenectomy patients. However, within the PDAC subgroup, there were fewer Clavien–Dindo ⩾ 3 complications after dexamethasone compared to no glucocorticoid administration.
Keywords
Context and relevance
While previous studies have presented mixed results on the role of perioperative corticosteroids in postoperative complications after pancreatoduodenectomy, we provide some further insights. We found that a single dose of intraoperative dexamethasone was associated with a reduction of severe Clavien–Dindo complications in pancreatic ductal adenocarcinoma patients. Dexamethasone also reduced the postoperative fever and C-reactive protein (CRP) levels in the general cohort although it did not show a significant impact on severe complications in overall cohort. Overall, dexamethasone demonstrated minimal adverse effects, supporting its use in pancreatoduodenectomy patients due to its anti-inflammatory and antiemetic properties. This study adds to the evidence for optimizing management in patients undergoing pancreatoduodenectomy.
Introduction
The enhanced recovery after surgery (ERAS) guidelines for pancreatoduodenectomy (PD) patients recommend that all PD patients should receive prophylaxis for postoperative nausea and vomiting (PONV). 1 An effective and inexpensive medicine reducing PONV is dexamethasone (DXM). Surgical patients who receive DXM experience a smoother recovery than those who receive a placebo. 2 In addition to the antiemetic effect, DXM also possesses analgesic and anti-inflammatory properties. DXM lowers the levels of proinflammatory cytokines and endoplasmic reticular stress, consequently diminishing the inflammatory response. 3 These anti-inflammatory effects could theoretically lead to a reduction in postoperative complications and infections. However, DXM also induces peripheral insulin resistance and, as a result, transient hyperglycemia, which could affect the innate immunity of the patients. 4 A meta-analysis indicated that, despite a short-term hyperglycemia, DXM did not increase postoperative infections or problems with wound healing after general anesthesia. 5
Studies on DXM in pancreatic surgery patients and its influence on postoperative complications have been somewhat contradictory. Sandini et al. pointed out that DXM was associated with fewer postoperative infectious complications, although it did not influence the overall morbidity in PD patients. Moreover, their study suggested that intraoperative DXM was associated with a better overall survival (OS) in pancreatic ductal adenocarcinoma (PDAC) patients. 6 However, in the recent PANDEX trial, intraoperative DXM did not lower the incidence of major postoperative complications in PD patients, although DXM showed a trend toward fewer reoperations and shorter hospital stay compared to placebo. 7
Despite the anti-inflammatory features of DXM, its influence on short-term and long-term outcomes in PD patients remains unclear. In addition, many prior studies have examined all PD patients, a group that is highly heterogeneous. This retrospective study aimed to (1) evaluate whether intraoperative DXM was associated with decreased postoperative complications in both PD patients and the PDAC subgroup; and (2) clarify whether DXM improved the OS in patients with PDAC.
Methods
Patient selection and perioperative care
We included all patients who underwent PD at Helsinki University Hospital from 2018 until 2021. We excluded patients who for any reason received other glucocorticoids than DXM perioperatively and patients whose anesthesiology record was missing. The Institutional Review Board of the Perioperative and Intensive Care Medicine at Helsinki University Hospital approved the study protocol on 30 May 2022. An ethics committee approval was not needed due to the retrospective design of our study.
In 2020, a recommendation for a 10 mg dose of DXM was added to our hospital’s treatment protocol for PD patients. However, throughout the entire study period, the administration of intraoperative DXM was determined at the anesthesiologist’s discretion. Patients received DXM during the early stage of surgery. The surgeon determined the prophylactic antibiotics according to their deliberation. Antibiotic treatment and surgical approaches, including surgical techniques, the use of stents and drains, and application of wound protectors, remained consistent throughout the research period. Postoperative care followed our hospital’s guidelines for PD patients including instructions for fluid management, postoperative nutrition, multimodal pain medication, and mobilization.
Outcomes
Primary endpoint of this study was the frequency of severe Clavien–Dindo complications in PD patients. Secondary endpoints included severe surgical complications, postoperative pancreatic fistulas (POPF), delayed gastric emptying, postoperative infections, mortality, and the length of hospital stay.
Data collection and definitions of postoperative complications
We collected patient data from the institutional electronic database including information on the preoperative characteristics, perioperative medications, postoperative outcomes, and in PDAC patients, the histopathology of the tumor. During the postoperative period, we recorded the laboratory parameters on the first five postoperative days (PODs), the patient’s body temperature, antibiotics administered, and the recovery of the patients. Fever was defined as ⩾38.0°C. CRP levels were measured daily on PODs 2–4, and thereafter as needed.
The postoperative clinical complications were determined according to the Clavien–Dindo classification, whereby grade III–V complications were regarded as severe. 8 In addition, we separately analyzed severe surgical complications, defined as any surgery-related complications requiring invasive intervention such as radiological drainage, endoscopy, or reoperation, excluding complications caused by internal medical conditions (e.g. cardiac or pulmonary problems). We applied the International Study Group of Pancreatic Surgery (ISGPS) definition for determining POPF (grade B–C) 9 and delayed gastric emptying (DGE, grade B–C). 10 We also analyzed biliary leakages, the length of hospital stay (LOS), and rehospitalization.
All postoperative infections were analyzed, including sepsis, pneumonia, urinary tract infection, wound infection, and abscesses. Sepsis was defined according to the third international consensus definitions for sepsis. 11 Pneumonia was diagnosed when a pulmonary infiltration was radiologically demonstrated, urinary tract infection when a urinary culture was positive, and abscess when seen radiologically. Blood cultures were obtained at the clinician’s consideration, typically when fever exceeded 38°C.
The follow-up time for clinical complications was 90 days and 30 days for infectious complications. Clinical complications included Clavien–Dindo complications, POPFs, DGE, and other surgical complications whereas infectious complications included sepsis, pneumonia, urinary tract infection, wound infection, and abscesses. We calculate the OS of PDAC patients from the date of first treatment (neoadjuvant or surgery) to the date when the patient was last known to be alive or to the time of death. The follow-up time for OS was until the end of November 2024.
In addition, we recorded the consistency of the pancreas from surgical reports and the main pancreatic duct (MPD) size from preoperative CT or MRI scans. We classified the patients as type A–D depending upon the MPD size and pancreatic texture using the ISGPS classification on the risk of a POPF. 12
Statistical analysis
All statistical analyses were performed using SPSS® Statistics (version 28; IBM, Inc., Armonk, NY, USA) and R (version 4.2.0; Foundation for Statistical Computing, Vienna, Austria). Continuous variables are presented as medians (interquartile range (IQR) or range). Statistical comparisons between groups were performed using the Mann–Whitney U test with continuous variables. Dichotomized and categorical variables are reported as numerals and percentages, and we employed the Fisher’s exact test or the Fisher–Freeman–Halton test for analyses. We performed analyses using the entire cohort and subgroup analyses for PDAC patients.
We used inverse probability weighting (IPW) based on propensity scores to reduce selection bias. Propensity scores were estimated using logistic regression with age and sex as covariates and treatment as the dependent variable. Stabilized IPW weights were calculated and applied in weighted logistic (svyglm, R survey package) or Cox regression models (svycoxph). Extreme weights (percentiles > 97.5% or <2.5%) were filtered out. This approach enables valid treatment effect estimation with robust standard errors.
Logistic regression was used for univariable and multivariable analyses for associations between severe Clavien–Dindo complications and independent variables. Interactions in the multivariable analyses were considered, but found non-significant. We performed the Kaplan–Meier analysis to calculate the OS in the PDAC patients, and Cox proportional hazard model to assess factors associated with OS. Severe surgical complications, intraoperative DXM, and Clavien–Dindo complications were used as time-dependent variables to address immortal time bias. We estimated the median time from operation to complication to be 5 days. All statistical tests were two-sided, with P < .05 considered statistically significant.
Results
During the study period (2018–2021), 596 patients underwent pancreatic surgery in Helsinki University Hospital, including 354 PDs. After excluding patients who received perioperative glucocorticoids other than DXM or whose anesthesiology record was missing, 319 patients were included in our analysis (Figure 1). DXM was administered to 178 patients intraoperatively, and 141 patients received no glucocorticoids during the perioperative period. In 2018, 16 patients (27.1%), in 2019, 20 patients (26.0%), in 2020, 65 patients (69.9%), and in 2021, 77 patients (85.6%) received intraoperative DXM. PDAC was the most common diagnosis (n = 166), and of these patients, 92 received intraoperative DXM and 74 no glucocorticoids.
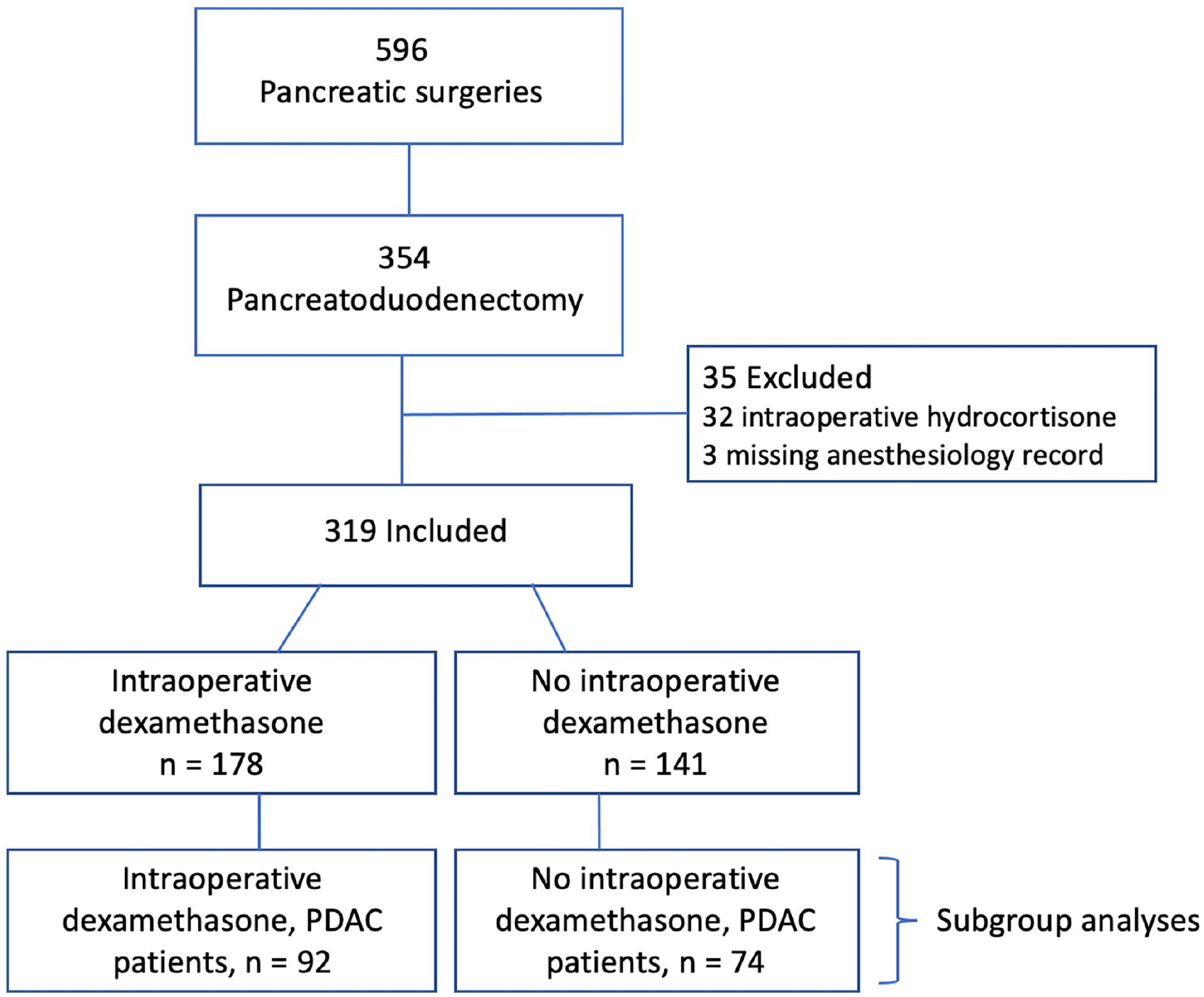
Flowchart of the patients included in the study. Study years were 2018–2021 and the data were collected from the anesthesia reports. Patients who did not receive intraoperative dexamethasone were not administered any other corticosteroid medications either.
Table 1 summarizes the preoperative and perioperative characteristics of patients. The patients’ characteristics were similar except that those who were not given DXM more often received intraoperative acetaminophen than patients who had received DXM. Tumor pathology and oncologic treatment of PDAC subgroup patients are presented in Supplementary Table 1.
Baseline and perioperative characteristics of the patients.
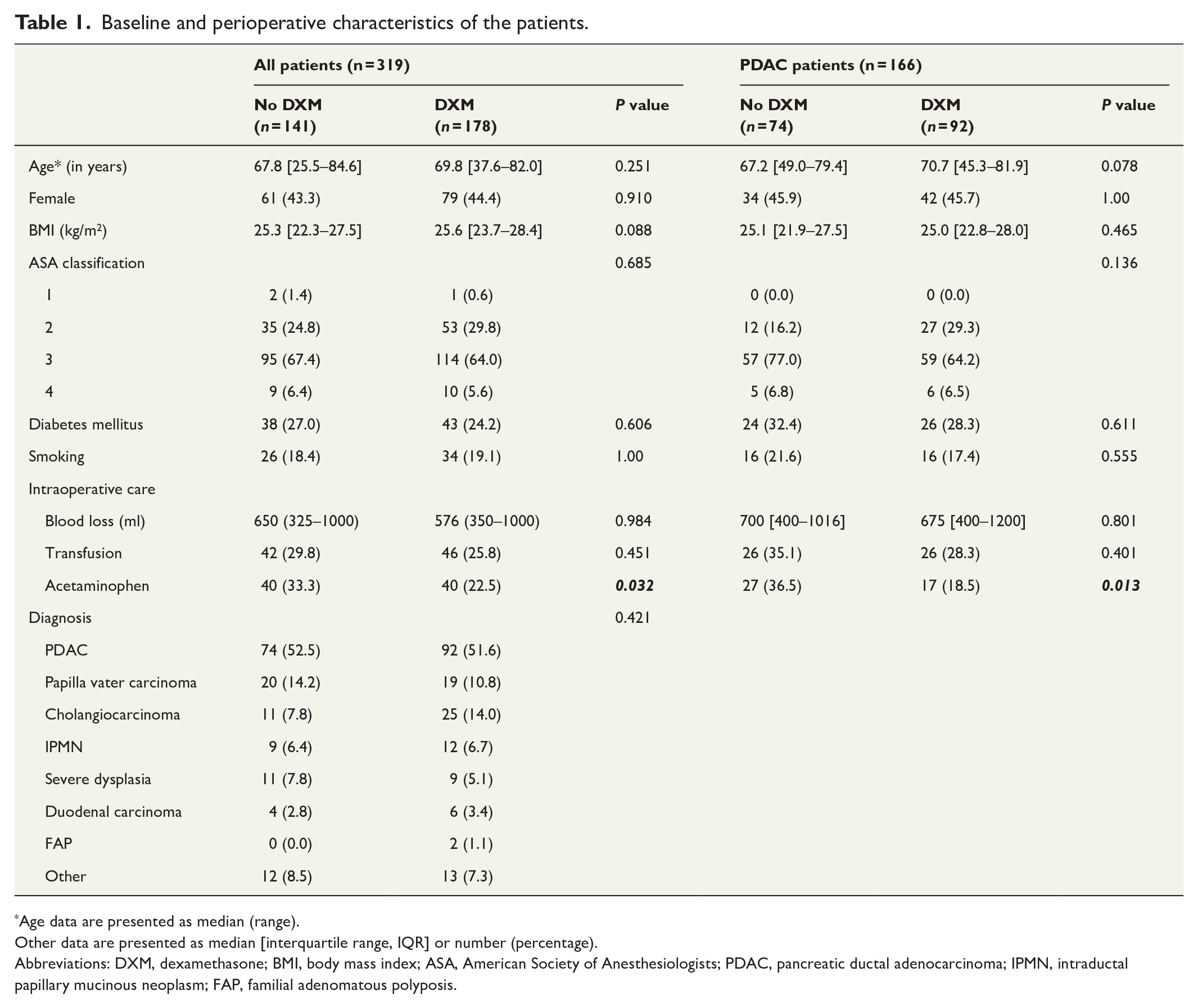
Age data are presented as median (range).
Other data are presented as median [interquartile range, IQR] or number (percentage).
Abbreviations: DXM, dexamethasone; BMI, body mass index; ASA, American Society of Anesthesiologists; PDAC, pancreatic ductal adenocarcinoma; IPMN, intraductal papillary mucinous neoplasm; FAP, familial adenomatous polyposis.
Postoperative clinical complications occurred at similar rates in both study groups (Table 2) but the postoperative CRP values remained lower in the DXM group during the first three PODs. Nevertheless, in the subgroup analyses of PDAC patients, severe Clavien–Dindo complications were more common in patients who had not received intraoperative DXM than in those given DXM (no DXM: n = 21 (28.4%) vs DXM: n = 13 (14.1%), P = 0.033); Table 2. Similarly, severe surgical complications more frequently occurred in PDAC patients not administered DXM (Table 2).
Postoperative outcomes among all patients and pancreatic ductal adenocarcinoma patients.
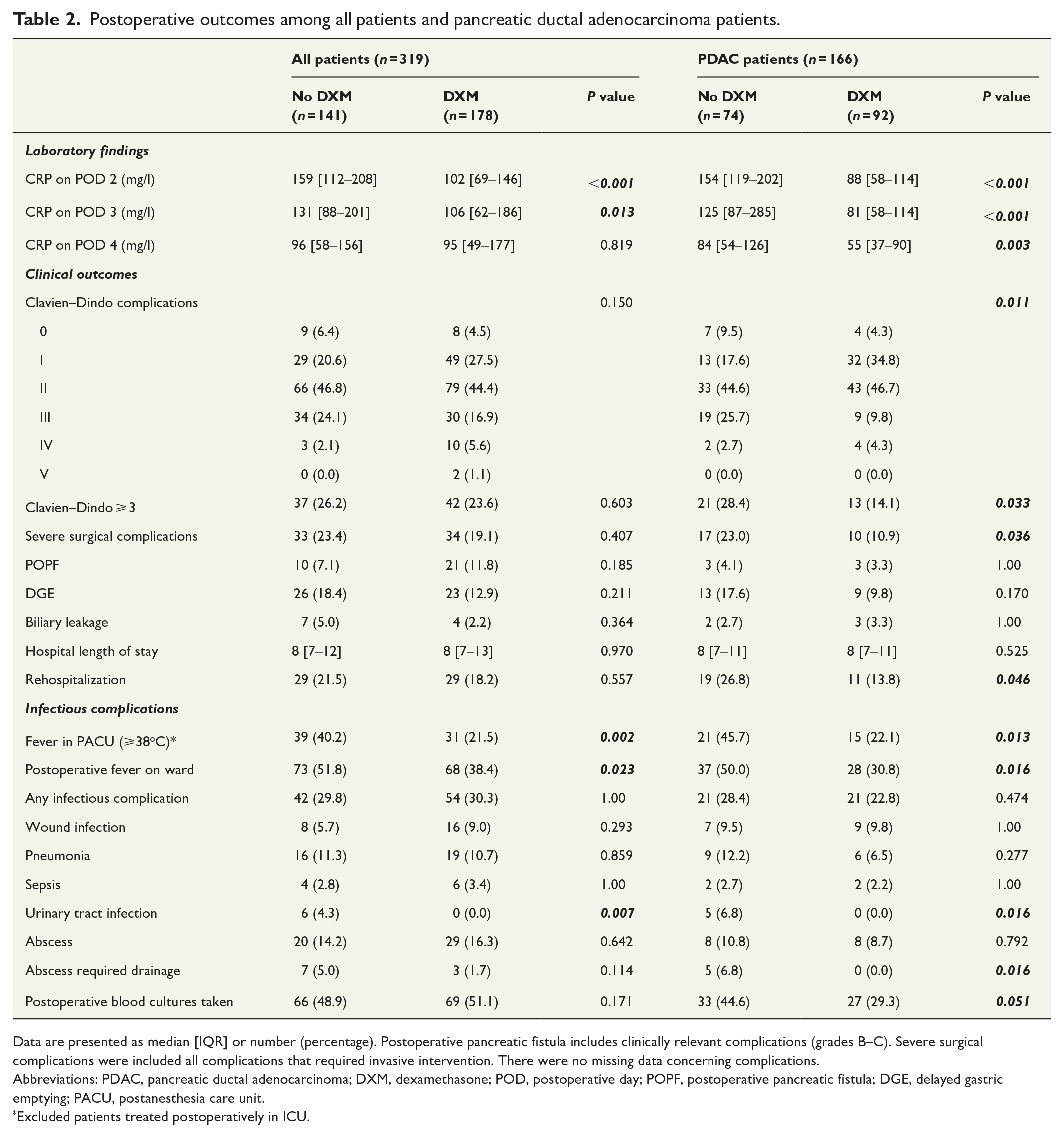
Data are presented as median [IQR] or number (percentage). Postoperative pancreatic fistula includes clinically relevant complications (grades B–C). Severe surgical complications were included all complications that required invasive intervention. There were no missing data concerning complications.
Abbreviations: PDAC, pancreatic ductal adenocarcinoma; DXM, dexamethasone; POD, postoperative day; POPF, postoperative pancreatic fistula; DGE, delayed gastric emptying; PACU, postanesthesia care unit.
Excluded patients treated postoperatively in ICU.
The multivariable analyses confirmed that intraoperative DXM was associated with a lower risk for severe Clavien–Dindo complications in PDAC subgroup (odds ratio [OR] 0.40 (95% confidence interval [CI] 0.18–0.91, P = 0.030; Table 3). Specific types of severe Clavien–Dindo complications and severe surgical complications are presented in Supplementary Tables 2 and 3. In addition, in PDAC patients, severe Clavien–Dindo complications reduced the proportion of patients who received adjuvant treatment (CD 0–2, n = 104 (78.8%) vs CD 3–5, n = 23 (67.6%), P = 0.040).
Univariable and multivariable analyses of severe Clavien–Dindo complications.
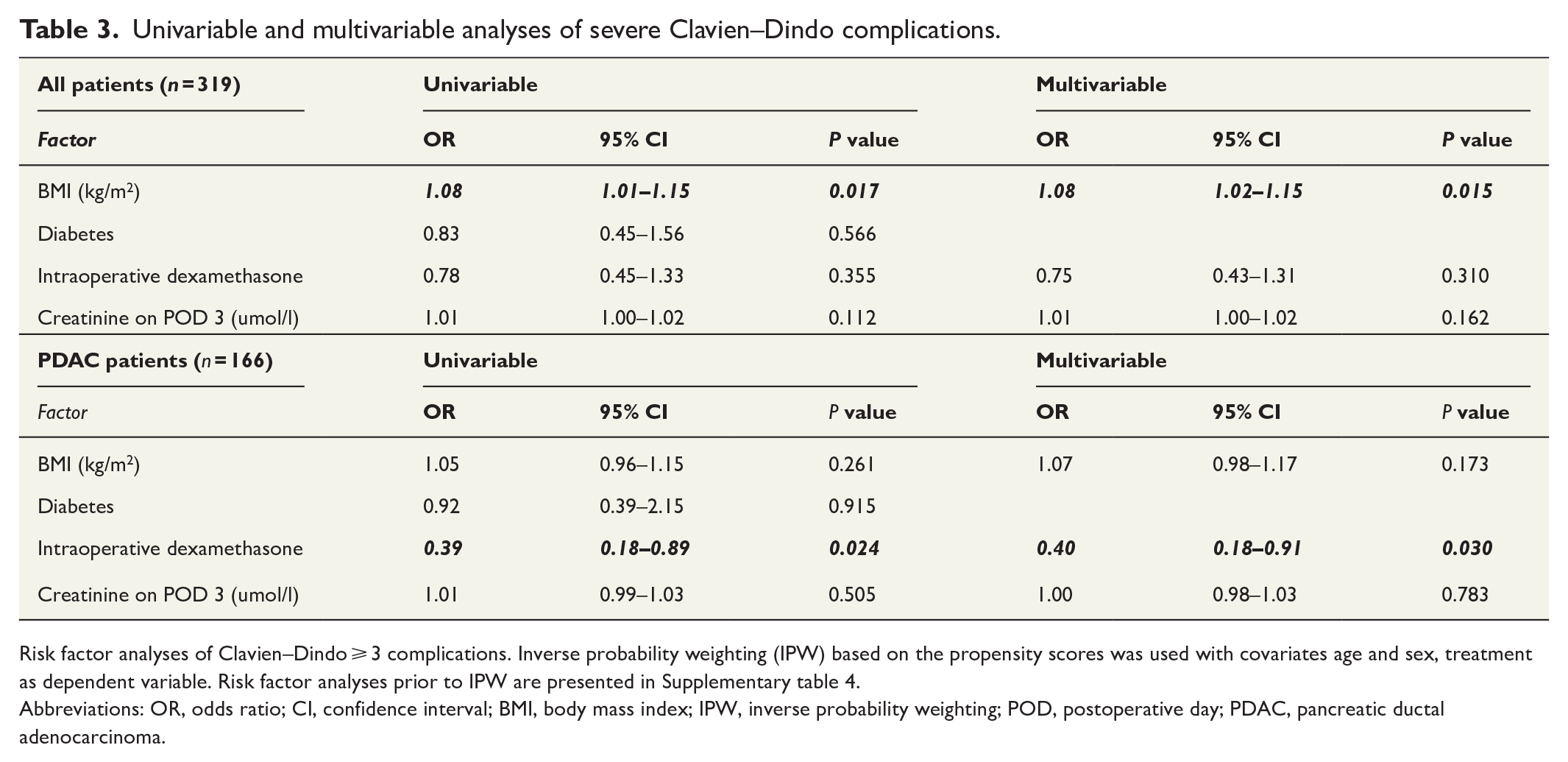
Risk factor analyses of Clavien–Dindo ⩾ 3 complications. Inverse probability weighting (IPW) based on the propensity scores was used with covariates age and sex, treatment as dependent variable. Risk factor analyses prior to IPW are presented in Supplementary table 4.
Abbreviations: OR, odds ratio; CI, confidence interval; BMI, body mass index; IPW, inverse probability weighting; POD, postoperative day; PDAC, pancreatic ductal adenocarcinoma.
Clinically relevant infectious complications were quite similar in both study groups (Table 2). Urinary tract infections and abscesses, which required drainage, occurred more frequently in patients not administered DXM. Postoperative fever (⩾38.0°C) was more common in patients who had not received intraoperative DXM. Blood cultures were frequently taken but bacteremia was rare with no significant difference between the patient groups (no DXM n = 4 (2.8%) vs DXM n = 8 (4.5%), P = 0.559). We observed no differences in the postoperative need for antibiotics.
We performed subgroup analyses of the whole cohort based on pancreatic texture and MPD size, and identified an inverse association between DXM and postoperative complications in type A patients (not-soft pancreatic texture and MPD > 3 mm, n = 121). In that patient group, severe Clavien–Dindo complications were more common in patients not administered intraoperative DXM (Table 4). We observed no differences in postoperative complications in any other subgroups.
Postoperative complications among ISGPS classification subgroups on the risk of a postoperative pancreatic fistula.
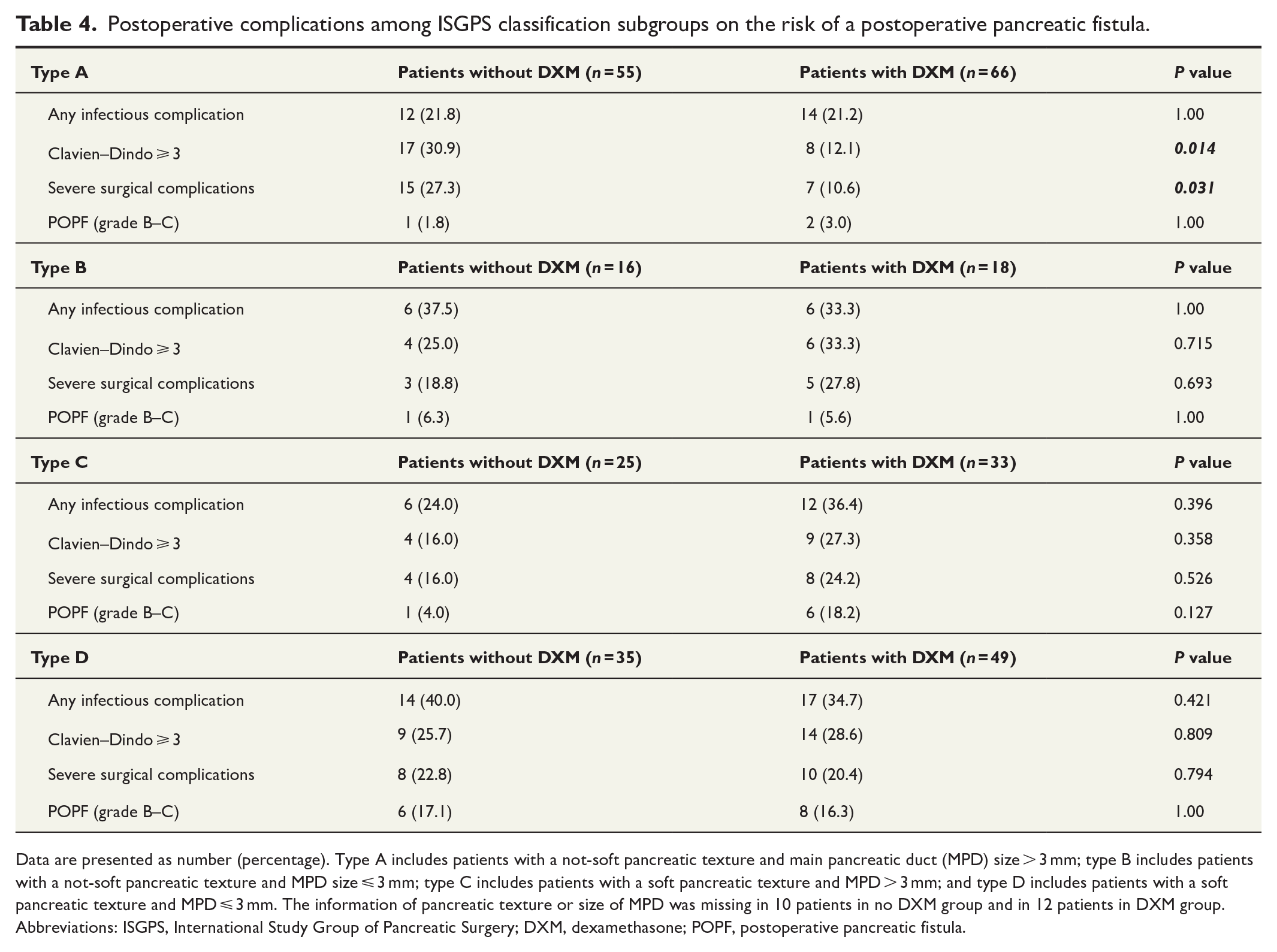
Data are presented as number (percentage). Type A includes patients with a not-soft pancreatic texture and main pancreatic duct (MPD) size > 3 mm; type B includes patients with a not-soft pancreatic texture and MPD size ⩽ 3 mm; type C includes patients with a soft pancreatic texture and MPD > 3 mm; and type D includes patients with a soft pancreatic texture and MPD ⩽ 3 mm. The information of pancreatic texture or size of MPD was missing in 10 patients in no DXM group and in 12 patients in DXM group.
Abbreviations: ISGPS, International Study Group of Pancreatic Surgery; DXM, dexamethasone; POPF, postoperative pancreatic fistula.
The administration of DXM was not associated with OS in PDAC patients who underwent PD surgery (Figure 2). Median OS was 32 months in patients who received DXM and 38 months in patients not receiving DXM (P = 0.4). Stage ⩾ IIB and occurrence of severe surgical complications were associated with poorer OS in PDAC patients (Supplementary Table 5).
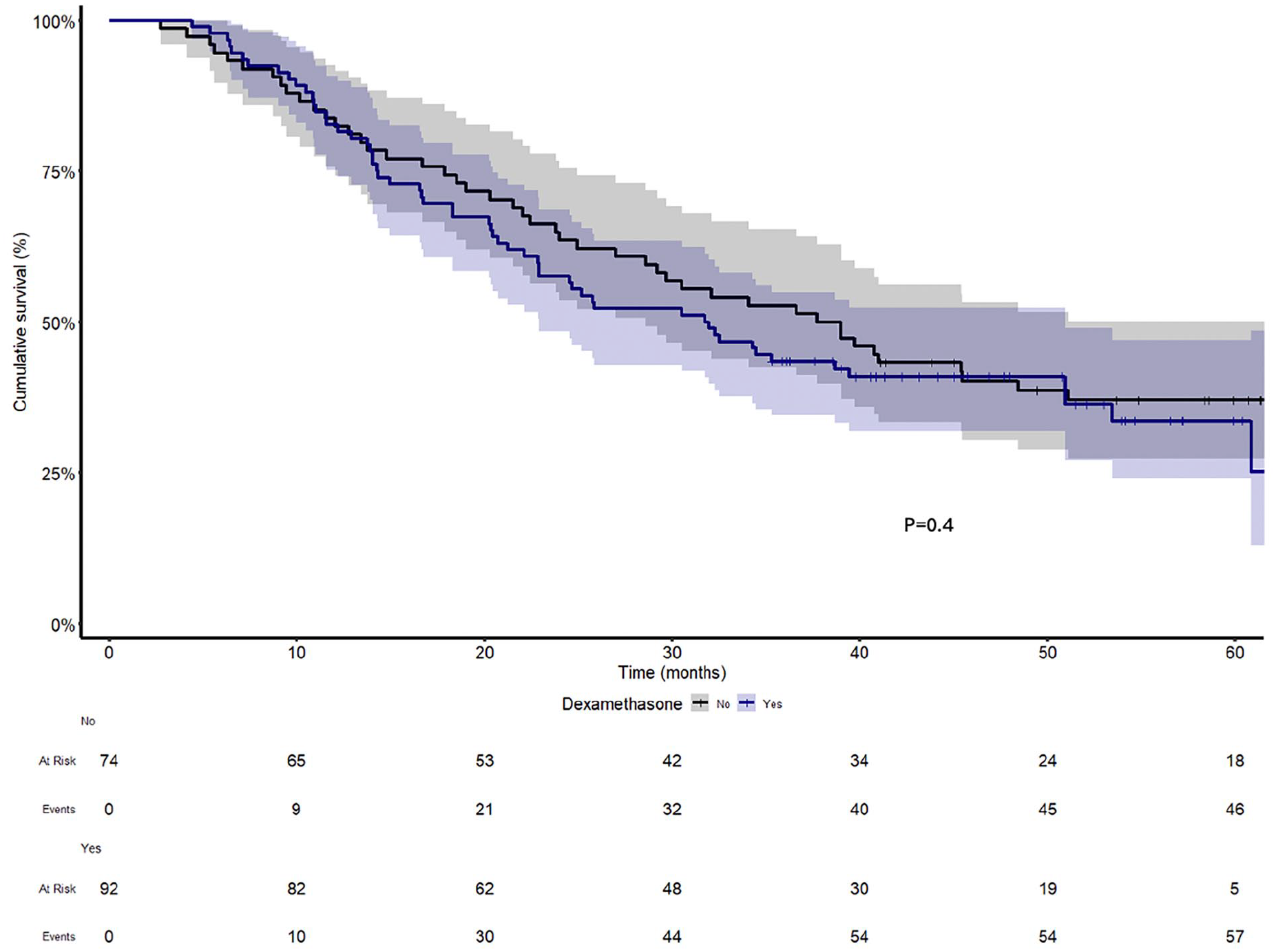
Overall survival of patients with a pancreatic ductal adenocarcinoma (PDAC). All patients underwent PD surgery. Kaplan–Meier curves indicate no difference in patients with and without intraoperative dexamethasone (P = 0.4).
Discussion
Although intraoperative DXM was not associated with improved postoperative outcomes in all PD patients, our study demonstrated that it was linked to fewer severe Clavien–Dindo complications in PDAC patients. In addition, DXM reduced the postoperative CRP levels and fever in all PD patients. Intraoperative DXM was not associated with better OS in PDAC patients.
In our hospital, intraoperative DXM was included in the treatment protocol for PD patients in the beginning of 2020 according to the general ERAS guidelines given its antiemetic, analgesic, and anti-inflammatory effects. 1 Surgical treatment and the operating surgeons remained consistent throughout the study period, with no differences in the administration of prophylactic antibiotics. Although a meta-analysis had demonstrated that a 4–5 mg dose of DXM was as effective in PONV prophylaxis as an 8–10 mg dose, 13 we chose to use a 10 mg dose since we aimed at a significant anti-inflammatory effect. An early increase in postoperative CRP levels likely results from inflammation rather than infection. In many studies, as in this study, perioperative glucocorticoid use resulted in lower postoperative CRP levels reflecting an anti-inflammatory effect. 14 – 16 Increased postoperative CRP levels have been associated with a higher incidence of pancreatic complications, 17 leading to growing interest in whether the anti-inflammatory properties of glucocorticoids could help reduce these complications.
Studies on the perioperative administration of glucocorticoids and their association with postoperative outcomes have generally yielded mixed results. Previous Finnish RCTs have suggested that perioperative hydrocortisone may reduce severe Clavien–Dindo complications in patients undergoing distal pancreatectomy or PD.18,19 These trials also linked hydrocortisone with a lower rate of clinically relevant POPF when administered to patients with >40% acini in the pancreatic transection line. 18 However, a recent RCT found no association between hydrocortisone and POPF in this same patient group. 20 A systematic review indicated that perioperative glucocorticoid use had a positive impact on morbidity in patients having undergone major pancreatic resections. 21 In contrast, a retrospective study from Newhook et al. 22 did not support this finding. A previous meta-analysis found no overall benefit of perioperative glucocorticoid administration on postoperative outcomes in PD patients but suggested that certain subgroups may experience positive effects of glucocorticoids on postoperative complications, 23 and these aspects warrant further research. This is one of the reasons we chose to assess the outcomes in the PDAC subgroup, alongside those in the overall PD cohort.
In this study, the association between intraoperative DXM and a reduction in postoperative complications was observed in PDAC patients, as well as in those with a wide MPD and not-soft pancreatic texture (type A). However, we cannot explain the underlying mechanisms as to why these patients appear to benefit from the anti-inflammatory effects of DXM. According to the ISGPS classification, type A patients are at the lowest risk for developing a POPF, whereas a narrow (⩽3 mm) MPD and a soft pancreatic tissue increases the risk for postoperative complications.12,24 The majority of PDAC patients (62.5%) were type A, so these subgroups partially overlapped. The positive association between DXM and reduced complications was observed in complications, which required invasive interventions, such as abscess drainage. A recent RCT demonstrated that a preoperative dose of DXM was associated with a reduction in similar complications in patients undergoing emergency laparotomy. 16 There have been concerns that corticosteroid administration may increase the risk for postoperative anastomotic leakage after lower gastrointestinal surgery, but these harmful effects were specifically associated with long-term use and high doses. 25 Although DXM induces a short-term hyperglycemia, no negative association with its use and infectious complications has been observed15,26,27 as the hyperglycemia can be easily controlled with insulin. A risk factor associated with severe Clavien–Dindo complications in the whole patient cohort was body mass index (BMI). Obesity itself promotes inflammation and dysregulates the autophagy of the cells. 28 In general, BMI ⩾ 25 kg/m2 seems to increase the risk for postoperative surgical complications and infectious complications in pancreatic surgery patients,29,30 which may be caused by more difficult surgical conditions and insulin-resistance associated with perioperative hyperglycemia.
This study confirmed our clinical experience of the early postoperative inflammatory reaction in PD patients. The postoperative temperature rose significantly more in patients not administered intraoperative DXM despite they more often received acetaminophen at the end of surgery. DXM has been shown to decrease postoperative fever on the first three PODs in noncardiac surgical patients, 26 and our findings support those results. Postoperative fever leads to unnecessary laboratory and radiological examinations and causes additional expenses. Due to fever, almost a half of our PDAC patients had blood cultures taken although bacteremia and sepsis were extremely rare. In the RCT by Chen et al., 7 PD patients who received DXM also had lower medical and drug costs compared to the placebo group.
The survival analysis of our study demonstrated that only well-known factors such as higher stage and severe complications had an influence on OS of PDAC patients. Our study failed to illustrate any effect of DXM on survival. Some previous studies indicated that a single dose of DXM significantly improved the OS of pancreatic cancer patients.6,31 Biochemical studies have demonstrated that the anti-inflammatory effects of DXM could inhibit the migration and tissue invasion of cancer cells and, therefore, impede disease progression, which indeed could have a positive effect on survival.32,33 There have also been concerns that immunosuppression caused by glucocorticoids could enhance the spread of cancer cells, as in vivo, DXM may decrease cell proliferation and lymphokine production and disturb the apoptosis of neoplastic cells.34,35 Nevertheless, a single dose of DXM has not been associated with changes in recurrence-free survival or OS in other malignancies.36,37
The major limitations of our study are attributable to the retrospective study design resulting in inevitable selection bias. The anesthesiologist responsible for the patient decided whether the patient was given DXM although the patient characteristics were similar in both study groups. However, the use of IPW strengthened the validity of our findings by reducing selection bias and confounding in this study. Although the surgical treatment remained consistent across the study years and perioperative care followed the hospital’s protocol, the details of treatment may have varied between the patients. In addition, the sample size of the PDAC subgroup was relatively small limiting any strong conclusions. The research team was not blinded to administration of DXM during the data collection.
Our study highlighted that DXM administration had minimal adverse effects in PD patients when a single intraoperative dose was given. In addition to its antiemetic effect, intraoperative DXM was associated with fewer severe complications in PDAC patients. The anti-inflammatory effect of DXM reduced postoperative fever, and therefore, decreased the redundant laboratory examinations. No associations with DXM and infectious complications were seen nor any impact on survival in PDAC patients. Further prospective studies on intraoperative DXM and short-term postoperative complications in different subgroups of PD patients are needed.
Supplemental Material
sj-docx-1-sjs-10.1177_14574969251371868 – Supplemental material for Intraoperative dexamethasone after pancreatoduodenectomy in pancreatic ductal adenocarcinoma: A retrospective cohort study
Supplemental material, sj-docx-1-sjs-10.1177_14574969251371868 for Intraoperative dexamethasone after pancreatoduodenectomy in pancreatic ductal adenocarcinoma: A retrospective cohort study by Piia Peltoniemi, Harri Mustonen, Katarina Johansson, Inkeri Lehto, Hanna Seppänen and Pertti Pere in Scandinavian Journal of Surgery
Footnotes
Acknowledgements
The authors thank Vanessa Fuller from the University of Helsinki Language Center and a colleague Nora Mattila for their help in proof reading of the manuscript.
Authors contributions
P.P., H.S., and P.P. conceived the study. P.P., K.J., and I.L. collected the relevant data. P.P. and H.M. analyzed the data. All authors participated in interpretation of the results. The manuscript was drafted by P.P. All authors critically revised the manuscript and approved the final version.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are grateful for research funds received from the Cancer Foundation Finland, the Sigrid Jusélius Foundation, Finska Läkaresällskapet, the Finnish Government Subsidy Funding, Helsinki University Hospital, the Paulo Foundation, the Mary and Georg C. Ehrnrooth Foundation, the Scandinavian Society of Anesthesiology and Intensive Care Medicine, Orion Research Foundation, and the Academy of Finland. None of the foundations were involved in the study design or execution.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
