Abstract
Background and aims:
The aim of this study was to compare the safety and efficacy of establishing a robotic-assisted thoracoscopic surgery (RATS) program in a low-volume center with mixed cardiothoracic practice and surgeons with different background experiences—video-assisted (VA) versus open thoracotomy (OT).
Methods:
Between January 2015 and June 2023, the center performed 460 anatomical lung resections. The RATS program was established in October 2021, and two surgeons, OT and VA, were selected as RATS surgeons. Before this, surgeon OT performed mostly open thoracic surgery, and surgeon VA had notably more thoracoscopic experience. The primary outcomes were the learning curves of surgeon OT and VA for lymph node yield, operative time, blood loss, and complications after starting the RATS program. A Risk-Adjusted Cumulative Sum (RA-CUSUM) method was used to determine learning curves.
Results:
Surgeons OT and VA showed similar learning curves in lymph node yield with surgeon OT having a peak at case number 23 and surgeon VA at 19. Operative time improved with surgeon VA after 15 cases and with surgeon OT after 30 cases. RA-CUSUM analysis showed no increased bleeding cases when comparing the baseline risk and the median bleeding values of the center. With both surgeons, there were significantly fewer overall complications when using RATS than with other surgical techniques.
Conclusion:
According to this study, training surgeons with either open or thoracoscopic background experience to perform robotic-assisted lung cancer surgery is safe and does not compromise short-term operative outcomes. This study is a retrospective registry study and has been approved by the Northern Ostrobothnia Research Ethics Committee with license number EETTMK 5/2019; hence, no clinical trial registry number is associated with this study.
Keywords
Context and Relevance
The implementation of robotic-assisted thoracoscopic surgery (RATS) in low-volume centers remains unexplored. In addition, no studies have assessed whether a surgeon’s prior experience influences the safety or effectiveness of transitioning to RATS. This study evaluates the learning curves of two surgeons with different backgrounds (experienced in video-assisted thoracoscopic surgery (VATS) or open thoracotomy) in a low-volume center. The surgeons showed similar learning curves in lymph node yield. In RATS procedures, the surgeons encountered less intraoperative bleeding and fewer overall complications compared to conventional techniques (VATS or open thoracotomy). These findings support the adaptation of RATS in low-volume centers, suggesting that RATS is both safe and feasible, even for surgeons with limited prior robotic experience, without compromising short-term outcomes.
Introduction
Robotic-assisted procedures have been utilized in various surgical fields for a relatively long time, for example, in urology, gynecology, gastrointestinal surgery, and in high-volume thoracic surgery centers.1 –5 Until recently, robotically assisted thoracic surgery programs were quite rare in low- to medium-volume hospitals. However, low- and medium-volume centers have, in increasing numbers, started to establish RATS programs of their own.6,7 The challenges of robotic-assisted surgery for especially low-volume centers are mainly economical but also include surgeon availability and training. As the use may be limited and the costs higher, the implementation of new technology requires careful consideration in low-volume centers.
Oulu University Hospital is a tertiary care center covering a large but sparsely populated geographical area in Northern Finland. It is a referral hospital for pulmonary malignancies, with an annual case load of approximately 50–70 operated lung cancers and can be considered a low- to medium-volume hospital depending on the perspective.8 –10 The hospital has a mixed cardiothoracic practice, where both cardiac and general thoracic procedures are performed by the same surgeons. Oulu University Hospital established an RATS program in late 2021, and two mixed-practice surgeons were trained to transition to the RATS program. There are only a few studies about transitioning from VATS to RATS in low-volume centers and none involving mixed practice surgeons.11,12 Similarly, to our knowledge, there have not been studies assessing if a surgeon’s operative background experience affects the safety and efficacy of transitioning to RATS, even though the potential impact of prior surgical experience has been considered in studies following a single-surgeon design.7,11,13
The aim and primary outcome of this study was to compare the learning curves of two surgeons with nearly identical experience in cardiac surgery while differing in their predominant prior experience in thoracic surgery: one with a background primarily in open thoracic surgery and the other in video-assisted (VA) thoracic surgery.
Methods
The study is a retrospective cohort study conducted at Oulu University Hospital. The cohort included all patients that underwent either lobectomy, bilobectomy, or segmentectomy during the study period from 1 January 2015 to 31 May 2023 in Oulu University Hospital. Patients who were operated on with wedge resections or pulmectomy were excluded. When analyzing lymph node yield and completeness of systematic lymph node dissection, metastasectomy cases were also excluded. The study was approved by the local hospital district and Northern Ostrobothnia Ethics Committee (EETTMK 5/2019). Due to the retrospective and registry-based nature of the study, the Northern Ostrobothnia Research & Ethics Committee provided a data permit that waived individual patient consent.
The RATS program was established in October 2021 and two surgeons, open thoracotomy (OT) and VA, were trained as RATS surgeons. Patients from October 2021 to May 2023 were operated on with RATS only by these two surgeons. Before the establishment of the RATS program, surgeon OT, who achieved a cardiothoracic specialist degree in 2018, performed 51 anatomical lung resections excluding pulmectomies. Of these, 16 (30.1%) were VATS operated, and the rest were performed via open thoracotomy. Simultaneously, surgeon VA, who also received a cardiothoracic specialist degree in 2018, performed a total of 96 anatomical lung resections excluding pulmectomies, of which 61 (63.5%) cases were VATS operated. In this study, the RATS surgeons’ transition from their previously used technique to RATS was compared individually by using risk-adjusted cumulative sum (RA-CUSUM) analysis.
All patients were retrospectively screened from electronic hospital records of Oulu University Hospital. Survival data and causes of death were obtained from the national Causes of Death register with 100% coverage for the years 2015–2021. The missing mortality data (2022–2023) was completed from electronic hospital records, although these admittedly do not provide 100% coverage.
Tumor characteristics were collected from electronic patient records and pathology reports. All non-small cell lung cancer (NSCLC) cases were staged according to the eighth edition of the TNM Classification of Malignant Tumors of the International Association for the Study of Lung Cancer. 14
Modified Charlson comorbidity index (CCI) was used to quantify comorbidities. 15 Current lung cancer treatment, hypertension, chronic atrial fibrillation, or hyperlipidemia was excluded as comorbidities. Complications were categorized using the Clavien-Dindo complication classification. 16 Complications were divided into groups where class IIIa or more were considered as a major complication and class I–II as minor complications. Overall complications included all the Clavien-Dindo classes. Operative time was defined as the time between a surgeon doing the first incision and closure of the last incision.
Systematic lymph node dissection was defined as at least three N2-stations collected, and in short-term results, lymph node yield was assessed as a total number of lymph nodes collected as reported in pathological analysis. In RA-CUSUM analysis, a cut-off value of less than seven lymph nodes collected was defined as an unsatisfactory lymph node yield. All electronic surgical reports were evaluated for any major intraoperative complications or technical errors that would otherwise not be classified or registered in the Clavien-Dindo classification, and they were categorized as intraoperative complications. These included a major tear in vascular structures resulting in bleeding, an unintended airway perforation, or significant damage to adjacent organs that required corrective action even though these complications did not compromise patient outcome.
RATS was performed using the daVinci XI surgical system (Intuitive Surgical, Inc, CA, USA). The procedure was accomplished via two 12 mm and two 8 mm robotic arm ports and one additional AirSeal® (CONMED Corporation; FL, USA) 12 mm port which was used to create CO2 insufflation with a target pressure of 6 mm Hg; the port entry was later extended into a 4–6 cm incision to remove the surgical specimen at the end of the procedure. VATS procedures were performed via three or four 5–10 mm ports and an anterior 4–6 cm incision. Incisions of thoracotomies were either anterolateral or posterolateral. Either robotic or manual staplers were applied when dividing pulmonary vasculature and parenchyma or bronchi.
Data were processed with SPSS software (v. 29.0.0.0 for Windows; SPSS Inc, IL, USA). The frequencies of the preoperative, intraoperative, and postoperative outcomes are presented by the surgical technique or RATS surgeon. For continuous variables, the skewness was assessed with the Kolmogorov–Smirnov test. Variables that followed the normal distribution were analyzed with the Independent Samples T-Test. Variables that followed the skewed distribution were analyzed with the Mann–Whitney U-Test.
The RA-CUSUM curves were calculated and drawn for lymphadenectomy, systematic lymph node station harvesting (at least 3N2 lymph node stations), operation time, significant bleeding, and overall complication, formed using R version 4.0.3 and Microsoft Excel for Mac version 15.29.1 (Redmond, WA, USA). First, predicted risks of each outcome were calculated using the total data set of 460 patients using multivariable logistic regression with pre-specified variables. Risk scores for age (<70 years/⩾70 years), CCI (0, 1, or 2 or more), body mass index (BMI; <18, 18–30, or >30), FEV1 (<60%, or 60% or more), and DLCO (< 60%, or 60% or more) were calculated. If the FEV1 or DLCO values were not available due to lack of respiratory function testing prior to surgery, imputation was completed by assuming that the patient had sufficient respiratory function and was categorized as >60%. These individual risks for outcomes were used to assess expected outcomes in RA-CUSUM in RATS resections. For lymph node outcomes, only primary resections excluding metastasectomies were considered. The curves depict the cumulative difference between the observed and expected outcome on the y-axis for surgeons OT and VA, equated as Si = Si − 1 + (Σi − ΣR); S0 = 0. In the equation, Si is the cumulative sum, Σi is the sum of events at procedure number i, and ΣR is the sum of expected events at procedure number i. In the RA-CUSUM equation, observations exceeding the expected risk of the outcome increase and observations below the expected risk decrease CUSUM.
The RA-CUSUMs were expected to increase in the learning phase before a plateau. After a plateau, the events occurred less often than expected, and the curve descended. A stable situation showed no clear pattern. Learning was interpreted to be complete at the end of the plateau phase, where a clear learning-plateau-improved performance-pattern was observed.
The primary endpoints were the surgeons’ CUSUM curves for operative time, intraoperative blood loss, lymph node yield, systematic lymph node dissection, major postoperative complications, and overall postoperative complications. The secondary endpoints included intraoperative complications and bleeding, conversion, overall complications (Clavien-Dindo), length of hospital stay, 30-day mortality, and total lymph nodes collected.
Results
Patient characteristics are shown in Table 1. In the overall cohort, a majority of patients had primary lung cancer (92.6%), while the rest underwent anatomical lung resections for metastasectomy. The most common histological diagnoses were adenocarcinoma (52.0%) followed by epidermoidal cancer (21.3%), metastasis, carcinoid, and then small cell lung cancer.
Baseline characteristics of patients who had non-pulmectomy anatomical lung resection between January 2015 and June 2023 in Oulu University Hospital, divided into groups by using surgical technique.
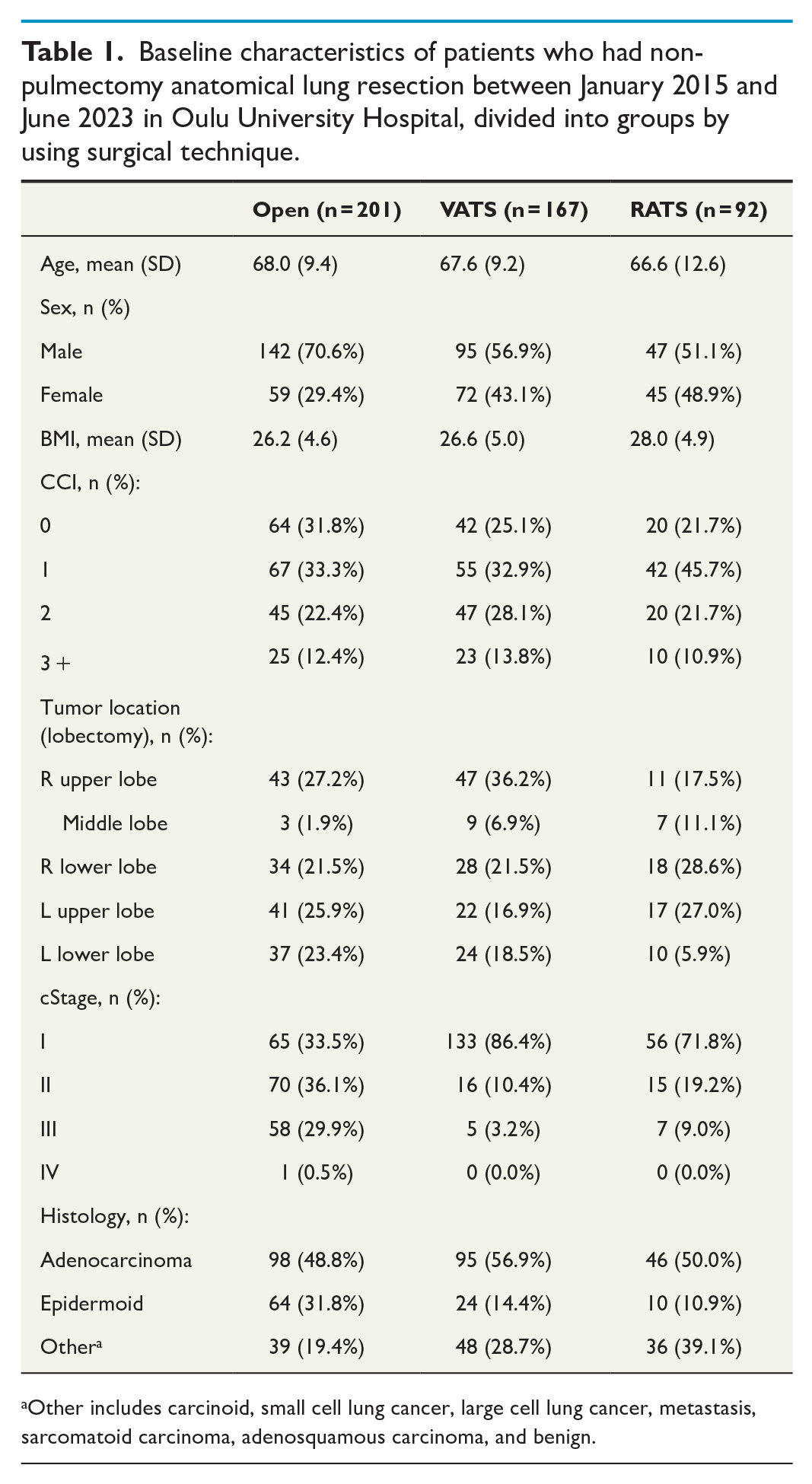
Other includes carcinoid, small cell lung cancer, large cell lung cancer, metastasis, sarcomatoid carcinoma, adenosquamous carcinoma, and benign.
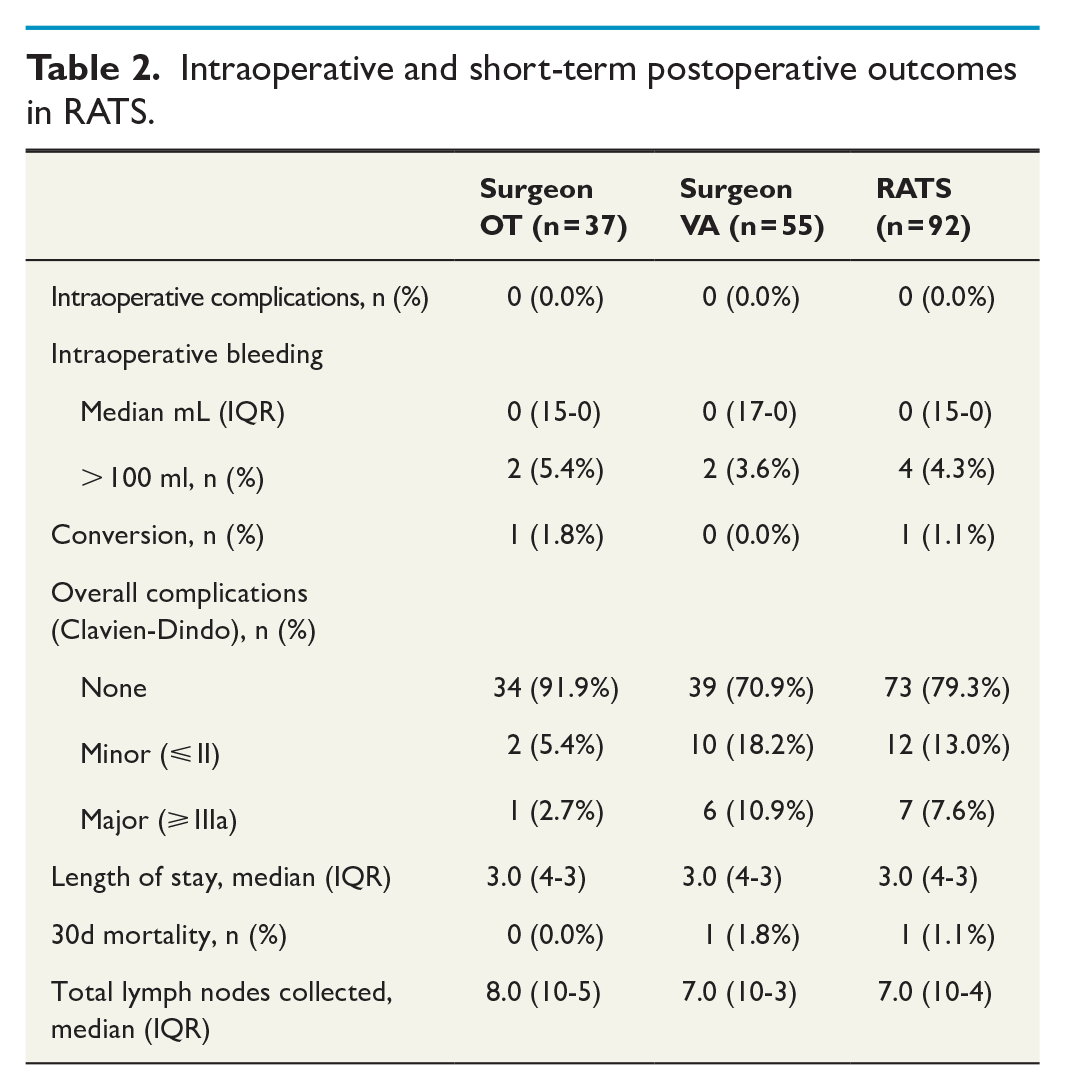
Intraoperative and short-term postoperative outcomes are presented in Table 2. No intraoperative complications were seen in the RATS group. The median intraoperative bleeding was 0 mL, and both surgeons had two cases where the intraoperative bleeding was more than 100 mL. There was only one conversion due to technical challenges caused by pleural adhesions. One patient needed a reoperation during their hospital stay and two others later (3.3%). The median length of the hospital stay was 3.0 (4.0–3.0) days. During the 30-day follow-up after surgery, one RATS patient (1.1%) died due to acute respiratory distress syndrome (ARDS).
Intraoperative and short-term postoperative outcomes in RATS.
For surgeons OT and VA, the main peak for the initial phase of the operative time was achieved after 30 and 15 cases, respectively (Fig. 1). The other phases were not as clearly definable, although for surgeon VA there was a non-monotonic curve until the 30th case followed by a slowly decreasing curve, representing the overcoming of the learning phase.
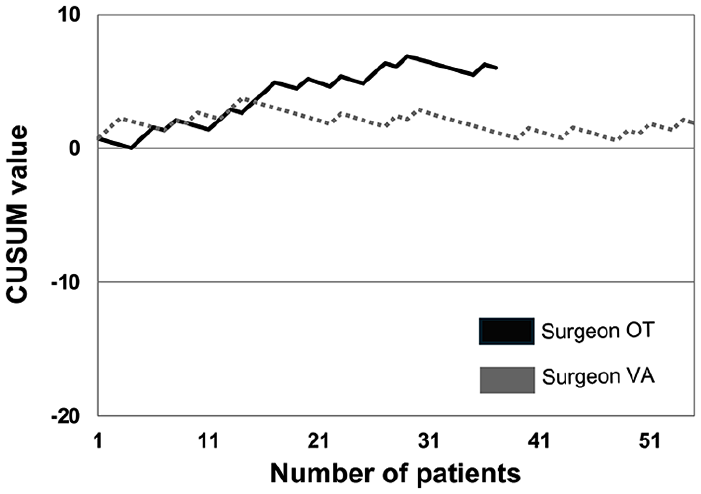
Risk-adjusted cumulative sum for operative time (overall) 75th percentile.
The CUSUM graphs for intraoperative bleeding showed similar trends between the surgeons OT and VA (Fig. 2). There was no clear peak for the initial phase, only a significant decrease in bleeding incidence since the start of the RATS program. Each surgeon had two peaks, the 27th and 37th procedures for OT and the 9th and 31st procedures for VA, that could be interpreted as isolated failures.
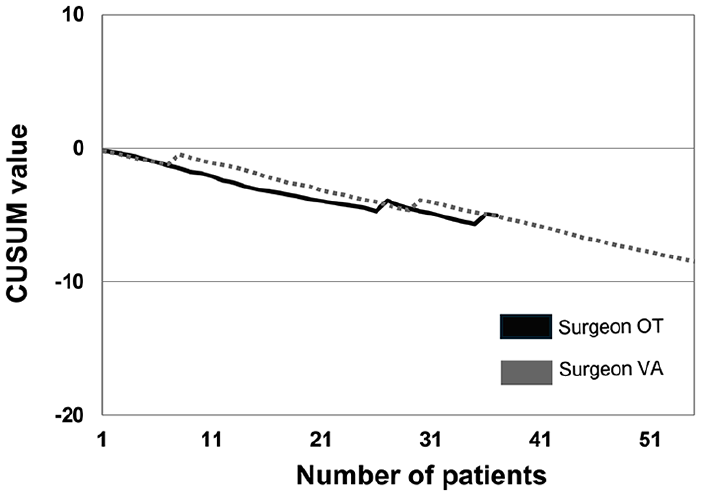
Risk-adjusted cumulative sum for intraoperative bleeding of more than 100 mL.
All three phases were identified from the CUSUM-LNY graphs for surgeons OT and VA: the initial phase until 23rd and 19th cases, which was followed by either a plateau phase for surgeon OT until the 30th case or a significant decrease in incidence for surgeon VA (Fig. 3a). After 37 cases surgeon VA’s graph showed a moderate increase.
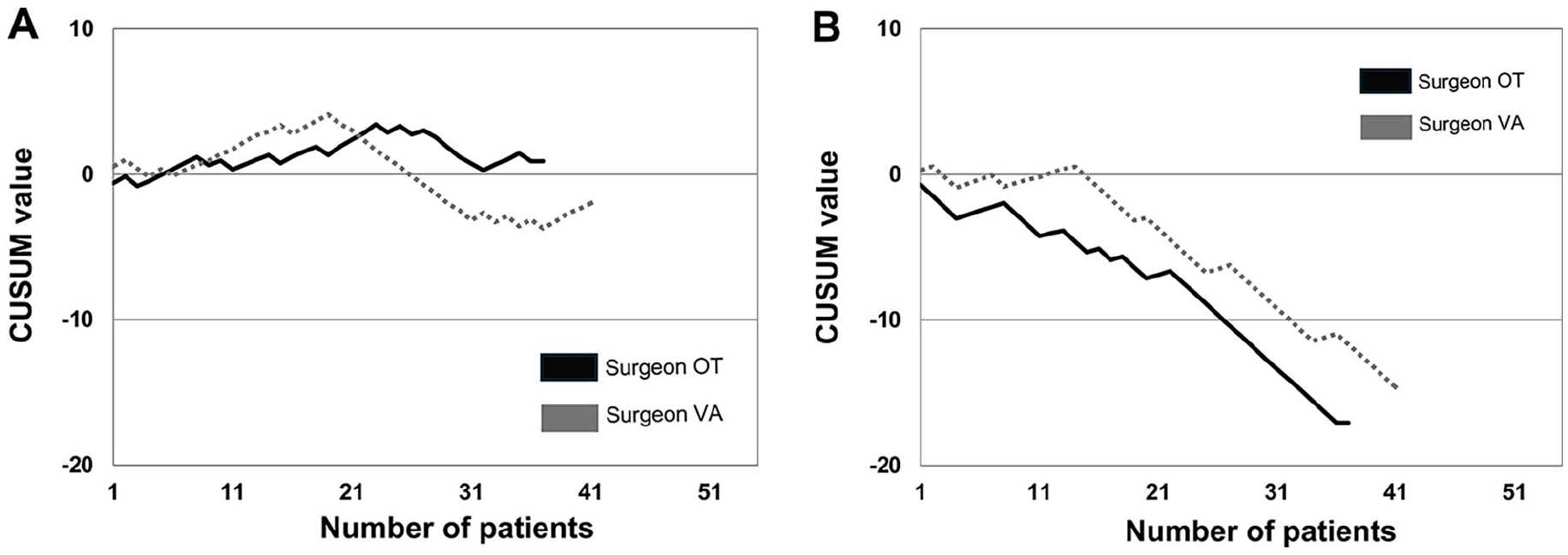
(a) Risk-adjusted cumulative sum for lymph node yield when operating on primary lung cancer. Satisfactory lymph node yield was defined as greater than or equal to eight. (b) Risk-adjusted cumulative sum for systematic lymph node dissection (at least three N2 stations sampled), when operating on primary lung cancer.
When implementing systematic lymph node dissection, surgeons OT and VA had a quite steady phase until the 8th and 14th cases, respectively, followed by a significant decreasing trend in CUSUM graphs (Fig. 3b).
There were no clearly definable phases for either surgeon when analyzing the major complications with the Clavien-Dindo classification scale (Fig. 4a). Surgeon OT had a single peak at the 9th case and surgeon VA at the 17th, 21st, 33rd, and 48th cases.
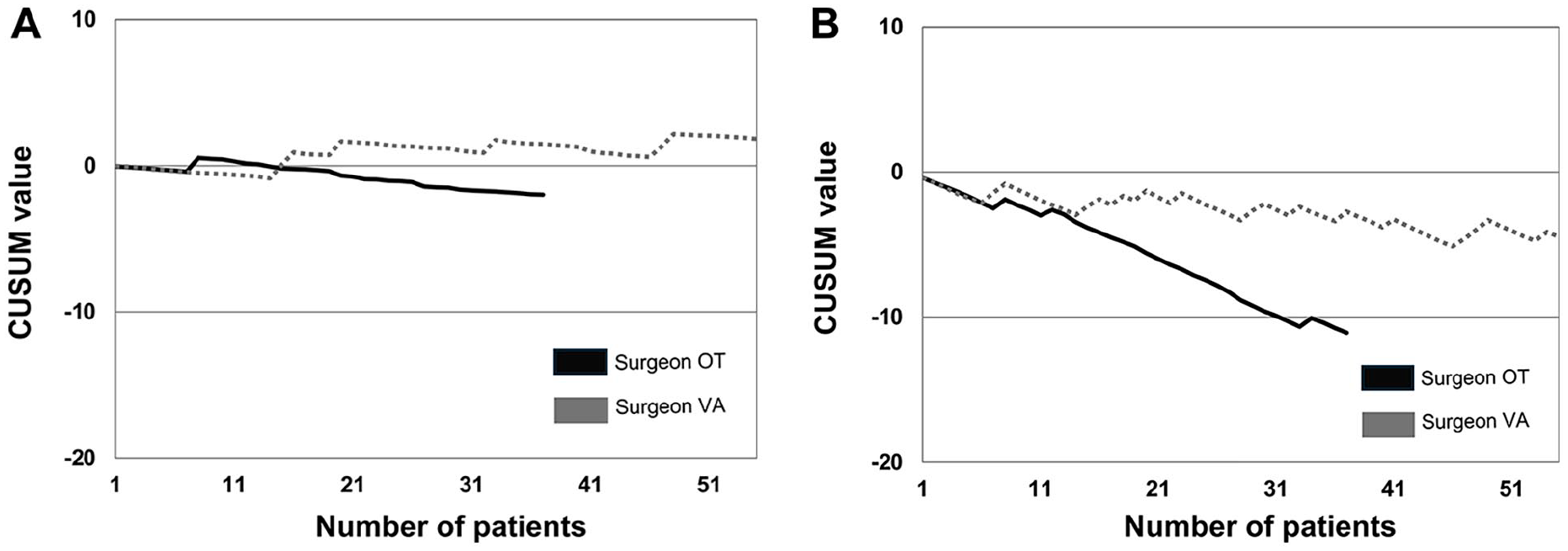
(a) Risk-adjusted cumulative sum for major complications using the Clavien-Dindo classification scale (IIIa and over is considered as major). (b) Risk-adjusted cumulative sum for overall complications.
The CUSUM curves for overall complications showed only decreasing curves; no other phases were identified (Fig. 4b). Surgeon VA had a steadily decreasing curve, whereas surgeon OT’s curve had only three peaks at the 9th, 12th, and 34th cases.
Discussion
Previous studies have not addressed if a surgeon’s background might affect the transition to operating with an RATS system. In this study, the learning curves for two surgeons were quite similar to each other despite one surgeon having significantly less VATS experiences, even though the surgeon with more thoracoscopic experience achieved the change point in some of the learning curves a few cases earlier than the surgeon OT. The most significant difference was seen in the operative time, where the change point was case number 15 for the surgeon with more VATS experience. This difference was most likely not clinically significant, as the real-time reduction was more pronounced on surgeon OT who had less VATS experience. Surgeon OT’s median operative time evolved from 189 (59.8) min to 144 (I62.0) min; change point after 30 cases, and surgeon VA’s from 158.0 (75.0) min to 167.0 (51.0) min; change point after 15 cases. The operative time was defined from the first incision until the final stitch. There was no statistically significant difference between the surgeons’ operation times, but the surgeon with more thoracoscopic experience reached the changing point in the learning curve faster (15 cases versus 30 cases), although the time reduction by definition was not clinically significant.
Among this study’s strengths is that both surgeons were of similar age with nearly identical surgical experience in the years before starting RATS surgery. The only major difference was that surgeon VA had more VATS experience. Furthermore, apart from surgeon VA performing more segmentectomies, there were no other differences in the procedure profile, even regarding the lobes being removed. Moreover, the cases were assigned between the surgeons without intentional allocation, and there was no substantial difference regarding the baseline characteristics such as age or cStage. There are also limitations, such as the limited sample size of individual surgeon cases, which can hide some selection bias that could affect the CUSUM curve. In addition, long-term follow-up is not included and could not have reasonably been done because of the inclusion of different types of cancers and some metastases.
In addition to operative time, lymph node yield is associated with more extensive and better intraoperative staging, and it is used as a surrogate for surgical quality. 17 The learning curves indicated that both surgeons achieved proficiency in collecting satisfactory lymph node yields and were able to steadily execute the systematic lymph node dissection since the start of the RATS program. This study is in line with a study by Zhou et al. 18 reporting that RATS was better than VATS for lymph node dissection.
It is important to acknowledge that the learning curve does not differentiate operational times between the removal of different lobes; lower and middle lobes can usually be considered technically more straightforward and therefore faster to operate, whereas upper lobectomies present a more complex anatomy. A recent study by Andersson et al. 11 showed decreasing operative time after 45 RATS cases with a median operative time of 178 min. In another study by Arnold et al., 19 the change point in the learning curve was achieved after 22 cases and the median operative time was 168 min, results in line with this study. Other studies indicate that operation times in RATS are longer when comparing operation times to thoracotomy or VATS.20 –22 The current study showed a similar trend with open thoracotomy being considerably faster than endoscopic procedures and VATS duration being slightly shorter than RATS.
The CUSUM graph for the major complications is quite even, which indicates that major complications (Clavien-Dindo ⩾ IIIa) are only individual cases in the RATS group. This is supported by the fact that when proceeding in mastering the new technique, more challenging cases are chosen to operate with RATS. There also were fewer overall complications in the RATS group than in the thoracotomy or VATS groups, although the significance of this can be debated, as minor Clavien-Dindo complications are very subjective and are easily affected by interpretation bias. Alternatively, major IIIa and upward complications are very objective by nature and not affected by the aforementioned bias. From the outset, both RATS surgeons had less intraoperative bleeding when comparing the intraoperative bleeding to the comparison cohort that included all thoracotomies and VATS. This finding may be due to multiple factors, such as the advantages of the robotic approach, the use of high-standard energy devices and staplers, or that the most complex cases are preferentially managed with an open approach. Reduced intraoperative bleeding together with the lower overall complication rate indicates that RATS is safe even with limited robotic experience.
According to our study, establishing an RATS program in a low-volume center with surgeons performing robotic-assisted lung cancer surgery with either open or thoracoscopic background experience is safe and does not compromise short-term outcomes.
Footnotes
Author contributions
Conceptualization and design: F.Y. Administrative support: F.Y. and T.M. Provision of study materials and patients: F.Y. and T.M. Collection and assembly of data: V.V., F.Y., T.M., and J.H.K. Data analysis and interpretation: all authors. Manuscript writing: V.V., F.Y., T.M., and J.H.K. Final approval of manuscript: all authors.
Data availability statement
The data analyses included in this article are available upon reasonable request to the corresponding author. However, due to the retrospective nature of this study, the study license (5/2019), and the Finnish healthcare data law, the raw data cannot be shared directly.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
