Abstract
Background:
The study aimed to analyze thyroid-stimulating hormone (TSH) levels quantitatively and investigate their potential correlation with the risk of incident atrial fibrillation (AF) in differentiated thyroid cancer (DTC) patients.
Methods:
DTC cases diagnosed between 1995 and 2015 in Stockholm, Sweden, were identified from the Swedish Cancer Registry. Medical records were scrutinized, and follow-up began 9 months post-surgery with tracking data until the earliest AF record, censoring, or 31 August 2022. TSH values were classified as unsuppressed (TSH > 0.5 mE/L), mildly suppressed (TSH 0.1–0.5 mE/L), or suppressed (TSH < 0.1 mE/L), with graphical analysis spanning up to a 10-year follow-up period. In addition, a nested case–control study assessed the impact of TSH category on incident AF. Additional data on cardiovascular risk factors were gathered.
Results:
Among 608 patients, approximately 78% maintained suppressed TSH levels for over half of their follow-up time. Notably, there was a decrease in the proportion of patients receiving long-term TSH suppression after 2013. Among 39 newly diagnosed AF cases, most were in the suppressed TSH category. Moreover, about half of these new AF patients had established cardiovascular risk factors prior to DTC diagnosis.
Conclusion:
DTC patients generally adhered to TSH suppression guidelines, with a decline observed in the proportion of suppressed TSH values following the adoption of individualized treatment in 2013. The study could not establish a clear link between TSH suppression and the risk of incident AF, highlighting the need for further investigation.
Context and Relevance
This study investigates the potential link between TSH suppression and the risk of AF in patients with DTC. Given that DTC treatment often involves TSH suppression to prevent recurrence, understanding its impact on cardiovascular health is important. The study finds that most patients adhered to TSH suppression guidelines, and a decline in suppression rates occurred after 2013. No clear connection between suppressed TSH levels and incident AF was, however, established.
Introduction
Differentiated thyroid cancer (DTC) accounts for over 90% thyroid cancer cases. 1 Patients with DTC have a favorable prognosis, with a 10-year overall survival rate exceeding 90%. 2 The standard treatment for DTC involves surgical removal of the thyroid gland, usually through a total thyroidectomy or a hemithyroidectomy in selected cases. Based on the estimated risk of recurrence,3,4 patients may additionally receive adjuvant radioactive iodine treatment (RAI). They will also require lifelong thyroid hormone replacement therapy with synthetic T4, levothyroxine, due to loss of the thyroid gland. To prevent stimulation of potential remnant cancer cells by thyroid-stimulating hormone (TSH), levothyroxine can be administered in doses that suppress TSH levels,1,4–6 leading to subclinical hyperthyroidism as a common side effect of the treatment.
Subclinical hyperthyroidism can potentially lead to adverse side effects such as osteoporosis 7 and AF.8–12 The increased risk of AF may be mediated by physiological changes, such as an elevation of the heart rate, increased myocardial contractility, and enlarged left ventricular size.13–19 In Sweden, lifelong TSH suppression was standard practice until the introduction of national guidelines in 2012 (further revised in 2017 and 2021) that emphasize individual customization of TSH suppression with regard to the risk of cancer recurrence (TSH < 0.1 mE/L for “high-risk” DTC, TSH: 0.1–0.5 mE/L for “intermediate-risk” DTC and TSH > 0.5 mE/L for “low-risk” DTC) and underlying cardiovascular status. 20 The existing literature on compliance to TSH suppression treatment is limited, as previous studies have used selective data with relatively short follow-up periods. 21
Cardiovascular disease among DTC patients, especially AF, is a clinical concern raised in several recent meta-analyses.22–24 Less attention has been paid to studying the connection between TSH values and adverse cardiovascular outcomes. One study showed that high doses of prescribed levothyroxine were associated with an increased risk of AF, 25 and another publication showed an association of suppressed TSH levels (<0.1 mE/L) with increased risk in mortality due to AF. 26 There is also research performed on the association between TSH levels and overall survival in DTC patients.27–29 Currently, there is no comprehensive investigation of the relationship between TSH suppression and AF incidence in a large, unselected DTC population examined over an extended follow-up period. More specifically, how the degree and duration of suppressed TSH levels affect the risk of cardiovascular disease in DTC patients.
In this population-based cohort study, the first aim was to evaluate TSH levels and their variations over time. Furthermore, the second aim was to investigate the association between the extent of TSH suppression and the risk of incurring AF in individuals treated for DTC.
Methods
Study population
The Swedish healthcare system functions through public funding, and each resident is assigned a unique personal identity number. This system facilitates the organization of medical records and establishes comprehensive nationwide registries. The registries include a range of information, such as hospital discharge diagnoses, cancer diagnoses, and the cause of death, on regional and population levels. Patients with DTC in the Stockholm region diagnosed between 1995 and 2015 who underwent a total thyroidectomy, 20 years or older at diagnosis, were identified through the national Swedish Cancer Registry (n = 1031). Cases of DTC microcarcinoma, misdiagnoses (anaplastic thyroid cancer, medullary thyroid cancer, poorly DTC, and benign tumors) and cases with insufficient information from medical records (such as missing operation date and follow-up information) were excluded. Medical records were scrutinized to collect information on patient-, disease-, and treatment characteristics, TSH values, and occurrence of AF. Baseline information on cardiovascular status was collected from the anesthesiologic assessment prior to total thyroidectomy. For newly registered AF cases, the status (yes/no) for the following disease-specific risk factors was also registered: hypertension, diabetes mellitus type 1 or 2, ischemic heart disease, peripheral critical vascular disease, cerebrovascular disease, and valvular heart disease.
TSH variability
To assess TSH variability, all registered TSH values from the date of surgery up to the 31st of August 2022 were retrieved. TSH values above 20 mE/L were excluded as they were stimulated by recombinant TSH. For analyses of TSH variability (not connection of TSH to AF), values were restricted from 9 months up to 10 years after the surgery date. All available TSH values were categorized into three levels based on the Swedish national guidelines: 20 “unsuppressed” (TSH < 20 mE/L and TSH > 0.5 mE/L), “mildly suppressed” (TSH 0.1–0.5 mE/L), and “suppressed” (TSH < 0.1 mE/L). Patients were assigned to one of the three TSH categories in which most of their follow-up time was recorded (more than 50%). This allocation was determined based on a series of TSH values, where patients stayed in a category until the next recorded TSH value during the follow-up period. The rationale behind this decision rule was to consider the variability of individual compliance with levothyroxine medication and the physician assessment of altered TSH targets. TSH values of each patient over the follow-up time were depicted in a spaghetti plot. The TSH categories for all TSH values were presented in two separate bar charts organized by follow-up time (regardless of year of diagnosis). The first bar chart encompassed TSH values registered before the introduction of national guidelines (1995–2012), and the second presents TSH values after the introduction of national guidelines (2013 or later).
AF and TSH
Baseline information on cardiovascular status was collected from the anesthesiologic assessment conducted prior to the total thyroidectomy operation. Follow-up on incidence in electrocardiogram (ECG) confirmed AF commenced 9 months after surgery and ended at the first record of AF, censored at the last retrievable medical record with cardiovascular status, or the 31st of August 2022, whichever occurred first.
All patients that developed AF during follow-up were included as cases. To assess the association between TSH suppression and the risk of AF, a nested case–control analysis was performed. For each case of AF, two controls were sampled among individuals who were still at risk of AF at that time point since diagnosis (i.e. matched by time since diagnosis or incidence-density sampling) and matched on age group at diagnosis (based on quartiles). The cases and controls were categorized based on which TSH category most of their follow-up time was recorded, as described above, but only counting time until the time of event or matching. The TSH categories were then compared between cases and controls by a conditional logistic regression, estimating hazard ratios (HR).
Ethics
This retrospective study has been approved by the Regional Ethical Board at Karolinska Institutet (Stockholm, Sweden), Dnr: 2014/714-31. It is not a clinical trial but a retrospective descriptive study, and according to the Swedish regulations, the need for informed consent was waived.
Results
Patient characteristics
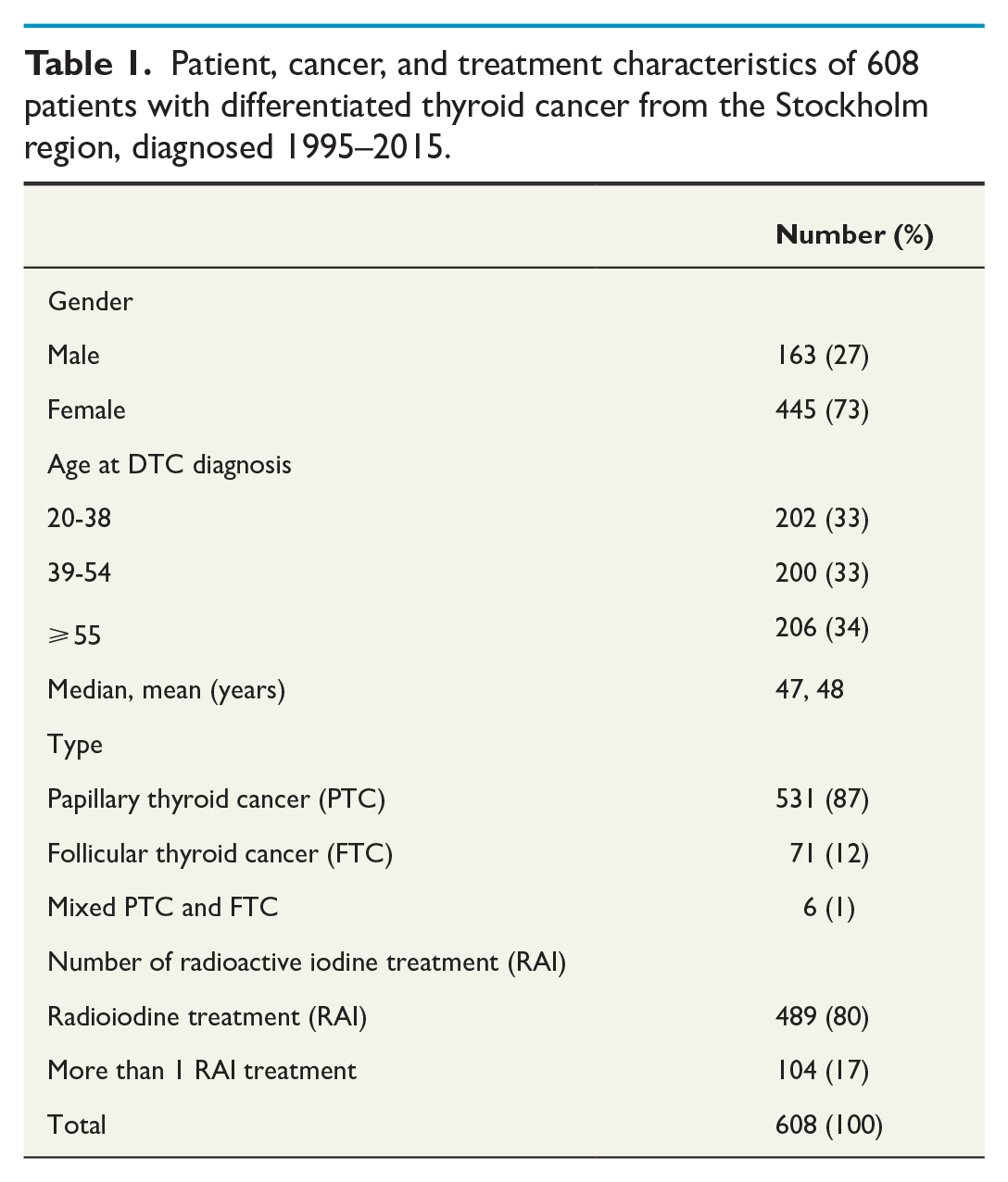
There were 1031 cases of DTC identified from the Swedish Cancer Registry. Out of these, the following cases were excluded: anaplastic thyroid cancer (n = 7), poorly DTC (n = 8), medullary thyroid cancer (n = 4), microcarcinoma (n = 133), misclassified benign disease (n = 27), cases where surgery was not performed (n = 8), medical records with incomplete information (n = 196), death within 9 months after surgery (n = 2), patients with a hemithyroidectomy as the only surgical intervention (n = 37), and finally one patient with missing TSH values (Fig. 1). The final cohort of 608 patients consisted of 531 (87%) patients with papillary thyroid cancer, 71 (12%) patients with follicular thyroid cancer, and six patients with mixed papillary and follicular cancer (Table 1). The vast majority (80%) was treated with radioactive iodine therapy (RAI) at least once, and 17% received RAI more than once. The median accumulated RAI dose was 3700 megabecquerel (MBq) with doses ranging from 800 MBq to 51,800 MBq. In total, 106 patients were treated with RAI and 81 with surgery due to cancer recurrence.
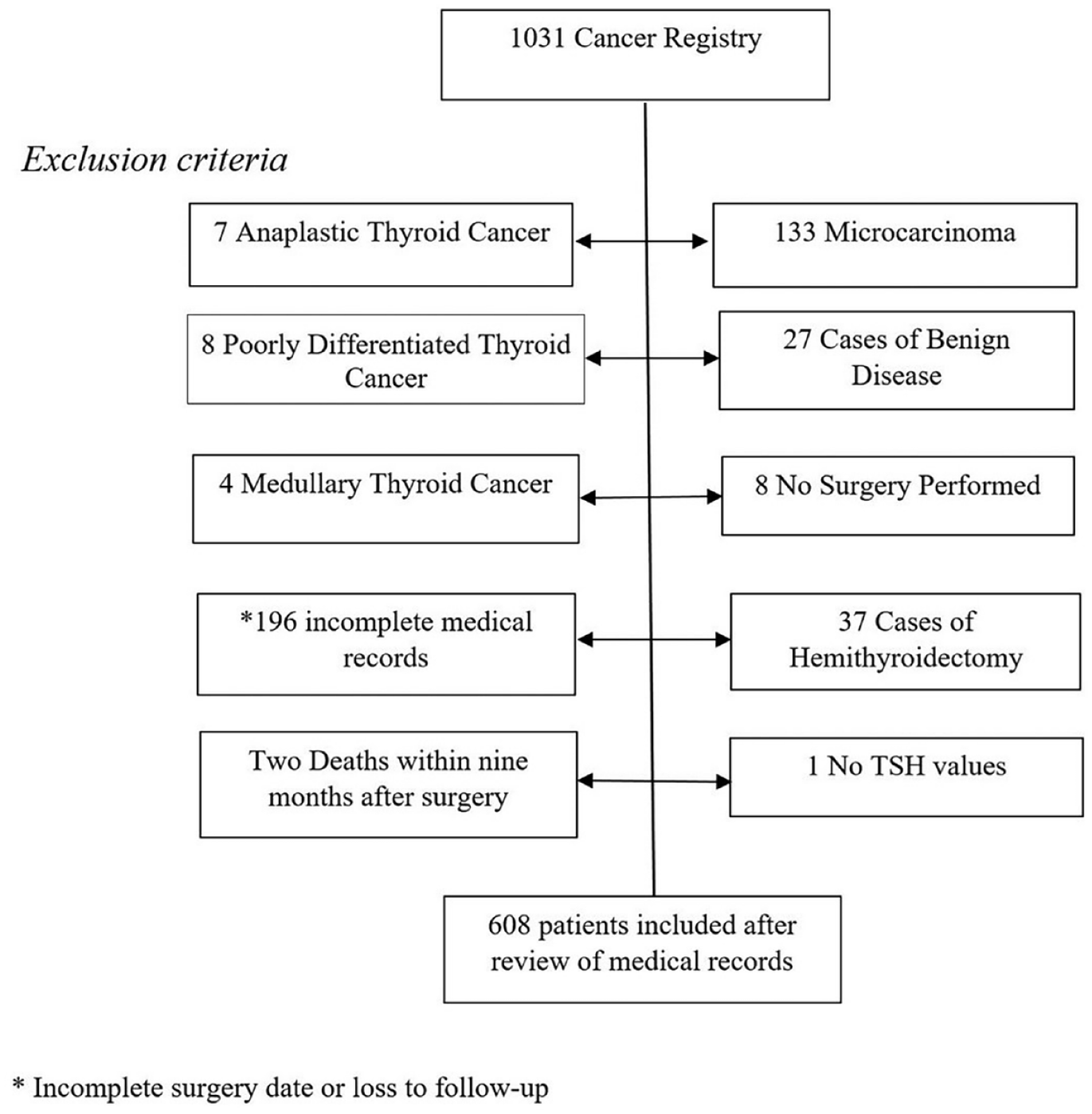
Flowchart outlines the exclusion criteria applied to 1031 patients with differentiated thyroid cancer identified in the Stockholm region from 1995 to 2015 using the Cancer Registry. Following the review of medical records, 608 cases were included in the study.
Patient, cancer, and treatment characteristics of 608 patients with differentiated thyroid cancer from the Stockholm region, diagnosed 1995–2015.
TSH values
Regarding analyses of TSH values, 606 individuals were included. Two individuals were excluded; one had only TSH values above 20 mE/L, and the other had only TSH values recorded 10 years after DTC diagnosis. During follow-up, 6810 (69%) registered TSH values were less than 0.1 mE/L, 1844 (19%) between 0.1 and 0.5 mE/L and 1208 above 0.5 mE/L (12%). About 78% of all patients were suppressed for at least half of their follow-up time. The maximum registered TSH value was on average 3.33 mE/L. Individual patient TSH values over time are depictured in a spaghetti plot (Fig. 2). The majority of the patients had suppressed TSH levels, which is illustrated by the density toward TSH levels of 0 on the y-axis. The pattern of TSH levels over time was similar for both sexes and for different age groups at DTC diagnosis (data not shown).
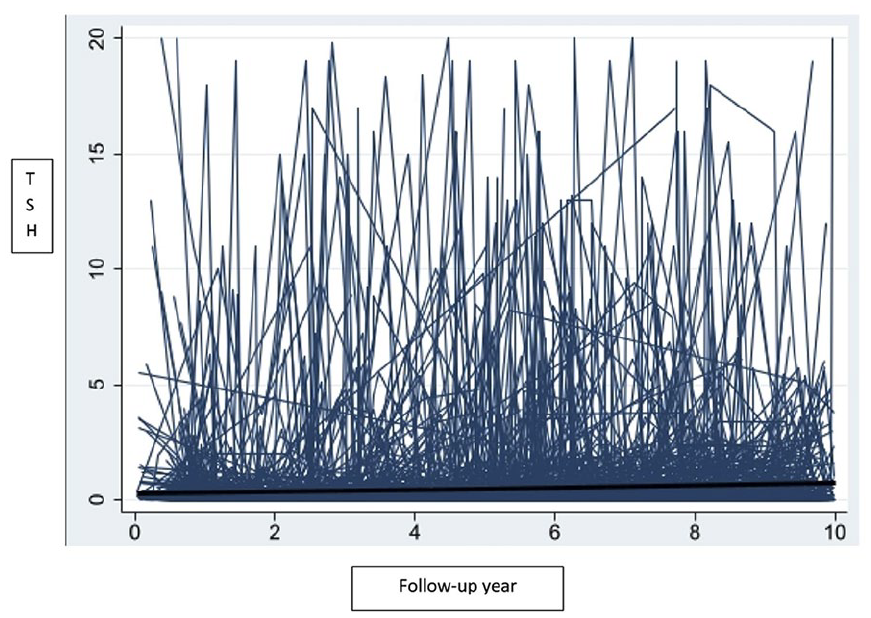
Spaghetti plot of thyroid-stimulating hormone (TSH) values (mE/L) over follow-up time in years (starting 9 months after surgery date) for 606 individual patients with differentiated thyroid cancer diagnosed 1995–2015 in the Stockholm region.
All registered TSH values are classified into the three TSH categories (“suppressed” TSH < 0.1 mE/L, “mildly suppressed” TSH: 0.1–0.5 mE/L, and “unsuppressed” TSH > 0.5 mE/L). These TSH categories were further categorized and graphically visualized by follow-up time in years, in two distinct time periods (Fig. 3).
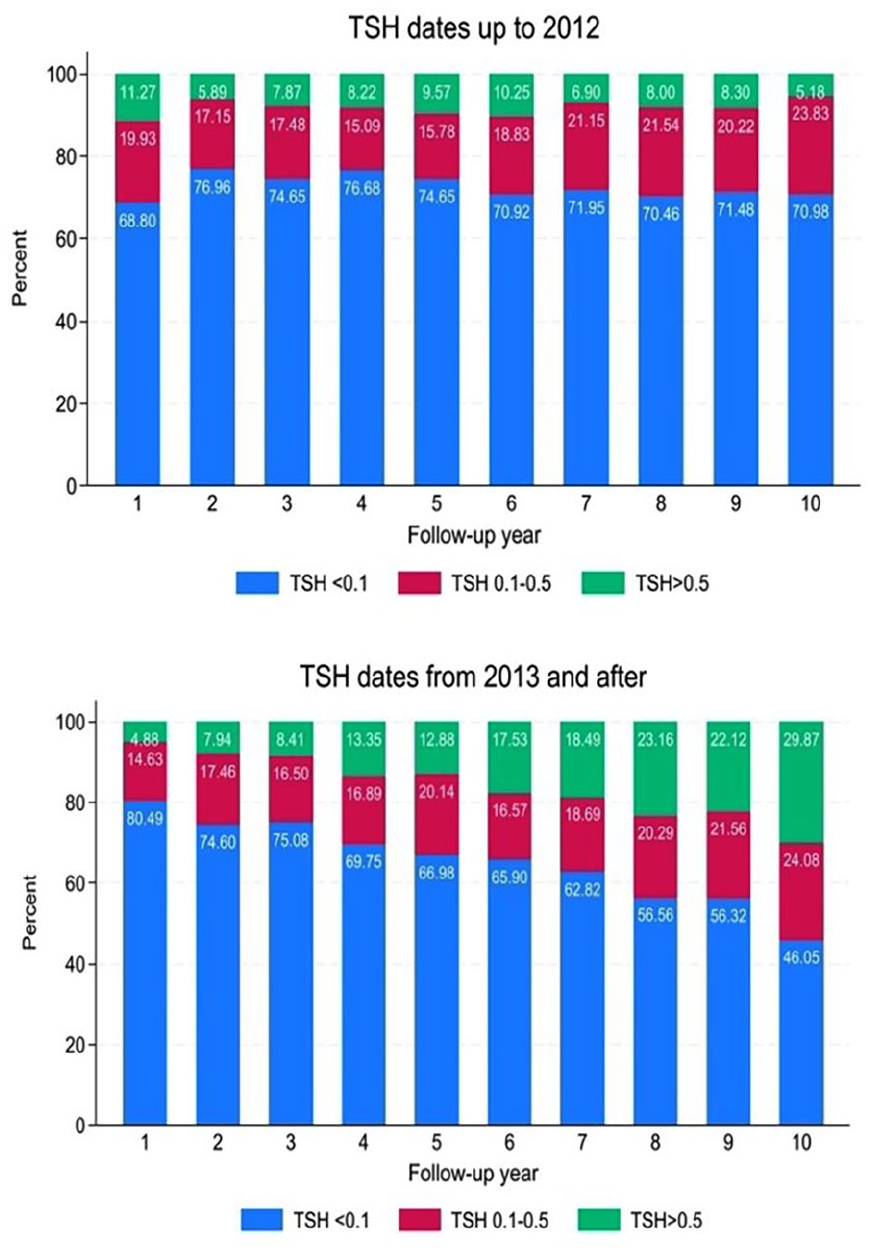
Proportion of thyroid-stimulating hormone (TSH) values in the categories (mE/L): “suppressed” (TSH < 0.1 mE/L), “mildly suppressed” (TSH: 0.1–0.5 mE/L), and “unsuppressed” (TSH > 0.5 mE/L) over follow-up year (commencing 9 months after surgery) for 606 individual patients with differentiated thyroid cancer diagnosed 1995–2015 in the Stockholm region.
TSH categories measured 1995–2012
During the first period involving TSH measurements prior to implementation of national guidelines (1995–2012), 5858 TSH values were registered. The “suppressed” category was consistently dominant for each calendar year, comprising between 69% and 79% throughout the entire 10-year follow-up. The second most prevalent TSH category, “mildly suppressed,” exhibited variations between 15% and 24% over the 10-year follow-up period. The “unsuppressed” category, being the least common, demonstrated the smallest proportion of TSH values, ranging from 6% to 11% across all 10 follow-up years. Throughout the 10-year follow-up period, the proportions of these three TSH categories remained relatively stable, displaying no significant increase or decrease.
TSH categories measured from 2013 onward
For TSH values measured in 2013 or later, 4004 TSH values were registered. The “suppressed” category remained the most common throughout the 10-year follow-up period; however, its proportion steadily declined from 80% in the first follow-up year to 46% in the 10th follow-up year. The “mildly suppressed” category varied from 15% in the first follow-up year to 24% in the 10th follow-up year, consistently ranking as the second most prevalent TSH category for the initial 5 follow-up years. In contrast, the “unsuppressed” category exhibited a gradual increase from 5% in the first follow-up year to 30% in the 10th follow-up year, becoming the second most prevalent category during the 5th to 10th follow-up years.
Risk of AF
During follow-up, 39 patients were diagnosed with AF, 30 of them belonged to the “suppressed” category (TSH < 0.1 mE/L), seven to the “mildly suppressed” category (TSH: 0.1–0.5 mE/L), and two individuals were assigned to the “unsuppressed” category (TSH > 0.5 mE/L) based on how their follow-up time was categorized. In the nested case–control study, 76 controls were selected, out of which 55 were categorized into the “suppressed” category. In the conditional logistic regression, where the “suppressed” category served as the reference group, the HRs of AF were not statistically significant, and the confidence intervals (CIs) were very wide. For the mildly suppressed category, the HR was 0.82 (95% CI = 0.29–2.31), and for the “unsuppressed” category, the HR was 0.41 (95% CI = 0.077–2.22). Cases with AF diagnosed during follow-up were on average 67 years old at DTC diagnosis and 75 years old at the time of AF. Furthermore, about half of these (19/39) had at least one of the following risk factors for AF prior to DTC diagnosis: hypertension (18/39), diabetes mellitus type 1 or 2 (3/39), ischemic heart disease (1/39), peripheral critical vascular disease and/or cerebrovascular disease (4/39), or valvular heart disease (2/39).
Discussion
This study provides a comprehensive analysis of the evolving TSH patterns during follow-up and explores the association between TSH levels and incident AF in patients diagnosed with DTC from 1995 to 2015. For TSH values recorded before 2013, the distribution of TSH patterns remains static over the 10-year follow-up period, with approximately 70% of measured TSH values classified as “suppressed,” 20% as “mildly suppressed,” and 10% as “unsuppressed.” Following the implementation of national guidelines effective since 2013, which prescribe levothyroxine to achieve individualized TSH targets based on factors such as cancer treatment response and patient-specific risk factors, notable variations in TSH patterns become evident. While “suppressed” remains the most prevalent category, its proportion steadily decreases from 80% in the 1st follow-up year to 45% in the 10th follow-up year. In contrast, the proportion of “unsuppressed” TSH values steadily increases from 5% to 30% during the same follow-up period. Importantly, these observed variations align with established treatment guidelines, supporting a reasoned inference. Despite occasional substantial deviations in individual TSH measurements from their respective targets, patients consistently demonstrate adherence to and alignment with national guidelines throughout the course of treatment.
Despite a long follow-up period and a sizable patient cohort, we were unable to draw definitive conclusions regarding TSH suppression and AF. This could be explained by the limited number of incident cases of AF but most of all due to the low variation between TSH categories, as over 70% of patients belonged to the suppressed category during follow-up. It is known that patients with DTC are at a higher risk of mortality and incidence in AF, which is most likely a consequence of TSH suppression, but a direct association has in most studies not been established9,10,12,22–24 except for one case–control study. 26 That study found that DTC patients with mean TSH levels < 0.1 Mu/L during follow-up had an increased risk of incurring AF compared with individuals without thyroid cancer matched by age, gender, and geographical location. Crucially, this conclusion did not account for the TSH values of the control group nor did it consider the duration of exposure within specific TSH categories. In our nested case–control study, the exposure status of TSH was defined by a different method, and the control patients were selected from the same population-based cohort as the cases. These methodological differences make it difficult to compare the studies, and with the previously discussed low variation in TSH exposure, we were not able to draw a conclusion regarding TSH suppression and the risk of AF.
Complementary analyses in our study revealed that about half of the patients who developed AF, had a pre-existing risk factor for AF prior to DTC diagnosis, and their average age at DTC diagnosis were higher compared to the rest of the cohort. These findings suggest that older patients with established cardiovascular risk factors are the ones at risk of developing AF. This rationale could help guide the individualization of TSH suppression treatment based on the aggressiveness of the cancer, underlying cardiovascular comorbidities, and age.
The regional population-based cohort study, characterized by an unselected sample, long follow-up period, and large patient material, provided several strengths that enhanced the reliability and generalizability of the findings. By including an unselected, population-based patient group, we were able to increase the external validity of our study and improve the representativeness of the results. In addition, the long follow-up period allowed us to track the incidence in AF over an extended period, which provided valuable insights into trends that may not have been apparent in studies with shorter follow-up. There are, however, two identifiable weaknesses in this study, namely the retrospective approach and the accuracy of medical records. It is important to interpret the dates of AF diagnoses with caution since these were the first records mentioned in the public medical record, potentially in some cases excluding earlier registrations from private caregivers (which, during the studied period and currently, constitute a minority in Sweden). Furthermore, it is known that patients may experience non-symptomatic episodes of AF, which may lead to a delayed or missed diagnosis.
This nested case–control study highlights the methodological challenge in establishing a potential association between TSH suppression treatment and AF. Given the widespread prevalence of TSH suppression in most patients, the reduced statistical variability of the independent variable mathematically diminishes the probability of capturing potential associations. Probably, if this study was to be repeated among patients diagnosed after the introduction of national guidelines (2012), a higher proportion of the cohort would be non-suppressed patients, leading to a higher variation of the exposure status. A randomized controlled trial would by construction capture more variability in TSH suppression categories but would face other issues. First, there was a long duration between DTC diagnosis and AF incidence, which in this study was approximately 7 years. Second, the TSH suppression treatment is proven to prevent cancer recurrence, at least in high-risk DTC,30,31 and a treatment randomization would therefore raise ethical issues. Possibly, pooling multi-center data could solve some of the above-mentioned challenges. Notably, the incidence of new cases of AF was limited, and no distinct association with TSH suppression treatment could be conclusively established.
DTC patients adhere to TSH suppression, and prior to 2013, TSH values were largely suppressed for extended follow-up durations. Following the implementation of individualized treatment in alignment with the 2013 national guidelines, the proportion of suppressed TSH values decreased over time. No definitive conclusions could be drawn on how TSH suppression affects the risk of incident AF.
Footnotes
Authors’ Note
This research paper is a revised version included in the corresponding author’s PhD dissertation. 32
Author contributions
M.Z.: Conceptualization of the study, methodology, responsible for data collection, conducted software analyses, results analyses, took the lead in writing the original draft, took the lead in the editing process of the manuscript, and took the lead in the review process. T.M.-L.A.: Conceptualization of the study, methodology, contributed to data collection, took the lead in software analyses, results analyses, contributed to writing the original draft, and contributed to the review process. C.H.: Conceptualization of the study, methodology, contributed to analyzing results, contributed to writing the original draft, and contributed to the review process. C.N.: Conceptualization of the study, methodology, contributed to analyzing results, contributed to writing the original draft, and contributed to the review process. C.I.L.: Conceptualization of the study, methodology, data collection, results analyses, contributed to writing the original draft, contributed to the review process, and main supervision of the project.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by grants from the Swedish state under the agreement between the Swedish government and the county councils, Foundation Stiftelsen för Kirurgiskt samarbete, Grönbergs Foundation, and the Swedish Society of Medicine.
