Abstract
Complicated colonic diverticulitis remains a frequent cause of hospital admissions in Western countries, associated with significant morbidity and health care utilization. Complications include abscess formation, perforation, sepsis, fistulas, and colonic stenosis, which may be present at initial presentation or develop during the disease course. Approximately 10%–20% of hospitalized diverticulitis cases are classified as complicated. Over recent decades, the management of this condition has undergone a paradigm shift—from routine surgical intervention to more individualized, evidence-based strategies emphasizing conservative treatment where appropriate. This review provides a comprehensive, clinically oriented summary of current diagnostic approaches, including the role of cross-sectional imaging, as well as non-operative and operative treatment options. It also outlines recommendations for follow-up, including indications for colonoscopy, and discusses ongoing controversies and future directions in the management of complicated diverticulitis.
Keywords
Introduction
By the age of 80, colonic diverticulosis affects up to 70% of individuals in Western populations.1,2 Symptomatic disease develops in approximately 4%–5% of those with diverticulosis, and complicated diverticulitis accounts for 10%–20% of hospitalized cases, posing a considerable burden on surgical services.3,4 The surgical management of complicated diverticulitis was first described by Mayo and colleagues in a case series in the early 20th century. 5 Historically regarded as a primarily surgical condition, complicated diverticulitis encompasses both acute presentations—such as abscesses, contained perforations, and free perforations into the abdominal cavity—and chronic manifestations, including fistulas and obstructions.
The Hinchey classification, based on intraoperative findings, traditionally guided surgical decision-making by dividing perforated diverticulitis into four stages. However, advances in radiologic imaging have enhanced diagnostic accuracy and introduced new non-operative treatment options. Consequently, most patients with complicated diverticulitis can now be managed without surgery. This paradigm shift has led to the development of numerous clinical and computed tomography (CT)-based classification systems. 6
In Scandinavia, a subset of these classifications is commonly applied in clinical practice (Table 1). This review provides a comprehensive, evidence-based, and clinically oriented overview of current diagnostic strategies, treatment options, and follow-up recommendations for complicated diverticulitis. For clarity, we have refrained from numbering stages and instead based our discussion on the widely accepted CT-based classification summarized in Table 2.
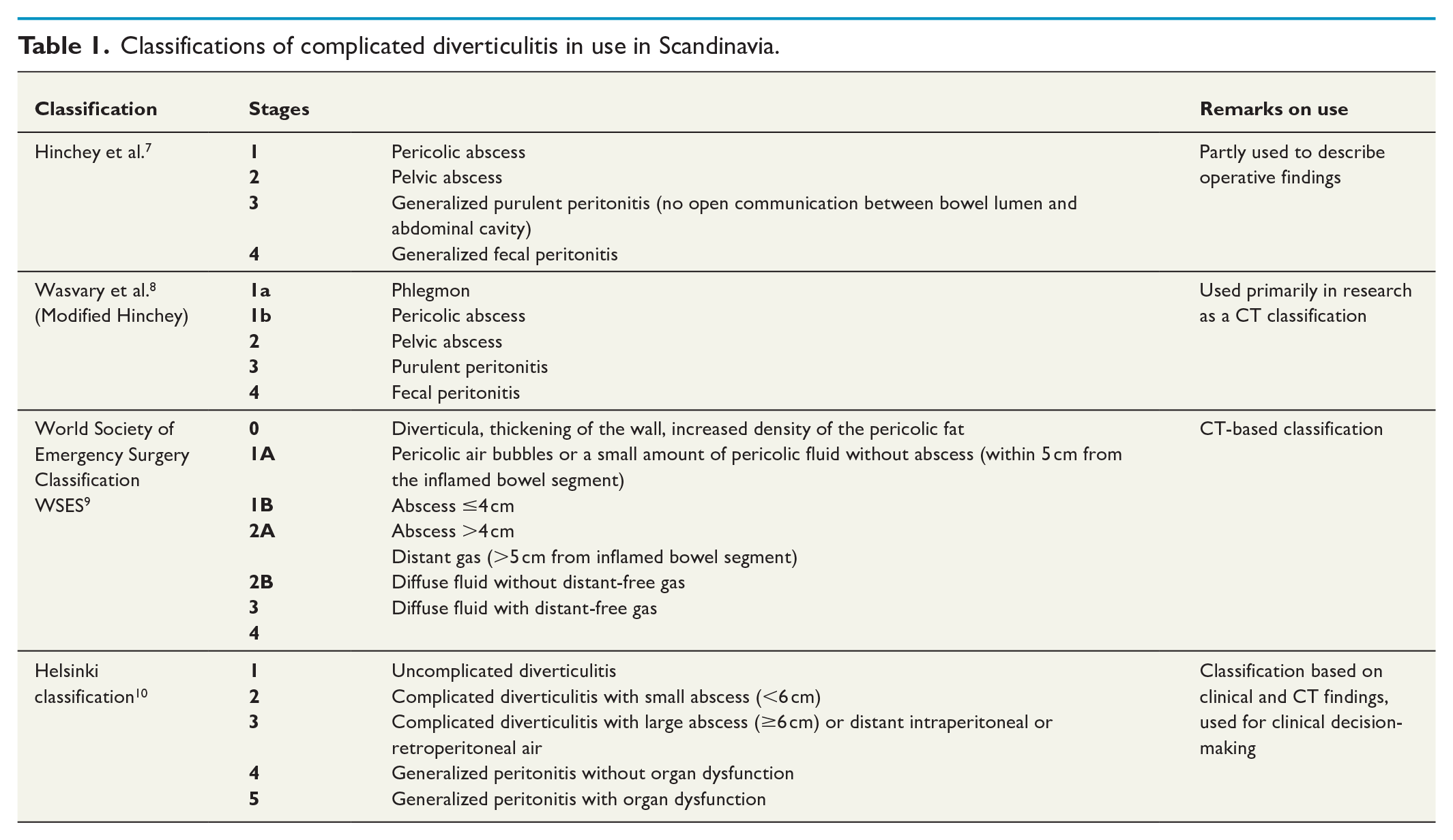
Classifications of complicated diverticulitis in use in Scandinavia.
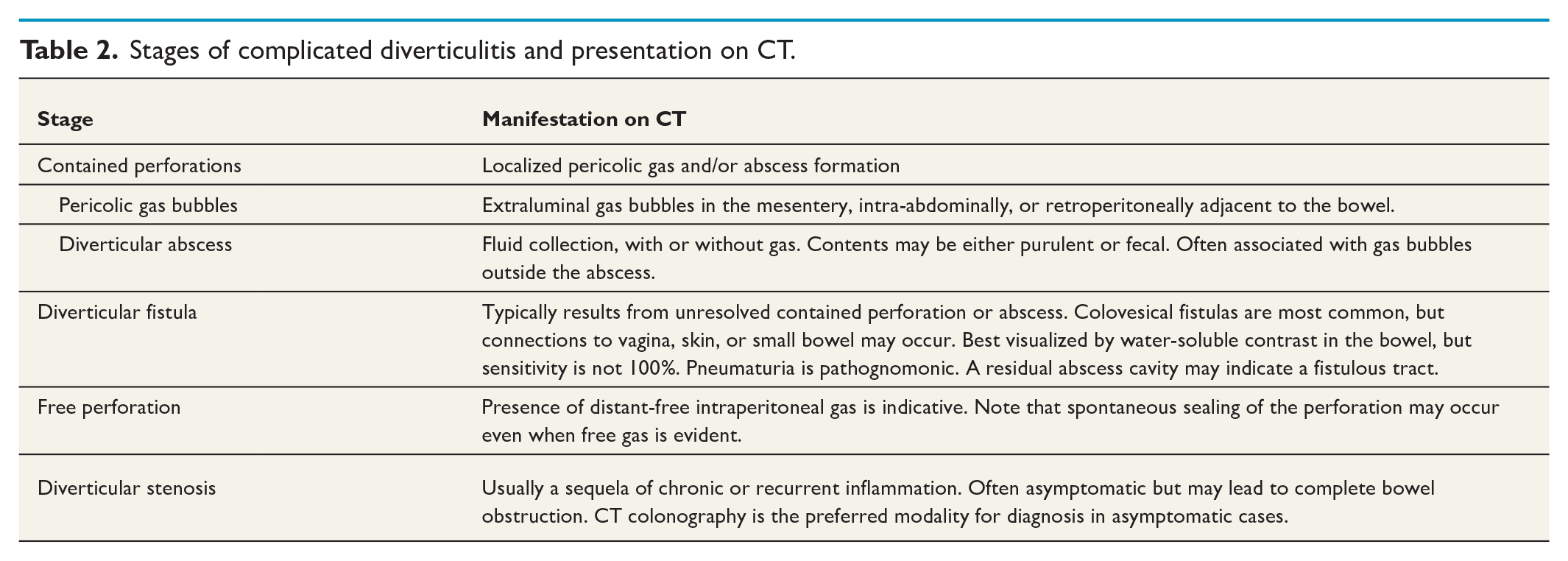
Stages of complicated diverticulitis and presentation on CT.
Diagnostic workup
A thorough medical history is essential, as several other conditions may mimic the symptoms of diverticulitis. The classical triad—left lower quadrant pain, altered bowel habits, and fever—is present in only a minority of patients with acute diverticulitis. Clinical examination and routine laboratory investigations are mandatory, yet history and physical findings alone are non-specific. The diagnostic accuracy of clinical assessment without imaging ranges from 45% to 72%.11 –13 This accuracy improves when left-sided abdominal tenderness is accompanied by the absence of vomiting and by a C-reactive protein (CRP) level >50 mg/L.12,14 A history of previous diverticulitis, in combination with clinical findings and an elevated CRP level, can raise the diagnostic accuracy to as high as 90%. 9 Several clinical scoring systems have been developed to improve diagnostic accuracy, but none have gained widespread use in routine practice.14,15 Distinguishing uncomplicated from complicated diverticulitis remains particularly challenging. 16 Although some prediction tools have been designed to exclude complicated disease, they have not been widely adopted in clinical settings.17,18
The gold standard to confirm a diagnosis of acute diverticulitis and assessing for complications is contrast-enhanced CT of the abdomen and pelvis.19,20 CT offers an excellent diagnostic performance, with a reported specificity of 99%–100% and sensitivity of 98%–99%. 21 Typical CT findings in acute diverticulitis include pericolic edema/fat stranding, colonic wall thickening, and the presence of diverticula. 22 CT manifestations of complications are summarized in Table 2.
CT with rectal contrast is generally not used in the acute setting due to its invasive nature and limited incremental value. However, it may be useful in selected cases for evaluating colonic fistulas following diverticulitis episodes. 23 Low-dose, non-contrast CT has been explored as an alternative but is less sensitive in detecting small pericolic or intramural abscesses. 24
Ultrasound (US) and magnetic resonance imaging (MRI) offer radiation-free imaging alternatives. The US is a cost-effective option, but its diagnostic accuracy is highly operator-dependent and may be limited in overweight or obese patients. 25 MRI is a valuable alternative in patients for whom CT is contraindicated, although its routine use is limited by cost, availability, and scan duration. MRI is less sensitive to extraluminal gas but may aid in distinguishing diverticular stenosis from cancer. 26
Contained perforations
In immunocompetent patients, diverticular perforations are often contained by adjacent anatomical structures such as the mesentery, bladder, omentum, or small bowel, depending on the perforation site. This containment can localize the infection, resulting in characteristic CT findings such as pericolic gas bubbles or a pericolic abscess.
Pericolic extraluminal gas
The presence of extraluminal gas in a patient with an acute abdomen typically indicates gastrointestinal perforation. Historically, detection of such gas on plain abdominal radiography often led directly to surgical intervention. With modern cross-sectional imaging, even small volumes of extraluminal gas are readily identified. In patients with acute diverticulitis, pericolic extraluminal gas bubbles are observed in approximately 15% of cases classified as Hinchey 1a 27 (Fig. 1). When pericolic gas is the sole sign of a perforation, the clinical course is generally favorable, and most patients can be managed non-operatively.28,29 However, current evidence for managing this subgroup without antibiotics is limited.30 –33 Therefore, in contrast to cases of uncomplicated diverticulitis, antibiotic therapy is still routinely recommended for patients with evidence of pericolic extraluminal gas.20,34
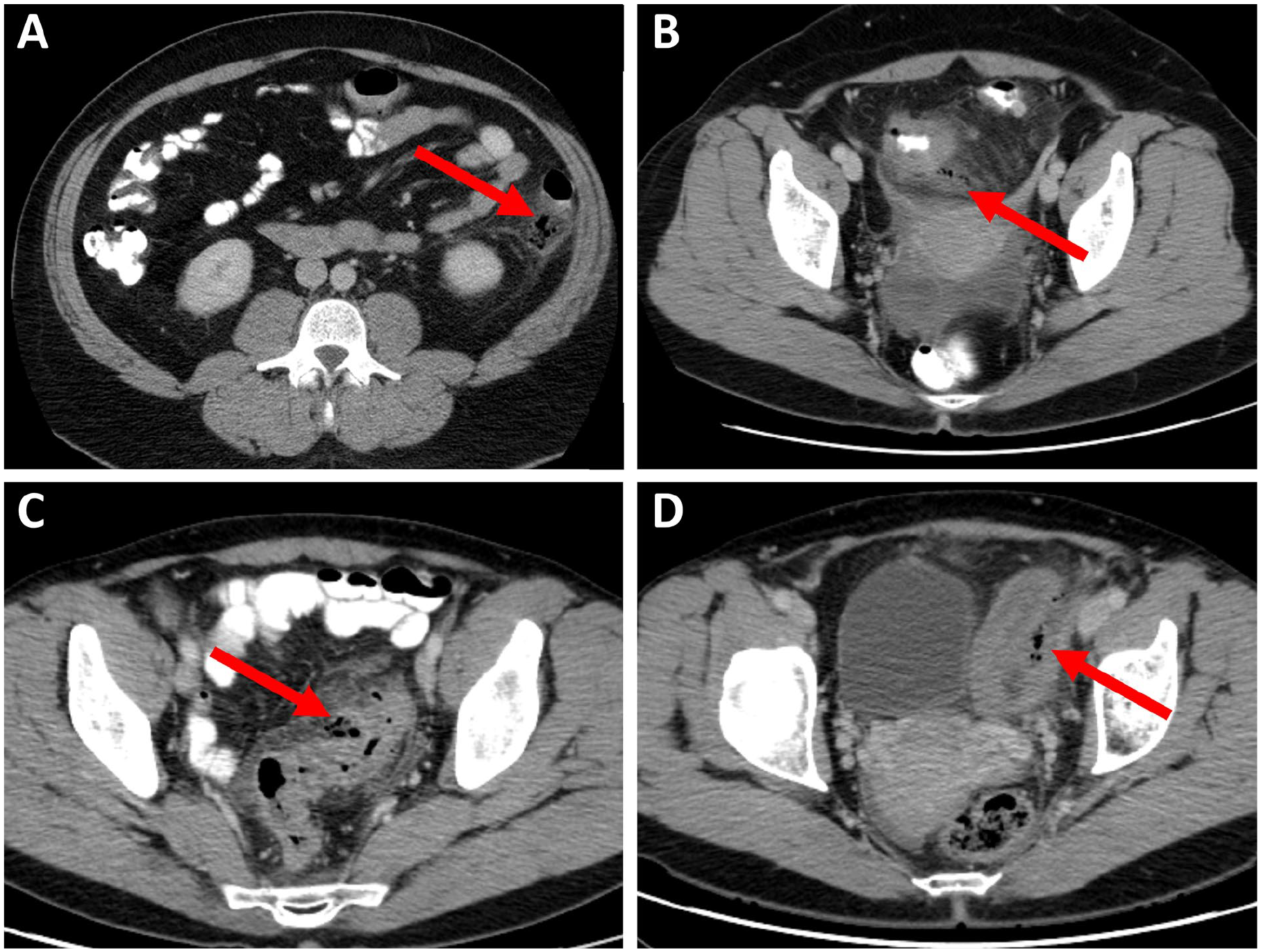
Diverticulitis with pericolic extraluminal air. Pericolic air bubbles marked with arrows in different locations: (A) retrocolic, (B) in the mesentery, (C) subserous pericolic fat, and (D) between bowel segments. Source: Pictures provided by Arnar Thorisson.
Diverticular abscesses
Abscess formation in the setting of acute diverticulitis may result from either a contained macroperforation or multiple microperforations of the colonic wall. These abscesses may communicate with the bowel lumen (Fig. 2), and their contents often include gas, pus, or fecal material. The natural course of diverticular abscesses is variable: some resolve spontaneously with or without antibiotics, while others may enlarge, leading to systemic infection, sepsis, or even death. Complications include spontaneous drainage into adjacent hollow organs or the skin, resulting in fistula formation, or rupture into the peritoneal cavity, causing generalized peritonitis. The clinical presentation is therefore highly heterogeneous, depending on the abscess size and anatomical location.
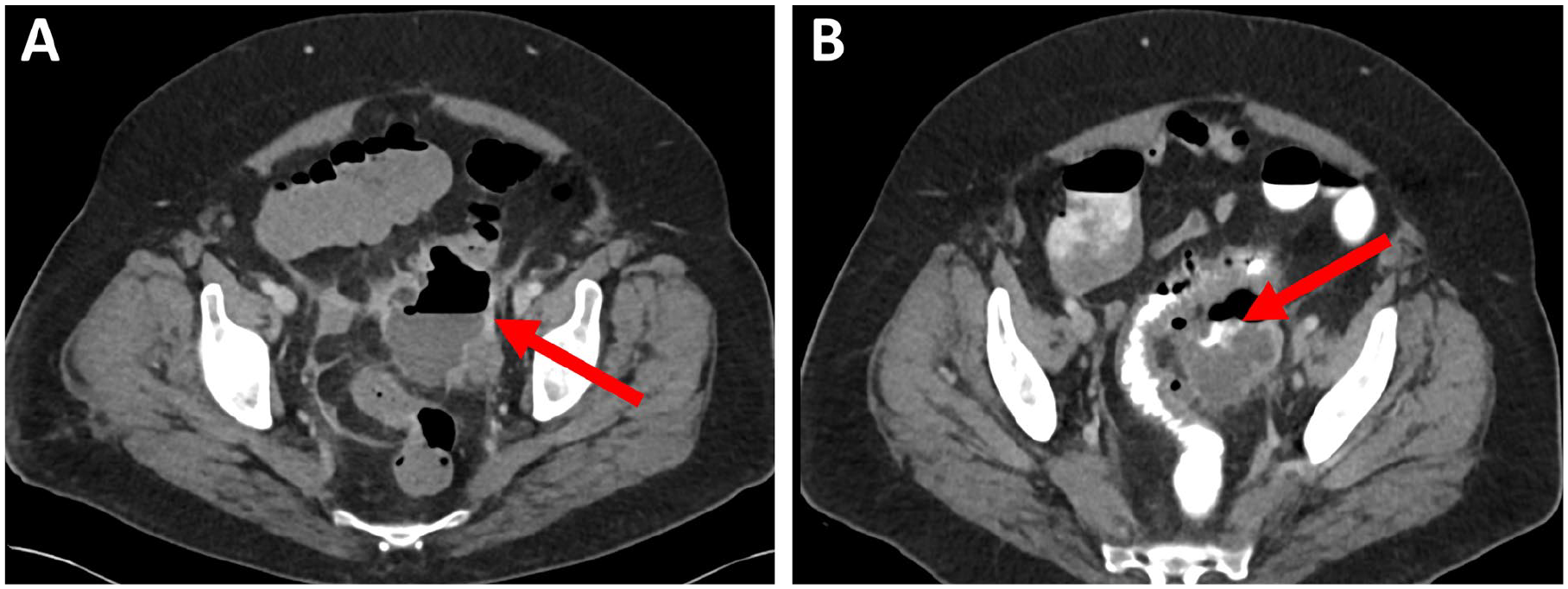
Pericolic abscess communicating with the bowel lumen: (A) abscess marked with arrow and (B) bowel communication visualized by contrast enema. Source: Pictures provided by Abbas Chabock.
Management strategies are tailored to the severity and include conservative treatment with antibiotics, image-guided percutaneous drainage, and surgery when necessary.
Small diverticular abscesses may resolve without any treatment. In one randomized trial evaluating non-antibiotic treatment for diverticulitis, patients with abscesses smaller than 5 cm were included. No significant difference in outcomes was observed between those treated with and without antibiotics (n = 26 no antibiotics, n = 16 antibiotics). 35 A post hoc CT review from another randomized trial also identified a number of patients with previously overlooked abscesses; the majority of those in the non-antibiotic group recovered without antibiotics. 33 Nevertheless, the overall evidence supporting non-antibiotic management of diverticular abscesses remains limited. There is therefore a general understanding that antibiotic treatment should be recommended regardless of abscess size.19,20,34,36
The best available evidence guiding interventional management of diverticular abscesses is derived from retrospective cohort studies. These include two large population-based registry studies37,38 and two multicenter studies based on individual patient file reviews.39,40 In addition, numerous single-center cohort studies have been published (Supplementary Table 1).
Abscess size is a key determinant in the likelihood of spontaneous resolution or successful conservative management with antibiotics. 41 However, accurately measuring abscess volume—the most relevant parameter—is often challenging in practice. Consequently, most studies report abscess dimensions using diameter alone, often without specifying whether this reflects the maximum, minimum, or mean measurement.
Clinical guidelines typically recommend image-guided percutaneous drainage for abscesses larger than 3–5 cm in diameter, which roughly corresponds to a volume of 15–65 mL assuming a spherical shape. However, the quality of evidence supporting this threshold is very low. 42 In practice, the decision to perform drainage should be individualized, weighing the potential benefits against procedural risks. This decision is primarily influenced by abscess size and anatomical accessibility. Drainage is generally recommended for well-accessible abscesses with a volume exceeding approximately 50 mL (diameter > 4–5 cm). Nonetheless, very large abscesses (>8 cm in diameter) frequently require surgical intervention for definitive management. 41
Conservative treatment of perforated diverticulitis with distant extraluminal gas
While diverticular perforation usually leads to generalized peritonitis requiring immediate surgery, a subset of patients with perforated diverticulitis may present with radiological evidence of free perforation—namely, distant extraluminal gas—without clinical signs of peritonitis. In such cases conservative management may be a viable option. Although the widely used Hinchey classification and its modifications 8 do not account for this clinical entity, more recent CT-based classification systems do recognize it (Table 1).6,10
Randomized controlled trials (RCTs) comparing conservative versus surgical treatment in patients with perforated diverticulitis and free distant gas are lacking, likely due to the rarity and heterogeneity of this presentation. The most comprehensive evidence to date stems from a recent meta-analysis of 209 patients across 12 observational studies conducted over three decades. 43 In this analysis, non-operative management—defined as avoidance of emergency surgery—was successful in about 70% of carefully selected patients, regardless of the presence of an associated abscess.
Notably, approximately 40% of patients with distal extraluminal gas also had concomitant abscesses, many of which were managed with percutaneous drainage. 44 Several studies identified the presence of peritoneal fluid 44 –47 and large volumes of distant-free gas44,47 as predictors of failed conservative treatment, likely reflecting more extensive or more persistent bowel perforations.
Overall, conservative treatment consisting of intravenous fluids and broad-spectrum intravenous antibiotics appears feasible in hemodynamically stable, immunocompetent patients with perforated diverticulitis and distant extraluminal gas, provided there is no clinical evidence of peritonitis or significant comorbidity. Close clinical observation is mandatory, as approximately one-third of patients may ultimately require emergency surgery if conservative management fails. 43 Whether conservative treatment offers superior outcomes compared to immediate surgery—particularly in terms of morbidity, length of stay, and recurrence—remains to be determined.
Follow-up after conservative treatment of diverticulitis
It is widely accepted that diverticulitis is not associated with an increased long-term risk of developing colorectal cancer (CRC). The elevated risk observed within the first year following a diverticulitis episode is generally attributed to initial misdiagnosis of a perforated or obstructing colorectal malingnancy. 48 Consequently, follow-up protocols have traditionally focused on short-term management rather than long-term surveillance.
With advancements in CT imaging, accumulating evidence suggests that routine colonoscopy may not be necessary in asymptomatic patients following an episode of uncomplicated diverticulitis. However, in cases of complicated diverticulitis managed non-operatively, follow-up colonic evaluation is recommended.19,20,36 The objectives of such follow-up are twofold: (1) to detect persistent or recurrent diverticular disease and (2) to identify CRC at an early stage.
Patients with complicated diverticulitis have a higher risk of underlying CRC; 49 estimates suggest a malignancy rate of approximately 6%–8%, even when CT imaging has been performed. 50 Although CT colonography offers improved patient tolerability and better diagnostic accuracy in identifying residual inflammation, abscesses, and fistulas, colonoscopy remains the preferred modality due to its superior sensitivity for detecting CRC and advanced adenomas. 51
The optimal timing for follow-up colonoscopy remains unclear. In patients with clinical or radiological suspicion of malignancy, early endoscopic evaluation within 1–2 weeks is advisable and considered safe in the absence of free intraperitoneal gas on CT. 52 For patients with diverticular abscesses, colonoscopy within 4 weeks may be appropriate due to the increased risk of an underlying neoplasia.6,53 In all other cases, colonoscopy is optimally performed 6–8 weeks after the acute episode, allowing time for inflammation to resolve and minimizing procedure-related risks. 54
Operative treatment of free perforation
Acute perforated diverticulitis with free intraperitoneal gas and sepsis remains a life-threatening condition, with reported mortality rates ranging from 10% to 25%. 55 Traditionally, surgical management—typically involving sigmoid resection with creation of a stoma, known as Hartmann’s procedure—has been advocated for Hinchey grades 3 and 4 disease, in addition to broad-spectrum antibiotics. Free perforation may lead to either purulent peritonitis (Hinchey 3) or fecal peritonitis (Hinchey 4), the latter carrying a worse prognosis due to extensive contamination.
Over the past decade, three European RCTs have evaluated laparoscopic lavage as an alternative treatment strategy for purulent peritonitis (Hinchey 3). These studies reported higher short-term reintervention rates in the lavage group, 56 but similar mortality and lower stoma prevalence, both in short- and long-term follow-up, compared to traditional resection.57 –60 Approximately 30% of patients initially treated with the laparoscopic lavage ultimately underwent sigmoid resection during long-term follow-up. Performing this resection electively, however, confers significant advantages, including a higher likelihood of primary anastomosis (Azhar, Johanssen and Sundstrom, 2021). 60 Real-world registry and cohort data have since confirmed that laparoscopic lavage is both safe and feasible in routine clinical practice.61,62
A key limitation of laparoscopic lavage lies in the difficulty of distinguishing between purulent and fecal peritonitis. While lavage is appropriate for Hinchey 3, fecal contamination (Hinchey4) requires immediate resection. In selected patients with Hinchey 3 or 4 disease undergoing sigmoid resection, primary anastomosis with or without diversion is increasingly considered feasible. However, the specific characteristics that predict successful outcomes in these patients remain to be clearly defined.
Recent comparative studies of Hartmann’s procedure versus resection with primary anastomosis in patients with purulent or fecal peritonitis have demonstrated no significant differences in mortality between groups. Importantly, when morbidity associated with reversal of Hartmann’s procedure is considered, primary anastomosis is associated with fewer long-term stomas and reduced overall morbidity.63,64 These studies, however, were underpowered, included both Hinchey 3 and 4 patients, failed to reach their intended sample sizes, and primarily included hemodynamically stable, immunocompetent patients. As such, Hartmann’s procedure should remain the standard of care for hemodynamically unstable or immunocompromised individuals.
Recently, damage control surgery has emerged as an alternative strategy for managing perforated diverticulitis in critically ill patients. While promising, further data are needed to determine its role in the management of acute perforated diverticulitis. 65 Minimally invasive techniques are considered safe in the acute setting when performed by experienced surgeons. Although no high-quality data exist comparing laparoscopic versus open resection in the emergency context, 20 diagnostic laparoscopy remains a valuable initial step. It allows for assessment of peritoneal contamination and, in appropriate cases, offers the opportunity for laparoscopic lavage in patients with purulent peritonitis.
Elective surgery after a conservatively managed episode of complicated diverticulitis
The previously widespread recommendation for routine elective sigmoid colon resection following an episode of complicated diverticulitis has been revised in recent international guidelines.6,19,20 While patients who experience diverticular abscesses are at increased risk of recurrence, most recurrent episodes can be managed non-operatively. 19 Current consensus emphasizes that the decision to proceed with elective resection should be individualized. Key considerations include the frequency and severity of recurrent episodes, the overall symptomatic burden, and the patient’s general health status and comorbidities. A tailored approach ensures that surgery is reserved for those who are likely to derive the greatest benefit, while avoiding unnecessary interventions in patients who can be managed conservatively.
Diverticular fistulas
Fistula formation in diverticular disease most commonly arises from an unresolved abscess, serving as a natural mechanism for the body to drain infection. Colovesical fistulas are the most frequent type, accounting for approximately 65% of all diverticular fistulas, followed by colovaginal fistulas, which comprise about 25%—especially in women with a history of hysterectomy. 66 Diverticular fistulas are, in fact, responsible for 75%–86% of colovesical fistulas.67 –69
The clinical presentation of diverticular fistulas varies depending on the affected organ. Colovesical fistulae typically manifest with recurrent urinary infections (reported in 57%–100% of cases) and often with pathognomonic symptoms such as pneumaturia (64%–75%) or fecaluria (51%–78%).67,70,71 Colovaginal fistulas may present with feculent or increased vaginal discharge and vaginal flatulence. Contrast-enhanced CT is the preferred diagnostic modality, demonstrating high diagnostic accuracy ranging from 61% to 100%.71,72 Water-soluble contrast enema can sometimes delineate the fistula tract, but the sensitivity is limited, with successful visualization in up to 64% of cases. 73 Functional tests, such as the poppy seed or charcoal test, offer the highest sensitivity for detecting colo- or enterovesical fistulas.71,72
Surgical resection of the affected bowel segment is the standard treatment for diverticular fistulas. However, in selected patients with minimal symptoms—particularly those who are frail—a conservative non-operative approach may be appropriate.67,74 Preoperative workup should include colonoscopy, and when relevant, cystoscopy or colposcopy of the involved organ to exclude malignancy.
A primary anastomosis with or without diverting stoma is generally preferred over Hartmann’s procedure. The choice of surgical strategy should be guided by patient-specific factors, including comorbidities, immunocompetence, overall condition, and intraoperative findings such as the degree of contamination and bowel wall edema. 74 In complex or emergency cases, a staged approach with proximal diversion prior to definitive resection may be advisable. Laparoscopy is generally considered safe in experienced hands, although conversion rates are likely higher than in other colorectal indications. 74
Diverticular stenosis
Chronic inflammation—whether due to smoldering low-grade inflammation or frequent recurrent episodes—may lead to the development of colonic stenosis. Such strictures present both diagnostic and therapeutic challenges. When malignancy can be excluded by endoscopy, treatment decisions should be guided by the severity of symptoms. In asymptomatic or mildly symptomatic patients, a conservative approach is generally preferred. Even in cases of significant luminal narrowing, symptomatic relief can often be achieved with the use of laxatives. However, diverticular stenoses may progress over time and eventually lead to bowel obstruction. In symptomatic patients, the decision to pursue surgical intervention should be individualized, taking into account the degree of functional impairment and patient’s overall condition and comorbidities.
If the stenosis is impassable by colonoscopy, it may be difficult to distinguish from CRC. Although CT colonography or MRI may provide additional information, definitive diagnosis may only be achieved following surgical resection.75,76 Surgery is clearly indicated in cases of suspected or confirmed bowel obstruction or when malignancy cannot be confidentially excluded.
In malnourished patients or those with a large inflammatory mass, a staged approach—with initial proximal diversion followed by delayed resection—may be most appropriate. In frail patients for whom curative resection is not feasible, a diverting stoma may serve as a palliative or permanent solution.
Endoscopic management techniques, such as balloon dilatation or stenting, have been described for benign colon stenosis. 77 However, they are less commonly employed in diverticular disease due to the typically long segment of affected bowel and an increased risk of perforation compared to malignant strictures. 78
Conclusion and future perspectives
Over the past few decades, the management of complicated diverticulitis has shifted from a predominantly surgical approach to one favoring conservative, individualized treatment strategies. 61 Ongoing areas of controversy include the role of antibiotics in early stages of complicated diverticulitis—such as cases with small abscesses or pericolic air—and the necessity of follow-up colonoscopy in these patients, a practice that may evolve further with the broader implementation of CRC screening programs.
In parallel, there is growing acceptance of non-operative management in selected immunocompetent, hemodynamically stable patients with Hinchey stage 1–3disease. As clinical decision-making becomes increasingly nuanced, improving the diagnostic accuracy of acute diverticulitis—particularly in distinguishing uncomplicated from complicated cases—remains a key objective for future research and innovation.
Supplemental Material
sj-docx-1-sjs-10.1177_14574969251343452 – Supplemental material for Changing paradigms in the management of complicated diverticulitis
Supplemental material, sj-docx-1-sjs-10.1177_14574969251343452 for Changing paradigms in the management of complicated diverticulitis by Johannes Kurt Schultz, Maziar Nikberg, Abbas Chabok, Arnar Thorisson, Johanna Sigurdardottir, Pamela Buchwald, Ville Sallinen and Najia Azhar in Scandinavian Journal of Surgery
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
