Abstract
Visceral artery aneurysms (VAAs) and pseudoaneurysms are rare but clinically significant vascular pathologies with potentially fatal complications including rupture. VAAs are often asymptomatic and frequently discovered incidentally during imaging performed for unrelated reasons. Their pathophysiology, clinical presentation, and management vary based on the etiology, anatomical location, and patient-specific factors. The prognosis for conservatively managed VAAs is generally favorable, with slow growth rates and low rupture risks. However, pseudoaneurysms, often associated with trauma or pancreatitis, carry a much higher rupture risk and typically require early intervention, regardless of size. Endovascular techniques have progressively replaced open surgery as the preferred treatment approach due to lower complication rates and comparable long-term outcomes. However, current knowledge is biased by the scarcity of high-quality evidence regarding the natural history, rupture risk, and optimal management of VAAs and pseudoaneurysms, largely due to the rarity of these conditions. Consequently, different international societies have provided recommendations with low strength and, in some cases, conflicting indications.
Keywords
Context and relevance
VAAs are rare but potentially fatal conditions. Data on the long-term outcomes of various interventions remain insufficient. Endovascular approaches are preferred because of their lower invasiveness. Long-term durability remains debatable, especially in younger patients or in those with a higher risk of recurrence.
Introduction
Visceral artery aneurysms (VAAs) and visceral artery pseudoaneurysms (VAPAs) are relatively rare but clinically significant conditions that can lead to life-threatening complications. Although these aneurysms are generally asymptomatic, rupture can result in significant morbidity and mortality. The pathophysiology, clinical presentation, and management strategies for these conditions vary depending on anatomical location, etiology, patient comorbidities, and clinical status.
Management strategies have evolved significantly in recent decades, with endovascular techniques increasingly complementing or replacing traditional open surgical approaches.
However, high-level studies on the natural history, rupture risk, and long-term comparison of different management strategies are still lacking, mainly owing to the rarity of these diseases. International societies have emphasized the critical need for more robust data, urging the scientific community to produce higher level evidence. This is essential because current guidelines are constrained by the limited quality of available data, resulting in weak recommendations.
General considerations
True aneurysms result from arterial degeneration involving all three layers of the arterial wall. According to the morphological classification, aneurysms can be defined as fusiform, when the entire circumference of the artery is affected by the dilative process, or saccular, when only part of the wall shows focal structural failure, resulting in an eccentric dilation and a neck that separates the aneurysmal sac from the lumen of the vessel. Pseudoaneurysms, also known as “false” aneurysms, originate from the disruption of the intimal and medial layers, are only bounded by thin adventitial tissue, and are typically encircled by a periarterial hematoma. The fragile structure of these lesions underlies a higher tendency to rupture than true aneurysms.
Etiology, prevalence, and natural history
The rarity of these conditions limits the development of solid evidence on their etiology, natural history, and treatment outcomes. In fact, the literature mainly consists of retrospective studies, as prospective or randomized trials are difficult to conduct in such rare conditions. Moreover, the vast majority of evidence on VAAs and VAPAs comes from broad studies often grouping both entities. In light of these considerations, the present paragraph focuses on reviewing the current knowledge on VAAs as a whole. Specific considerations for single visceral branches are examined in the following paragraphs.
VAAs are rare conditions with an estimated prevalence of 0.01%–0.2%.1–3 Atherosclerosis, smoking habit, fibromuscular dysplasia, and collagen diseases have been associated with the development of true VAAs.1–3
The natural history of VAAs seems to be benign, with slow growth rates and few ruptures during follow-up, as reported in the literature. Annual growth rate for VAAs is reported to be low (0.06–0.6 mm/year) for conservatively managed patients.2,4 In a large retrospective study of 138 VAAs, the vast majority (108 VAAs) were treated conservatively with no adverse events related to VAA, even in those more than 2 cm in diameter. Patients presenting with a previously unknown ruptured VAA are unlikely to present a lesion less than 2 cm in diameter.2,4 The limited evidence on the natural history of VAAs in the literature can be misleading, sometimes resulting in conflicting recommendations from different international societies. Notably, the natural history of unruptured VAAs should not be inferred from the characteristics observed in ruptured VAAs.
Diagnosis
In recent times, VAAs are mostly diagnosed by chance during imaging performed for other reasons. Different imaging modalities have been described for the diagnosis and follow-up of VAAs. Doppler ultrasound (DUS) and contrast-enhanced ultrasound (CEUS) may be a useful non-invasive and radiation-sparing tool, but face a low diagnostic accuracy due to the location of VAAs, which are often difficult to explore and concealed by abdominal fat and bowel gas. 5 Computed tomography angiography (CTA) is the most common diagnostic option for VAAs, allowing for precise location and imaging reconstruction for preoperative evaluation and planning of endovascular interventions. Magnetic resonance angiography (MRA) can be an alternative in patients with contraindications to perform CTA examination. 6 Considering the association of VAAs with other abdominal and peripheral aneurysms, patients diagnosed with VAAs are indicated for screening for other aneurysms, especially those with collagen diseases and fibromuscular dysplasia. 7
Threshold for repair
The European Society for Vascular Surgery (ESVS) addressed the issue of VAAs in the 2017 guidelines on Management of the Diseases of Mesenteric Arteries and Veins. 1 Intervention for asymptomatic mesenteric artery aneurysms has been suggested for diameters ≥2.5 cm, whereas repair was recommended for symptomatic or ruptured aneurysms of any size. The authors identified certain features associated with a higher risk of rupture, justifying surgical intervention regardless of the aneurysm size. These features include aneurysms of the pancreaticoduodenal, gastroduodenal, or intra-parenchymal hepatic arteries, women of childbearing age, and liver transplant recipients.
The Society of Vascular Surgery (SVS) 7 and the Italian Society for Vascular and Endovascular Surgery (SICVE) 8 have recently issued clinical guidelines for the management of VAAs, 2020 and 2023 respectively. The thresholds for repair issued by the three societies are displayed in Table 1 and further discussed in the text.
Dimensional threshold for elective repair in asymptomatic visceral aneurysms according to different international societies.
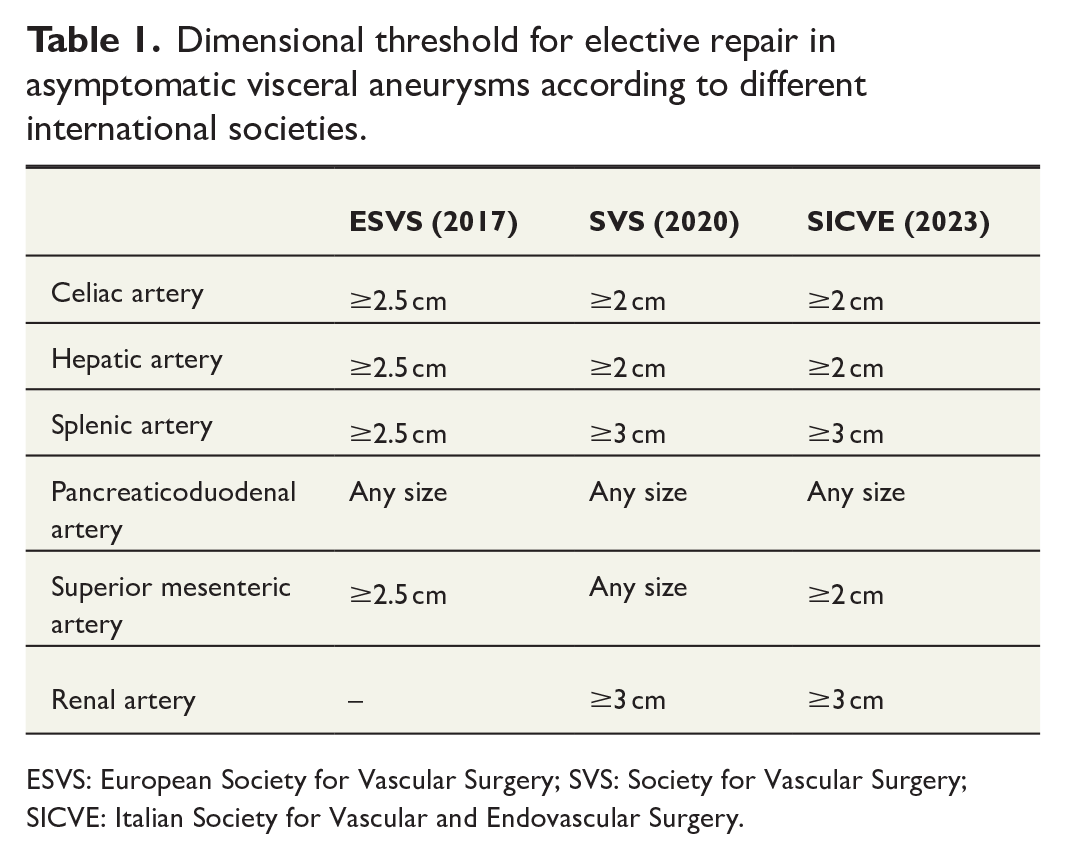
ESVS: European Society for Vascular Surgery; SVS: Society for Vascular Surgery; SICVE: Italian Society for Vascular and Endovascular Surgery.
Interventions
Interventions for VAAs and VAPAs may entail both open and endovascular options, with several treatment strategies developed over time. A large retrospective analysis showed a progressive increase in the number of repairs per year for VAAs in the last decade, with the majority of cases now treated through an endovascular approach. 9 For both endovascular and open surgery, the challenging setting in treating ruptured VAAs reflects the higher rates of perioperative mortality when compared with elective repair of non-ruptured VAAs. 10
Open intervention may be conducted by vein/prosthetic bypass, aneurysm resection, or aneurysm ligation, with or without en bloc resection of the adjacent organ. Laparoscopic and robotic surgeries for the treatment of VAAs and VAPAs have also been reported. A recent meta-analysis of robotic surgery for VAA reconstruction in 53 patients reported 0% perioperative and aneurysm-related mortality and a low reintervention rate (3.6%) over a median follow-up of 9 months. 11
Endovascular techniques have been reported as the treatment of choice in patients with a favorable anatomy. Preserving the target vessel patency is always preferable, with several available techniques including aneurysm exclusion using covered or flow-diverting stent grafts, coil/glue embolization, or stent/balloon-assisted embolization.2,12,13 Proximal and distal occlusion through vascular plugs may not be considered a first-line strategy because of its possible consequences on distal organ perfusion.12,13
Both open and endovascular approaches have been associated with high technical and clinical success, and overall survival rates. However, different studies have found an advantage for the endovascular approach in terms of lower complication rates and better long-term survival.14,15 A retrospective study on 9260 interventions for VAAs found that open intervention was associated with longer hospitalization and was an independent predictor of perioperative mortality and complications. 9
Follow-up
Patients with VAAs should be periodically followed after the diagnosis of VAAs in both cases of surgical intervention and conservative management. Postoperative surveillance should assess the technical success and patency of the treated vessel over time. Follow-up studies may be conducted using DUS or CTA based on the patient’s anatomy and the intervention performed. DUS/CEUS and MRA have been described as valid tools for follow-up of patients treated by coil embolization of VAAs because of their ability to reduce imaging artifacts of coils visible in CTA examinations 16 (Fig. 1). In cases where CEUS or MRA is unavailable, based on our experience, it may be reasonable to follow up such patients through plain CT scan monitoring for sac enlargement and indicating angiography in case of growth. For patients treated conservatively, yearly imaging has been indicated with the chance of becoming less frequent in cases of size stability.
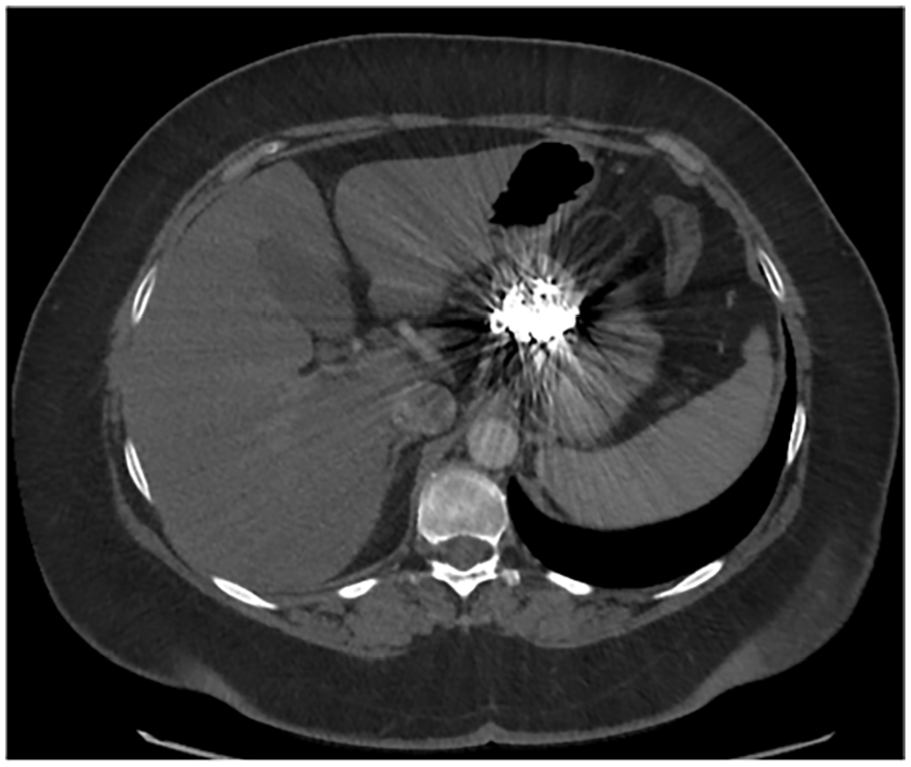
Coil embolization causes significant image artifacts in postoperative CTA examinations.
Pseudoaneurysms
Knowledge of pseudoaneurysms is mainly derived from broad studies on VAAs and VAPAs combined, whereas data specifically focused on VAPAs only come from case reports or small retrospective cohort studies.
False aneurysms are usually reported in patients with a history of abdominal trauma, abdominal surgery, other invasive diagnostic procedures, and local infectious or inflammatory diseases (especially pancreatitis).1–3 A suggestive medical history and imaging of focal and irregular lesions with otherwise normal arteries should lead the clinician to consider the diagnosis of VAPAs (Fig. 2). The hepatic artery and celiac branches are the most common locations of VAPAs.10,17 False aneurysms do not share the same benign natural history as VAAs. Different studies have reported a vast majority of VAPAs, in some cases up to 89%, presenting as ruptured with a consequent increase in mortality.10,17
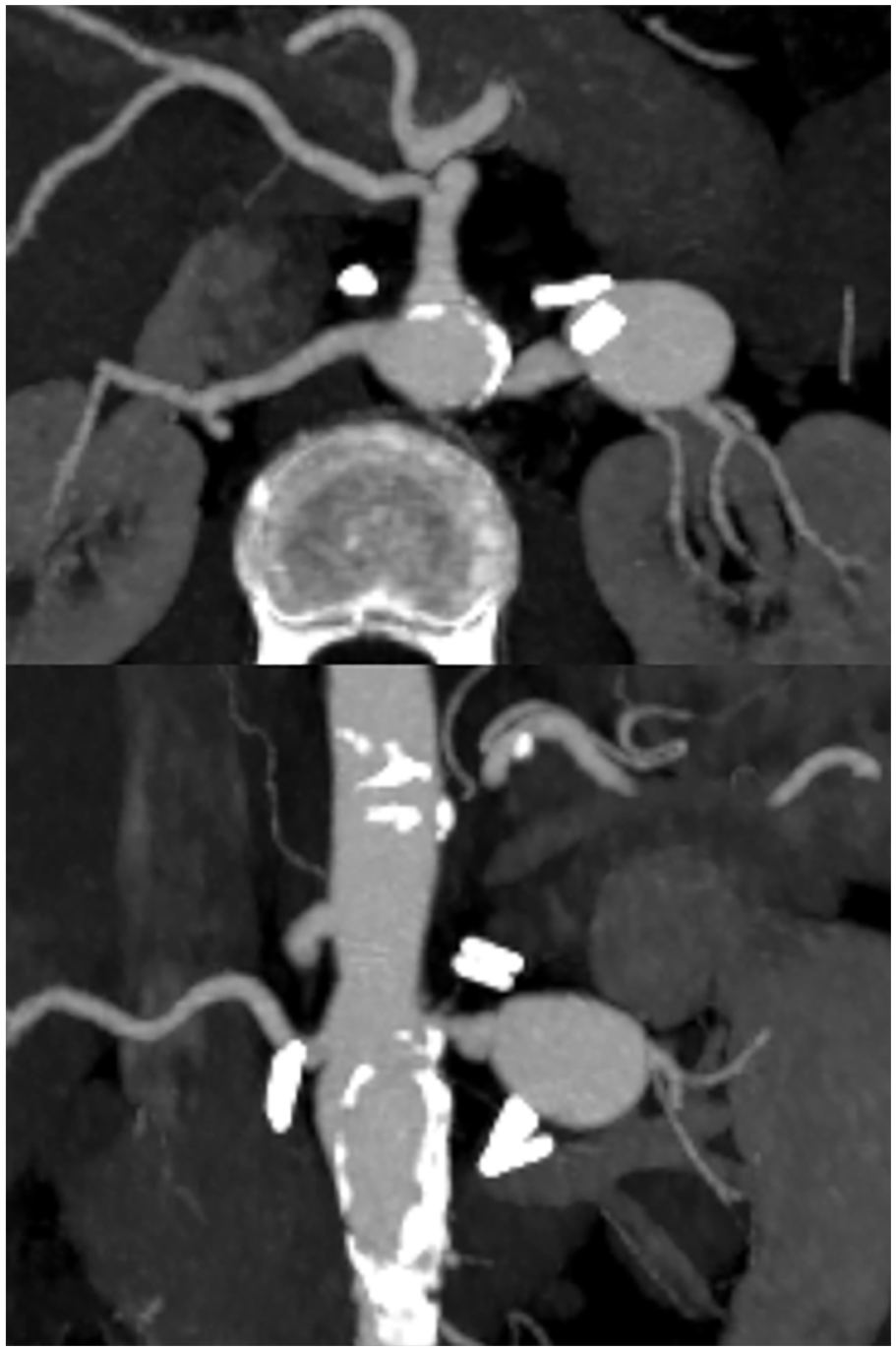
Intact pseudoaneurysm of the left renal artery in CTA multiplanar reconstructions. The patient underwent an aorto-bifemoral bypass associated with aortic and renal endarterectomy for aorto-iliac obstructive disease 2 years before. Surgical clips are visible in the picture. An association of evocative medical history and imaging must call into question the presence of a false aneurysm.
Transarterial embolization is reported to be the most frequent interventional strategy,18,19 with high technical success rates (97%) and an early clinical success rate of 82%, as reported in a meta-analysis. 18 Exclusion of the lesion through covered stents has also been described with 80% technical and clinical success, even in a population of patients with high rates of septic complications at the time of intervention. 20
Due to their high rupture risk, pseudoaneurysm affecting any visceral branch is recommended for surgical management irrespective of the size.1,7,8
Splenic artery aneurysms
Splenic artery aneurysms (SAAs) are the most common type of VAAs, accounting for approximately 60% of cases. They represent the third most common intra-abdominal aneurysmal pathology, after aortic and iliac aneurysms. Concomitant intra-abdominal aneurysms have been reported with variable rates of abdominal aortic aneurysms (3%–7%) and other VAAs (3%–4%). 21
SAAs are more prevalent in women (60%–80%) and appear to be commonly associated with smoking habit, fibromuscular dysplasia, connective tissue disorders, pregnancy, and liver cirrhosis.21–23 The combination of wall weakening, increased arterial pressure, and portal hypertension seems to underlie the pathogenic mechanisms at the basis for SAA formation during pregnancy and liver cirrhosis.24,25 Atherosclerosis seems to have less influence on SAAs than on other arterial aneurysms and mycotic etiology is rare. 23
Natural history and consequent rupture rate of SAAs are still scarcely defined. The growth rate of SAAs ranges from 0.16 to 0.63 mm/year. 23 Portal hypertension has been described as an independent risk factor for an accelerated growth rate, 4 but this association is still debated. 26
A 20 years’ experience with 217 patients reported a mean size of 2.1 cm in 168 conservatively managed patients. During follow-up, growth occurred in only 10% of cases (mean: 0.6 mm/year), but no rupture and only three interventions because of aneurysm growth were reported. 21 However, ruptured SAAs are associated with a high mortality rate. Historical mortality approaching 25% seems to have decreased in recent years, at rates of approximately 7%. 27 Women of childbearing age are reported to be more prone to SAA rupture with a mean diameter similar to non-ruptured cases (21.6 versus 28.2 mm). 24 Ruptures of SAA in pregnant women occur mainly in the third trimester of pregnancy and are associated with high maternal (26%) and fetal mortality (50%). 24
Endovascular and open surgical techniques have been described for the treatment of SAAs in elective and emergency settings. Open surgery involves arterial reconstruction or bypass exclusion of the aneurysm and may be associated with splenectomy, especially in urgency/emergency settings. 22 Moreover, open surgery is reported to be the most commonly used approach for giant splenic aneurysms, defined by a diameter >5 cm (Fig. 3). 28 In such cases, vascular reconstruction has been associated with additional resection of adjacent organs in rare cases of fistula formation or tight adherence. 28 Laparoscopic and robotic surgeries have been described as alternatives for the elective repair of SAAs. These approaches demonstrated no reinterventions or death and low rates of conversion (4.8%) to open surgery and postoperative complications (10%) according to a recent systematic review including 107 patients. 29 However, laparoscopic and robotic SAA treatment did not spread, as per other surgical specialties, favoring endoluminal techniques. Endovascular repair is the treatment of choice in cases of favorable anatomies including proximal and saccular aneurysms. It is performed through techniques that preserve vessel patency, such as coil/glue embolization for saccular aneurysms, stent-assisted embolization, or covered stent placement. 27 A systematic review showed a significantly higher mortality rate after open repair (5.1% versus 0.6%); however, this data may be influenced by the higher frequency of rupture cases in the open repair group. 30 A recent meta-analysis of 523 SAA interventions reported low short- and long-term mortality and complication rates in both open and endovascular approaches. However, lower reintervention rates have been reported for open surgery (0%) than for the endovascular approach (7%). 3
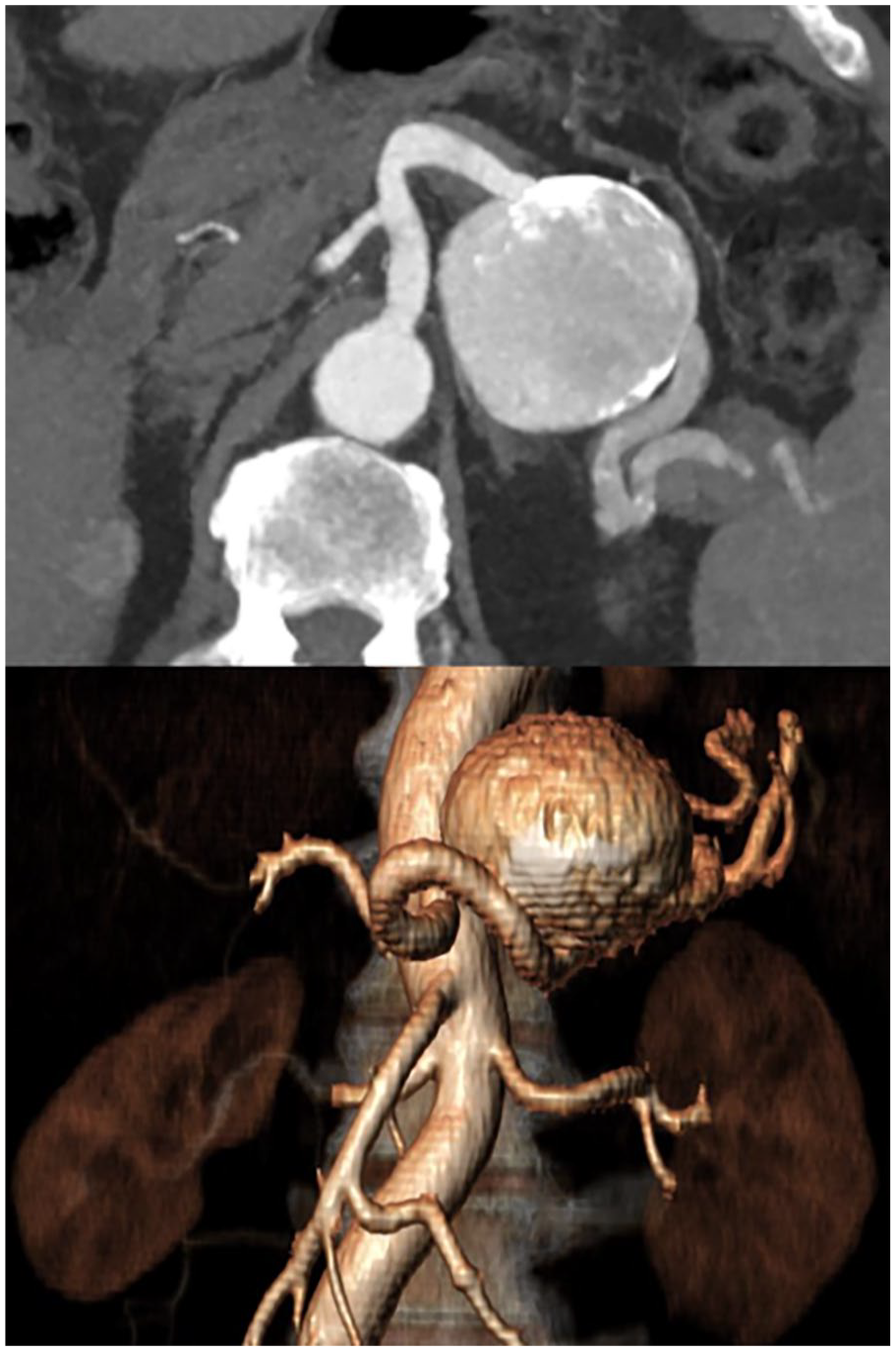
Reconstructions of a giant true aneurysm of the medial segment of splenic artery after CTA examination.
SVS 7 and SICVE 8 share the same indications for SAA management. Interventional management is recommended for pseudoaneurysms of any size, ruptured/symptomatic true aneurysms of any size, asymptomatic true aneurysms >3 cm or with documented rapid growth, and asymptomatic true aneurysms of any size in women of childbearing age. The choice between open or endovascular approach may be based on anatomical criteria and surgical risk of the patient.
Renal artery aneurysms
Renal artery aneurysms (RAAs) are reported with an estimated incidence of 0.1%–1% in the general population, with slight differences in autopsy, arteriogram, or computed tomography rates.31,32 RAAs typically occur in the sixth decade of life and are more commonly seen in female patients, probably due to their association with fibromuscular dysplasia. 32 The right renal artery is more commonly affected by aneurysmal disease. Bilateral aneurysms have been reported at rates of 10%–20%. Saccular morphology is frequently described, and its location is frequently reported to be at renal artery bifurcation.6,33
Follow-up studies have shown that the natural history of RAAs is characterized by an annual growth rate of 0.6–0.8 mm/year during follow-up. A recent study has not identified morphology and calcification as factors influencing growth rates. 6
Although mostly asymptomatic, RAAs can present with hypertension, flank/abdominal pain, hematuria, or renal infarction due to distal embolization. About two thirds of the patients with RAAs present with hypertension.6,33,34 Coexisting stenotic disease of the renal artery is reported in 7%–66% of cases, yet the pathophysiology of RAAs and hypertension is not clear. However, treatment of the aneurysm has been proven beneficial in most patients in different publications, even when not associated with arterial stenosis.12,6
The risk of rupture of RAAs is still debated. Although past reports found rupture rates up to 14%–30%, recent data seem to prove RAAs as a more benign condition.6,32,34 A large multi-institution cohort study of 865 RAAs followed over a mean of 29 months showed a rupture rate of 0.3% (mean diameter at rupture 3.7 cm) at baseline and no rupture in conservatively treated patients (mean diameter: 2.7 cm) over time. Among RAAs >3 cm, the rupture rate was 18%. 6 In a recent retrospective cohort study on 338 RAAs, only 34% of patients experienced aneurysm growth (mean rate of 0.23 mm/year) with no rupture recorded during a 41-month follow-up. 33 Even if infrequent, rupture of RAAs may lead to a life-threatening hemorrhage. Mortality rates have been reported with a favorable tendency to decrease, with first reports as high as 80% and now being reported at rates of approximately 10%.32,34
Pregnant women have been associated with higher rupture rates of RAAs, usually in the third trimester, with consequent high rates of maternal and fetal mortality. Although the true incidence of rupture in pregnant women is still unclear, reports of small aneurysm rupture can be found in the literature. 35 A historic large cohort study reported maternal and fetal death rates of 56% and 82%, respectively. 36 Despite a notable decrease in maternal mortality has been proven in most recent publications, lowering to 5.1%, fetal mortality did not show a significant decrease over time, reported at a 37.8% rate. 37
Surgical management of RAAs involves both open and endovascular repair. Open repair of RAAs includes in situ repair or ex vivo repair with auto-transplantation. Both techniques are mostly described for mid- and distal aneurysms, and have shown similar rates of morbidity, mortality, and patency during follow-up.38–40 A recent meta-analysis of 199 kidney auto-transplantations for RAAs showed a perioperative technical success rate of 99.5% and a low rate of surgical complications (5.6%). 41 Endovascular repair has become the preferred treatment in feasible cases owing to its minimally invasive nature. Several techniques (covered stent graft, flow-diverting stents, coil embolization, stent/balloon-assisted coil embolization) have been described for treatment of RAAs, mainly when affecting the proximal portion of the affected artery or in case of a saccular morphology allowing to maintain patency of the renal artery.13,42,43 A recent qualitative systematic review of 454 RAAs treated by endovascular means found a technical success rate of 97% and perioperative moderate/severe adverse event rate of 6.7%. Over a mean follow-up of 21 months, a low rate of sac reperfusion (0.9%), incomplete occlusion (0.7%), and aneurysm enlargement (0.2%) was reported. 13 A recent meta-analysis comparing open and endovascular approaches included 1279 RAAs and showed no significant differences in short- and long-term mortality. However, reintervention rates were significantly higher for endovascular approach than for open approach (16% versus 3%). 3
SVS 7 and SICVE 8 share the same indications recommending intervention for asymptomatic RAAs ≥3 cm. Pseu-doaneurysms, asymptomatic true aneurysms in pregnant or women of childbearing potential, and symptomatic aneurysms are suggested for treatment regardless of the size following their higher tendency to rupture. Endovascular approach is suggested for proximal lesions with favorable anatomy, whereas open strategies may be considered for more distal lesions.
Hepatic artery aneurysms
The incidence of hepatic artery aneurysms (HAAs) ranges from 0.002% to 0.01%.44,45 Atherosclerosis is recognized as the main cause of HAAs, with fibromuscular dysplasia, connective disorders, and mycotic, the latters being minor causes.44–46 HAAs are mostly documented in male patients, during the sixth decade of life, in extra-hepatic location and presenting as a single lesion.44–46
As for the other VAAs, solid data on natural history and consequent rupture risk are still lacking. Most HAAs are asymptomatic at the diagnosis. Right upper quadrant abdominal pain, elevated liver markers, and obstructive jaundice have been described, alone or in association, in symptomatic patients.44,45 In a large retrospective study of 43 HAAs, 16% of patients presented with rupture at a mean size of 5.2 cm. Rupture was more common in female patients (40%) and in those with vasculitis (67%). The majority of patients (51%) were managed conservatively with a baseline diameter of 2.1 cm and no rupture occurred during follow-up (mean: 3.9 years). 46
HAA management involves either open surgical repair or endovascular treatment. In both cases, preservation of arterial flow is always preferable upon ligation/occlusion. When performing open surgery, aneurysm can be resected and the artery reconstructed with end-to-end anastomosis, interposition graft, or aorto-hepatic artery bypass. Endovascular strategies consist of coil/glue embolization or covered stent graft apposition. A non-comparative retrospective study of 26 patients treated for HAAs reported that the majority underwent open surgery (65.4%), and only two cases of postoperative complication have been reported. 47 Low and comparable short- and long-term mortality for open and endovascular repair has been reported in a meta-analysis of 228 HAAs, with the latter technique demonstrating higher reintervention rate (40%). 3
SVS 7 and SICVE 8 guidelines suggest a diameter ≥2 cm as the threshold for surgical management of asymptomatic HAAs. Both societies highlight the possibility of intervention in asymptomatic aneurysms of any size with documented vasculopathy/vasculitis etiology, due to their tendency to rupture. Moreover, surgical intervention is recommended for ruptured or symptomatic HAAs or pseudoaneurysms of any size.
Celiac artery aneurysms
Celiac artery aneurysms (CAAs) are historically recognized as a rare location for VAAs, although their diagnosis has been reported at higher rates in recent studies, 23%–46% of all VAAs.2,4
Publications on the natural history, rupture risk, and effectiveness of surgical management of CAAs are scarce; therefore, solid evidence is lacking. Historically, CAAs were reported to have high rupture rates, 87% before 1950 dropping at 13% in 1950–1970, and consequent high mortality rates. 48 However, more recent retrospective studies have addressed the issue that the growth rate of CAAs may be low, infrequent, and have a nil rupture rate during follow-up.2,4
A recent meta-analysis reported only 87 cases, mainly treated by endovascular approach, with low rates of perioperative complications with either endovascular or open approach. However, the paucity of cases made it impossible to conduct inferential analysis. 3
Historically reported rupture rates still influence the indications from international societies. SVS 7 and SICVE 8 suggest 2 cm as a threshold for intervention in asymptomatic CAAs. In all pseudoaneurysms or symptomatic/ruptured true aneurysms, intervention is recommended regardless of the size.
Superior mesenteric artery and inferior mesenteric artery aneurysms
Superior mesenteric artery aneurysms (SMAAs) are reported with a frequency of 3%–18% among VAAs in different studies.2,3,10 Early publications reported a significant rate (up to 30%) of SMAAs of mycotic origin, especially in patients aged <50 years with endocarditis, along with a higher prevalence in male patients. 49 However, more recent studies have observed that non-mycotic causes (dyslipidemia, polyarteritis nodosa, and collagen diseases) are still the most common etiology of SMAAs. Mycotic causes remain important, especially in patients with endocarditis positive cultures for Streptococcus.50,51 Aneurysmal degeneration following spontaneous isolated mesenteric artery dissection has been also described as a possible etiology, and is usually associated with a high tendency to rupture. 52 Despite being a minority of cases, symptomatic aneurysms usually present with abdominal pain. Pulsatile mass, fever, nausea/vomiting, jaundice, or gastrointestinal hemorrhage are also described.49–51
As previously said, data on the natural history and rupture risk of SAAs are limited. A retrospective series including six SMAAs showed a mean growth of 2 mm with one rupture (diameter 3.6 cm) during a mean follow-up of 50 months. 2 Another study reported no significant growth (initial diameter: 16.5 mm) and no ruptures of 13 SMAAs over a mean follow-up of 57 months. 4 However, rupture of SMAAs is a life-threatening condition with a reported operative mortality of up to 38%, and is reported to be more common in SMAAs of mycotic origin.50,51
A recent meta-analysis reported only 95 published cases of SMAA intervention, 57 of which involved open surgery. 3 The mortality rates have been significantly lower in comparative series evaluating endovascular versus open approach, 0% versus 17% respectively,53,54 but the paucity of cases hindered accurate inferential analysis. 3
The scarcity of data on the natural history and operative outcomes of SMAAs has led to different interpretations of current evidence from international societies. SVS guidelines 7 suggest treating all asymptomatic true and false SMAAs, irrespective of size. Notably, this indication included both mycotic and non-mycotic aneurysms, possibly explaining the precautionary recommendations. On the other hand, SICVE guidelines 8 appear more prone to conservative management, with a suggested threshold of 2 cm for surgical treatment of SMAAs. However, mycotic and post-dissection aneurysms are recommended for treatment of any size because of their recognized tendency to rupture.
Inferior mesenteric artery aneurysms (IMAAs) are extremely rare conditions. 2 Actual knowledge of the pathology mainly comes from sparse case reports. Collagen disorders have been reported as a possible etiology, 55 but multiple publications have described IMAAs in patients with chronic steno-occlusion of other visceral branches (celiac trunk, hepatic artery, SMA) or the iliac axes.56,57
All the reported cases were asymptomatic, and both open and endovascular approaches have been described. However, owing to the paucity of cases and the absence of cohort studies, it is impossible to draw conclusions about this pathology. No specific guidelines for IMAAs are currently available.
Gastroduodenal artery and pancreaticoduodenal artery aneurysms
Gastroduodenal artery aneurysms (GDAAs) and pancreaticoduodenal artery aneurysms (PDAAs) are rare conditions, accounting for 1%–2% of all VAAs. 2 They are often described in patients with stenosis or occlusion of the celiac artery or it branches, also known as Sutton–Kadir syndrome.58,59 The association with median arcuate ligament syndrome has been described as well. 60 It should be noted that these entities are rare, but the VAA in the presence of upstream stenosis classification has been recently proposed. 60 This anatomical classification takes into account the aneurysm and stenosis location. However, the timing of either revascularization or ligament release as well as aneurysm exclusion remains vague with conflicting evidence leaving the surgical plan to the treating physicians and mostly decided on case-by-case basis.59,60 Pseudoaneurysms of the GDA and PDA have been reported to be more frequent than true aneurysms in some studies. 61
GDAAs and PDAAs are more prone to rupture, even at smaller diameters (<10 mm). 2 A retrospective analysis of 59 true PDAAs reported that 32% of cases presented with rupture at a mean size of 16.4 mm, whereas asymptomatic patients presented with a mean size of 19.4 mm. 62 The surveillance cohort consisted of eight cases (mean diameter: 13.1 mm), and no rupture was reported during a 5.5-year follow-up. 62 Another publication on 264 SAAs identified PDAAs and GDAAs as independent risk factors for rupture ( Odds ratio 11.2). 63 As for other VAAs, mortality in patients presenting with rupture has been described to be higher than elective surgery (10%–12.5% versus 1.3%–5.6%).62,64
Both endovascular and open approaches have been described for surgical management of PDAAs and GDAAs. Endovascular management has progressively gained more importance and it is now reported as the treatment of choice in several publications.3,62,65 A comparative study was conducted on 11 surgical treatments for PDAAs and reported high technical success in both open and endovascular approaches (100% and 91%, respectively) with a higher perioperative death rate for open surgery. 65 An additional publication on 37 interventions for PDAAs reported a mortality rate of 5.6% and 10.5% after repair of intact and ruptured aneurysms, respectively. Reintervention was needed in 28% of the patients. 62
The SVS 7 and SICVE 8 guidelines recommend early intervention for GDAAs and PDAAs due to their high rates of rupture. In fact, both societies recommend intervention for GDAAs and PDAAs and pseudoaneurysms of any size.
However, interventions of any size have been debated. Despite their high frequency of rupture, the natural history of unruptured aneurysms is still unclear, and interventions at any size may expose patients to unnecessary operative risks.
Jejunal, ileal, and colic artery aneurysms
Jejunal, ileal, and colic artery aneurysms are rare conditions, representing <1% of all VAAs.3,10
The literature on these conditions consists mainly of case reports and limited cases in wider studies on grouped VAAs. Therefore, it is impossible to establish a precise natural history and rupture risk of these aneurysms. Different cases have been associated with collagen disease, polyarteritis nodosa, and other autoimmune diseases.66,67 Although ileal and jejunal aneurysms are often accidentally diagnosed as asymptomatic lesions, colic artery aneurysms have been reported to be associated with abdominal pain in most cases. Rupture rates are not well established but seem to be higher in colic artery aneurysms, with a reported mortality of 20%–50%.49,68
Owing to their rarity, management strategies for jejunal, ileal, and colic artery aneurysms are not well standardized. However, endovascular treatment via coil embolization was preferred in recent reports.69,70
The SVS 7 and SICVE 8 guidelines share the same treatment recommendations. Asymptomatic jejunal and ileal artery aneurysms should be considered for interventional management in cases with a diameter ≥2 cm. Asymptomatic colic artery aneurysms should be considered for treatment at any size because of their frequent presentation with rupture.
Conclusions
VAAs are rare, but potentially fatal conditions. Research on this topic remains challenging because of the rarity of the pathology. The majority of studies are retrospective single-center cohort studies or case series, resulting in insufficient data regarding both the natural history and appropriate management of these pathologies. Furthermore, data on the long-term outcomes of various interventions remain insufficient. Nowadays, endovascular approaches are preferred because of their lower invasiveness and better outcomes in terms of morbidity and mortality compared to open surgery. However, long-term durability remains debatable, especially in younger patients or in those with a higher risk of recurrence. Open surgical repair remains the mainstay in aneurysms involving a distal artery of end-perfusion organs or in anatomical scenarios not suitable for endovascular repair. The goal of VAA repair should be to exclude the aneurysm by preserving the correct flow to the organs, with both endovascular and open approaches.
An additional challenge in the management of these lesions is the determination of appropriate follow-up protocols, both for patients treated conservatively (due to an uncertain natural history) and for those who have undergone intervention. Indeed, certain interventions, such as VAA coil embolization, can significantly impede aneurysm visualization during routine follow-up examinations, thereby complicating the selection of optimal imaging modalities.
Given the paucity of evidence in the literature, current guidelines lack robust recommendations and exhibit considerable variation among professional societies. For instance, there is no consensus regarding the optimal size threshold for intervention in asymptomatic aneurysms, and these thresholds are likely to be revised in accordance with the forthcoming ESVS guidelines, which are expected in 2025. An enhanced comprehension of the natural history of asymptomatic aneurysms, long-term outcomes of different interventions, and optimal strategies for follow-up imaging would facilitate evidence-based decision-making. Ultimately, a multidisciplinary approach involving radiologists, vascular surgeons, and interventionists is essential to provide optimal care for patients with VAAs.
Footnotes
Author contributions
Nicola Leone: Conceptualization; Formal analysis; Methodology; Project administration; Writing—original draft preparation; Writing—review & editing.
Mattia Migliari: Investigation; Methodology; Writing—original draft preparation; Writing—review & editing.
Giovanni F. Baresi: Formal analysis; Investigation; Writing—original draft preparation; Writing—review & editing.
Stefano Gennai: Conceptualization; Writing—review & editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
