Abstract
Lymphedema occurs when the lymphatic system is malformed, leaking or is damaged by infection or cancer treatment. Enlarged extremities due to extensive fluid retention and adipose tissue deposition, recurrent cellulitis, and the requirement for hard-to-wear compression garments lead to impaired limb function and decreased quality of life. This narrative review aims to elucidate classification, diagnostic tools, and conservative management strategies leading the path to patient selection for microsurgical reconstructive procedures or volume reduction procedures. The surgical approaches include reconstructive procedures such as lymphaticovenous anastomosis (LVA) and lymph node transfer and reductive procedures, mainly liposuction and occasionally excisional surgery. Comparing the efficacy of lymphedema surgeries is fully dependent on standardized and reproducible outcome measures. A growing body of knowledge has evolved pointing to the benefits of reconstructive microsurgery of the lymphatic system with or without surgical volume reduction. Tissue engineering and stem cell research leads the way for new approaches. However, several questions remain emphasizing the research needs. Ongoing multicentre trials aiming to elucidate the efficacy and accessibility of surgical treatments for this condition are on their way.
Introduction
Lymphedema is a chronic and progressive condition characterized by the accumulation of protein-rich fluid and tissue swelling due to impaired lymphatic drainage. It affects millions of people worldwide and can significantly impact quality of life. 1 Lymphedema can be categorized into primary and secondary forms. Primary lymphedema is caused by congenital abnormalities in the lymphatic system and can present at various ages, while secondary lymphedema arises from damage to the lymphatic system most often due to surgery for cancer treatment, or infection. Secondary lymphedema tends to develop several months after the injury to the lymphatic system, and the risk continues in subsequent years.
Minimizing the extent and morbidity of surgery to draining lymph node basins without affecting recurrence or survival rates is an evolving area of cancer management. The high incidence of breast cancer, long survivorship, and the fact that one in five breast cancer patients will develop arm lymphedema means that breast cancer-related lymphedema is the leading cause of lymphedema referrals in western health care settings. 2
For breast cancer staging, a huge leap forward was the introduction of sentinel lymph node biopsy. Further advances in breast cancer care include omission of completion lymph node dissections in favor of radiotherapy, the use of targeted axillary dissection after neo-adjuvant chemotherapy, delayed sentinel lymph node biopsy in ductal carcinoma in situ with the aid of superparamagnetic iron oxide nanoparticles (SPIONs) or Indocyanine green (ICG) marking and the avoidance of sentinel lymph node biopsy in selected patients with low-risk cancers.3–7 Similarly, management of the draining lymph node basins in melanoma has become less morbid with the introduction of sentinel lymph node biopsy, avoidance of completion lymph node dissection in favor of surveillance, and the introduction of immunotherapy and targeted treatments.8–10
The management of lymphedema involves a multidisciplinary approach, including conservative therapies such as complete decongestive therapy (CDT), as well as surgical interventions in selected cases. This review aims to provide an overview of modern surgical treatments for lymphedema, patient selection for these techniques, evidence, and future directions.
Clinical assessment and outcome measures
Patients referred to a tertiary lymphedema service generally have an established clinical diagnosis, often with a lymphoscintigraphy showing impaired lymphatic drainage (Table 1). At the tertiary center, the focus is on lymphatic function assessment, volume assessment, extremity content assessment, and patient-reported outcomes measures (PROMs). ICG lymphography, high-resolution ultrasound, and magnetic resonance (MR) lymphography are useful in confirming the diagnosis, aiding patient selection for different surgical procedures, and assessing clinical outcomes of surgery (Table 1). The center may also support patients in adjusting and maintaining conservative treatments such as optimal compression garment use, weight control, and physical activity. The clinical assessments are completed with limb volume assessments by circumferential measurements and/or perometry and bioimpedance assessing fluid content. Standard photographs and three-dimensional (3D) photography are also commonly used (Table 1).
Clinical and indocyanine green (ICG) lymphography staging systems for lymphedema.

ISL: International Society of Lymphology; ICG: indocyanine green.
Importantly, PROMs are an essential tool in assessing the impact of interventions on health-related quality of life. Efforts are underway to find a consensus on which PROMs should be emphasized in lymphedema to standardize outcomes. Some of those currently used are listed in Table 1. 13 In addition, the number of episodes of cellulitis is a commonly used outcome measure in lymphedema studies. 14
Classification
Accurate classification of lymphedema is essential for assessing disease severity, guiding suitability for surgery and using research outcomes to guide clinical decision-making. Classification can be based on the clinical picture (International Society of Lymphology System) or ICG imaging (MD Anderson ICG staging) (Table 2). Different areas of the extremity may exhibit different severity levels/stages of lymphedema.
Summary of assessment and outcome measures.

ICG: indocyanine green; MRI: magnetic resonance imaging.
Conservative management
Conservative management remains the cornerstone of treatment for lymphedema. Early and adequate compression treatment can significantly reduce progression of lymphedema. 1 High costs, variability in the quality of therapy, and patient compliance can hinder the efficacy of CDT. The CDT aims to reduce swelling, improve lymphatic circulation, prevent complications such as cellulitis, and enhance overall quality of life. 25 While CDT can effectively manage mild-to-moderate lymphedema, surgical interventions may, however, be beneficial as an adjunctive treatment to reduce symptoms, prevent progression of disease, address cases where compression is difficult or not possible, and for advanced cases to reduce limb volume.
Surgery
Surgery for lymphedema can be broadly characterized as reconstructive procedures and reductive procedures. Reconstructive procedures focus on restoring lymphatic function and fluid drainage while reductive procedures aim to reduce tissue volume. Patient selection is based on different factors including lymphedema stage, residual lymphatic function, patient factors (comorbidity, etc.), and patient needs as well as the potential morbidity of the procedure.
Reconstructive procedures
LVA
LVA bypasses the defective lymphatic system by connecting functional lymphatic channels distal to the affected lymph node basin to adjacent subdermal veins (Figs 1 and 2). LVA surgery is generally indicated for early-stage lymphedema (International Society of Lymphology (ISL) stages 0–2).
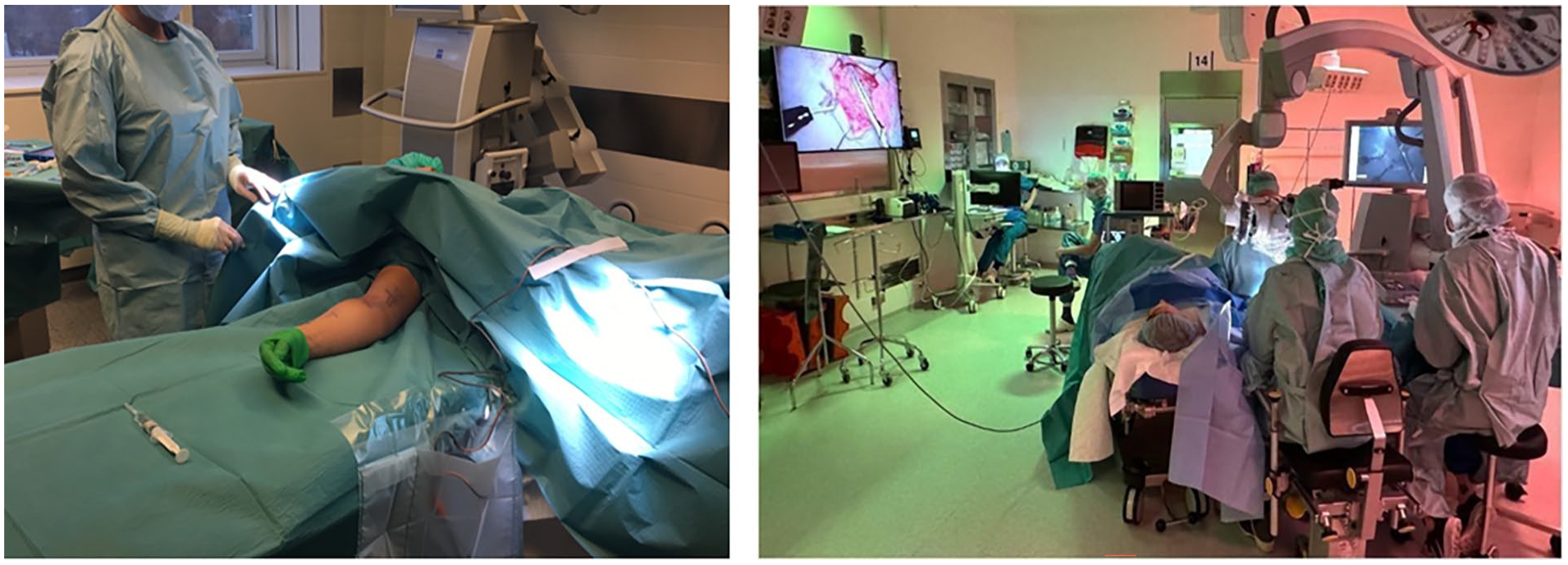
Operative setup for upper limb LVA being performed under local anesthetic giving the patient the opportunity to observe the procedure on the screen if they wish.
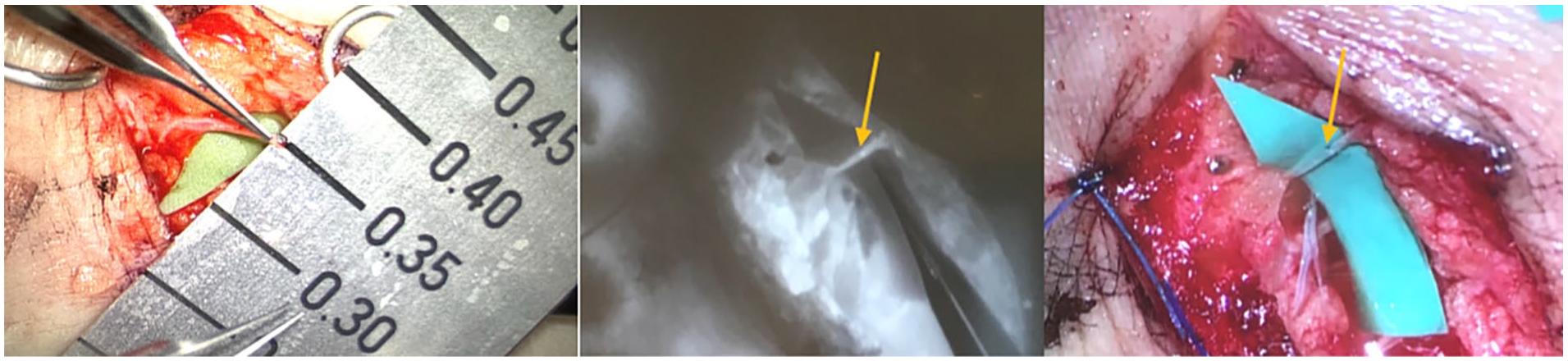
Intraoperative images of lymphatic channel measuring 0.35 mm (left), intraoperative ICG showing lymphovenous flow (yellow arrows) after anastomosis (middle) and intraoperative picture (right) of the same completed LVA (normal light).
The use of supermicrosurgical techniques involving anastomosing vessels less the 0.8 mm in diameter for LVAs was developed by Koshima in the 1990s and has become widespread. 26 An alternative microsurgical technique of inserting multiple lymphatic channels end to end into the lumen of a vein or end to side and securing with a U-shaped suture has also been described. 27
Potential LVA sites are identified preoperatively using ICG lymphography and/or high-resolution ultrasound. An ideal site is characterized by a transition zone where a functioning channel ends, turning into dermal backflow on ICG lymphography. The LVAs can be carried out under local anesthetic meaning that it can be performed on an outpatient basis and could be used for co-morbid patients. A 2- to 3-cm skin incision is made over the planned LVA site, and careful subdermal dissection is performed under the microscope with the aid of intraoperative ICG lymphography or patent blue dye to identify suitable lymph channels (Fig. 2 and Supplemental Video 2). Adjacent venules with a favorable direction of flow and low pressure are chosen. Anastomotic technique can be decided based on the caliber and number of vessels available and the desire to maintain lymphatic channel continuity. Common options for anastomosis are illustrated in Fig. 3. Compression is applied post-operatively which is important in overcoming venous back flow and to optimize lymphovenous flow. 28
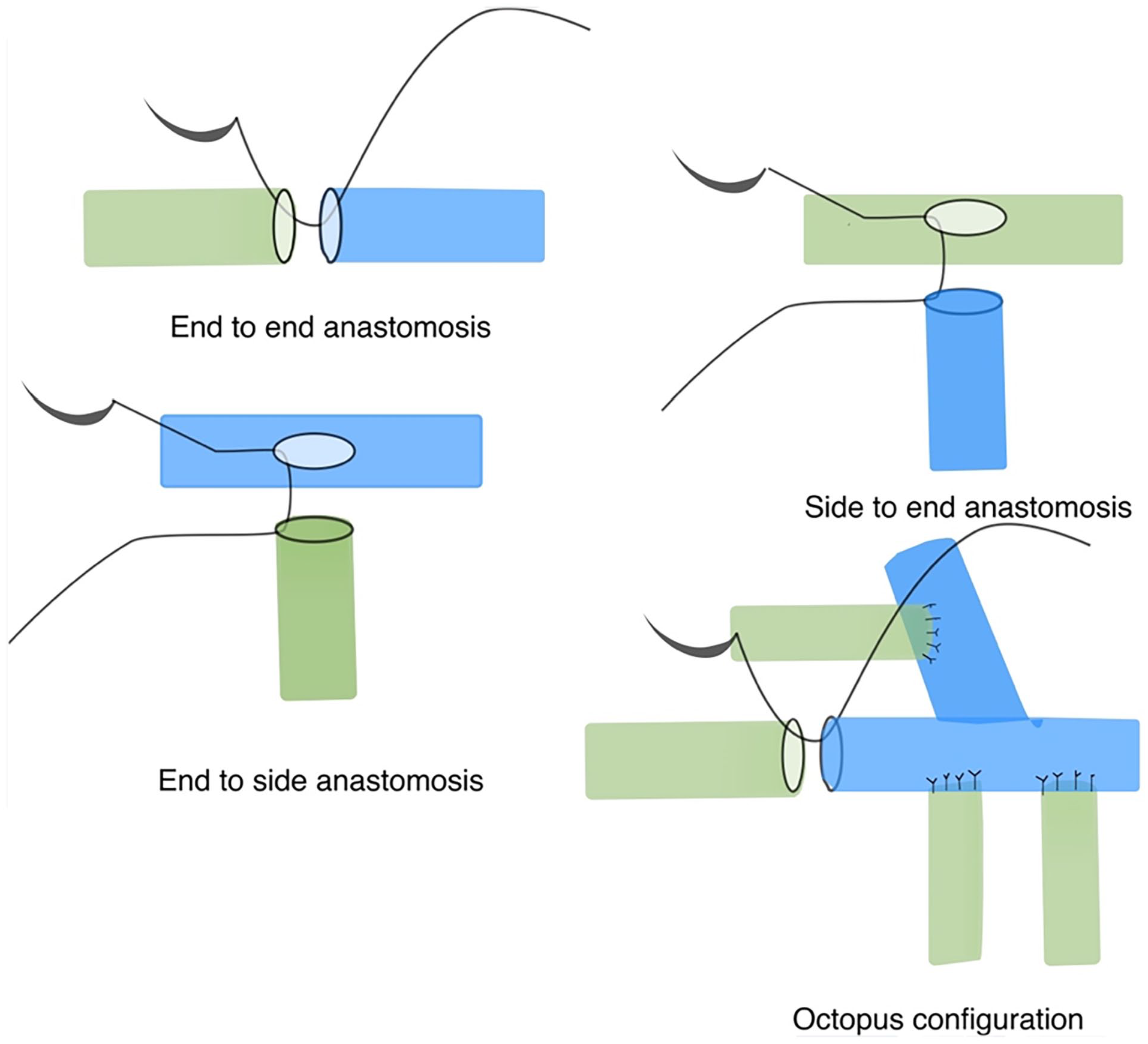
Various configurations of LVAs.
Although promising results showing reduction in limb volumes and a reduced requirement for compression garments have been reported for many years from pioneers of these techniques, there has been a requirement for high-quality randomized control trials (RCTs) to critically analyze the impact on quality of life, symptoms, and cellulitis episodes.27–31
There is evidence from one RCT comparing combined LVAs and CDT with CDT alone for patients with secondary lower extremity lymphedema showing a reduction in the incidence of cellulitis. 32 This trial reported no significant difference in thigh circumference or pain. Interim results from a multicentre RCT in the Netherlands also comparing LVAs and CDT with CDT alone showed improved health-related quality of life and a higher rate of discontinuation of compression garments at 6 months. 33
Further RCTs are ongoing including the PRELUDE trial (NCT04579029) in the United Kingdom comparing LVAs combined with compression with compression alone, the N-LVA study (NCT06082349) in the Netherlands comparing LVAs with sham surgery and the LYMPHA-Trial that is an international multi-center trial comparing any surgical intervention with compression. 34
LVA can also be performed prophylactically at the same time as lymphatic clearance. 35 Lymphatics are identified at the time of lymph node dissection with the aid of reverse lymphatic mapping, clipped and later anastomosed to an adjacent vein. These may be performed end to end, end to side, or using an intussusception technique where multiple lymphatics are placed into the lumen of a single outflow vein and held in place with a horizontal mattress suture. 36 There are several published case series using this technique reporting lower rates of lymphedema at early follow-up compared with historical cohorts.35–38 A study with longer-term follow-up, however, found similar rates of lymphedema in prophylactic LVA-treated and untreated groups. 39 An RCT is underway with promising early results reported on quality of life and compression use, which should help to guide the use of immediate lymphatic reconstruction. 40 The search for predictive factors for lymphedema development is essential to guide the indications for prophylactic interventions so that overtreatment is avoided.
Technical challenges with performing LVAs may include difficulty in performing a patent anastomosis of very small vessels. Robot-assisted supermicrosurgery aims to overcome the limitations of surgeon dexterity by eliminating tremor. The da Vinci system, which is the most used surgical robot worldwide, was developed for endoscopic surgery. The instruments are large, and its limited resolution and magnification are not optimized for supermicrosurgery. As a solution, two different systems have been developed that are the Microsure system from the Netherlands and the Symani system from Italy. These have been optimized to eliminate tremor and scale motion as the surgeon movement is copied by the robot with a scaled factor. Both systems have been trialed for performing LVAs, and next-generation robots are being developed with further improvements and applications (Fig. 4).41–43
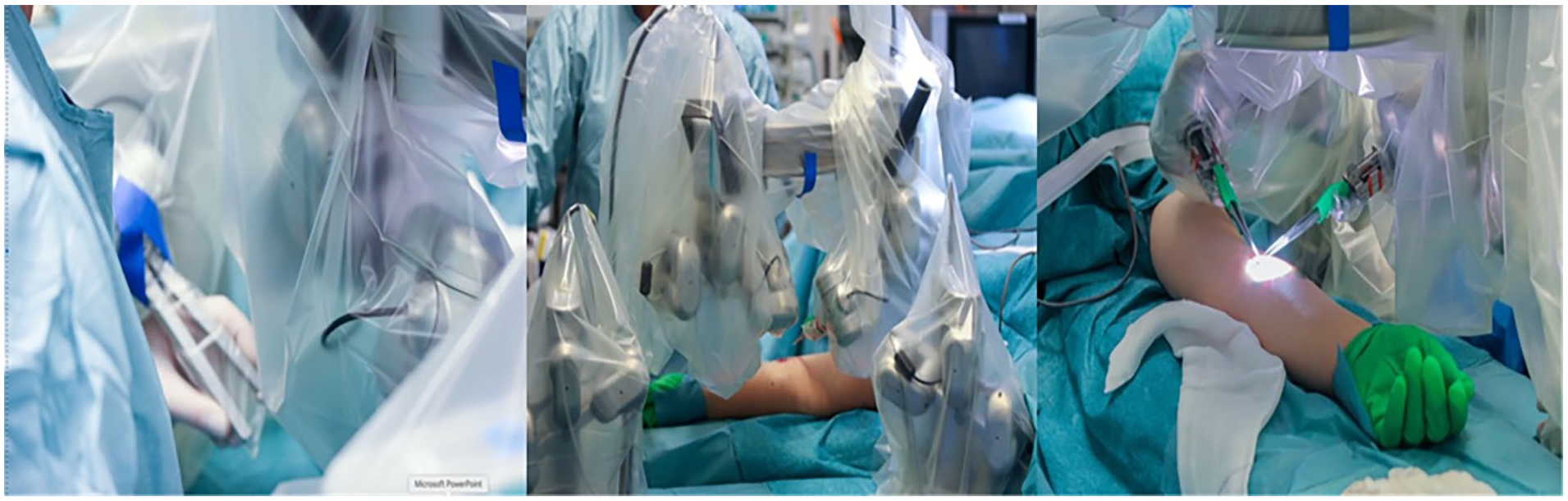
Intraoperative setup for robotic LVA using Microsure system.
Lymph node transfer
Lymph node transfer involves harvest of lymph nodes as a microvascular free flap and transfer to the affected limb. Potential lymph node donor sites include the axilla, cervical region, groin, and omentum (Fig. 5). This technique does not include any anastomosis between the lymphatic vessels and venous system but only standard microsurgical venous and artery anastomosis for circulation of the transplanted tissue. The lymph node transfer technique rather relies on the process of lymphangiogenesis. It can be performed when no functional lymphatic channels can be identified to perform LVAs. Importantly, in cases of breast reconstruction with abdominal free tissue, that is, the deep inferior epigastric perforator (DIEP) flap, lymph node transfer to the axilla can be combined with the flap reconstruction with minimal increase in duration of the surgery and donor-site morbidity (Fig. 6). 44
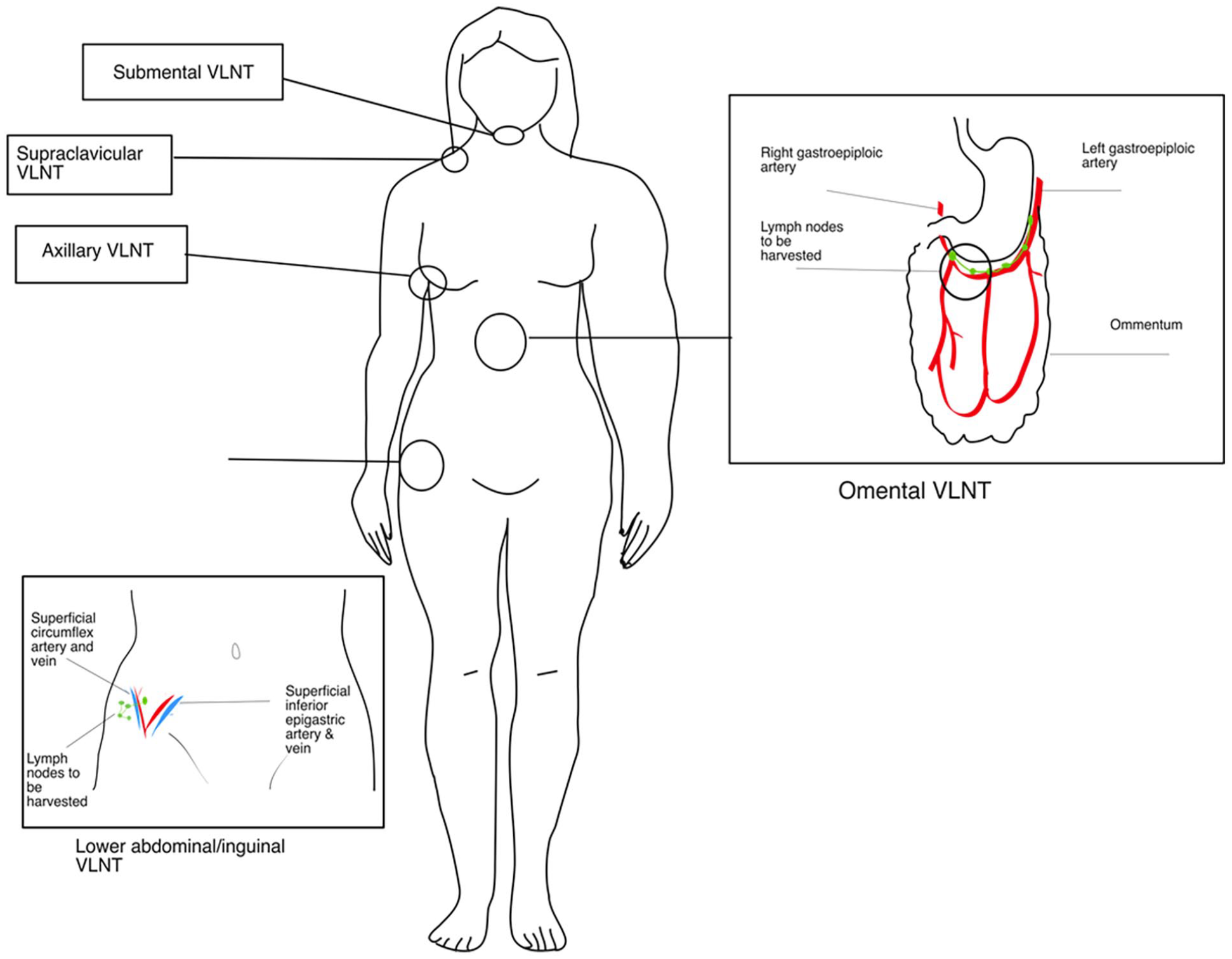
Various donor site for vascularized lymph node transfer (VLNT)
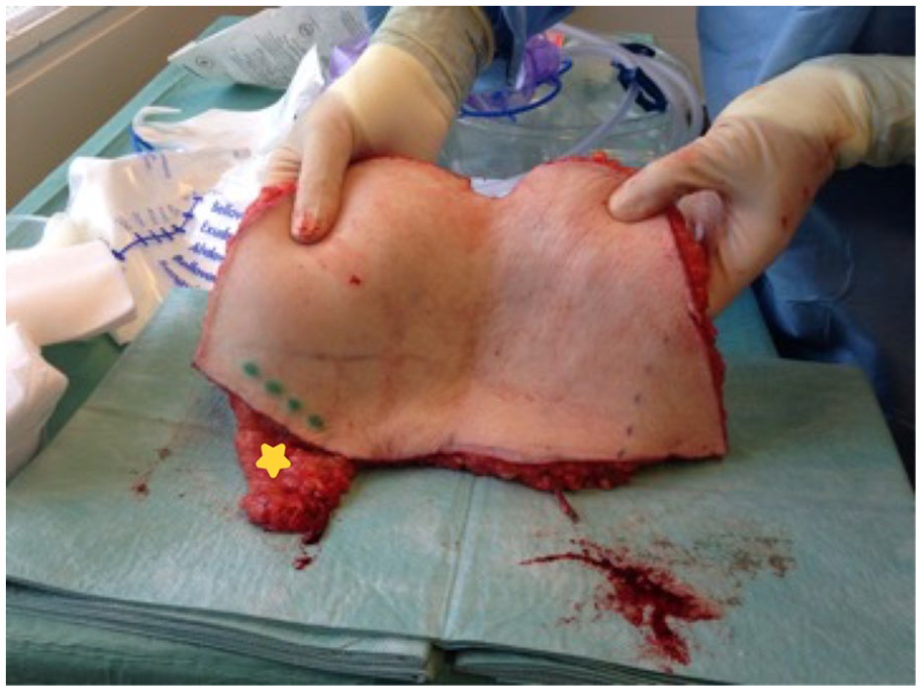
Combined DIEP free flap (from abdomen) and vascularized lymph node transfer with lymph nodes (yellow star) harvested from right inguinal region above the groin to be used for breast reconstruction and concomitant treatment of ipsilateral arm lymphedema. The DIEP is formed into a breast, and the tissue with the lymph nodes is positioned in the axilla after scar release.
The method of action of lymph node transfer is thought to be due to several contributing factors. In cases of secondary lymphedema where the flap transfer is to a previous lymph node basin, such as the axilla, an extensive scar release and excision should be performed as this may affect mobility and venous as well as lymphatic drainage. The transferred lymph nodes produce growth factors, such as vascular endothelial growth factor C (VEGF-C), that promote lymphangiogenesis. 45 There is evidence from experimental animal models that integration of flap and recipient site lymphatics occurs when the flap is transferred early after lymph node clearance. 44 In addition, there is evidence of neolymphangiogenesis from histological samples taken from the area of flap transfer in patients 1 year after surgery. 46 Radiological evidence of flow between afferent lymphatics and transferred lymph nodes has moreover been demonstrated on lymphoscintigraphy. 47 The transferred lymph nodes may also have an immunological function acting to prevent infections in the lymphedematous limb. 48
An alternative technique is to transfer the lymph node flap distally in the lymphedematous limb, for example to the wrist or ankle. Distal transfers also work via lymphangiogenesis, acting as a pump by diverting lymph fluid into the venous circulation. 49 When transferring the combined DIEP and lymph node flap, the DIEP vessels are anastomosed to the internal mammary vessels and a second anastomosis of the pedicle (superficial circumflex iliac artery/vein or superficial inferior epigastric artery/vein) supplying the lymph node flap to axillary vessels, usually the thoracodorsal vessels or if these are not available to the lateral thoracic or subscapular vessels. 50
Dionyssiou et al. 51 have demonstrated reduced incidence of cellulitis and improved functional status in an RCT comparing lymph node transfer with conservative management for breast cancer-related lymphedema.
A rate of 1.6% of donor-site lymphedema has been reported with the harvest of inguinal lymph nodes in combination with a DIEP flap. 52 To reduce the risk of iatrogenic lymphedema in the limb distal to the lymph node donor-site, reverse lymphatic mapping with technetium or ICG is recommended, as well as limiting the dissection in the inguinal region. 53 This aims to identify the lymph nodes draining the extremity that must be preserved to minimize the risk of donor-site lymphedema.
There is an ongoing RCT (LYNT study, ISRCTN10851194) comparing scar release and fresh tissue transfer to scar release and lymph node transfer in patients planned for DIEP breast reconstruction and who have lymphedema of the ipsilateral arm.
Ablative procedures
Liposuction
In established chronic lymphedema, there is hypertrophy of adipose and fibrous tissues, and as a result, reductive procedures such as liposuction or excision surgery can be indicated to reduce this excess. 54 Liposuction for lymphedema was reported by O’Brien et al. 55 Tourniquet use or a tumescent technique with infiltration of adrenaline solution in areas where a tourniquet cannot be applied can minimize blood loss and enable safe large-volume liposuction. 54 The procedure can be performed under general anesthetic or regional block. Multiple small incisions are used to perform power-assisted liposuction to the limb (Fig. 7 and Supplemental Video 3). A compression garment is made preoperatively sized to the contralateral unaffected limb, and compression is continued lifelong to maintain the volume reduction achieved by liposuction.
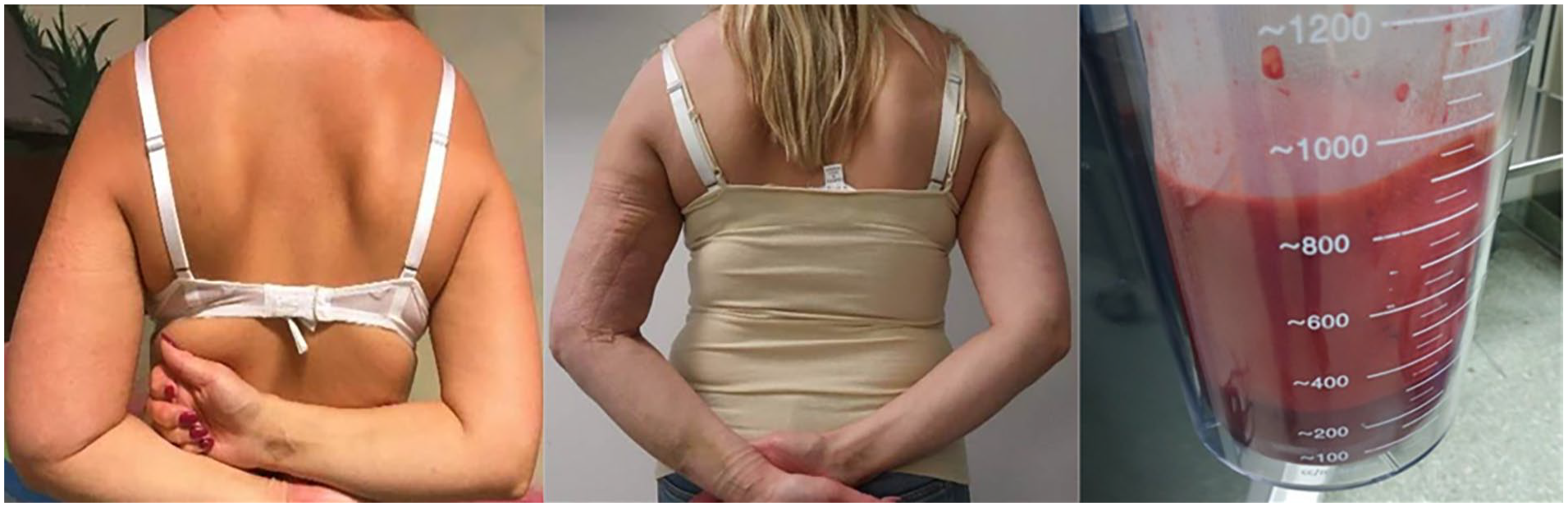
Pre-operative (left) and 4 weeks post-operative (middle) photographs of patient with breast cancer-related lymphedema and 600 mL excess upper limb volume treated with liposuction. Picture on right showed lipoaspirate removed.
A combination of liposuction and compression garments has been shown to produce long-term reduction in excess limb volume, improved limb function, and quality of life.56–58 In a systematic review, the average reduction in excess volume with liposuction was 96.6%. 59 Extensive scarring occurs after liposuction limiting the repeated use of this technique. Therefore, it is recommended to remove all excess adipose tissue in one operation.
Excisional procedures
The Charles procedure, first described in 1912, involves radical resection of skin and subcutaneous tissue of the extremity to the deep fascia followed by skin grafting. While the Charles procedure can achieve significant volume reduction, there are significant risks of graft failure, delayed healing, infection, and hematoma as well as extensive scarring and sensory changes. 59 It has been superseded by the introduction of liposuction as a less invasive option for limb volume reduction in chronic lymphedema. Select cases may have an indication for excisional surgery, such as removal of overhanging skin to improve function or in settings with limited resources where access to more expensive equipment is not available.
Combining techniques
Although reconstructive techniques work to improve the lymph drainage of the limb and prevent worsening of lymphedema, they do not address any pre-existing excess of fibroadipose tissue. Therefore, some centers have proposed combining liposuction with lymph node transfer or LVA in a single session60–62 or performing multiple LVAs and lymph node transfers followed 2 weeks later by liposuction avoiding the sites of LVA and lymph node transfer. 63 In cases where there is a clear indication for lymph node transfer after axillary clearance, radiotherapy, and a lymphoscintigraphy with a high transport index in combination with severe accumulation of lymphatic fat in the limb, there may be an indication to combine a reconstructive procedure with liposuction. 64
The challenges with deciding whether to combine these techniques or use them sequentially include concerns about injury to the LVA or lymph node transfer with circumferential liposuction, difficulty with applying high-grade compression after combined procedures and extensive scarring after liposuction limiting subsequent LVAs. 61 On the contrary, lymphatics may be easier to visualize in the combined setting once fibroadipose tissue is reduced. 60 In addition, combining LVAs and lymph node transfer in the same sitting has also been reported. 65
There is a lack of consensus currently of the practice of combining techniques, and some recommend waiting between performing procedures. 66 Although several small case series have demonstrated the technical feasibility of combining techniques, a more thorough evaluation with RCTs is required to develop appropriate treatment regimens. Furthermore, performing multiple procedures at the same time challenges the evaluation of the therapeutic effect of the different parts of the procedures performed and which technique is leading to a particular outcome.
Patient selection
Patient selection for a particular technique varies across institutions with some advocating an initial 3 to 6 months of CDT and offering surgery to those who respond poorly respond or have recurrent episodes of cellulitis. 67 The author’s own local approach has been to offer the procedure with the lowest morbidity, that is LVAs, to motivated patients. They should have had a minimum of 6 months of compression treatment and are otherwise fit for surgery, and the LVA procedure needs to be technically feasible. Patients are subjected close follow-up of clinical outcomes including limb volumes, episodes of cellulitis, and PROMs. Standardized protocols and review timepoints are used including assessments with and without compression garments pre-operatively and at 1 year. In cases where there is a poor response, there is the possibility to proceed to more invasive procedures such as lymph node transfer or liposuction.
Future
To optimize outcomes of reconstructive techniques for lymphedema, efforts have been made to combine regenerative therapies with surgery. VEGF-C is an important growth factor in lymphangiogenesis and has shown promise in animal studies at promoting integration of lymph node transfer to the recipient site. Results from an RCT are currently underway evaluating the effect of combining VEGF-C gene therapy with lymph node transfer. 68 Other areas of research include the use of adipose-derived stem cells combined with scar release in the axilla for breast cancer-related lymphedema and the development of a collagen conduit for treatment of lymphedema.69,70
Future research in lymphedema surgery should focus on optimizing patient selection criteria including those for prophylactic surgery, refining surgical techniques, standardizing outcome measures, and exploring novel approaches such as robotics and regenerative therapies. Randomized controlled trials comparing different surgical interventions and long-term follow-up studies are awaited to establish evidence-based guidelines and improve the accessibility of surgical treatments for lymphedema.
Lymphedema surgery represents a promising option for patients with refractory or advanced lymphedema, offering the potential to alleviate symptoms, reduce complications, and improve quality of life. While surgical interventions have shown encouraging outcomes, further research is warranted to enhance their efficacy, safety, and accessibility. Collaboration between clinicians, researchers, and patients is essential to advance the field of lymphedema surgery and address the unmet needs of individuals living with this debilitating condition.
Footnotes
Acknowledgements
Maria Mani has been a medical advisory board member of Microsure since 2023.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Informed consent was obtained for all patient images included.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

