Abstract
Background and aims:
There is no consensus on which Lisfranc injuries can be treated non-operatively. The aim of the study was to compare non-operative treatment and open reduction and internal fixation (ORIF) in the treatment of non-displaced Lisfranc injuries.
Materials and methods:
This study was a multicenter randomized controlled trial (RCT) conducted at two hospitals in Finland between 19 March 2012, and 20 December 2022, with a target sample size of 60 patients. The primary outcome was Visual Analogue Scale Foot and Ankle (VAS-FA) at 2 years. The secondary outcomes included VAS-FA pain, function, and other complaints subscales and the American Orthopedic Foot & Ankle Society (AOFAS) Midfoot Scale. All outcomes were measured at 6 months, 1 and 2 years.
Results:
Altogether 27 patients with computed tomography (CT)-confirmed non-displaced Lisfranc injuries were enrolled in this trial resulting in an underpowered trial. In patients with non-displaced Lisfranc injuries, the mean VAS-FA overall score in the non-operative group was 96.1 [confidence interval (CI): 91.5–100] and 91.8 [86.9–96.7] in the ORIF group at 2 years with no statistically significant difference between the groups (mean between-group difference (MD) 4.3 [CI, −2.4 to 11], Cohen’s d = 0.706) in this underpowered RCT.
Conclusion:
There was no difference in VAS-FA between non-operative and ORIF in patients with non-displaced Lisfranc injuries, but the trial is underpowered to draw robust conclusions.
Introduction
Lisfranc injury, as known as injury on the tarsometatarsal (TMT) joint covers broad spectrum of injuries from subtle distensions to open fracture dislocations.1–5 The incidence of Lisfranc injuries based on computed tomography (CT) is around 9 to 14/100,000 person-years.6,7
There is no high-quality evidence-based consensus on the treatment of Lisfranc injuries. 8 Suggested by retrospective studies, displacement or a bony avulsion fracture (“Fleck sign”) between the medial cuneiform and the second metatarsal (MT) bone in weightbearing radiographs or stress test are considered to be a sign of unstable midfoot injury, indicating that these injuries should be treated operatively to achieve higher functional outcomes and lower risk for post-traumatic arthritis.5,9–14 The threshold for clinically relevant displacement has been suggested to be 2 mm,11,15 yet studies are based on weightbearing radiographs, which is shown to have lower sensitivity and specificity than CT. 16 Based on a retrospective cohort study it is possible to have good outcomes if there is no displacement in the CT. 17 Nevertheless, there is no consensus on the injury classification or treatment of Lisfranc injuries based on CT.6,17,18
Although the treatment of Lisfranc injuries has changed over the years, the importance of achieving anatomic reduction has been the main goal for operative treatment.5,19 Open reduction and internal fixation (ORIF) of the affected TMT joints have been the primary surgical method for displaced Lisfranc injuries. ORIF have been traditionally performed with screws, yet more recent studies have also introduced comparable results with temporary bridge plating (BP).20–23 Even though BP has been suggested to be more cartilage friendly than transarticular screws, BP led to high prevalence of post-traumatic arthritis in randomized controlled trial (RCT) study by Stodle (2020), suggesting that the cause of the arthritis is the injury rather than fixation. 23
This RCT compared non-operative and operative treatment (ORIF) for non-displaced Lisfranc injuries. Our hypothesis was that non-operative treatment and ORIF yields similar functional results after 2 years of follow-up.
Materials and methods
The study is a prospective, randomized, national multicenter trial. The patient recruitment was conducted in Tampere University Hospital and Seinäjoki Central Hospital from 19 March 2012 to 20 December 2022. The protocol of this trial was approved by the ethical review board of Pirkanmaa Healthcare District and was previously published. 24 All patients gave written informed consent.
Participants
We enrolled patients sustaining an acute Lisfranc joint injury who were treated at the emergency departments of the study centers. All patients with clinical suspicion (pain, swelling, plantar ecchymosis, or gap sign) or typical findings on plain radiographs (“fleck sign,” avulsion or fracture) of Lisfranc injury led to CT where the diagnosis and morphology of the injury were confirmed. Eligible patients were referred to the outpatient clinic, where one of the clinicians involved in the study met the patients and continued the recruitment process.
Inclusion criteria:
Non-displaced (<2 mm) fractures affecting TMT joints II and III.
No or minimal displacement (distance <5 mm) between medial cuneiform and base of MT II.
No fractures affecting TMT joints IV and V.
Exclusion criteria: Patients aged under 18 or over 60, open fractures, extra-articular MT fractures, extremely comminuted fractures with bone loss and poor chance of gaining proper fixation with screws, polytrauma patients, unable to co-operate (dementia, alcohol use, etc.), significant neuropathy or some other neurological condition, diabetes, rheumatoid arthritis, severe circulatory disorder of the lower limb, diagnostic delay more than 14 days, a previous foot injury or surgery of the injured foot, pregnancy, and patients who refuse to participate.
Randomization
We used computerized program to generate random allocation sequence with 1:1 allocation ratio and a block size of 10. The treatment allocations from the randomization were sealed in opaque envelopes prepared by a research nurse with no involvement in the clinical care of patients. The envelopes were kept locked room at all study centers. The envelopes were opened in numerical order after patient enrolment was confirmed by the research physician.
Interventions
Non-operative treatment
Non-operative treatment was conducted with cast-immobilization and non-weightbearing for 6 weeks. The cast was changed at 1- and 2-week follow-up visits. Since cast removal at 6 weeks a walking boot was prescribed and the patients were advised to half-weightbearing for next 2 weeks and after that reaching weightbearing as tolerated. At 10-week follow-up, patients were allowed to use normal shoes and walk as tolerated.
ORIF
The surgical procedures were performed by experienced foot and ankle surgeons of the study group. Only the affected and instable TMT joints were fixed. The first incision was made between MT I–II and the second incision (if necessary) at the base of MT IV. Open anatomical reduction and screw fixation of the affected TMT I–III joints were be performed with 4.0 cannulated screws (DePuySynthes©, Stryker©). In the surgery, a “home run screw” from second MT base to the medial cuneiform bone was regularly used. Fixation screws were removed only if they caused symptoms.
All patients received antibiotic prophylaxis. Surgery was performed under tourniquet at 280–300 mmHg pressure.
Postoperative treatment
Postoperative care was identical to non-operative treatment, that is, 6 weeks of non-weightbearing cast-immobilization and then 4 weeks of walking boot and gradual weightbearing. Stitches were removed and cast changed in 2 weeks. The cast was removed in 6 weeks. Deep vein thrombosis prophylaxis and analgesic medication was prescribed individually.
Outcomes
The primary outcome was the Visual Analogue Scale Foot and Ankle (VAS-FA) overall score measured at 24 months. VAS-FA has been shown to be a valid patient-reported outcome measure to measure the function and symptoms of foot and ankle25,26 (score range, 0–100; 0 denotes extreme disability and 100 no disability; minimal clinically important difference (MCID) is 6.8 for total VAS-FA score, and 9.3 for the Pain, 5.8 for the Function, and 5.7 for the Other complaints subscales. 27 VAS-FA and AOFAS Midfoot Scale were collected from all participants throughout the trial. When the trial began The AOFAS Midfoot Scale was planned to be the primary outcome measure, 24 yet it was later shown to be insufficient to distinct patients’ function due to high amount of ceiling values. 28 During this trial, the validity of the Finnish version of VAS-FA was shown to be high 25 and the MCID value was presented 27 and thus VAS-FA measured at 24 months was chosen to be the primary outcome measure. Both instruments were collected from the patients throughout the trial.
The secondary measured outcomes at 6, 12, and 24 months were VAS-FA pain, function and other complaints subscales, AOFAS Midfoot Scale (0–100, 0 extreme disability, 100 no disability, MCID 12.4 29 ), 30 number of secondary operations (implant removal, secondary arthrodesis) and number of other treatment-related complications.
The questionnaires were administered at each follow-up visit (6, and 12 and 24 months after randomization). Data regarding demographic and clinical characteristics were collected at baseline.
Clinical and radiological evaluation was performed at each follow-up. Anteroposterior, oblique, and lateral views were used. Fracture fixation and its possible failures as well as delayed unions were recorded.
After the primary completion of the trial, a member of the study group collected data on complications, adverse events, and reoperations from medical reports. Adverse events were categorized as serious or minor. Minor adverse events included wound numbness, hyperesthesia, infection, and delayed healing. Major adverse events included deep vein thrombosis, secondary operations, and death.
Power analysis
Power analysis was based on AOFAS Midfoot Scale. Assuming a 10-point difference in the mic score and a standard deviation (SD) of 12 points, the estimated sample size is 23 patients per group (delta = 10, SD = 12, alpha = 0.05, power 0.8). With assumed 20% drop-out rate in all groups, the total patients needed would be 56 patients. Due to block randomization in blocks of 10, 60 patients was the planned sample size.
Based on the results of the study, we updated the power calculations, and the required sample size did not differ from our original calculations with 23 patients per group (Supplement 2).
We were unable to reach the previously planned sample size due to challenges with recruiting, and therefore this trial remains to be underpowered. As the recruitment was carried for more than 10 years, we decided to end the trial as it was clear that we are unable to reach the proposed power.
Statistical analysis
The baseline characteristics were reported as mean with SD, median with quartiles or as count with proportion. The primary comparison between groups was performed using a repeated-measures mixed-model (RMMM). Patients were included as random factors and study group and time of follow-up (6, 12, and 24 months) were included as fixed factors. The model included interactions between study group and time of follow-up. The model was used to evaluate the treatment effect as the absolute difference between the groups in primary and applicable secondary outcome measures (mean with 95% confidence intervals (CIs) and p value). Effect size was calculated as Cohen’s d, where d = 0.2 represents small, d = 0.5 medium and 0.8 = large effect size. 31 The data were analyzed according to the intention-to-treat principle, assuming the patients change group during the study. Imputations of missing data were not conducted. The statistical analysis was performed with R version 4.0.3 (R Foundation for Statistical Computing, Vienna, Austria).
Results
Characteristics of the patients
Altogether 27 patients gave written consent to participate and were included in the non-displaced study stratum. Of these patients, 15 (56%) were allocated to non-operative treatment and 12 (44%) to ORIF. Total of six patients were lost to follow-up at 1 year and total of eight patients at 2-year follow-up; therefore, 21 patients were analyzed at 1-year and 19 patients at 2-year follow-up point (Fig. 1). The baseline characteristics of patients in each group are presented at Table 1.
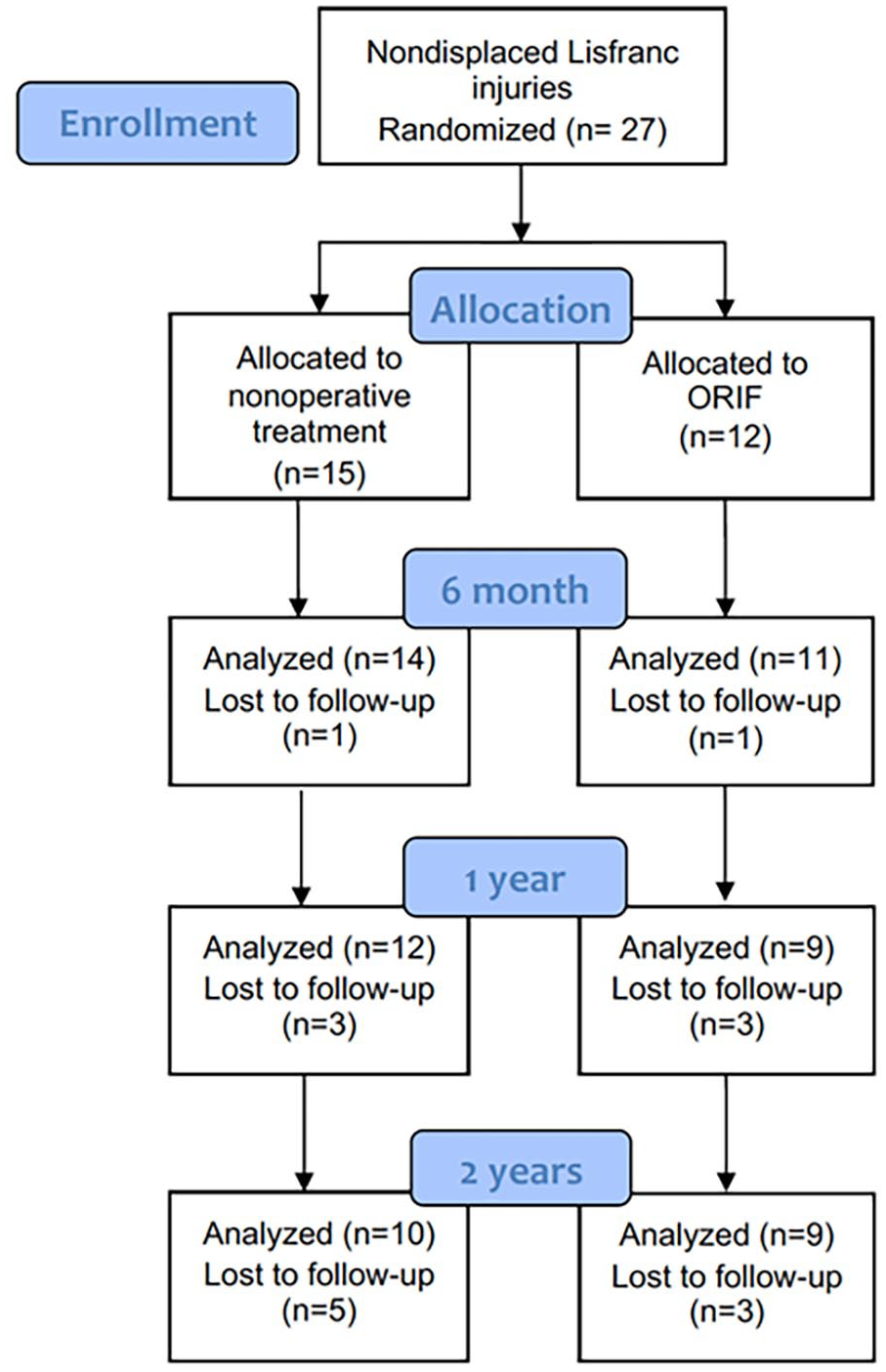
Flow chart.
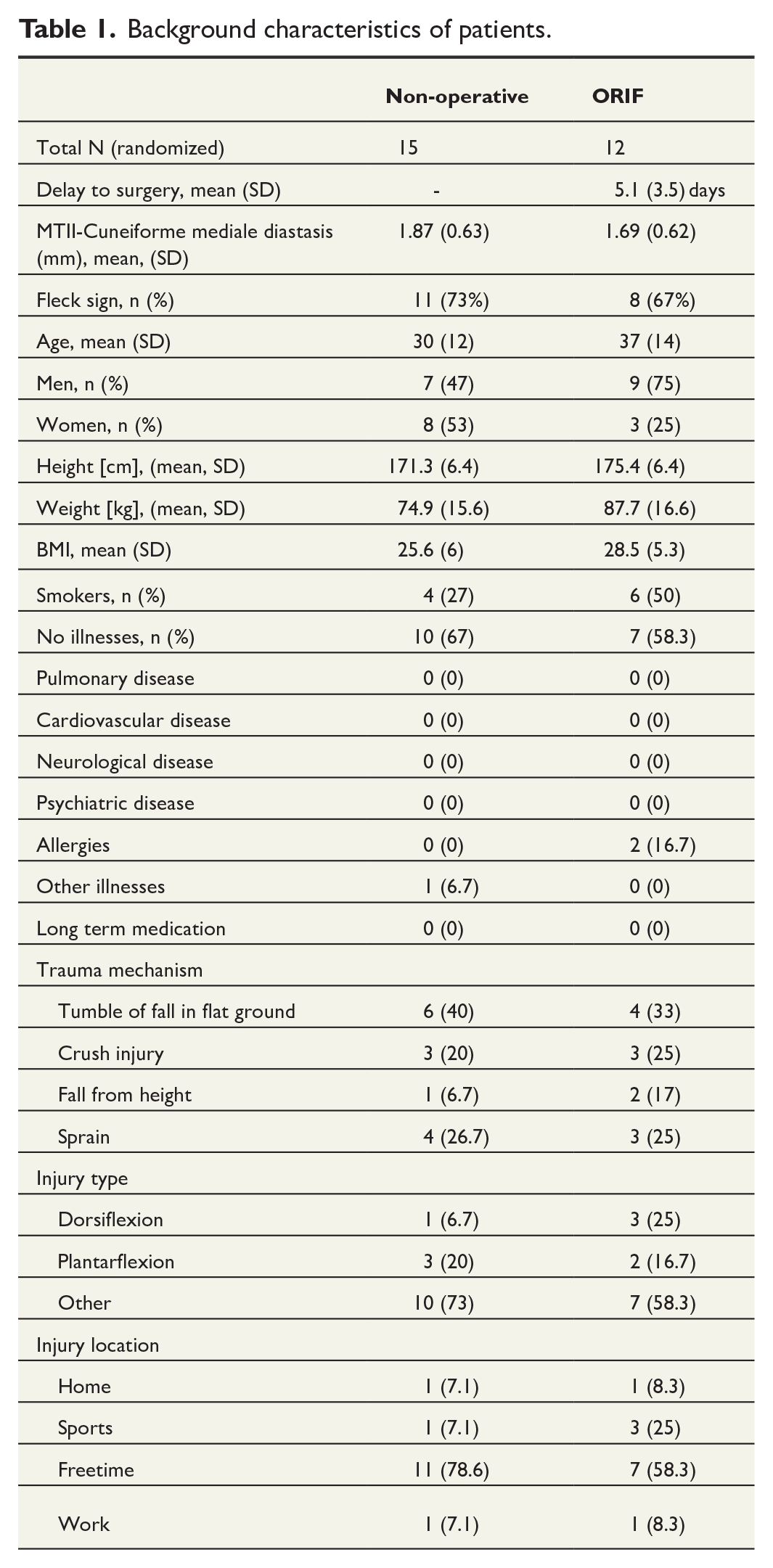
Background characteristics of patients.
Primary outcomes
The mean VAS-FA Overall score in the non-operative group was 96.1 [CI 91.5–100] and 91.8 [86.9–96.7] in the ORIF group at 24-month follow-up (Fig. 2). There was no difference in VAS-FA Overall scores (mean between-group difference (MD) 4.3 [CI, −2.4 to 11], Cohen’s d = 0.706, Table 2) at 24-month follow-up.
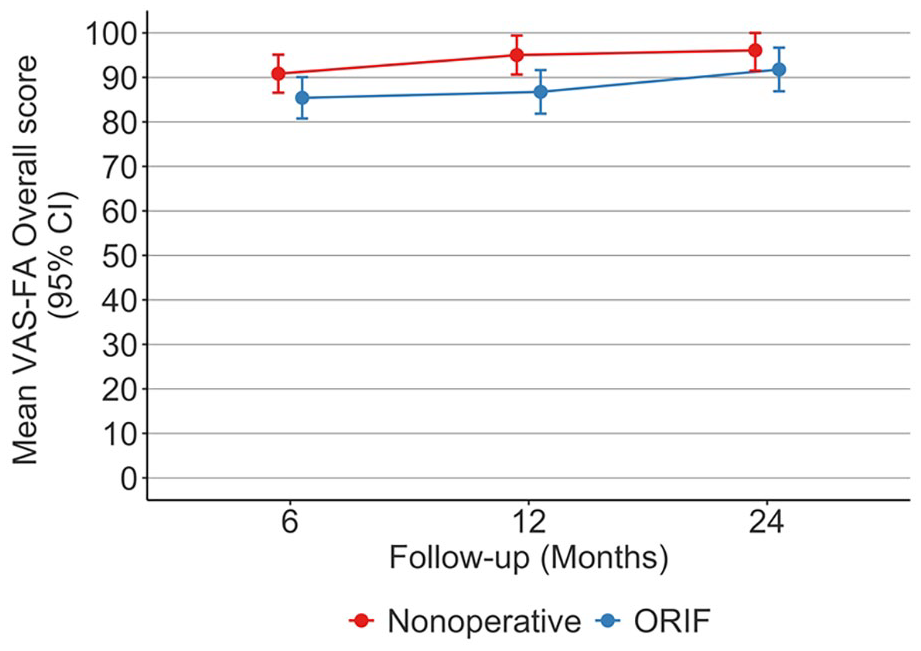
Mean (95% CI) VAS-FA Overall scores of non-displaced Lisfranc injuries at 6, 12, and 24 follow-up.
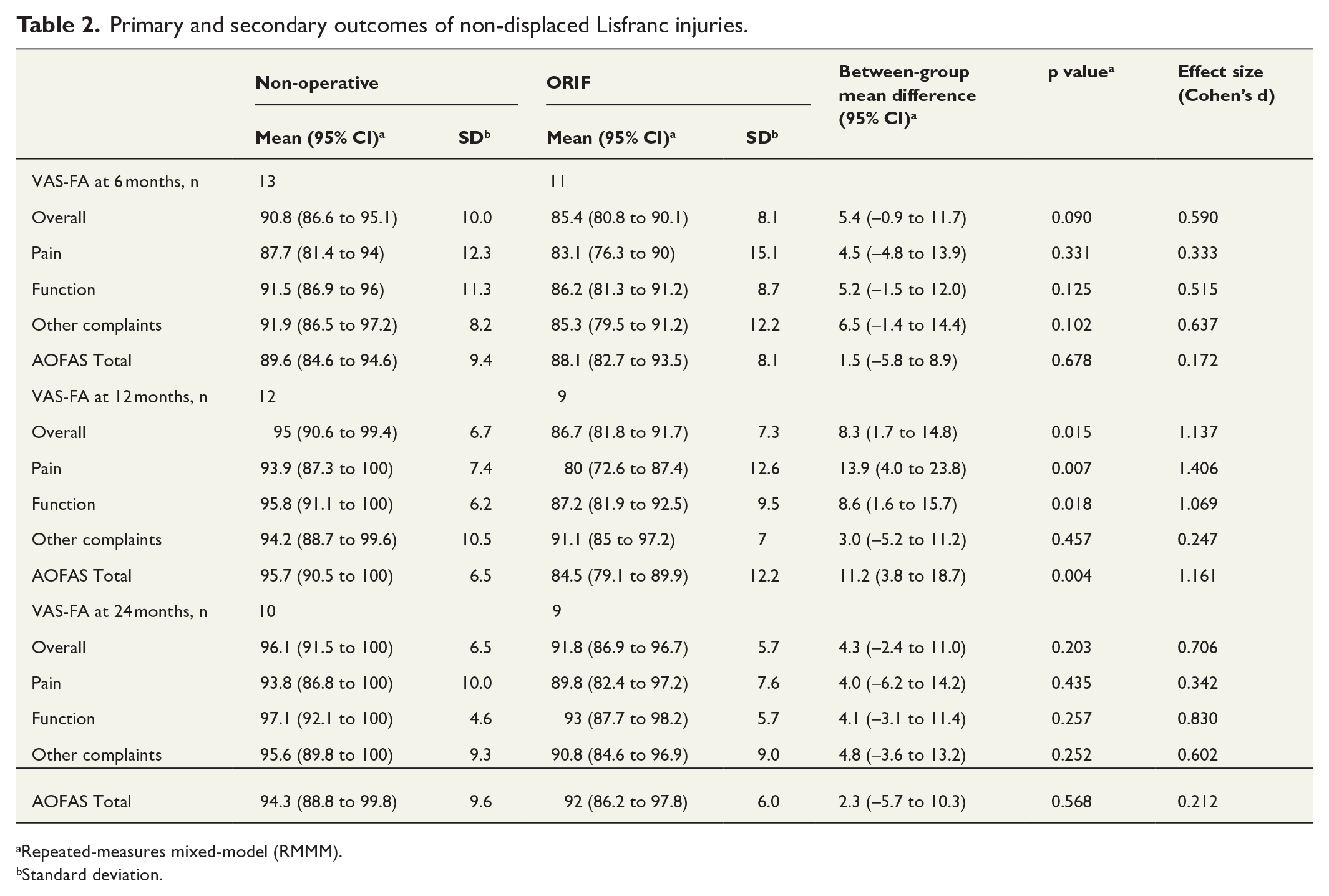
Primary and secondary outcomes of non-displaced Lisfranc injuries.
Repeated-measures mixed-model (RMMM).
Standard deviation.
Secondary outcomes
There were no differences at 6-month follow-up in any of our outcome measures; VAS-FA Overall (MD 5.4 (CI: –0.9 to 11.7)), Pain (MD 4.5 (CI –4.8 to 13.9)), Function (MD 5.2 (CI −1.5 to 12.0)) or Other Complaints (MD 6.5 (CI –1.4 to 14.4)) subscales, or in AOFAS Midfoot Scale (MD 1.5 (CI –5.8 to 8.9)) (Table 2).
The mean VAS-FA Overall score in the non-operative group was 95.0 (CI, 90.6–99.4) and 86.7 CI, 81.8–91.7) in the ORIF group at 1 year (Fig. 2). Non-operative treatment resulted in higher VAS-FA Overall scores (MD 8.3 (CI, 1.7–14.8), Table 2) at 1 year. Non-operative treatment resulted higher scores in VAS-FA Pain (MD 13.9 (CI 4.0–23.8)) and Function (MD 8.6 (CI 1.6–15.7)) subscales, as well as AOFAS Midfoot Scale (MD 11.2 (CI 3.8–18.7)) at 1 year. There was no difference in VAS-FA Other Complaints (MD 3.0 (CI −5.2 to 11.2)) subscale at 1 year.
There were no differences at 24-month follow-up in any of our outcome measures; VAS-FA Pain (MD 4.0 (CI −6.2 to 14.2)), Function (MD 4.1 (CI −3.1 to 11.4)) or Other Complaints (MD 4.8 (CI −3.6 to 13.)) subscales or in AOFAS Midfoot Scale (MD 2.3 (CI –5.7 to 10.3)) (Table 2).
Complications
One patient in non-operative group was diagnosed and treated due to deep vein thrombosis before the 6-week follow-up (Table 3). Two patients underwent screw removal due to pain, one after 8 months and second after 12 months after the primary operation. Both patients reported that the pain ended after the screw removal. None of the non-operatively treated patients underwent operative treatment. Examples of pre- and post-treatment X-rays of patients treated non-operatively and operatively are provided in Fig. 3A, B.
All minor and major adverse events.

Two screw removals in ORIF group were performed routinely at 12 months opposite to our planned protocol.

(A) X-rays of a study patient with a non-operatively treated Lisfranc injury. Left: Primary x-ray taken at the emergency department. Right: Follow-up x-ray taken after 2 years. (B) X-rays of a study patient with an operatively treated Lisfranc injury. Left: Pre-operative x-ray taken at the emergency department. Right: Post-operative x-ray taken after 2 years.
Discussion
Based on this RCT we did not find evidence of a difference in VAS-FA between non-operative and ORIF in patients with non-displaced Lisfranc injuries. Non-operatively treated patients had slightly better functional results at 1 year, yet the trial is underpowered to draw meaningful conclusions.
The literature does not provide any previous prospective randomized controlled studies comparing non-operative and operative treatment of Lisfranc injuries. Current knowledge is based on a few case-series11,32 and retrospective studies.9,13,33,34 It has been proposed that the main factor to support operative treatment is the displacement or a bony avulsion fracture between the medial cuneiform and the second MT bone in weightbearing radiographs or stress test. 17 However, these methods are flawed and CT should be the preferred primary diagnostic method. 16 Still, there are no consistent methods to evaluate the displacement in CT. Recent studies have explored the use of weight bearing CT to detect instability of Lisfranc injuries, 35 yet there is still a problem regarding the weightbearing with injured and painful foot. Our inclusion criteria for the non-displaced injuries were chosen to include subtle instability but to exclude clearly displaced and obviously unstable injuries. It has been shown that immediate weightbearing may lead up to 54% of displacement rate, 34 and thus our hypothesis was, that subtle instability will heal non-operatively, if the non-operative protocol restricts weight bearing for long enough to ligaments and capsule to heal and to restore the stability of the joint. This protocol was used in our previous retrospective study, which showed good functional outcomes after non-operatively treated Lisfranc injuries after 2–6.5 years. 17 The present prospective randomized study shows, that non-operative treatment may lead to slightly better functional outcomes than ORIF at 1 year. The difference in VAS-FA Overall (8.2 points, minimal important change (MIC) 6.8), Pain (13.9 points, MIC 9.3), and Function (8.6 points, MIC 5.7) subscales were higher than the reported MIC values. 27 This finding may partly be a result from the fact that patients who undergo foot surgery often sustain issues with the wound, such as neuropathical pain, numbness, and irritation. The problems may still be present at 1 year but after that the symptoms will heal. Thus, it seems that screw fixation of these relatively stable (“stable enough to be treated non-operatively”) injuries do not result in any better outcome. We acknowledge that the trial is underpowered to draw strong conclusions.
In the non-operative group, only one complication, a deep vein thrombosis was reported. None of the patients underwent surgery during our 2-year study period. On the contrary, two of the patients treated with ORIF underwent screw removal surgery. It has been reported that failed treatment may lead to substantial disability, deformity, and dysfunction requiring secondary arthrodesis. 9 However, in our 2-year study period, none of the non-displaced Lisfranc injuries required a secondary arthrodesis as all patients healed well. This finding was similar to a previous retrospective study with longer follow-up, where only 2% of the non-operatively treated patients underwent secondary arthrodesis, and the included injuries were more severe (more affected TMT joints with less than 2 mm of displacement) than in our current RCT. 17
Protocol deviations
The study protocol for this trial was previously published. 24 This study report variated from the previously published protocol in two ways. First, we decided to change the primary outcome from AOFAS Midfoot Scale due to VAS-FA due to multiple studies raising concerns on the validity and reliability of the AOFAS Midfoot scales’ measurement properties. 28 Second, we decided to change the primary analyses from t-test or Mann–Whitney U test to RMMM. This change was done, as RMMM takes the changes in each patient into account and is currently thought to be more suitable than traditional methods such as t-test or Mann–Whitney U test in trials with repeated measures. 36 We performed sensitivity analysis to test if the change of analysis method had an influence on the primary outcomes, which showed no changes in any outcome measures (Supplement 1A).
Limitations
The main limitation of this trial was that we were unable to reach the previously planned sample size, and therefore this trial remains to be underpowered. Main reasons for this are problems with the recruitment process in our clinics, and second, the attitude of patients toward surgical treatment, and they preferred non-surgical treatment instead or they were unwilling to commit to study follow-ups for 2 years. Originally, the study was also planned to be conducted at two other university hospitals and one central hospital. However, these centers were unable to begin the recruitment process, which slowed the recruitment timeline compared to what was initially planned. In our catchment area, the incidence of TMT joint injuries was 9.2 per 100,000 person-years, resulting in approximately 50 injuries per year. 6 Of these, two-thirds were non-displaced injuries, so a significant number of patients were not included. In addition, our strict inclusion criteria excluded many injury patterns that could have been comparable enough to be included. As the recruitment was carried for more than 10 years, we decided to end the trial as it was clear that we are unable to reach the proposed power. Furthermore, the limited sample size resulted in an imbalanced distribution of participants across the study groups. Particularly noteworthy is the observation that individuals undergoing non-operative treatment had a mean age of 7 years less than those undergoing ORIF. This discrepancy in age distribution introduces a possible source of bias, which could potentially favor non-operative treatment, as it is associated with enhanced tissue healing in younger patients.
Conclusion
Based on this multicenter RCT, we did not find difference in VAS-FA between non-operative and ORIF in patients with non-displaced Lisfranc injuries at 2 years, but the trial is underpowered to draw robust conclusions.
Supplemental Material
sj-docx-1-sjs-10.1177_14574969241295585 – Supplemental material for Operative versus non-operative treatment for non-displaced Lisfranc injuries: A two-center randomized clinical trial
Supplemental material, sj-docx-1-sjs-10.1177_14574969241295585 for Operative versus non-operative treatment for non-displaced Lisfranc injuries: A two-center randomized clinical trial by Ville Ponkilainen, Heikki Mäenpää, Heikki-Jussi Laine, Nikke Partio, Olli Väistö, Janne Jousmäki, Ville M. Mattila and Heidi Haapasalo in Scandinavian Journal of Surgery
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics
Ethical approval for this study was obtained from Pirkanmaa Healthcare District, Finland.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Competitive State Research Financing of the Expert Responsibility area of Tampere University Hospital.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
