Abstract
Background:
Antibiotic therapy is currently considered a safe and effective treatment alternative for computed tomography (CT)-confirmed uncomplicated acute appendicitis with recent studies reporting good results on both oral antibiotics only and outpatient management. Furthermore, there are promising pilot results on uncomplicated acute appendicitis management with symptomatic treatment (placebo). This trial aims to assess whether both antibiotics and hospitalization can be safely omitted from the treatment of uncomplicated acute appendicitis.
Methods:
The APPAC IV (APPendicitis Acuta IV) trial is a randomized, double-blind, multicenter noninferiority clinical trial comparing oral moxifloxacin with oral placebo in an outpatient setting with a discharge directly from the emergency room (ER). Adult patients (18–60 years) with CT-confirmed uncomplicated acute appendicitis (absence of appendicolith, abscess, perforation, tumor, appendiceal diameter ⩾15 mm on CT, or body temperature >38 °C) will be enrolled in nine Finnish hospitals. Primary outcome is treatment success at 30 days, that is, the resolution of acute appendicitis resulting in discharge from the hospital without appendectomy during the 30-day follow-up evaluated using a noninferiority design with a noninferiority margin of 6 percentage points. Noninferiority will be evaluated using one-sided 95% confidence interval of proportion difference between groups. Secondary endpoints include postintervention complications, recurrent appendicitis after the 30-day follow-up, duration of hospital stay, admission to hospital and reason for admission, readmissions to emergency department or hospitalization, VAS pain scores, quality of life, sick leave, and treatment costs. The follow-up after discharge from the ER includes a phone call at day 1, and at 3–4 days, 30 days, and 1, 3, 5, 10, and 20 years. Those eligible patients, who decline to undergo randomization, will be invited to participate in a concurrent observational cohort study with follow-up at 30 days, and 1 and 5 years.
Discussion:
To our knowledge, APPAC IV trial is the first large randomized, double-blind, noninferiority multicenter clinical trial aiming to compare oral antibiotics and placebo for CT-diagnosed uncomplicated acute appendicitis in an outpatient setting. The study aims to bridge the major knowledge gap on whether antibiotics and hospitalization or both can be omitted in the treatment of uncomplicated acute appendicitis.
Trial registration:
The study protocol has been approved by the Clinical Trials Information System (CTIS) of the European Medicines Agency (EMA), study number: 2023-506213-21-00 and the trial has been registered in ClinicalTrials.gov, NCT06210269.
Keywords
Summary
A study protocol of a double-blind, multicenter, APPAC IV RCT comparing oral moxifloxacin with placebo in an outpatient setting—can antibiotics and hospitalization both be omitted in the treatment of uncomplicated appendicitis?
Context and relevance of the randomized clinical trial
Antibiotic management of CT-diagnosed uncomplicated acute appendicitis is considered a safe and effective alternative to surgery. However, there is a major knowledge gap on the actual role of antibiotics in the treatment of uncomplicated acute appendicitis. Recent promising results on both symptomatic treatments alone in a pilot double-blind randomized clinical trial (RCT) and frequent outpatient management in a larger RCT question both the need for antibiotics and hospitalization. This APPendicitis Acuta IV (APPAC IV) study protocol, to our knowledge, is the first large randomized, double-blind, noninferiority multicenter clinical trial aiming to compare oral antibiotics and placebo for CT-diagnosed uncomplicated acute appendicitis in an outpatient setting. The study aims to bridge the major knowledge gap on whether antibiotics and hospitalization or both can be omitted in the treatment of uncomplicated acute appendicitis.
Background
For over a century, surgical removal of the appendix has been considered the only treatment option for acute appendicitis with appendectomy being one of the most common emergency surgeries. This century-old belief of emergency appendectomy for all appendicitis patients has recently been overturned as we now know that complicated and uncomplicated appendicitis are two different diseases epidemiologically, clinically, and also microbiologically 1 all suggesting different pathophysiology.2,3 Although laparoscopic appendectomy is considered a low-risk operation, the modern knowledge of postoperative complication rate, 4 equal patient satisfaction and quality of life (QOL), 5 costs of surgery, and hospitalization6 –8 all advocate for further assessment and optimization of nonoperative treatment for uncomplicated acute appendicitis.
For the first time during the COVID-19 pandemic, antibiotic treatment for uncomplicated acute appendicitis was recognized in the guidelines as a feasible treatment alternative by the American College of Surgeons. 9 Based on multiple RCTs, the majority of patients with uncomplicated acute appendicitis can be treated with antibiotics alone avoiding unnecessary surgeries.4,10 –15 As most appendicitis cases (approximately 70%–80%) are uncomplicated, this results in major cost savings both at short-term 6 and long-term follow-up, 8 despite using intravenous antibiotics requiring hospitalization and several days of hospital stay due to the novelty of antibiotic approach at the time of these studies. 16 The recent APPAC II trial suggested that oral antibiotics alone could be a feasible treatment alternative for CT-confirmed uncomplicated acute appendicitis,17,18 further encouraging evaluating outpatient management, 19 which was already shown feasible by the large CODA RCT. 4 Outpatient treatment would markedly further decrease the costs of nonoperative treatment 8 and possibly increase patient satisfaction. 4
To further improve patient selection and subsequently the success rate of nonoperative treatment for uncomplicated acute appendicitis, accurate differential diagnosis is essential, focusing on ruling out complicated acute appendicitis. Computed tomography (CT) is the most accurate diagnostic method in diagnosing both acute appendicitis and distinguishing between appendicitis severity, and it is the gold standard imaging for appendicitis.20 –24 A recent assessment of potential preintervention findings associated with nonresponsiveness to antibiotics in the treatment of CT-confirmed uncomplicated acute appendicitis in APPAC II trial, appendiceal diameter ⩾15 mm on CT, and a body temperature ⩾38 °C, have been added as exclusion criteria for the current APPAC IV trial to further optimize the patient selection for nonoperative treatment of uncomplicated acute appendicitis. 25 Also, the presence of an appendicolith has been shown to be associated with a more complicated course of the disease,4,15,26 and it has already been an exclusion criteria in all of the previous APPAC trials.10,17,27
Acute appendicitis and acute diverticulitis seem to be clinically very similar diseases for both the uncomplicated and complicated forms of these two diseases suggesting a common underlying pathogenesis. 2 As there are several trials suggesting that uncomplicated diverticulitis can resolve spontaneously even without antibiotics,28 –31 supportive care alone may be a viable treatment option for uncomplicated appendicitis as well.27,32 The pilot double-blind RCT APPAC III 27 and a single-blinded RCT by Park et al. 32 both compared placebo and antibiotic treatment in patients with uncomplicated acute appendicitis, showing that the lack of antibiotic superiority compared to placebo warrants a large randomized double-blind noninferiority trial to assess the role of antibiotics in the treatment of uncomplicated acute appendicitis. Antibiotic resistance threatens effective prevention and treatment of infections warranting studies on rational use of antibiotics. Abandoning possibly unnecessary broad-spectrum antibiotics would prevent adding to the growing problem of antimicrobial resistance. 33
This APPAC IV randomized double-blind multicenter clinical noninferiority trial comparing oral moxifloxacin with placebo in an outpatient setting aims to bridge the knowledge gap of the role of antibiotics in the treatment of uncomplicated acute appendicitis, assessing whether antibiotics and hospitalization or both can be omitted in uncomplicated acute appendicitis.
Methods
Study design
The APPAC IV trial is a multicenter, double-blind, placebo-controlled RCT comparing oral moxifloxacin therapy with oral placebo in an outpatient setting in the treatment of CT-confirmed uncomplicated acute appendicitis. Trial enrolls patients from nine major Finnish hospitals including four University hospitals (Turku, Tampere, Oulu, and Kuopio) and five central hospitals (Pori, Seinäjoki, Lahti, Jyväskylä, and Mikkeli). The protocol adheres to the SPIRIT (Standard Protocol Items: Recommendations for Interventional Trials) statement. 34 A flowchart of the trial is shown in Fig. 1.
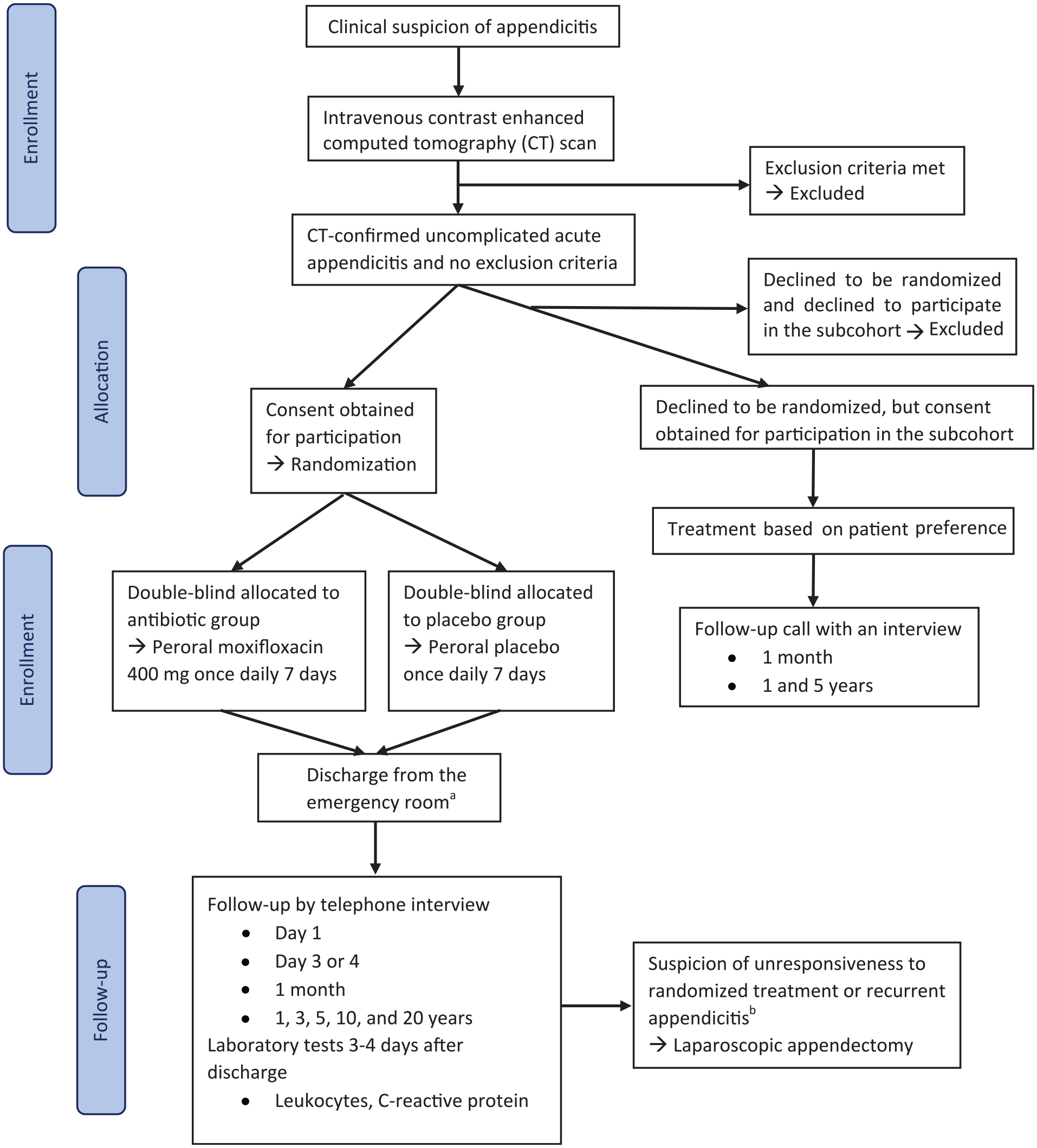
APPAC IV flow diagram.
Patient selection and diagnostic procedures
All adult patients (aged 18–60 years) in the emergency department (ED) with a clinical suspicion of acute appendicitis are evaluated for trial recruitment, and the baseline data of all patients with a CT-diagnosed appendicitis will be collected at the ED by a doctor on call. The patients with an intravenous contrast-enhanced CT-confirmed uncomplicated acute appendicitis are evaluated for eligibility for the trial. Patients will undergo CT imaging using the lowest possible radiation dose evaluated as diagnostically adequate by the study-center radiologist on call. A radiological diagnosis of uncomplicated acute appendicitis is defined as having an appendiceal diameter >6 mm with a thickened, contrast-enhanced wall along with periappendiceal edema and/or minor fluid collection, and no features of complicated appendicitis (presence of appendicolith, perforation, abscess, suspicion of tumor, or appendiceal diameter ⩾15 mm). The CT findings will be evaluated using a standardized CT scan report sheet (Table 1). Patient temperature is measured; they are physically examined and assessed by an attending surgeon or a specialist on call. Laboratory tests (hemoglobin, white cell count, creatinine, C-reactive protein (CRP), potassium, and in female patients also the human chorionic gonadotropin), electrocardiogram, and prior to pain medication Visual Analog Scale (VAS) pain scores will be recorded. The study inclusion and exclusion criteria are presented in detail in Table 2.
The standardized CT scan report for assessment of CT findings.

The detailed inclusion and exclusion criteria of the study.

Randomization and blinding
After written informed consent, patients will be randomized by the attending surgeon on call in each study hospital with a 1:1 equal allocation ratio to receive either moxifloxacin or placebo in a double-blind study design. A safety statistician will perform the randomization procedure using random permuted blocks. These will be blinded to the investigators and the study statistician. The complete randomization listing will be made available only to the safety statistician. To ensure patient safety in emergency situations, all study hospitals will have the double-blinded randomization codes for the locally randomized patients available in a sealed envelope in the hospital. This envelope contains the specific information about the randomized medicine received by each participant, allowing this information to be easily available in case of an emergency, for example, anaphylactic reaction. Enrolled patients are randomized using the Research Electronic Data Capture (REDCap) online database by the attending surgeon in each study hospital. The randomized treatment will be initiated in the emergency room (ER). The randomized patients will be discharged from the ER with the oral double-blind medications with no hospital stay unless they are evaluated to require follow-up at the hospital or if they have social or geographical reasons advocating overnight hospital stay. Pain medication will be prescribed according to standard hospital protocol recommended to be consisting of paracetamol and nonsteroidal anti-inflammatory drugs (NSAIDs) according to clinical practice guidelines. The oral moxifloxacin and placebo drugs are identical by appearance, taste, and smell. Both drugs are in colored oral gelatin capsules. Patient receives a plastic medicine bottle with identical labeling containing the randomized drug and is instructed to take it once daily for 7 days.
Prospective subcohort of eligible patients unwilling to participate in the randomized study
Eligible patients with CT-confirmed uncomplicated acute appendicitis declining to participate in the randomized trial will be given a separate informed consent to participate in a prospective subcohort, who will be treated according to their own preference and joint decision-making with the surgeon on call. Based on previous trials,10,17,27 this prospective subcohort is estimated to have several hundred patients. Similarly to the randomized patients, clinical history, physical examination, VAS pain score, and laboratory tests will be registered in the database including a follow-up at 30 days and 1 year. In addition, this prospective subcohort will be used to analyze the treatment costs as most likely the majority of the patients in this subcohort will undergo appendectomy, allowing the comparison of costs for nonoperative treatment and appendectomy.
Sample size calculation
Calculations are based on noninferiority test for two binomial proportions. Sample size was calculated from an estimated 30-day success rate of 93% for antibiotics.17,32 The hypothetical difference between groups was set to zero, 32 and noninferiority margin was set to 6 percentage points based on clinical review and previous studies. 17 Since the adverse effects of antibiotic treatment are usually very mild for the patient, there are no large disadvantages of using antibiotics to treat uncomplicated appendicitis, and therefore, the clinically important difference between the treatments is not very large and the noninferiority margin of 6 percentage points is quite narrow. The same noninferiority margin was used also in the APPAC II trial 17 to compare oral versus intravenous, followed by oral antibiotics. We estimated that a total of 448 patients would yield a power of 0.8 (1 − β) to establish whether placebo therapy is noninferior to antibiotics using a one-sided significance level (α) of 0.05. With an estimated dropout rate of 10%, a total of 498 patients, 249 patients per group, will be enrolled in the study. Targeted minimum sample size per study hospital will be 40 patients. Sample size calculations were performed using Power procedure in SAS System for Windows, Version 9.4 (SAS Institute Inc., Cary, NC, USA).
Interventions
The antibiotic regimen and duration of oral moxifloxacin 400 mg for 7 days are selected based on previous proven efficacy in the APPAC II trial 17 and are considered as the “standard” treatment in the noninferiority study design. Oral placebo will be administered according to an identical once daily for 7 days protocol with the antibiotic therapy group.
Follow-up
The clinical follow-up for outpatient treated patients will include a phone call at day 1 after discharge from the ER by a study nurse. The phone call at day 3 or 4 includes laboratory tests (white cell count, CRP). Telephone follow-up will include an interview with inquiry of pain (VAS score) and assessment of overall recovery (fever, general condition, need of pain medication), a questionnaire of any drug adverse effects or possible appendectomy. Patients will be instructed to return to ER if they have high fever, general condition weakens, or they have a need of repeated analgesics. A follow-up call will be also performed at 30 days after the discharge and at 1, 3, 5, 10, and 20 years.
If a patient is suspected of unresponsiveness to the administered treatment based on the evaluation of the surgeon on call or suspected to have a recurrent acute appendicitis based on clinical findings, the patient will undergo laparoscopic appendectomy according to the study protocol without repeated imaging. If symptoms are atypical or vague based on the clinical evaluation, it is possible to include imaging in the diagnostic procedure based on the decision by the surgeon on call. Surgical findings and histopathology of the appendix will be recorded in the database.
Outcome parameters and statistical analysis
Primary outcome
The primary outcome of the APPAC IV trial is the 30-day treatment success defined similarly as in the previous APPAC trials, that is, resolution of acute appendicitis resulting in discharge from the hospital without the need for appendectomy during the 30-day follow-up. All study outcomes are presented in Table 3.
Detailed study primary, secondary, and subcohort outcomes.
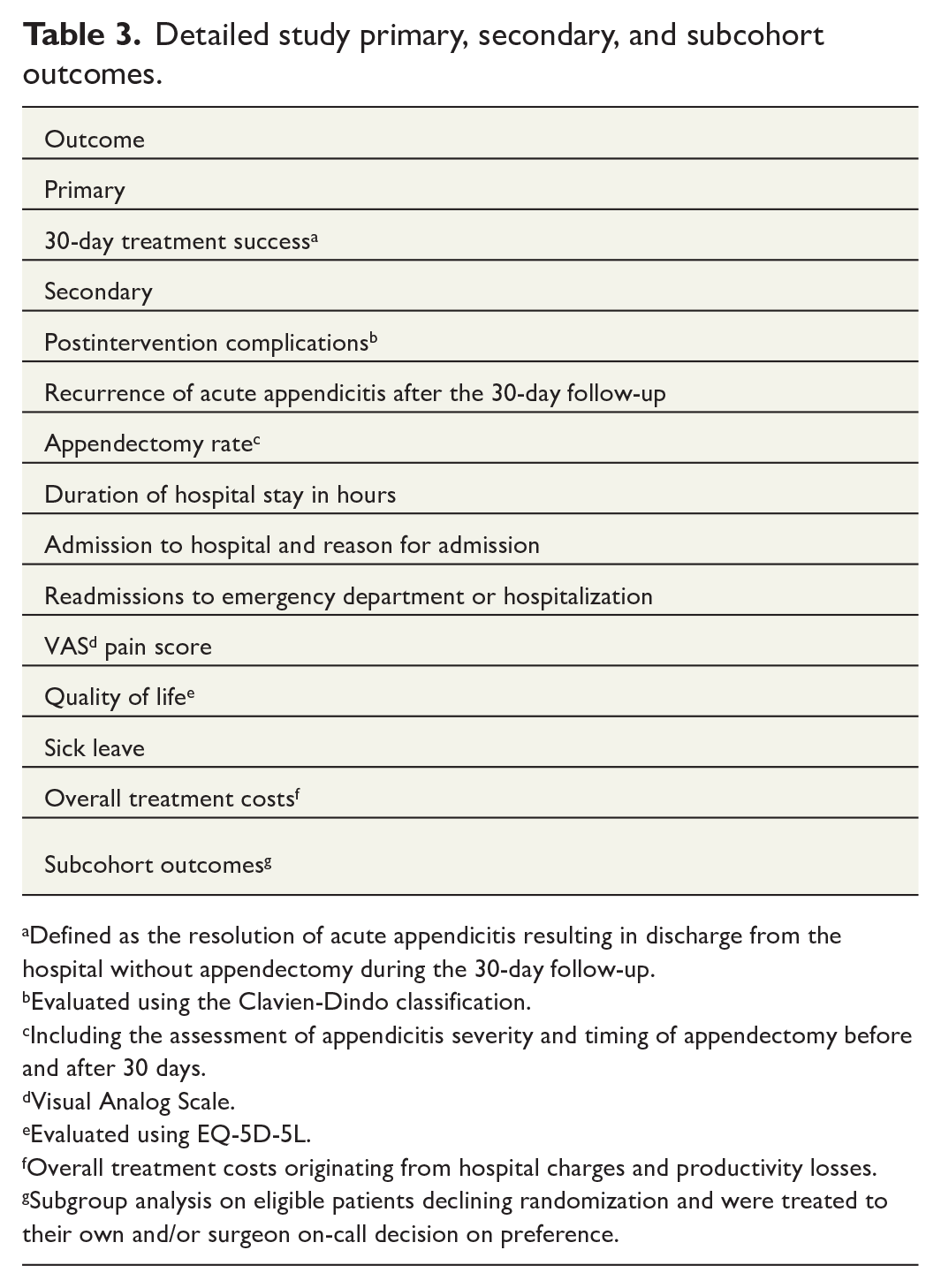
Defined as the resolution of acute appendicitis resulting in discharge from the hospital without appendectomy during the 30-day follow-up.
Evaluated using the Clavien-Dindo classification.
Including the assessment of appendicitis severity and timing of appendectomy before and after 30 days.
Visual Analog Scale.
Evaluated using EQ-5D-5L.
Overall treatment costs originating from hospital charges and productivity losses.
Subgroup analysis on eligible patients declining randomization and were treated to their own and/or surgeon on-call decision on preference.
Secondary outcomes
Secondary endpoints include postintervention complications (Clavien-Dindo classification), 35 recurrence of acute appendicitis and appendectomy rate after the 30-day follow-up including the assessment of appendicitis severity, duration of hospital stay in hours, admission to hospital and reason for admission, readmissions to ED or hospitalization, VAS pain scores, QOL (EQ-5D-5L), sick leave, and overall costs originating from hospital charges and productivity losses in the treatment of uncomplicated acute appendicitis. Baseline QOL will be evaluated at the first follow-up phone call at day 1. All adverse events or symptoms related to randomized treatment comparable with grade 1 or higher in the Clavien-Dindo classification 35 for postoperative complications and any symptoms resulting in discontinuation of the randomized treatment will be classified as treatment-associated adverse events. Potential prognostic factors predicting initial unresponsiveness to the randomized treatment will be evaluated.
Subcohort outcomes
A subcohort analysis will be performed comparing the treatment outcomes, treatment costs, complications, length of hospitalization, QOL, and length of sick leave of the randomized patients (as a combined group as well as the two treatment arms separately) with the corresponding outcomes of the eligible patients, who declined to be randomized and were subsequently treated according to their own preference and/or joint decision based on the alternatives suggested by the surgeon on-call. Within this subcohort, patients who were treated with initial appendectomy and patients treated with initial antibiotics will also be separately evaluated.
Data collection
Study data will be collected and managed using REDCap tools hosted at University of Turku.36,37 REDCap is a secure, web-based software platform designed to support data capture for research studies, providing an interface for validated data capture, audit trails for tracking data manipulation and export procedures, and interoperability with external sources. Data collection includes evaluated duration of symptoms prior to entering the ED, pain evaluated with VAS score, tympanic temperature (°C), weight and height with body mass index (BMI), CT findings, laboratory results, profession, length of sick leave, and the length of hospital stay. If the patient needs monitoring at hospital ward, the reason for hospitalization is recorded. For the hospitalization and possible appendectomy, the length of hospital stay, VAS score, profession, length of sick leave, and operative details are recorded at discharge. True appendicitis recurrence rate (appendicitis at histopathology) following initial successful nonoperative treatment as well as the final clinical diagnosis for all operated patients will be evaluated based on the intraoperative and histopathological findings.
To ensure both thorough data collection and the ability to evaluate selection bias, all the patients presenting with suspected acute appendicitis at the research hospitals will be recorded in the database for the baseline information. For the excluded patients or patients declining to participate, no additional data besides the baseline data of the primary appendicitis episode and treatment will be recorded. For patients withdrawing their consent at any time during the trial, the data will be collected only up to that time point. For patients declining randomization, but willing to participate in the observational prospective cohort, the follow-up data will be collected at 30 days and 1 year.
Safety monitoring and interim analysis
To ensure patient safety in the placebo treatment group, an interim analysis will be conducted after enrolment of 100 patients. For the interim analyses, a safety statistician with independent trial safety monitoring committee will calculate the point estimate of the success rate proportion at 30 days in each group. If the proportion in at least one of the groups is below 60%, the study will be terminated. Only the safety statistician and the independent safety monitoring committee members will know the exact proportions. The investigators and the study statistician will only be informed of the continuation or the termination of the study. No statistical tests will be conducted in interim analysis, and therefore, no corrections to the p-values are needed in the final analyses of study. In addition to the interim analysis, the safety monitoring committee may be asked to meet ad hoc, if evaluated necessary by the researchers.
Statistical hypothesis
The primary objective of the APPAC IV study is to demonstrate noninferiority between per oral placebo and antibiotics in treating contrast-enhanced CT-confirmed uncomplicated acute appendicitis. The primary outcome is treatment success at 30 days evaluated using the following statistical hypotheses
H0: p1 − p2 > 6
H1: p1 − p2 ⩽ 6
where p1 is the success of treatment proportion of antibiotic group and p2 for placebo group, and p1 − p2 is the difference between groups (antibiotics–placebo).
Statistical analyses
In the statistical analyses of primary outcome, noninferiority of placebo versus antibiotics in treatment success at 30 days, will be evaluated using a one-sided 95% confidence interval (CI) of proportion difference between antibiotic and placebo groups, and one-sided Wald test for noninferiority with an α level of 0.05. Noninferiority margin for difference is 6 percentage points. Treatment success at 1, 5, 10, and 20 years will be analyzed similarly as the primary outcome.
Categorical variables of the study will be characterized by treatment using frequencies and percentages. For continuous variables, means and standard deviations or medians with range and 25th and 75th percentiles will be used. The secondary outcomes will be analyzed using chi-squared test or Fisher’s exact test in case of categorical variables, and for continuous variables, independent samples t-test or Mann–Whitney U-test will be used. The recurrence of acute appendicitis after the 30-day follow-up will also be analyzed using survival analysis methods. The assumptions of all tests used in the statistical analyses will be checked for justification of the analyses. For the secondary outcomes, two-sided p-values will be used. The study site differences will be evaluated in statistical models, and if major differences are detected, more complicated statistical models will be used in the analyses of primary and secondary outcomes. If needed, mixed models will be used to be able to incorporate study site as a random factor in the model. The p-values less than 0.05 will be considered statistically significant. The analyses will be based on the intention-to-treat (ITT) principle (all randomized patients excluding possible erroneously randomized patients with CT diagnosis of complicated appendicitis). For the primary outcome, in cases of patients lost to follow-up, missing data will be collected from hospital registries, if possible, and therefore, no imputation of missing values will be needed. For secondary outcomes, the subjects with missing data will automatically be excluded from the analyses of the variables in concern. Statistical analyses will be performed using SAS System for Windows, Version 9.4 or later (SAS Institute Inc., Cary, NC, USA).
Ethics and dissemination
The data of APPAC IV contain sensitive personal data of the patients, and it will be managed according to the EU General Data Protection Regulation (GDPR) and judicial requirement according to the Finnish laws. All patients in the data have given their permission to the use of their patient information in the research. Owner of the data is The Wellbeing Services County of Southwest Finland. APPAC IV team have permission to use the data in the research, and it is the owner of the research results.
The APPAC IV trial is designed according to the standard PICOT criteria: Patient/Population, Intervention, Comparison, Outcome, and Time element. To obtain the highest possible internal validity, APPAC IV trial execution adheres to the EQUATOR Network guidelines (www.equatornetwork.org): SPIRIT for trial protocols, CONSORT for trial reporting, and CHEERS for economic evaluations.
Trial is approved by Clinical Trials Information System (CTIS) of the European Medicines Agency (EMA). The trial has been registered in a clinical trial registry (ClinicalTrials.gov) before initiation of patient enrolment (NCT06210269). The trial will be performed in accordance with the Declaration of Helsinki. The clinical trial will be conducted in compliance with the protocol, with the Regulation (EU) 536/2014 and with the principles of good clinical practice. Patients are informed of their right to withdraw from the study without explanation at any time.
The results will be published in international peer-reviewed scientific journals and by presentations at international and/or domestic conferences.
Discussion
APPAC IV trial aims to evaluate whether antibiotics or hospitalization or both can be omitted in the treatment of uncomplicated acute appendicitis. This study is a direct continuum to previous APPAC studies,10,17,27 showing that contrast-enhanced CT-confirmed uncomplicated acute appendicitis is not, in fact, a surgical emergency. The knowledge gap of the role of antibiotics needs to be bridged before true implementation of the nonoperative treatment in the treatment paradigm of uncomplicated acute appendicitis. Another major issue warranting further assessment is the feasibility of nonoperative outpatient management. Only after establishing both the role of antibiotics and the need of hospitalization, the nonoperative treatment can be optimized similarly to optimized surgical treatment of laparoscopic appendectomy.
The current treatment paradigm of appendectomy for all appendicitis patients requires a substantial amount of healthcare resources worldwide with acute appendicitis being one of the most common surgical emergencies. In previous trials, the treatment costs of antibiotic therapy have been lower compared to those patients treated with initial surgery despite including the up to 3-day hospitalization.10,14,19,38 To our knowledge, this is the first large trial with systematic outpatient treatment for uncomplicated acute appendicitis, and therefore, the patients are closely followed by multiple calls and laboratory tests to ensure patient safety. The large pragmatic CODA trial previously reported that there were no differences in patient with outpatient antibiotic treatment in complication rates compared to patients initially admitted to hospital. Instead, the key to decrease complication rates appears to be optimizing patient selection by ruling out complicated acute appendicitis by, for example, excluding patients with an appendicolith. 4 If outpatient management proves to be a feasible treatment choice either with or without antibiotics, it would inevitably lead to markedly increased further cost savings when both hospitalization and surgery could be avoided. To enable the comparison of the costs between nonoperative treatment and appendectomy, a prospective observational subcohort of eligible patients declining randomization will be included. These patients will choose their preferred treatment in a joint decision-making with the surgeon on call, and the hypothesis is that most of these patients will undergo appendectomy, allowing the comparison of laparoscopic appendectomy with the randomized treatment arms. This subcohort enrolment will also aid in assessing potential selection bias due to the possibility of comparing randomized patients with the patients declining randomization.
In order to have an antibiotic regimen with a well-known efficacy and risk profile in the antibiotic arm, moxifloxacin was selected having a proven efficacy based on the previous APPAC II trial both at short-term 17 and longer-term follow-up. 18 A wide usage of a broad-spectrum antibiotic, such as moxifloxacin, is not optimal regarding the global threat of antibiotic resistance. If the study hypothesis is true and placebo is noninferior to antibiotic therapy, patients worldwide could be offered a treatment alternative for uncomplicated acute appendicitis without unnecessary antibiotics. On the contrary, if the study results show that antibiotics are needed for the treatment of uncomplicated acute appendicitis, the next future trial should address to use a less-broad spectrum antibiotic to optimize the antibiotic therapy. Bridging this knowledge gap of the role of antibiotics can only be achieved by a large double-blind noninferiority trial such as the APPAC IV.
It should be emphasized that the treatment of complicated acute appendicitis requires emergency appendectomy39,40 excluding the patients with periappendicular abscess most likely best treated with interval appendectomy.41,42 The treatment success of antibiotic or symptomatic treatment is the highest among a selected group of patients with an uncomplicated disease. There are still no unified standardized criteria for defining uncomplicated and complicated appendicitis.40,43 To further optimize patient selection, a prespecified secondary analysis of the APPAC II RCT assessed prognostic factors for nonresponsiveness to antibiotics, and these were incorporated in the APPAC IV inclusion and exclusion criteria, further adding to patient safety and higher treatment success. These rather strict criteria inevitably exclude a large group of appendicitis patients from the trial. However, especially considering the outpatient approach of this study and optimization of nonoperative treatment, it is necessary to rule out complicated appendicitis with high sensitivity. It is reasonable to target this with the expense of specificity; a false-positive diagnosis of complicated appendicitis will only lead to laparoscopic appendectomy for an uncomplicated appendicitis, which is an accepted treatment alternative. As CT is currently the most accurate diagnostic imaging method to distinguish uncomplicated and complicated acute appendicitis, it will be used as the mandatory diagnostic tool in this study. 44 Diagnostic CT imaging is instructed to be performed with the lowest radiation dose possible without reducing the diagnostic accuracy. Although CT is currently considered the most accurate diagnostic tool for appendicitis, scoring systems combining imaging findings to clinical parameters have been showing promising results and might provide added diagnostic accuracy in the future.45,46
There are some limitations in this study that have already been acknowledged. The main weakness is the limited assessment of the possible role of NSAID in the resolution as this administration cannot be standardized. However, as the pain medications are used according to standard clinical practice, the use of NSAIDs will most likely be somewhat similar between the randomized groups. Another major challenge is the acute care surgery setting of the trial as the patient enrolment in the busy ED environment always poses a challenge to conducting RCTs potentially having an effect on the recruitment schedule.
Footnotes
Acknowledgements
In this multicenter trial, there is a multitude of unmentioned essential contributors of this trial. In addition, we acknowledge all supporting residents, surgeons, radiologists, emergency medicine physicians, and nurses. The surgical departments of the following Finnish Hospitals contribute to the execution of this trial: University Hospitals of Turku, Oulu, Tampere, and Kuopio and Central Hospitals of Pori, Jyväskylä, Mikkeli, Lahti and Seinäjoki.
Author contributions
All authors have contributed to the design of this trial protocol. PS is the chief investigator. PS has initiated the project. The protocol was drafted by PS, which was refined by JH and SH. Statistical advice was provided by SH. HL was responsible for drafting the protocol manuscript, which was refined by JH, PS, and SH. All authors have read and approved the final manuscript. The APPAC collaborative study group consists of all local investigators who are responsible for execution of the trial and valid data gathering.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the European Union (ERC, APPAC IV, 101125245). Views and opinions expressed are, however, those of the author(s) only and do not necessarily reflect those of the European Union or the European Research Council Executive Agency. Neither the European Union nor the granting authority can be held responsible for them. The work was also funded by the Sigrid Jusélius Research Foundation and Research Council of Finland (Academy of Finland) for P.S. and a government research grant awarded to Turku University Hospital (EVO Foundation) for P.S. The research nurses and statisticians will be covered by the funding of the APPAC study group. H.L. reports receiving personal grants from TYKS Foundation, Turku University Foundation, and The Wellbeing Services County of Satakunta. J.H. reports receiving personal grants from the Orion research foundation and the Mary and Georg C. Ehrnrooth Foundation.
