Abstract
Background:
Carotid endarterectomy (CEA) carries a risk of perioperative stroke, particularly during carotid cross-clamping. While neurological monitoring is reliable for patients under regional anesthesia, alternative options are needed for those unsuitable for regional anesthesia. Near-infrared spectroscopy (NIRS) is commonly used during CEA, but its diagnostic accuracy varies, particularly under general anesthesia.
Methods:
A systematic review with meta-analysis was performed to assess the diagnostic accuracy of intraoperative NIRS in detecting clamp-associated hemodynamic cerebral ischemia in patients under general anesthesia. MEDLINE, Google Scholar, and Web of Science were searched for studies that compared NIRS with the occurrence of immediate or early postoperative neurological deficits in patients undergoing CEA under GA. Meta-regression was performed to explore causes of heterogeneity.
Results:
A total of 28 studies involving 5729 patients were included. The results show that NIRS has a summary sensitivity of 47.5% and a summary specificity of 90.3% in diagnosing clamp-associated cerebral ischemia under general anesthesia, with an area under the ROC curve (AUC-ROC) of 0.85. The presence of a contralateral carotid occlusion (CCO) increased summary sensitivity while smoking history the presence of CCO and smoking history decreased summary specificity. For postoperative stroke prediction, NIRS has a summary sensitivity of 49.5% and summary specificity of 88.5%, with an AUC-ROC of 0.85.
Conclusions:
NIRS is a specific but not highly sensitive tool for detecting cerebral ischemia during CEA, and its use in combination with more sensitive neuromonitoring methods is recommended, in order to guide intraoperative neuroprotective strategies.
Context and Relevance
The meta-analysis included 28 studies involving 5729 patients undergoing CEA under general anesthesia. NIRS demonstrated a summary sensitivity of 47.5% and a summary specificity of 90.3% in diagnosing clamp-associated hemodynamic cerebral ischemia. The area under the ROC curve (AUC-ROC) for NIRS was 0.85. The presence of contralateral carotid occlusion (CCO) increased sensitivity.
What this paper adds
There is no gold standard for intraoperative neurological monitoring during carotid endarterectomy under general anesthesia. Non-invasive cerebral perfusion measurements have been considered since not every patient is a candidate for intervention under regional anesthesia. Near-infrared spectroscopy (NIRS) quantifies the regional oxygen saturation in the frontal lobe, which correlates with brain perfusion. Its accuracy in detecting brain ischemia has only been reported in a few studies and is highly variable across the literature. This study systematically reviews the diagnostic accuracy of intraoperative NIRS in patients submitted to carotid endarterectomy under general anesthesia. Meta-analysis was performed to determine the summary sensitivity, specificity, positive and negative likelihood ratios, and diagnostic odds ratio. The reference standard used was the occurrence of neurological deficits during recovery from anesthesia. A secondary meta-analysis was performed to determine the accuracy of NIRS in predicting postoperative stroke. This study used clinically meaningful reference standards, highlighting the performance of NIRS as a monitoring tool in carotid endarterectomy in everyday practice.
Introduction
Carotid endarterectomy (CEA) is the gold-standard procedure used to treat both symptomatic and asymptomatic carotid artery stenosis.1,2 However, there is a potential risk of perioperative stroke during CEA, with up to 1.2%–3% of previously asymptomatic patients being affected.3,4 The majority of these strokes are embolic in nature, while 10% are believed to be hemodynamic strokes occurring due to cerebral ischemia during carotid cross-clamping. 5 Intolerance to carotid cross-clamping may occur in up to 12% of patients undergoing CEA under regional anesthesia. 6
In patients undergoing CEA under regional anesthesia, the most reliable method to identify critical cerebral ischemia during carotid cross-clamping is clinical neurological monitoring, commonly known as the “awake” test.1,7 Nevertheless, regional anesthesia is not suitable for all patients, and some surgeons prefer operating patients on general anesthesia, thus alternative options for neuromonitoring are required. Several monitoring techniques have been used with general anesthesia, but none is as accurate as the “awake” test. 8
Near-infrared spectroscopy (NIRS) measures the absorption of light in the infrared spectrum to determine the levels of oxygenated hemoglobin. 9 Although widely used during CEA, alone or in conjunction with other strategies, it has been shown not to be particularly accurate, despite its high specificity in detecting cerebral ischemia during CEA under regional anesthesia.10,11 In the case of general anesthesia, its diagnostic accuracy is harder to determine, given the lack of a gold-standard. 12
The primary aim of this study is to perform a systematic review of the literature on the diagnostic accuracy of intraoperative NIRS in detecting clamp-associated hemodynamic cerebral ischemia in patients undergoing CEA under general anesthesia. Furthermore, a meta-analysis was performed to determine the pooled diagnostic accuracy of intraoperative NIRS in detecting clamp-associated hemodynamic cerebral ischemia in CEA under general anesthesia. The occurrence of neurological deficits during recovery from anesthesia (NDRA) was used as the reference test. Secondary aims included the determination of the pooled diagnostic accuracy of NIRS using other neuromonitoring tests as the reference, and the ability of intraoperative NIRS to predict postoperative ischemic stroke.
Methods
This systematic review was conducted in accordance with the Preferred Reporting Items for a Systematic Review and Meta-analysis of Diagnostic Test Accuracy Studies (PRISMA-DTA) statement. 13 The review protocol has been registered at https://www.Crd.york.ac.uk/PROSPERO/, ID: CRD42021231782.
Eligibility criteria
Prospective or retrospective observational studies assessing patients submitted to CEA under general anesthesia with intraoperative NIRS (index test) were included. Eligible studies had to provide data on the number of patients who presented with immediate or early postoperative neurological deficits (reference test); the intraoperative NIRS cutoff for the detection of cerebral ischemia; and the number of patients with a positive and negative reference test who had a positive and negative index test (to compute diagnostic accuracy parameters).
Search strategy
The electronic databases MEDLINE, Google Scholar, and Web of Science were searched up to 1 June 2022. The following query was used for MEDLINE (with adaptations for other databases): (“Endarterectomy” (MeSH Terms) OR “Endarterectomy, Carotid” (MeSH Terms) OR “carotid endarterectomy” (Text Word) OR “Carotid Endarterectomies” (MeSH Terms)) AND (“NIRS” (All Fields) OR “near infra-red spectroscopy” (All Fields) or “NIS” (All Fields) or “Oximetry”). No language restrictions were applied.
After duplicate removal, two reviewers (L.D.G. and J.R.N.) independently screened the titles and abstracts of preselected studies. Subsequently, references were selected for inclusion based on full-text evaluation by the same reviewers. Any disagreements were resolved by a third independent reviewer (M.F.M.). The search strategy further included cross-referencing.
Data extraction
Data concerning study design, patient characteristics, procedural variables and results were independently extracted by two reviewers using a purpose-built form. Variables whose data were extracted included the year of publication, country and center of recruitment, study design, recruitment time, sample size, participants’ age and gender distribution, frequency of cardiovascular comorbidities, and carotid symptomatic status. In addition, data related to procedural protocols was retrieved, as well as the diagnostic cutoff value of NIRS used in each study, number of participants with positive and negative NIRS test, and number of participants submitted to CEA. Further collected variables include the number of patients with NDRA and number of patients with postoperative stroke or transient ischemic attack.
Assessment of study quality
Study quality was assessed by two independent authors (J.R.N. and L.D.G.) based on the QUADAS-2 risk-of bias-tool for diagnostic test accuracy. 14 Discrepancies were resolved by a third author (M.F.M.).
Statistical analysis
Heterogeneity was evaluated through the I2 Q statistic-related measure of variance, and a diagnostics random-effects model was preferred to obtain summary sensitivity, specificity, positive and negative likelihood ratios, and diagnostic odds ratio. Forest plots were used to display results from the meta-analysis, where the measure of effect for each study is represented by a square and the respective areas are proportional to study weight. A random-effects model was performed to compute the diagnostic test’s summary ROC curve. Meta-regression was used to explore heterogeneity through a random-effects model, with the diagnostic odds ratio as the dependent variable. A p value inferior to 0.05 was considered statistically significant for all analyses. Computations were conducted using Stata 16.1, OpenMeta [Analyst], and MetaDiSc 1.4. 15
Results
Search results
The search yielded 504 references, of which 363 were screened by abstract and title after elimination of duplicates. Through screening, 69 references were deemed eligible for full text appraisal, and 28 were selected for inclusion in this review (Fig. 1), comprising a total of 5729 patients and 5760 interventions under general anesthesia.13,16 –39
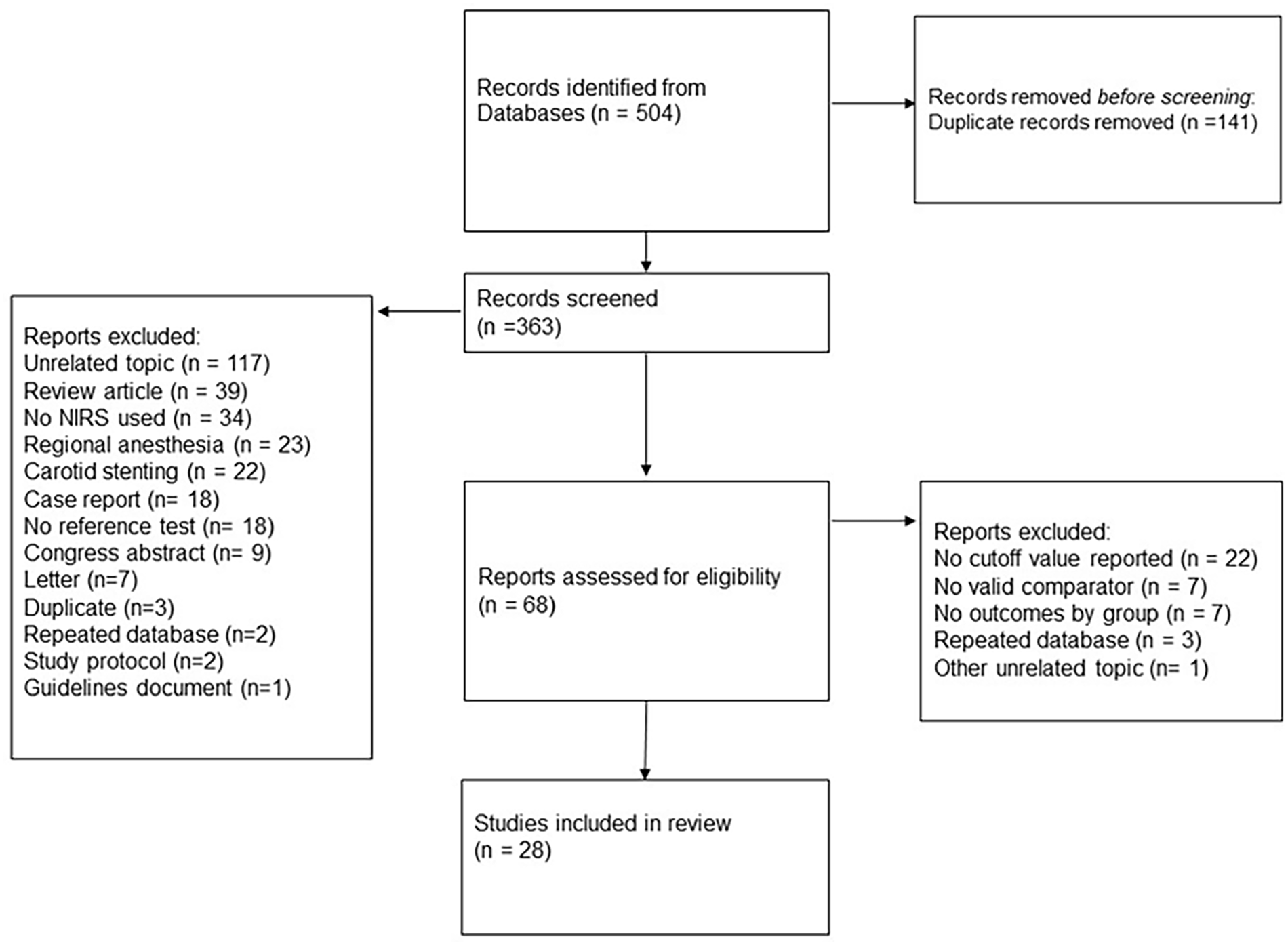
PRISMA flow diagram illustrating reference selection process.
The characteristics of each study are displayed in Table 1. Study sample size varied between 19 and 654 patients. Mean patient age ranged between 59 and 73.5 years. The percentage of male patients across studies varied between 59% and 86%. The prevalence of symptomatic carotid stenosis across studies varied between 0% and 100%. The percentage of patients with contralateral carotid occlusion (CCO) ranged between 0% and 51% (Table 1).
Characteristics of the studies and summary clinical data of the patients included in the systematic revision and meta-analysis.
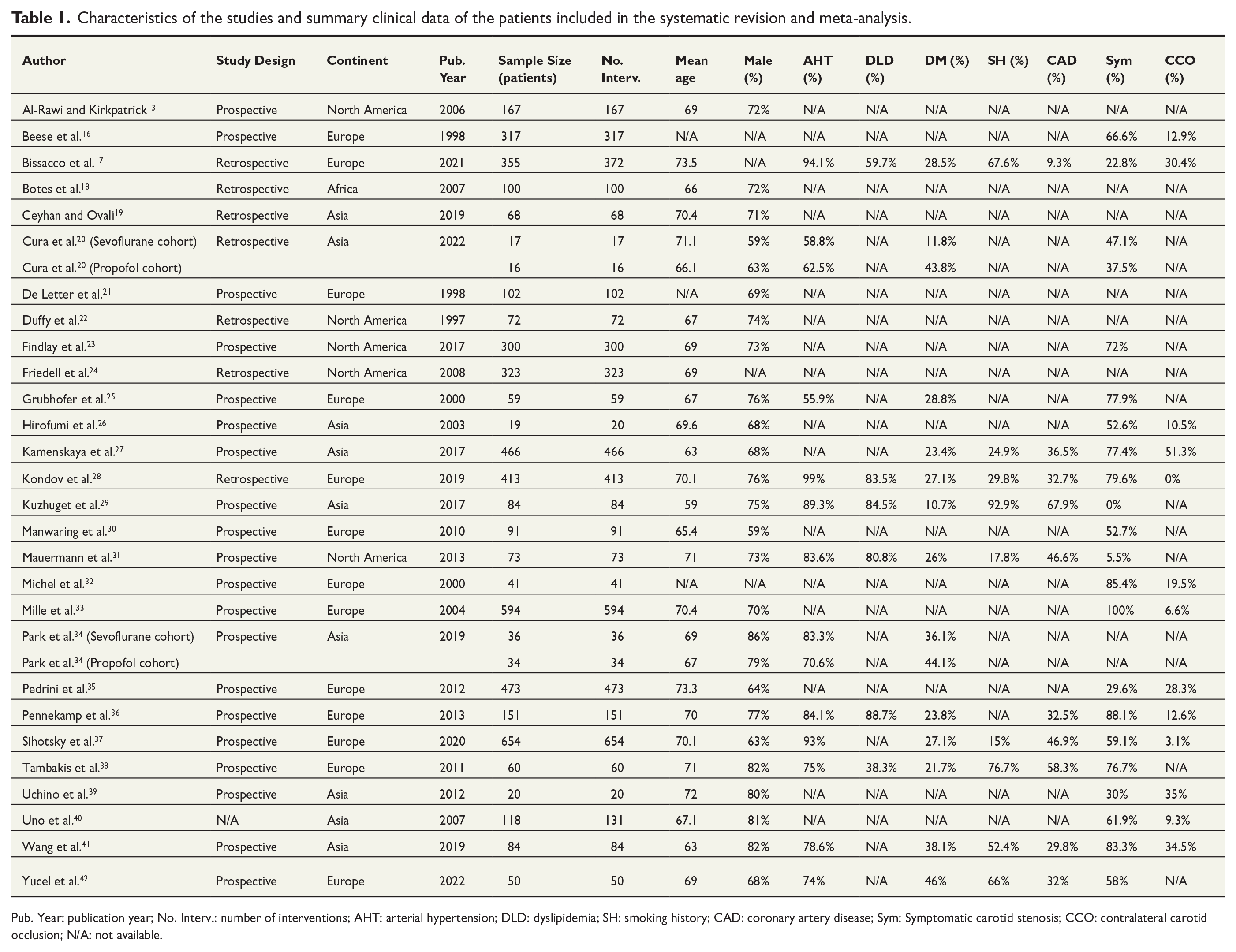
Pub. Year: publication year; No. Interv.: number of interventions; AHT: arterial hypertension; DLD: dyslipidemia; SH: smoking history; CAD: coronary artery disease; Sym: Symptomatic carotid stenosis; CCO: contralateral carotid occlusion; N/A: not available.
While most studies used a selective shunting protocol (n = 23), only one study used routine shunting, and three studies reported a “never shunt” protocol or excluded shunted patients from the sample. The rate of shunt use in the studies which used it selectively varied between 0% and 32%.
The majority of studies (n = 23) reported using the INVOS™ (Somanetics Corp, Troy. MI, USA) system. Other NIRS systems used included the NIRO 300 (Hamamatsu Photonics, Shizuoka, Japan), Masimo Cerebral Oxymeter (Masimo Corporation, Irvine, CA, USA), Equanox 7600 (Nonin Medical Inc, Plymouth, MN, USA) and Foresight (CAS Medical Systems, Irvine, CA, USA). The location of the oximetry probes was the frontal or frontoparietal region in 21 studies and the temporal region in one study. Bilaterally placed probes were used in 16 studies, while six studies reported using only one probe ipsilateral to the side of the intervention (Supplemental Table S1).
Risk of bias
The QUADAS-2 risk of bias assessment is displayed on Fig. 2. Overall, most studies were considered as having a low risk of bias in the index test and flow and timing domains. In the patient selection domain, approximately half of the studies were considered as having an unclear risk of bias, mostly due to the authors not specifying whether patients had been consecutively selected. Most studies were deemed as having high risk of bias in the standard reference domain, as only one study specified whether NDRA or stroke had a hemodynamic or an atherothrombotic cause. 40
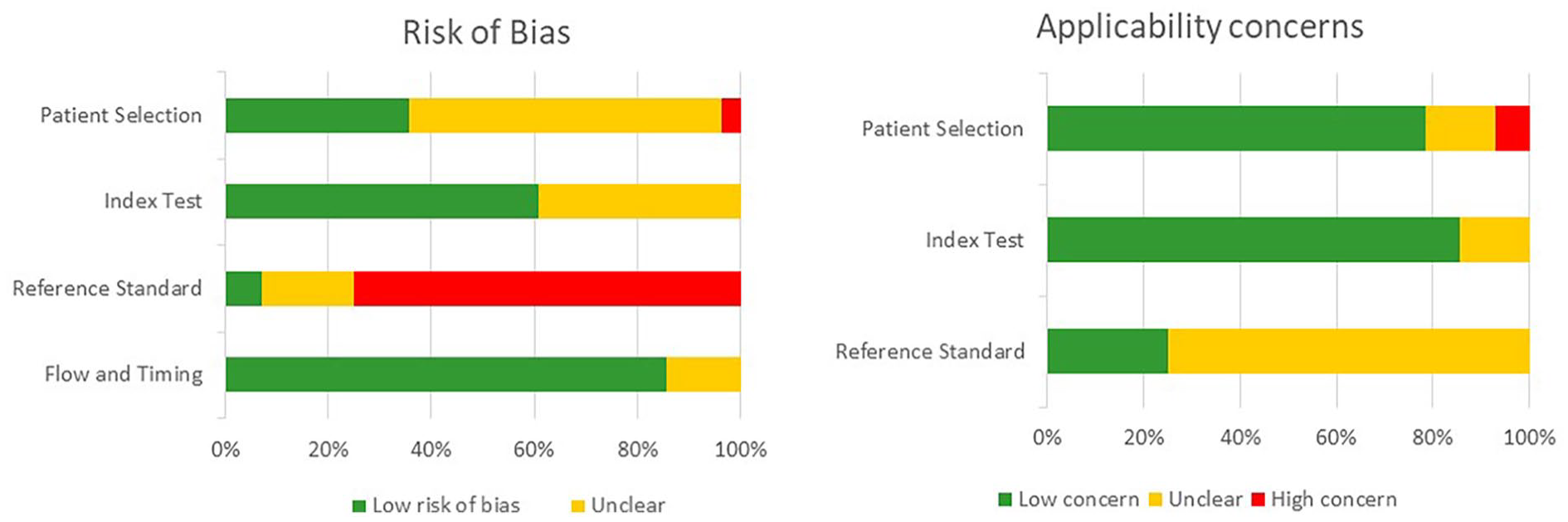
QUADAS-2 risk of bias assessment.
Diagnostic accuracy of NIRS
Neurological deficits during recovery from anesthesia
Eight studies17,23,24,33,35,39,40,42 were used to determine summary sensitivity, specificity, positive and negative likelihood ratios and diagnostic odds ratio of NIRS compared to the occurrence of NDRA. Ten studies were included in the calculation of the summary ROC curve and 15 studies in the meta-regression.17,19,22 –24,26,28,33,35,36,38 –42
The reported NIRS decrease cutoff (∆NIRS) for the diagnosis of carotid clamping-related cerebral ischemia reported varied between 10% and 25% across all studies, and the incidence of NDRA ranged from 0% to 5.6%. Overall, there were a total of 49 patients who presented NDRA, of which 24 had NIRS drop beyond the cutoff values defined in each study. The sensitivity and specificity of NIRS varied between 0%–100% and 26.3%–99.2% among all studies when using NDRA as reference. The positive and negative predictive values varied between 0%–40% and 80.9%–100%, respectively. The positive and negative likelihood ratios and the diagnostic odds ratio ranged 0–47.6 and 0–1.07, and 0–24.8, respectively (Supplemental Table S2).
Meta-analysis of diagnostic accuracy was performed for studies with a diagnostic cutoff ∆NIRS of 20%. The partial AUC-sROC was 0.85 (Fig. 3A) with a summary sensitivity of 47.5% (95% confidence interval (CI) 31.9–63.7, I2 = 0, p = 0.456) and a summary specificity of 90.3%, (95% CI 71.8–97.1, I2 = 97.94%, p < 0.001). The summary positive likelihood ratio (LR+) was 6.121 (95% CI 2.327–16.100, I2 = 89%, p < 0.001), while the summary negative likelihood ratio (LR−) was 0.472 (95% CI 0.364–0.613, I2 = 0%, p = 0.860) (Fig. 4). The summary diagnostic odds ratio (DOR) was 12.218 (95% CI 5.242–28.469, I2 = 12.43%, p = 0.333) (Fig. 5A).
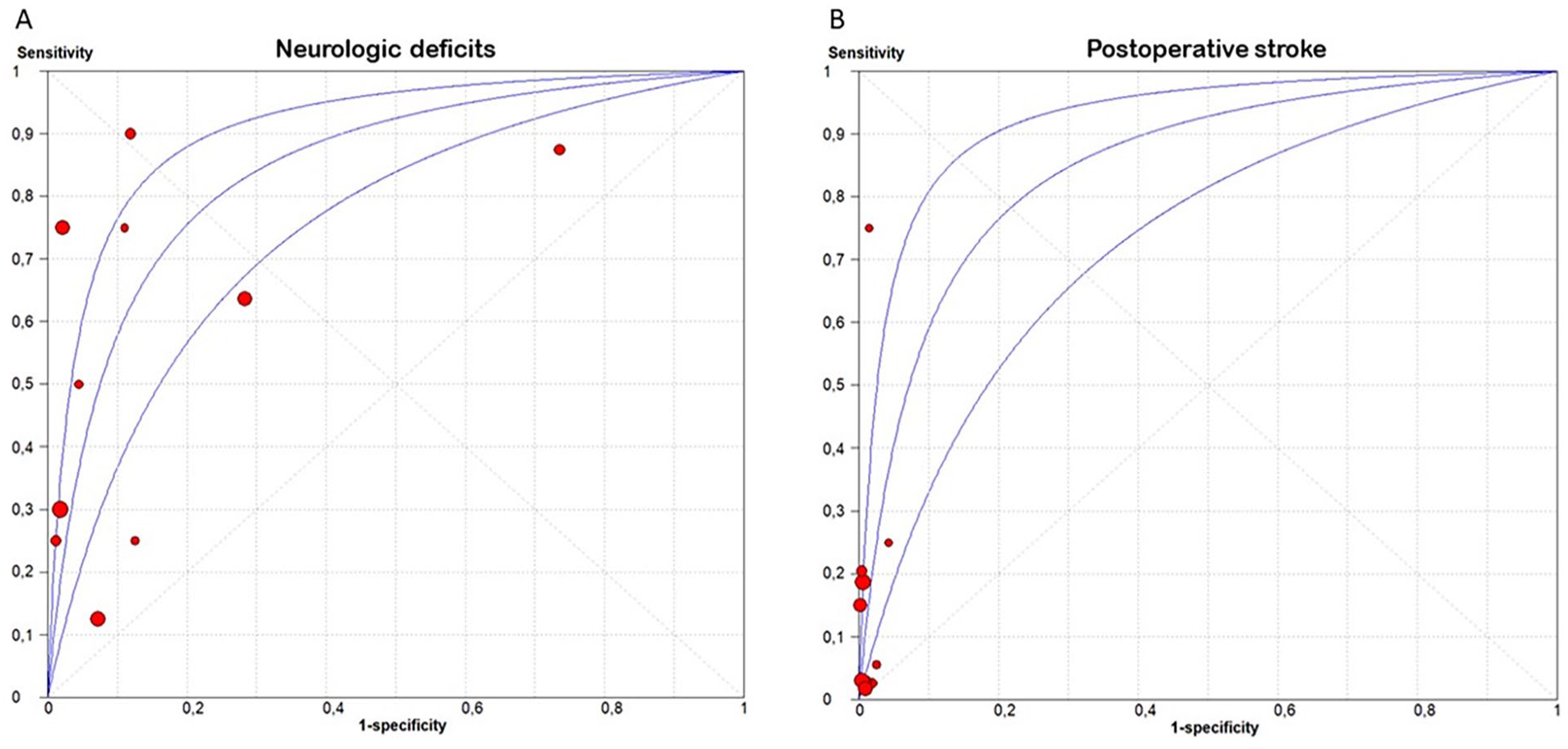
Summary receiver operating characteristic curves for the performance of NIRS in diagnosing clamp-associated cerebral ischemia (A) and predicting postoperative stroke (B).
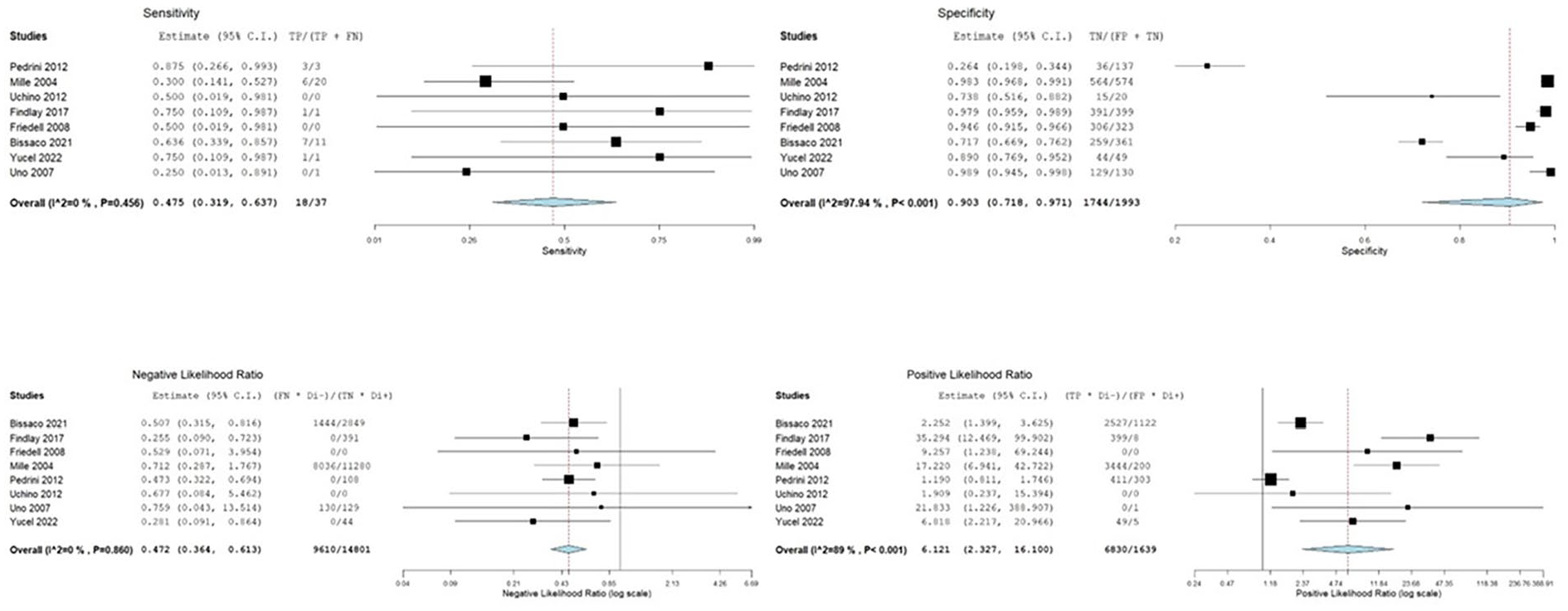
Forest-plots of sensitivity, specificity, positive and negative likelihood ratios of NIRS in diagnosing clamp-associated cerebral ischemia.
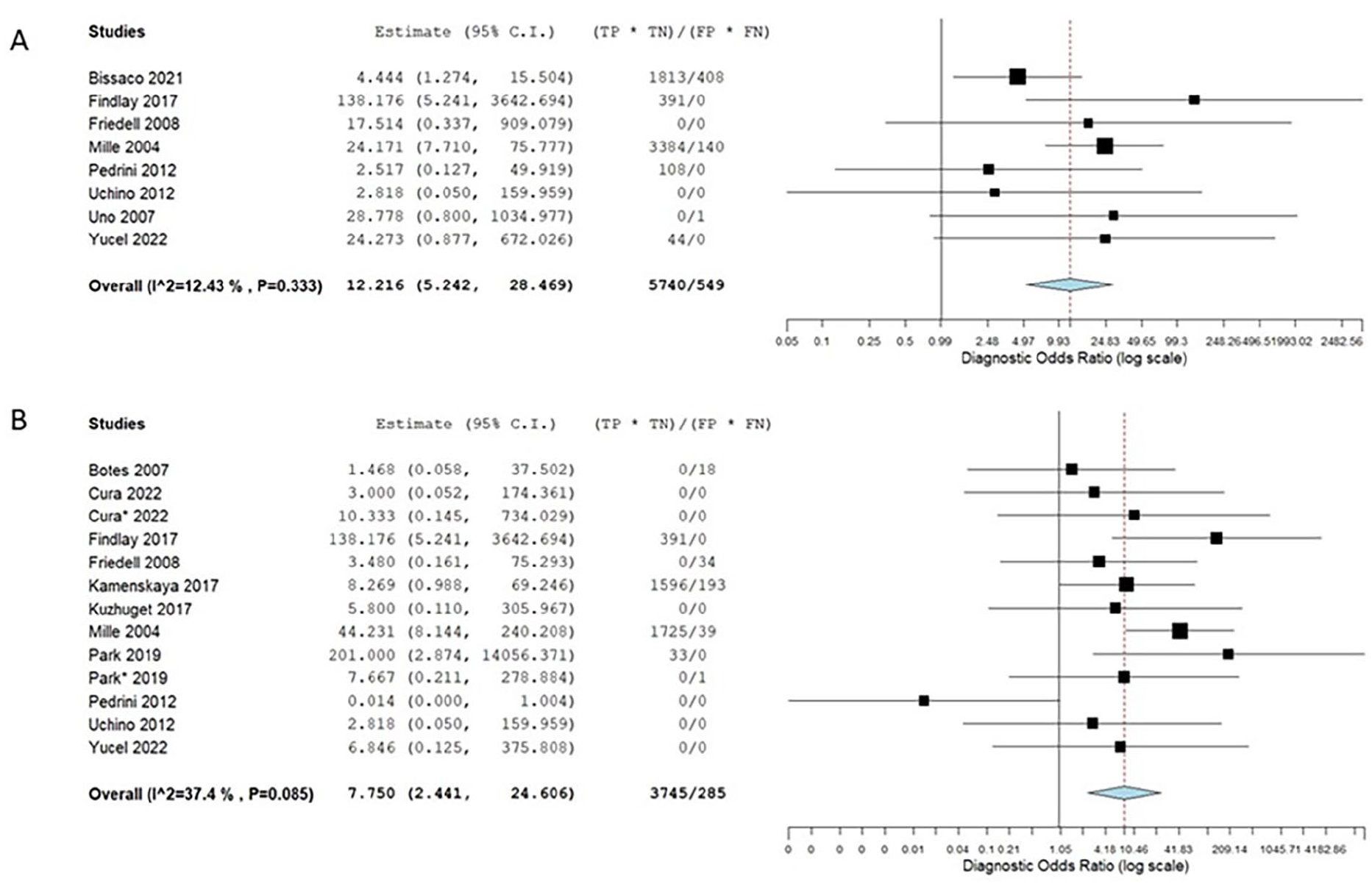
Forest plots of diagnostic odds ratio of NIRS in diagnosing clamp-associated cerebral ischemia (A) and predicting postoperative stroke (B).
Meta-regression was performed to assess variables accounting for heterogeneity in diagnostic accuracy across studies. There was no considerable threshold effect observed (β = 0.02 (95% CI –0.16 to 0.21), p = 0.814). Only the NIRS equipment used accounted for interstudy heterogeneity in DOR (β = –0.81 (95% CI –1.53 to –0.09), p = 0.028). The presence of CCO was associated with higher summary sensitivity (β = 6.03 (95% CI 0.56–11.50), p = 0.031). Variables accounting for interstudy heterogeneity in specificity included smoking history (β = –2.94 (95% CI –4.86 to –1.01), p = 0.003) and the presence of CCO (β = –8.43 (95% CI –14.03 to –2.82), p = 0.003), both of which decreased summary specificity (Supplemental Table S4).
Postoperative stroke
Eleven studies18,20,23,24,27,29,33 –35,39,42 were included in the determination of summary sensitivity, specificity, positive and negative likelihood ratios and diagnostic odds ratio of NIRS compared to postoperative stroke. Nine studies were included in the calculation of the summary ROC curve and nineteen in the meta-regression.18 –20,22 –24,26 –30,33 –36,38,39,41,42
Overall, there were 53 postoperative strokes, of which 30 had non-positive NIRS diagnostic values, according to the threshold defined in each study. The sensitivity and specificity of NIRS for the prediction of postoperative strokes varied between 0%–100% and 75%–100%, respectively. Positive and negative predictive value varied between 0%–19% and 76%–100%, respectively. The positive and negative likelihood ratios and diagnostic odds ratio varied between 0–49.9 and 0–1.2, and 0–44.2, respectively (Supplemental Table S3).
Meta-analysis was performed in order to determine the value of NIRS in predicting postoperative stroke. Studies with a diagnostic cutoff ∆NIRS of 20% were included. The partial AUC-sROC was 0.85 (Fig. 3B) with a summary sensitivity of 49.5% (95% confidence interval (CI) 30.9 to 68.3, I2 = 0, p = 0.727) and a summary specificity of 88.5% (95% CI 74.6–95.3, I2 = 96.22%, p < 0.001). The summary LR+ was 4.742 (95% CI 1.965–11.439, I2 = 80.02%, p < 0.001), while the summary negative LR− was 0.812 (95% CI 0.220–3.002, I2 = 92.03%, p < 0.001) (Fig. 6). The summary diagnostic odds ratio (DOR) was 7.750 (95% CI 2.441–24.606, I2 = 37.4%, p = 0.085) (Fig. 5B).
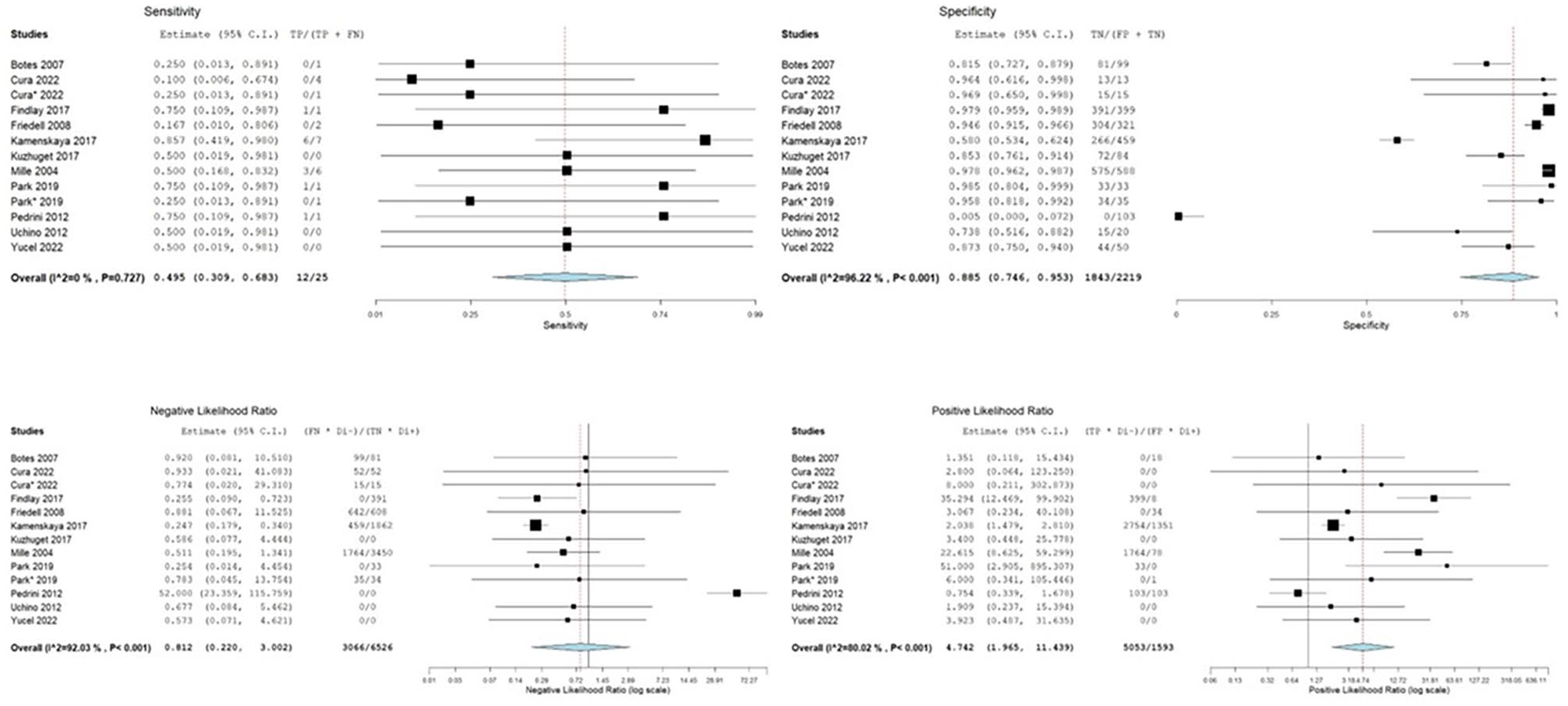
Forest-plots of sensitivity, specificity, and positive and negative likelihood ratios of NIRS in predicting postoperative stroke.
There was no considerable threshold effect observed (β = 0.02 (95% CI –0.17 to 0.28), p = 0.625). The presence of a symptomatic carotid stenosis was associated with higher DOR (β = 3.96 (95% CI 0.83–7.08), p = 0.013). The type of study design was the only factor accounting for heterogeneity in summary sensitivity (β = –1.35 (95% CI –3.39 to –0.51), p = 0.008). Variables accounting for interstudy heterogeneity in specificity included arterial hypertension (β = 3.07 (95% CI 0.90–5.23), p = 0.006) and the presence of CCO (β = –6.07 (95% CI –11.41 to –0.72), p = 0.026) (Supplemental Table S4).
Other neuromonitoring tests
A total of 16 studies compared NIRS against other neuromonitoring tests (Supplemental Table S5). Due to the small number of studies for each neuromonitoring modality, it was not possible to perform meta-analysis of diagnostic accuracy.
Five studies compared NIRS against electroencephalography (EEG).18,21,31,36,40 Two studies utilized a ∆NIRS diagnostic cutoff value of 20%, yielding sensitivity and specificity values of 0% and 100% and 87.2%–99.1%, respectively. One study, utilizing a ∆NIRS cutoff of 16%, obtained a sensitivity and specificity of 94% and 96%, respectively. 36
When compared against sensory evoked potentials (SEP), two studies using a ∆NIRS cutoff of 10% obtained sensitivity values of 50% and 83%, and specificities of 94% and 96%, respectively.22,32 With a ∆NIRS cutoff of 20%, two studies found a sensitivity of 0% and 7% and a specificity of 99% in both studies.16,40 Friedell et al. 24 compared NIRS against changes in EEG and/or SEP, reporting a sensitivity of 75% and a specificity of 98%.
In addition, two studies compared NIRS against transcranial Doppler (TCD), using a ∆NIRS cutoff of 12% and 13%, respectively.25,41 The sensitivity obtained was 75% and 100%, while the specificity was 92% and 87%, respectively. Uno et al. 40 reported a sensitivity and specificity of 0% and 99%, respectively, for a ∆NIRS cutoff of 20%.
Stump pressure was used in the standard reference in two studies, yielding sensitivity values of 86% and 76%, and specificity values of 91% and 81%, respectively.30,38 Finally, one study compared NIRS with cerebral function monitoring (CFM) and reported a sensitivity and specificity of 100% and 93.2%, respectively. 13
Discussion
This study suggests that intraoperative NIRS has an estimated meta-analytical summary sensitivity of 47.5% and summary specificity of 90.3% in diagnosing clamp-associated hemodynamic cerebral ischemia in patients undergoing CEA under general anesthesia.
The present findings are similar to the results on patients undergoing intervention under regional anesthesia from two previous meta-analyses.10,11 These studies demonstrated that NIRS has a low sensitivity and high specificity in regional anesthesia. However, no previous study has focused on its diagnostic accuracy in patients under general anesthesia.
Heterogeneity was negligible for sensitivity and considerable for specificity, which was addressed through meta-regression. The presence of a contralateral carotid occlusion both increased summary sensitivity and decreased summary specificity. The most plausible explanation for this finding is the compromised collateral network and compensatory mechanism that comes into play during carotid cross-clamping.43,44 In a previous study, patients undergoing CEA with a contralateral carotid occlusion had greater drops in oximetry values ipsilateral to the intervention side after clamping. 45 Therefore, local hemispheric oxygen saturation seems to be more sensitive to a sudden decrease in perfusion. Contralateral carotid occlusion has also been associated with adverse postoperative events, including in-hospital death, stroke, and myocardial infarction.8,46 One study has shown that patients with an incomplete Circle of Willis characterized by an isolated middle cerebral artery had an over 10-fold risk of suffering an immediate post-CEA neurologic event.47,48 Nonetheless, it has been suggested that even in patients with a contralateral carotid occlusion, CEA can be safely performed without using a shunt. 49
Smoking accounted for a decrease in specificity. One possibility revolves around the increased carboxyhemoglobin levels found in smokers. 50 Carbon monoxide has a higher affinity for hemoglobin than oxygen, which could result in lower baseline NIRS values and greater drops in NIRS after clamping, even in the absence of cerebral ischemia. 51 However, one study was unable to show changes in NIRS values after CO inhalation in healthy volunteers. 52 Smoking also leads to cerebral vasoconstriction and decreased local blood flow, which could explain this finding. 53
Most studies reported using different models of the INVOS™ oximeter.16 –30,32 –39 Some studies have compared the INVOS™ oximeter with other models, yielding discrepant values, although none have been performed in patients undergoing CEA.54 –56
A selective shunting protocol was used by most studies, which were based on a varied range of indications. This field of research holds significance due to the small yet relevant number of patients who experience hemodynamic cerebral ischemia during carotid cross-clamping. 57 The implementation of shunts could theoretically mitigate this concern effectively, despite the inconclusive evidence. 58 Surprisingly, the use of shunt did not affect diagnostic accuracy in meta-regression.
The estimated summary sensitivity of intraoperative NIRS in predicting postoperative stroke was low (49.5%). It is important to note that no study provided information on the cause of stroke. As only a minority of strokes following CEA are hemodynamic in nature,59,60 it is likely that intraoperative NIRS is not able to detect the majority of postoperative strokes. This, coupled with the relatively low incidence of postoperative strokes, accounts for the sensitivity values observed across studies. The presence of a contralateral carotid occlusion was also associated with lower specificity. Symptomatic carotid stenosis was associated with higher diagnostic odds ratio value, possibly due to microembolization from an unstable plaque and ensuing disruption of collateral networks, and lower tolerance to cross-clamping. 61
This study has some limitations, mainly related to the absence a gold standard reference test to confirm the diagnosis of clamp-related cerebral ischemia. There is no information on the etiology of NDRA, some of which may not have a hemodynamic cause. Also, the definition of NDRA is not cut clear, and might vary between centers, especially in prospective and retrospective studies. The use of shunts may also affect the relation between NIRS values and the occurrence of neurologic symptoms and ischemic events. The duration of post-clamping decrease of NIRS values and magnitude of normalization with shunt insertion are often unexplored variables that could also influence perioperative outcomes. Some bias may also be present due to the impossibility of blinding reviewers to the results of the index and standard tests.
In contrast, this study has some strengths. The selection criteria were inclusive, not excluding on the basis of language or time of publication. A considerable number of studies were included, amounting to a total of 5760 interventions. The large sample of studies also enabled the evaluation of source heterogeneity in diagnostic accuracy through meta-regression.
In conclusion, NIRS is a highly specific but not sensitive neuromonitoring test to detect clamp-associated hemodynamic cerebral ischemia during CEA under general anesthesia. Therefore, it may not be reliable enough to be used as the exclusive method for this end or to guide intraoperative neuroprotective strategies. Its high specificity may be used in conjunction with more sensitive neuromonitoring methods in validated protocols.
Supplemental Material
sj-docx-1-sjs-10.1177_14574969241282611 – Supplemental material for The diagnostic accuracy of intraoperative near-infrared spectroscopy in carotid artery endarterectomy under general anesthesia: A systematic review with meta-analysis
Supplemental material, sj-docx-1-sjs-10.1177_14574969241282611 for The diagnostic accuracy of intraoperative near-infrared spectroscopy in carotid artery endarterectomy under general anesthesia: A systematic review with meta-analysis by Luís Duarte-Gamas, Mariana Fragão-Marques, Petar Zlatanovic, José P. Andrade and João Rocha-Neves in Scandinavian Journal of Surgery
Supplemental Material
sj-docx-2-sjs-10.1177_14574969241282611 – Supplemental material for The diagnostic accuracy of intraoperative near-infrared spectroscopy in carotid artery endarterectomy under general anesthesia: A systematic review with meta-analysis
Supplemental material, sj-docx-2-sjs-10.1177_14574969241282611 for The diagnostic accuracy of intraoperative near-infrared spectroscopy in carotid artery endarterectomy under general anesthesia: A systematic review with meta-analysis by Luís Duarte-Gamas, Mariana Fragão-Marques, Petar Zlatanovic, José P. Andrade and João Rocha-Neves in Scandinavian Journal of Surgery
Supplemental Material
sj-docx-3-sjs-10.1177_14574969241282611 – Supplemental material for The diagnostic accuracy of intraoperative near-infrared spectroscopy in carotid artery endarterectomy under general anesthesia: A systematic review with meta-analysis
Supplemental material, sj-docx-3-sjs-10.1177_14574969241282611 for The diagnostic accuracy of intraoperative near-infrared spectroscopy in carotid artery endarterectomy under general anesthesia: A systematic review with meta-analysis by Luís Duarte-Gamas, Mariana Fragão-Marques, Petar Zlatanovic, José P. Andrade and João Rocha-Neves in Scandinavian Journal of Surgery
Supplemental Material
sj-docx-4-sjs-10.1177_14574969241282611 – Supplemental material for The diagnostic accuracy of intraoperative near-infrared spectroscopy in carotid artery endarterectomy under general anesthesia: A systematic review with meta-analysis
Supplemental material, sj-docx-4-sjs-10.1177_14574969241282611 for The diagnostic accuracy of intraoperative near-infrared spectroscopy in carotid artery endarterectomy under general anesthesia: A systematic review with meta-analysis by Luís Duarte-Gamas, Mariana Fragão-Marques, Petar Zlatanovic, José P. Andrade and João Rocha-Neves in Scandinavian Journal of Surgery
Supplemental Material
sj-docx-5-sjs-10.1177_14574969241282611 – Supplemental material for The diagnostic accuracy of intraoperative near-infrared spectroscopy in carotid artery endarterectomy under general anesthesia: A systematic review with meta-analysis
Supplemental material, sj-docx-5-sjs-10.1177_14574969241282611 for The diagnostic accuracy of intraoperative near-infrared spectroscopy in carotid artery endarterectomy under general anesthesia: A systematic review with meta-analysis by Luís Duarte-Gamas, Mariana Fragão-Marques, Petar Zlatanovic, José P. Andrade and João Rocha-Neves in Scandinavian Journal of Surgery
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by National Funds through FCT—Fundação para a Ciência e Tecnologia, I.P., within CINTESIS, R&D Unit (reference UIDB/4255/2020) and within the scope of the project RISE, Associated Laboratory (reference LA/P/0053/2020).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
