Abstract
Background:
Blunt thoracic aortic injury (BTAI) is associated with considerable mortality and morbidity. Thoracic endovascular aortic repair (TEVAR) has essentially replaced open aortic repair (OAR) with superior outcome. The objective of this study was to evaluate the early and intermediate-term outcomes in patients sustaining BTAI treated with TEVAR, and to evaluate the prevalence and impact of left subclavian artery (LSA) coverage.
Methods:
This retrospective analysis includes patients undergoing TEVAR for BTAI between February 2006 and December 2022 at the Helsinki University Hospital, Finland. The primary endpoints were 30-day and 2-year mortality, technical success of stent-graft system deployment, and procedural and device-related complications. The secondary endpoints pertained to reintervention events: conversion to OAR, re-TEVAR, any endovascular/surgical reinterventions for optimal stent-graft function, or any reinterventions during follow-up.
Results:
A total of 38 consecutive BTAI patients treated with TEVAR were included in the analyses. Median patient age was 45.5 years (range, 15–79) and 66% were male. The median follow-up period was 39 months. Technical success was 100%, 30-day mortality was 0%, and 2-year mortality was 11% in the study cohort. Coverage of LSA without revascularization (13/18) resulted in one postoperative nondisabling stroke (1/13), no paraplegia, and one had ischemic left arm findings (1/13). Only one patient required reintervention (LSA embolization; 1/38).
Conclusion:
In this institutional series, we provide further evidence in favor of TEVAR for BTAI treatment. We demonstrated that TEVAR is linked to highly favorable outcomes in the short and intermediate term, and coverage of LSA without revascularization was quite well tolerated.
Keywords
Context and relevance
Blunt thoracic aortic injury (BTAI) is a life-threatening injury necessitating urgent treatment. Due to improved outcomes, thoracic endovascular aortic repair (TEVAR) has been preferred over open aortic repair. However, TEVAR is not without its own set of complications, including spinal cord ischemia, postoperative ischemic stroke, and acute arm ischemia. We evaluated the outcomes of patients with BTAI undergoing TEVAR, including mortality rates, technical success, and device- and procedure-related complications. We also assessed the impact of left subclavian artery (LSA) coverage and necessity for reinterventions. Our findings suggest that TEVAR is associated with excellent technical success, low mortality rates at 30 days and 2 years, infrequent need for reinterventions, and a low incidence of neurologic complications with or without LSA coverage.
Introduction
Blunt thoracic aortic injury (BTAI) is the second leading cause of mortality associated with blunt trauma, 1 often caused by motor vehicle collisions or falls from considerable heights. Approximately 80% of patients die at the scene, 2 and emergent medical treatment is crucial to prevent early in-hospital mortality. Most BTAIs occur in the proximal ascending aorta but are seldom encountered due to fatalities at the scene. Aortic isthmus injuries are most observed since the ligamentum arteriosum tethers the aorta next to a relatively mobile arch during deceleration. Open aortic repair (OAR) has previously been the mainstay of treatment for BTAIs but linked to a 16.7% postoperative mortality and 8.3% postoperative paraplegia rate. 3 Thoracic endovascular aortic repair (TEVAR) has been increasingly used and has essentially replaced OAR.
However, procedural and device-related hazards can influence the feasibility and long-term effects of the intervention when compared to OAR. An important consideration during TEVAR is the need to cover the left subclavian artery (LSA). LSA coverage may reverse the flow of blood in the vertebral artery, which could result in blood being “stolen” from the cerebral posterior circulation, resulting in vertebrobasilar ischemia. It can manifest clinically as vertebrobasilar insufficiency (vertigo, headaches, diplopia, blindness, vomiting, ataxia) or posterior cerebral stroke. Furthermore, because the LSA facilitates blood flow to the ipsilateral arm, its coverage may result in ischemia and claudication symptoms in the arm.
We reviewed our 16-year single-center institutional experience of the endovascular approach in BTAI. Our primary objective was to evaluate the early and intermediate-term outcomes following TEVAR for BTAI, with a special emphasis on LSA coverage without revascularization.
Methods
Patient selection and study design
A retrospective analysis was conducted on electronic medical records of TEVAR patients at Helsinki University Hospital between February 2006 and December 2022. Notably, 38 consecutive patients surviving to hospital with BTAI and subsequently undergoing TEVAR were identified. Previously serving 1.8 million people, the Helsinki University Hospital Trauma Unit currently serves 2.15 million people from 2018 onwards. It is a referral center for all major traumas in that area. Data were collected in an electronic datasheet and checked for accuracy while conforming to TEVAR standards. 4 New Injury Severity Scores (NISS) were retrieved from the trauma registry database. Due to the retrospective nature of the study, the Institutional Review Board waived informed patient consent.
Definitions
Clinicians graded BTAI injuries according to the Society of Vascular Surgery (SVS) classification criteria: 5 Grade I (intimal tear), Grade II (intramural hematoma), Grade III (pseudoaneurysm), and Grade IV (free rupture).
Spinal cord ischemia (SCI) is a new onset periprocedural transient or permanent neuromotor or sensory dysfunction in the lower extremities. Stroke was defined as a focal or global neurological impairment lasting more than 24 h and visualized as cerebral infarction or hemorrhage through neuroimaging. Exertion-induced ischemia of the arm is defined as claudication. Systolic blood pressure < 90 mmHg, heart rate > 100–130 beats per minute, or no significant response to extensive transfusion or vasopressors in shock defines hemodynamic instability.
Technical success was defined as successful access and deployment of the stent-graft system at the site of injury without repeat interventions or conversion to OAR, and no signs of Type I endoleak. Early endoleak is detected during completion angiography or computed tomography angiography (CTA) after TEVAR and delayed endoleak in follow-up imaging. Treatment success was defined as the absence of delayed Type I or Type III endoleak or worsening of the aortic injury after the index procedure or reinterventions.
Post-discharge TEVAR involves vascular and clinical examinations with CTA assessments at 1-, 6-, and 12-month intervals, and yearly thereafter. Our standard practice is to follow young patients throughout their lives. Follow-up intervals can be extended for 2–3 years after a patient’s condition stabilizes after a certain period, typically 5 years.
Outcome measures
All-cause 30-day and 2-year mortality, the success of stent-graft deployment, and postoperative procedural and device-related complications were the primary outcome measures. The secondary endpoints were conversion to OAR, repeat TEVAR, or any other surgical or endovascular reintervention to maintain optimal stent-graft function.
Diagnostics and operative procedure
On hospital admission, hemodynamically stable trauma patients received a full-body CTA scan to confirm injuries, measure aortic anatomy, and grade injury severity. NISS assessed the severity of whole-body trauma.
TEVAR was conventionally conducted under general anesthesia (95%) via transfemoral access as previously described in detail. 6 Stent-grafts were deployed with fluoroscopic guidance and a 2-cm proximal landing zone was ensured before completion aortography to rule out endoleak. When necessary, LSA was intentionally covered (42%), for optimal graft fixation and sealing. Revascularization of LSA before coverage (8%) was made at the surgeon’s discretion, based on patency of Circle of Willis and dominance of either vertebral artery. We aimed for a slightly reduced activated clotting time (ACT) of 180–250 s, in accordance with the SVS guidelines 5 since systemic heparinization has been debated in polytrauma patients. Postoperatively, heparin was fully reversed with protamine.
Statistical analysis
Statistical analyses were performed using SPSS Statistics software version 26.0 (IBM Corp., Armonk, NY). Categorical data are presented as counts with percentages in parentheses and continuous data as medians with ranges or interquartile ranges in parentheses. The Kaplan–Meier curve was used for overall survival analysis. The hazard ratio and risk factors contributing to all-cause 2-year mortality were determined with a multivariate Cox proportional hazard regression model with statistical significance set at p-value less than 0.05.
Results
Patients’ characteristics
Over the study period, 38 patients underwent TEVAR for BTAI treatment and were analyzed (Fig. 1). Most patients (66%) were males, and the median age was 45.5 (range 15–79) years. A notable proportion of patients had a history of alcohol or drug abuse (37%) and hypertension (26%). Table 1 shows baseline characteristics of all patients. Motor vehicle collision was the most frequent aortic injury mechanism, accounting for 55% of cases. The median NISS was 41 (range, 17–66) and all patients (n = 38) presented with concomitant non-aortic injuries (Supplementary Table 1). With respect to BTAI severity, 5 grade III constituted 76% of the BTAIs. An aortic isthmus injury was the most common (92%) (Table 1).
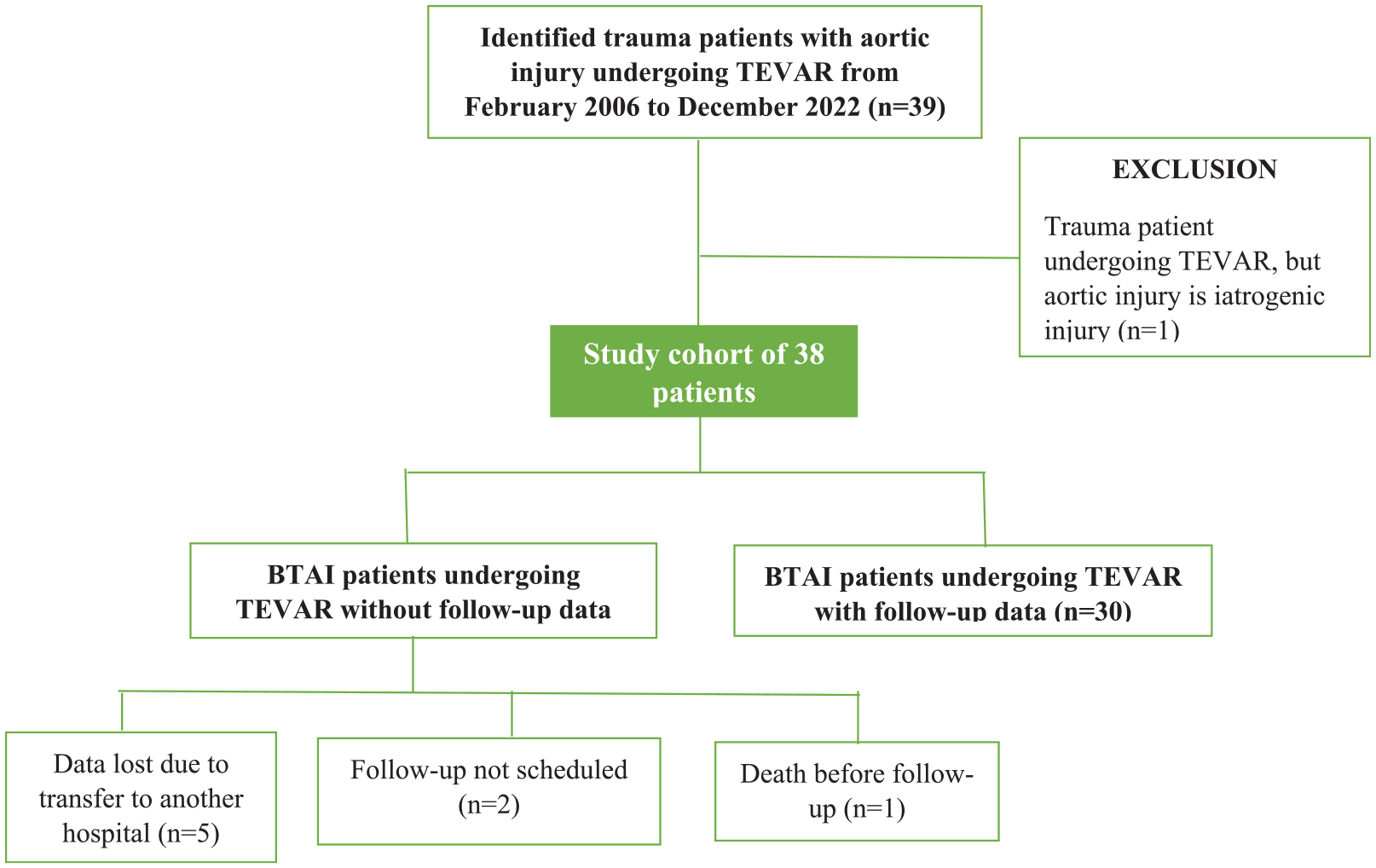
Study flowchart. Patients undergoing TEVAR for BTAI were divided into groups with (n = 30) and without (n = 8) follow-up computer tomography angiography (CTA) data.
Baseline characteristics of patients undergoing TEVAR for BTAI (n = 38).
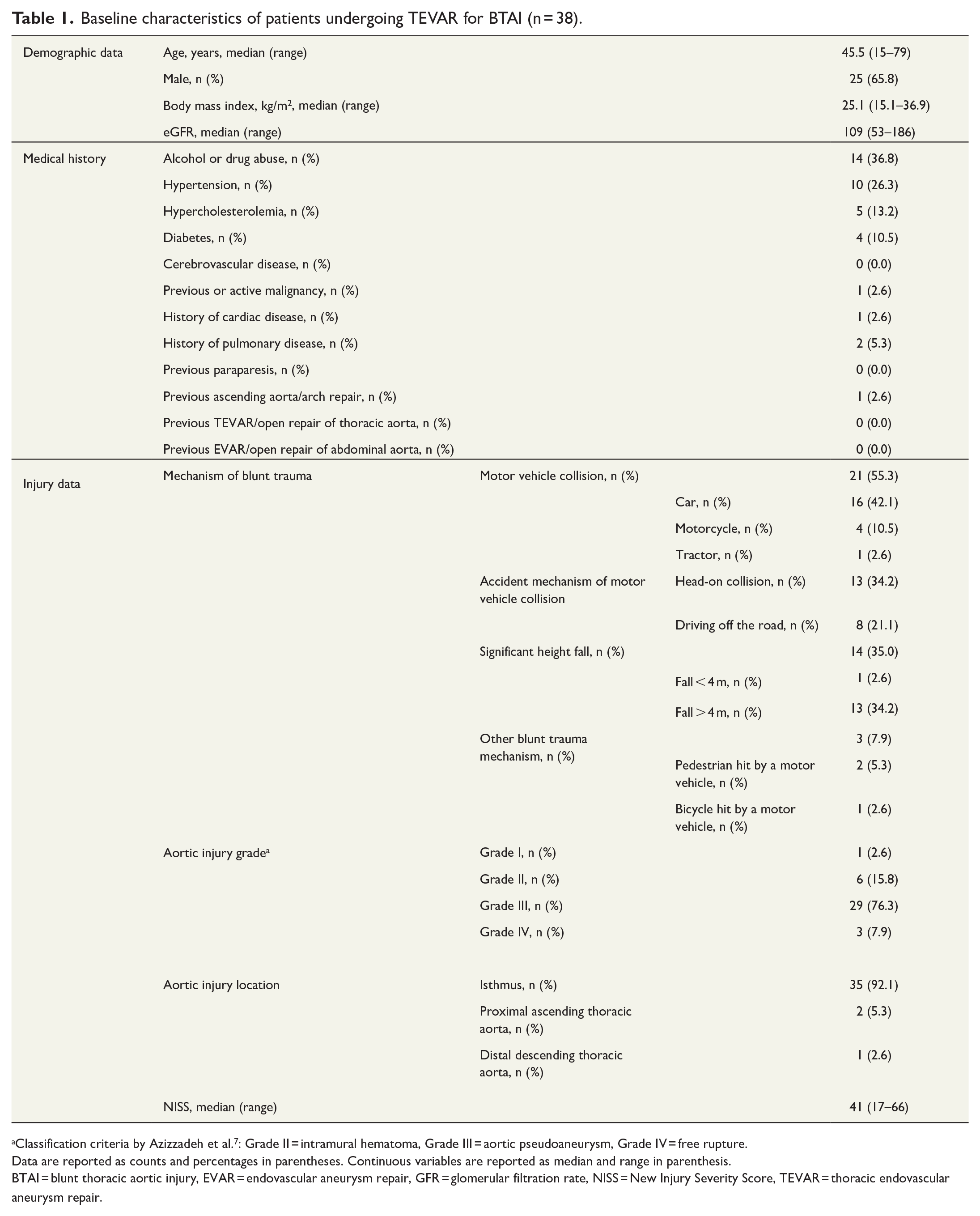
Classification criteria by Azizzadeh et al. 7 : Grade II = intramural hematoma, Grade III = aortic pseudoaneurysm, Grade IV = free rupture.
Data are reported as counts and percentages in parentheses. Continuous variables are reported as median and range in parenthesis.
BTAI = blunt thoracic aortic injury, EVAR = endovascular aneurysm repair, GFR = glomerular filtration rate, NISS = New Injury Severity Score, TEVAR = thoracic endovascular aneurysm repair.
Treatment methods and early outcomes
Table 2 summarizes operational and clinical utility data. The majority of TEVARs were performed under general anesthesia (n = 36; 95%), and most stent-graft systems were deployed urgently (87%), which was defined as TEVAR within 24 h. Among the three types of stent-grafts used (Table 2), Gore cTAG (W.L. Gore and Associates, Flagstaff, AZ) was the most prevalent (n = 26). Stent-grafts were most often deployed in proximal landing Zones 2 (58%) and 3 (34%). The median length and diameter of the stent-graft were 100 (range 100–200) and 28 (range 21–40) mm (Table 2).
Operative and clinical utility data of patients undergoing TEVAR for BTAI (n = 38).
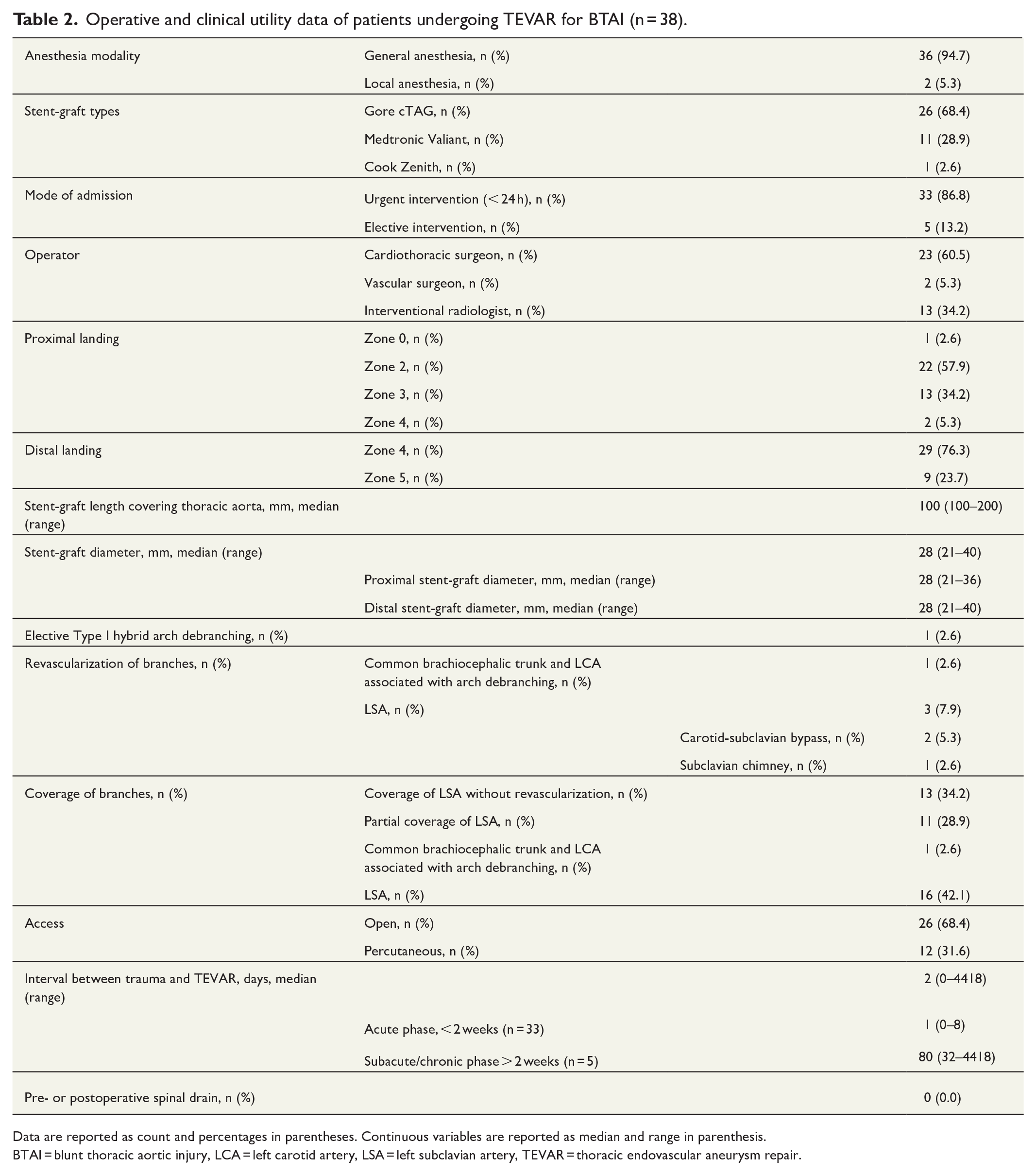
Data are reported as count and percentages in parentheses. Continuous variables are reported as median and range in parenthesis.
BTAI = blunt thoracic aortic injury, LCA = left carotid artery, LSA = left subclavian artery, TEVAR = thoracic endovascular aneurysm repair.
In total, 16 patients (42%) required LSA coverage, of whom 3 required revascularization (19%). LSA revascularization in 2/3 patients was due to dominant left vertebral arteries (Fig. 2). Pre-emptive carotid-subclavian (CS) bypass was performed in two patients and chimney bypass in the third. Type II endoleak was encountered in one patient (3%) secondary to LSA coverage and it was treated with LSA embolization.
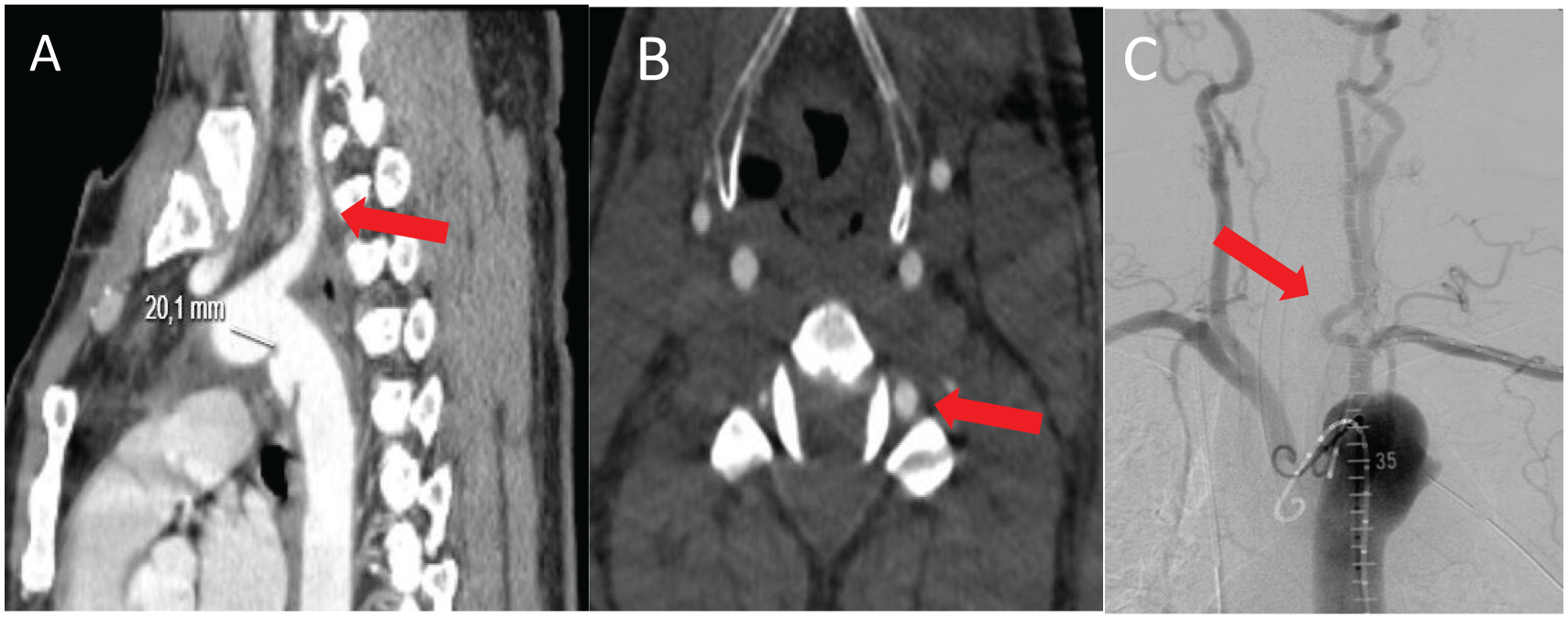
Images of patients (n = 2) undergoing LSA revascularization. (A) Pre-operative CTA scan of the thoracic aorta of patient with CS-bypass, displaying a dominant left vertebral artery (marked with red arrow). (B) Pre-operative axial CTA scan of the same patient (A). (C) Aortography image of patient undergoing chimney stenting. Dominance of left vertebral artery is marked with red arrow (A to C).
Two patients presented with proximal aortic injury. The first patient presented with a small intimal tear proximal to the innominate artery and another intimal tear at the isthmus. The patient underwent a multiple-step procedure: elective type I hybrid arch debranching with reconstruction of the brachiocephalic trunk and left carotid artery with a Dacron Y-prosthesis, followed by CS-bypass and TEVAR from Ishimaru Zone 0 2 days later.
The second patient had similarly Grade I injury proximally to the innominate artery, and a BTAI grade II at the isthmus. The intimal tear resolved with close monitoring, whereas the intramural hematoma necessitated TEVAR.
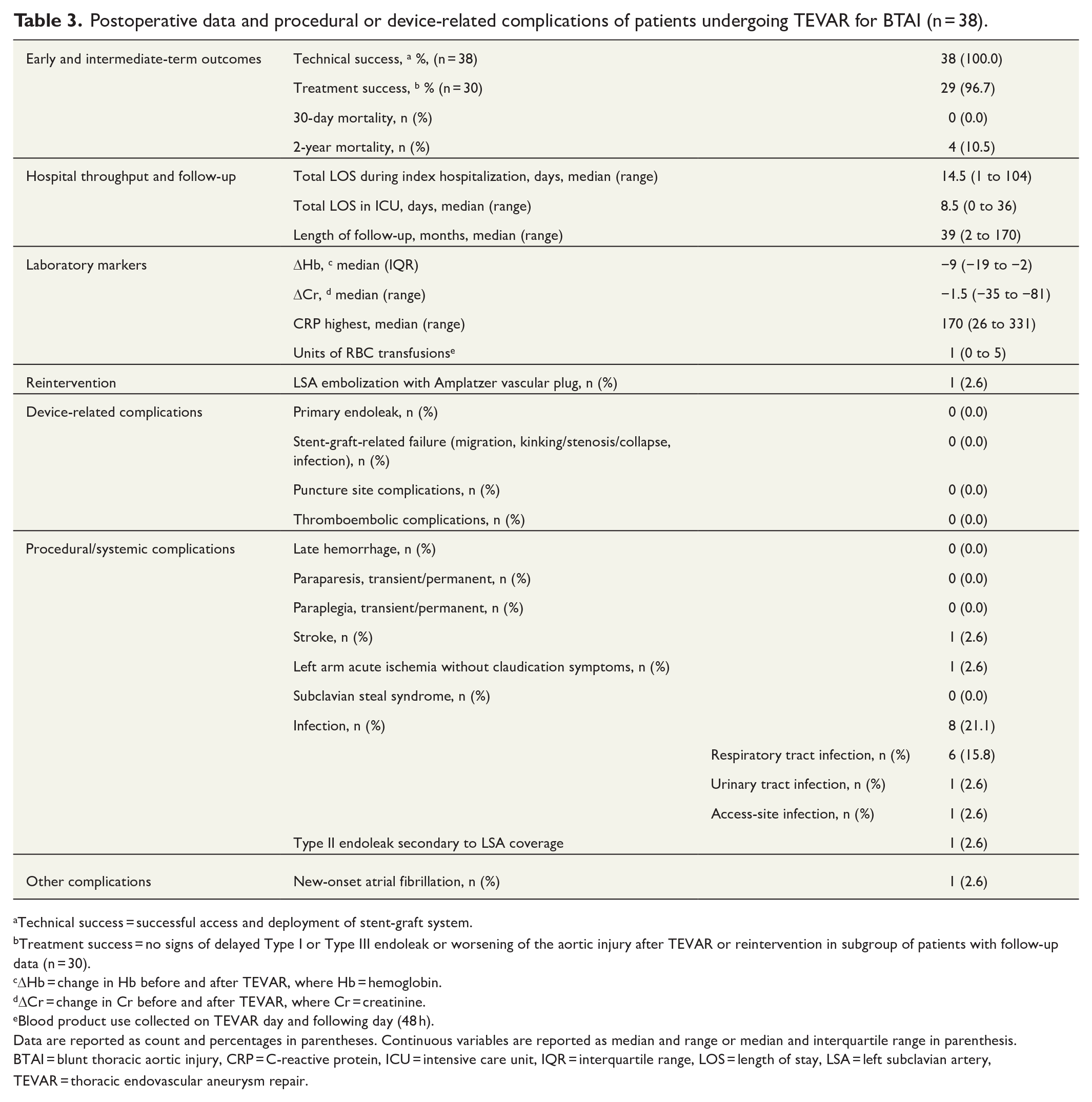
Postoperative data are in Table 3. Technical success was 100.0% in the study cohort. The median length of stay in the intensive care unit (ICU) was 8.5 (range, 0–36) days and the median length of stay of the index hospitalization was 14.5 (range, 1–104) days.
Postoperative data and procedural or device-related complications of patients undergoing TEVAR for BTAI (n = 38).
Technical success = successful access and deployment of stent-graft system.
Treatment success = no signs of delayed Type I or Type III endoleak or worsening of the aortic injury after TEVAR or reintervention in subgroup of patients with follow-up data (n = 30).
ΔHb = change in Hb before and after TEVAR, where Hb = hemoglobin.
ΔCr = change in Cr before and after TEVAR, where Cr = creatinine.
Blood product use collected on TEVAR day and following day (48 h).
Data are reported as count and percentages in parentheses. Continuous variables are reported as median and range or median and interquartile range in parenthesis.
BTAI = blunt thoracic aortic injury, CRP = C-reactive protein, ICU = intensive care unit, IQR = interquartile range, LOS = length of stay, LSA = left subclavian artery, TEVAR = thoracic endovascular aneurysm repair.
The median follow-up period was 39 (range, 2–170) months. After discharge, follow-up with CT imaging was incomplete in eight patients (21%). The treatment success was 97% (29/31) in the subgroup (n = 30) with CTA follow-up data.
No patients died within 30 days of TEVAR and the 2-year all-cause mortality rate was 11% (4/38) (Table 3). In Fig. 3, the Kaplan–Meier curve shows overall survival for the study cohort. At time points of 2.5, 3, 6.5, and 8 months, the respective means ± the standard errors of cumulative proportion of survival were 97 ± 3%, 94 ± 4%, 90 ± 5%, and 86 ± 7%, respectively. Supplementary Table 3 shows the results of Cox regression analysis. The significant risk factors associated with all-cause 2-year mortality were age at time of operation (hazard ratio (HR) = 1.240; 95% confidence interval (CI) = 1.014–1.476, p = 0.016) and LSA coverage without revascularization (HR = 41.651; 95% CI = 1.035–1676.265, p = 0.048).
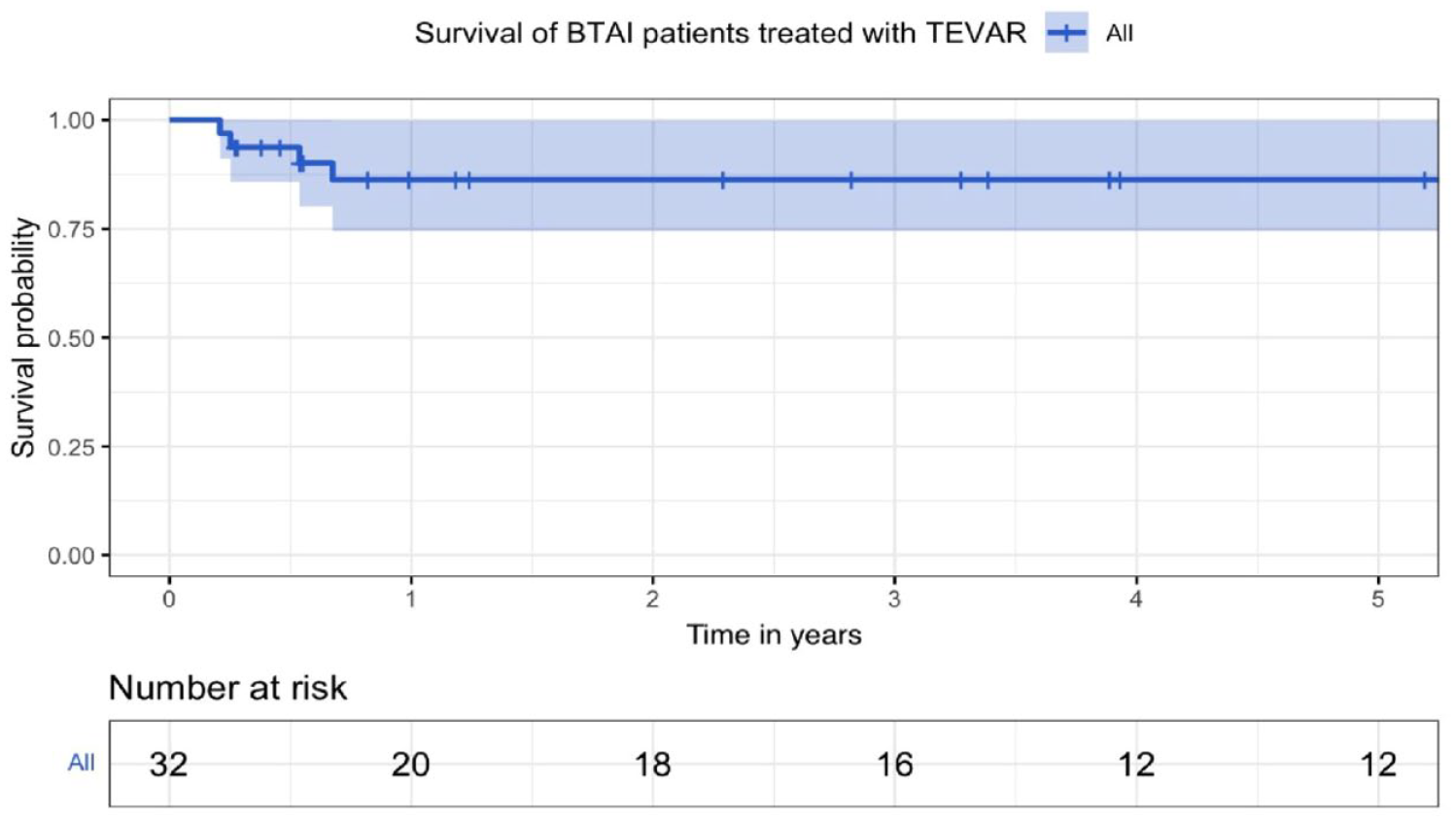
Kaplan–Meier survival estimates of BTAI patients treated with TEVAR in long-term follow-up.
Procedural and device-related complications
Procedure- and device-related complications of TEVAR are summarized in Table 3. Spinal cord ischemia and subsequent paraparesis/paraplegia were not observed (0%). Clinical follow-up revealed no late hemorrhage, arm claudication, or subclavian steal syndrome (0%). Stroke occurred in one patient (3%). After LSA coverage without revascularization, this patient developed nondisabling posterior brain infarction due to hypoplastic posterior communicating arteries and an incomplete Circle of Willis (Fig. 4).
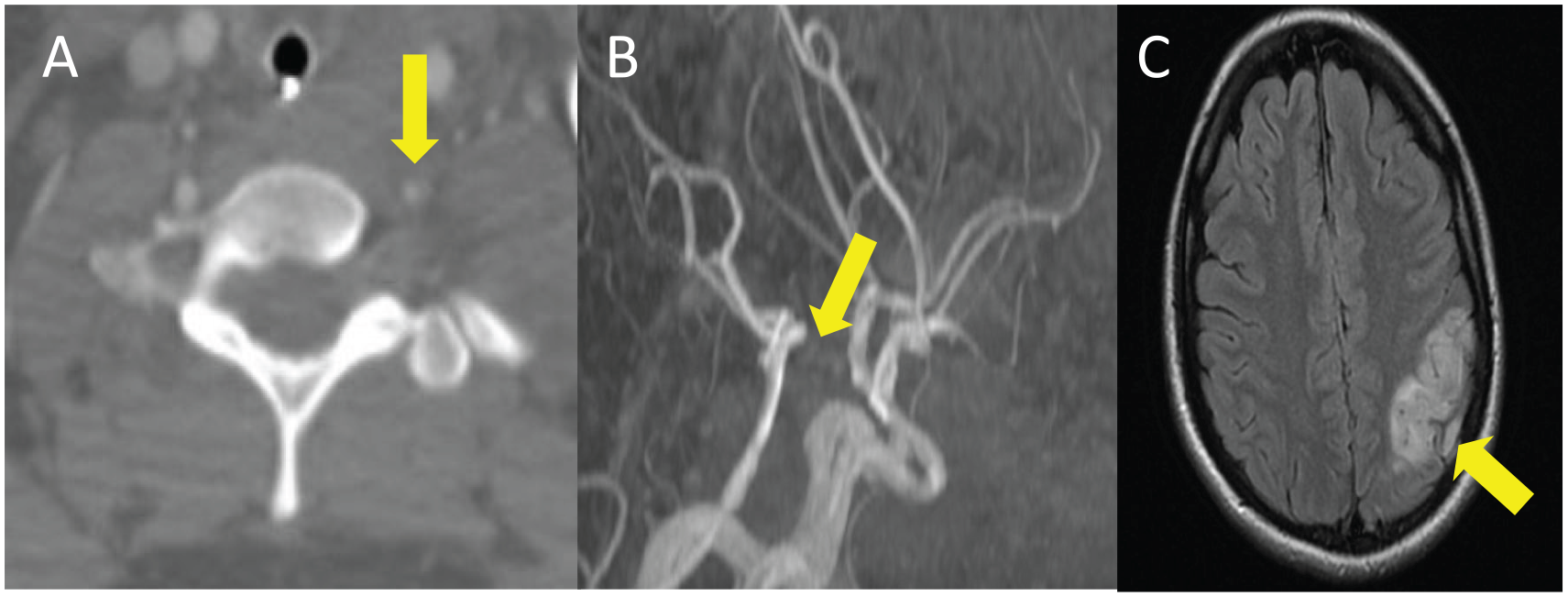
Images of a patient undergoing LSA coverage without revascularization and developing postoperative ischemic stroke. (A) Non-dominant left vertebral artery (marked with yellow arrow) as seen on axial CTA scan. (B) Circle of Willis is not patent due to absent/hypoplastic posterior communicating arteries (marked with yellow arrow) as seen on CTA scan in sagittal plane. (C) Postoperative posterior brain infarction (marked with yellow arrow) as seen on MRI scan.
Following coverage of LSA and vertebral artery origins due to unfavorable vascular anatomy, one patient (3%) developed asymptomatic arm ischemia, which manifested itself as nonpalpable distal pulses and prominent coldness. Claudication was not observed. Doppler ultrasound showed flow in the brachial artery, possibly due to collaterals, but not in ulnar and radial arteries. Arm ischemia resolved in a few days without additional intervention.
Among postoperative systemic complications, infection accounted for 21%, with pneumonia accounting for 16%. No primary endoleaks at procedure completion or stent-graft failures were detected (0%).
Reinterventions
A technical success rate of 100% was achieved, with no conversion to OAR or re-endografting. However, the aforementioned case of detected type II endoleak in follow-up required embolization of the LSA with a 12-mm Amplatzer vascular plug within 89 days of the primary procedure.
Discussion
A summary of our main findings is as follows: (1) Endovascular BTAI repair is associated with low mortality in the first 30 days and 2 years and excellent technical success; (2) neurological complications were low with and without LSA coverage, and (3) only a small percentage (3%) of patients were reintervened on.
TEVAR was approved by the Food and Drug Administration (FDA) in 2005 for thoracic aortic disease, 8 and for traumatic aortic injuries in 2012. There has been a shift from OAR to TEVAR due to excellent clinical results, and hence, the 2011 guidelines from SVS suggest that TEVAR should be preferred over conventional OAR or nonoperative management in BTAI. 9 In a previous meta-analysis based on 17 retrospective studies 9 and additional comparative studies,10 –13 the TEVAR group had significantly lower 30-day mortality, ranging from 2.1% to 8%. Our 30-day mortality rate is 0.0%, which is in line with the work by Okadome et al. 14
Although our study cohort’s median NISS (41, range 17–66) indicates severe injury, our 10.5% 2-year mortality rate is low compared to Kuorikoski et al.’s 15 22% 1-year mortality rate for blunt trauma in a large trauma population. This difference may be explained by the fact that in their study, some died prior to TEVAR due to other injuries or stent-grafting was not executed due to poor overall prognosis. In addition, trauma increased mortality up to 12 years, 15 so short-term mortality is debatable as an outcome measure.
CTA has become the primary diagnostic modality for BTAI. Our treatment regimen depends on injury grade. 5 Treatment of concomitant life-threatening injuries and initial hypertension management are prioritized to minimize rupture risk (Grade IV). We use urgent TEVAR (within 24 h from injury) for Grade II–IV injuries, while nonoperative management is considered for Grades I and II. Aortic imaging, close monitoring, and control of systolic blood pressure with a target of < 100 mmHg are recommended because injury progression is possible. Grade III aortic injuries receive TEVAR expediently, usually within 48 h, depending on the patient’s prognosis.
No patient had SCI or paraplegia, and none underwent cerebrospinal fluid (CSF) draining. It compares favorably with Hellgren et al.’s 16 0.0% paraplegia and paraparesis rates. However, Abdou et al. 17 found higher rates of 1.9% SCI and 0.2% paraplegia. It is noteworthy that these adverse outcomes may be attributed more to CSF drainage than TEVAR itself if performed on the same day. They used selective spinal drains in 1.6% of their patients. Recent research on complex aneurysms 18 suggests that prophylactic spinal drains can reduce the risk of SCI in TEVAR patients, but complications such as SCI following CSF drainage cannot be excluded. Extending results from this study to BTAI management requires caution given the differences in patient populations and clinical contexts.
Fossaceca et al. 19 reported a technical success rate of 100.0% using TEVAR, and we obtained similar results. Access and delivery of TEVAR can be hindered by tortuous arterial anatomy, reduced vessel caliber, and circumferential arterial wall calcification. Fernandez et al. 20 reported ruptured iliac arteries in 8.9% of TEVAR patients, 66% of whom had heavy arterial calcification. Comparatively, arterial calcification (n = 1) or iliac tortuosity (n = 2) was rare in our study cohort (Supplementary Table 2). TEVAR is usually performed in an urgent setting, but accurate pre-procedure planning is essential.
A sufficient landing zone may require LSA coverage. 2011 SVS guidelines 5 recommend selective revascularization for managing the LSA, but controversy persists. LSA coverage without revascularization does not increase neurologic deficits in BTAI-TEVAR patients, according to the study by Kruger et al. 21 However, this contradicts the findings of several literary reports.22 –24 Our two patients experiencing symptoms after LSA coverage without revascularization did not have left vertebral artery dominancy and did not undergo pre-emptive LSA revascularization. Detailed investigation of the Circle of Willis may have prevented the nondisabling stroke. As long as dominant left vertebral artery and incomplete Circle of Willis are excluded during TEVAR for BTAI, coverage of the LSA without revascularization is safe. LSA vascularization may be considered if arm claudication persists postoperatively.
Our revascularization guidelines for LSA are patient-tailored, considering various factors, such as long aortic segment coverage, a dominant left vertebral artery/occluded right vertebral artery, or patent left internal mammary artery for coronary artery bypass graft. As CTA includes supra-aortic vessel imaging, it influences decision-making. It is not routine to image cerebral vessels preoperatively. LSA revascularization is affected by concomitant neck injuries, cervical collar use, and surgeon preferences.
In this study, LSA coverage without revascularization seemed to be associated with increased risk of 2-year all-cause mortality. However, as the wide CI describes the uncertainty of the estimate, the correlation cannot be determined from these data. In fact, both of the two (2/13) who died within 2 years of LSA coverage without revascularization, had other causes of death: The first patient was diagnosed with rectal cancer a few months before passing away. The second patient died of diffuse axonal injury confirmed by a head CT scan. Thus, we do not believe LSA coverage without revascularization to negatively impact TEVAR outcomes.
A recommendation for early TEVAR within 24 h by SVS for BTAI is adhered to by 87% of our BTAI patients. TEVAR timing remains a complex issue, according to recent research. An early TEVAR is associated with higher short-term mortality,25 –27 but shorter ICU stays.25,26 Delayed TEVAR was linked to reduce in-hospital mortality, but higher acute kidney injury incidence. 26 The findings underline the need for further exploration of TEVAR timing in BTAI.
There is emerging interest in thoracic branch endografting (TBE), which is not yet available at our institution. BTAI patients, many of whom are young, may have smaller diameter proximal aortic and clustering supra-aortic branches, limiting TBE’s applicability. Only 12.5% of patients met anatomical criteria in one study, 28 and only 32% in another. 29 Enhanced endovascular repair technologies and refined device designs, combined with changes in inclusion criteria, may expand TBE’s use.
Several prior studies fall short of achieving our median follow-up period of 39 months. Reinterventions in follow-up are rare, as evidenced by a meta-analysis encompassing 11 studies 30 reporting a collective estimate of 1.2% reinterventions. One patient (2.6%) in our report required LSA embolization for type II endoleak, which was not detected at TEVAR completion but rather 89 days later. We stress the significance of long-term follow-up after TEVAR.
Limitations
Its retrospective design and lack of a control group constrain this study. In addition, single-center data limited sample size. Furthermore, patient age < 18 years was not exclusory in our study, with our youngest patient being 15 years old. The thoracic aorta of young patients is usually smaller, leading to oversizing complications and inadequate graft apposition. As a final note, this study only included BTAI patients who had undergone TEVAR, so it may not fully represent all BTAI patients. The reason for this may be that some patients died before undergoing TEVAR, precluding their inclusion.
Conclusion
We demonstrate that TEVAR is effective for treating BTAI and yields favorable outcomes in the short and intermediate term despite coverage of the LSA without revascularization. TEVAR had excellent technical success, low mortality, and infrequent complications. During follow-up, only a few reinterventions were performed. After excluding dominant left vertebral artery and incomplete Circle of Willis, we believe that coverage of LSA during TEVAR for BTAI is safe.
Supplemental Material
sj-docx-1-sjs-10.1177_14574969241255242 – Supplemental material for 16-year outcomes of blunt thoracic aortic injury treated with thoracic endovascular aortic repair: A single-institution experience
Supplemental material, sj-docx-1-sjs-10.1177_14574969241255242 for 16-year outcomes of blunt thoracic aortic injury treated with thoracic endovascular aortic repair: A single-institution experience by Tasnia Rahman, Lauri M. Halonen, Lauri Handolin, Tatu Juvonen, Mikko Jormalainen and Sebastian Dahlbacka in Scandinavian Journal of Surgery
Supplemental Material
sj-docx-2-sjs-10.1177_14574969241255242 – Supplemental material for 16-year outcomes of blunt thoracic aortic injury treated with thoracic endovascular aortic repair: A single-institution experience
Supplemental material, sj-docx-2-sjs-10.1177_14574969241255242 for 16-year outcomes of blunt thoracic aortic injury treated with thoracic endovascular aortic repair: A single-institution experience by Tasnia Rahman, Lauri M. Halonen, Lauri Handolin, Tatu Juvonen, Mikko Jormalainen and Sebastian Dahlbacka in Scandinavian Journal of Surgery
Supplemental Material
sj-docx-3-sjs-10.1177_14574969241255242 – Supplemental material for 16-year outcomes of blunt thoracic aortic injury treated with thoracic endovascular aortic repair: A single-institution experience
Supplemental material, sj-docx-3-sjs-10.1177_14574969241255242 for 16-year outcomes of blunt thoracic aortic injury treated with thoracic endovascular aortic repair: A single-institution experience by Tasnia Rahman, Lauri M. Halonen, Lauri Handolin, Tatu Juvonen, Mikko Jormalainen and Sebastian Dahlbacka in Scandinavian Journal of Surgery
Supplemental Material
sj-docx-4-sjs-10.1177_14574969241255242 – Supplemental material for 16-year outcomes of blunt thoracic aortic injury treated with thoracic endovascular aortic repair: A single-institution experience
Supplemental material, sj-docx-4-sjs-10.1177_14574969241255242 for 16-year outcomes of blunt thoracic aortic injury treated with thoracic endovascular aortic repair: A single-institution experience by Tasnia Rahman, Lauri M. Halonen, Lauri Handolin, Tatu Juvonen, Mikko Jormalainen and Sebastian Dahlbacka in Scandinavian Journal of Surgery
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: S.D. received grants from the Dorothea Olivia, Karl Walter, and Jarl Walter Perklén Foundation and the Medical Society of Finland (Finska Läkaresällskapet).
Consent to publication
The Institutional Review Board approved conduction of this study and waived informed patient consent requirements.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
