Abstract
Background:
One of the most common complications of total thyroidectomy is post-operative transient or persistent hypoparathyroidism that can cause symptomatic hypocalcaemia. To prevent this complication, shorten the period of hospitalization and reduce morbidity, routine supplementation of oral vitamin D and calcium has been suggested. This systematic review and meta-analysis aims to critically assess the association between pre-operative calcitriol supplementation and post-operative hypocalcaemia.
Methods:
Randomized controlled trial studies were identified by searching PubMed, Scopus, and Google Scholar databases up to 30 March 2023. Screening of titles, abstracts, and full texts of articles were performed, and data were extracted for a meta-analysis.
Results:
This meta-analysis includes data from nine randomized controlled trials with a total of 1259 patients but with significant heterogeneity. The results demonstrate that calcium levels were higher in patients who had pre-operative calcitriol supplementation, with a weighted mean difference (WMD) 0.18 (95% confidence interval (CI) = 0.00, 0.37). Pre-operative calcitriol supplementation did not lead to significant changes in parathyroid hormone (PTH) levels, with WMD −0.49 (95% CI: −1.91, 0.94).
Conclusion:
Pre-operative calcitriol supplementation leads to higher calcium levels, but the high heterogeneity of the included studies (79% to 98.7%) could affect the results.
Context and Relevance
This systematic review meta-analysis aims to critically assess the association between pre-operative calcitriol supplementation and post-operative hypocalcaemia after total thyroidectomy. Briefly, 792 articles were screened after conducting a primary search in various databases. After exclusions, nine articles were included in the final analysis encompassing a total of 1259 patients. This meta-analysis shows that although calcitriol consumption before the procedure is associated with remarkable higher calcium level, it does not change the parathyroid hormone level significantly.
Introduction
Hypocalcaemia due to hypoparathyroidism caused by injury or devascularization of parathyroid glands is one of the most frequent complications of total thyroidectomy. The incidences of transient and permanent hypocalcaemia are reported 6.9% to 75% and 0% to 8%, respectively.1,2 Hypocalcaemia usually presents 24–48 h after surgery, and it is mild and transient (i.e. lasting less than 6 months) in most cases. It may be asymptomatic when serum calcium levels are mildly decreased or symptomatic with patients showing Trousseau’s and Chvostek’s signs, as well as having muscle spasms. 3
Despite all attempts to establish risk factors for development of hypocalcaemia and hypoparathyroidism after thyroidectomy,4 –13 it remains difficult to predict which patients will need calcium and vitamin D supplementation after surgery. Oral vitamin D and calcium supplementation have been suggested to reduce the risk of developing symptomatic hypocalcaemia and to accelerate hospital discharge after thyroid surgery. Routine post-operative treatment with oral calcium and vitamin D could moreover be a practical approach to decrease the risk of severe hypocalcaemia, since symptomatic hypocalcaemia typically occurs 24 h to several days after operation. Furthermore, this would reduce the need for multiple blood sampling to monitor serum calcium levels and costs of lengthy hospitalization.14 –16
Several authors thus suggest early discharge and routine administration of oral calcium and/or vitamin D supplementations for all patients irrespective of parathyroid gland function. Such an approach, however, results in overtreatment of many patients and could lead to a delay in diagnosing and proper treatment of hypoparathyroidism.17,18
The aim of the present systematic review and meta-analysis was to evaluate the impact of routine oral administration of calcitriol on hypocalcaemia after total thyroidectomy.
Methods
This systematic review investigated the impact of oral administration of calcitriol to prevent symptomatic hypocalcaemia after total thyroidectomy. A protocol was submitted to OSF (https://osf.io/btprz/links) using a checklist-based approach for the study design, screening, and data selection. The PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-analysis) was followed.
Search strategy
A general search was performed in March 2023 using Google Scholar, Scopus, and PubMed (Medline) databases. The search was based on the title, abstract, and using the advanced search strategy with suitable operators and tags for each database (Supplemental Table S1). Two researchers separately went through the titles and abstracts of the selected studies after removal of duplicates. Studies suitable for the project and meeting the inclusion criteria based on full text were selected.
Inclusion and exclusion criteria
Inclusion criteria were those randomized controlled trials (RCTs) analyzing the impact of oral calcitriol use on preventing symptomatic hypocalcaemia after total thyroidectomy. Studies using other forms of vitamin D were excluded as were retrospective studies, reviews, case reports, case series, letters to the editor, and posters.
Quality assessment
To assess the quality of studies, the Cochrane risk of bias assessment tool was used. Full text of the studies were obtained and checked by two persons separately to see if they fully met the inclusion criteria and quality. Any disagreement was resolved by consensus. A data extraction list was generated, and the data were extracted separately by two researchers.
Statistical analysis
Stata version 13.1 (Stata Corp, College Station, TX, USA) was used to conduct the meta-analysis. Results were reported as weighted mean difference (WMD) with a 95% confidence interval (CI), illustrated in a Forest plot. For evaluating heterogeneity among the entered studies, the I2 statistic was calculated and in the case of verified heterogeneity (I2 > 50%), a random effects model was applied. Furthermore, by sensitivity analyses, stability of results was assessed. Eventually, to investigate the potential publication bias, funnel plot symmetry plots and Egger’s regression analysis were used.
Result
Briefly, 792 articles were screened after conducting a primary exhaustive search in PubMed, Scopus, and Google scholar databases with duplicates initially removed. Out of these, 543 articles were subsequently removed based on title and/or abstract. This left 249 potentially relevant studies to be read in full text. Of those, 232 records were further eliminated as they did not meet the inclusion criteria. Finally, 17 RCT records remained and were assessed based on using the Cochrane risk of bias assessment tool. After elimination of eight articles due to poor quality and outcome interest, nine RCTs were used in the final analysis including 1259 patients (Figs 1 and 2).
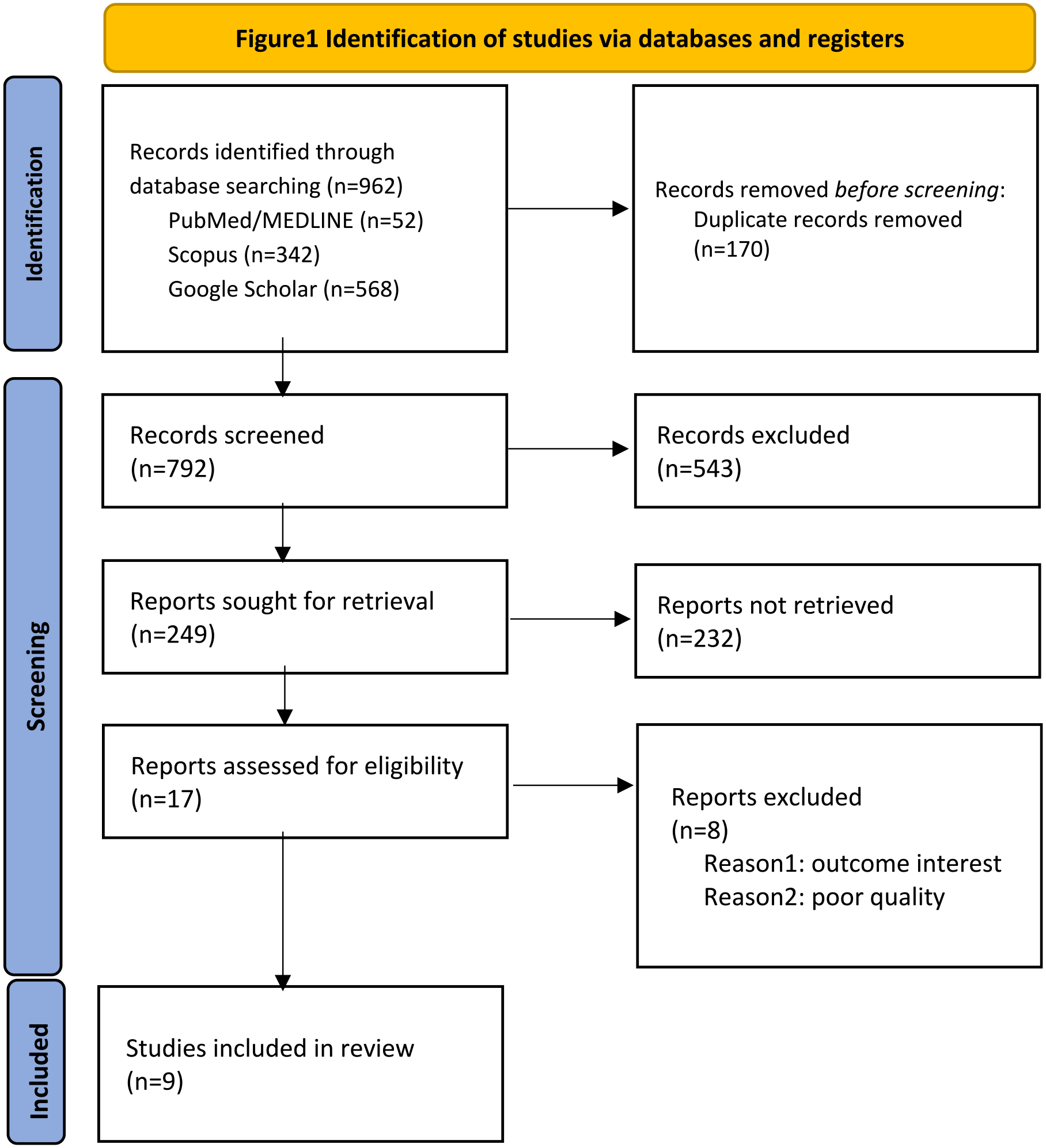
Identification of studies via databases and registers.
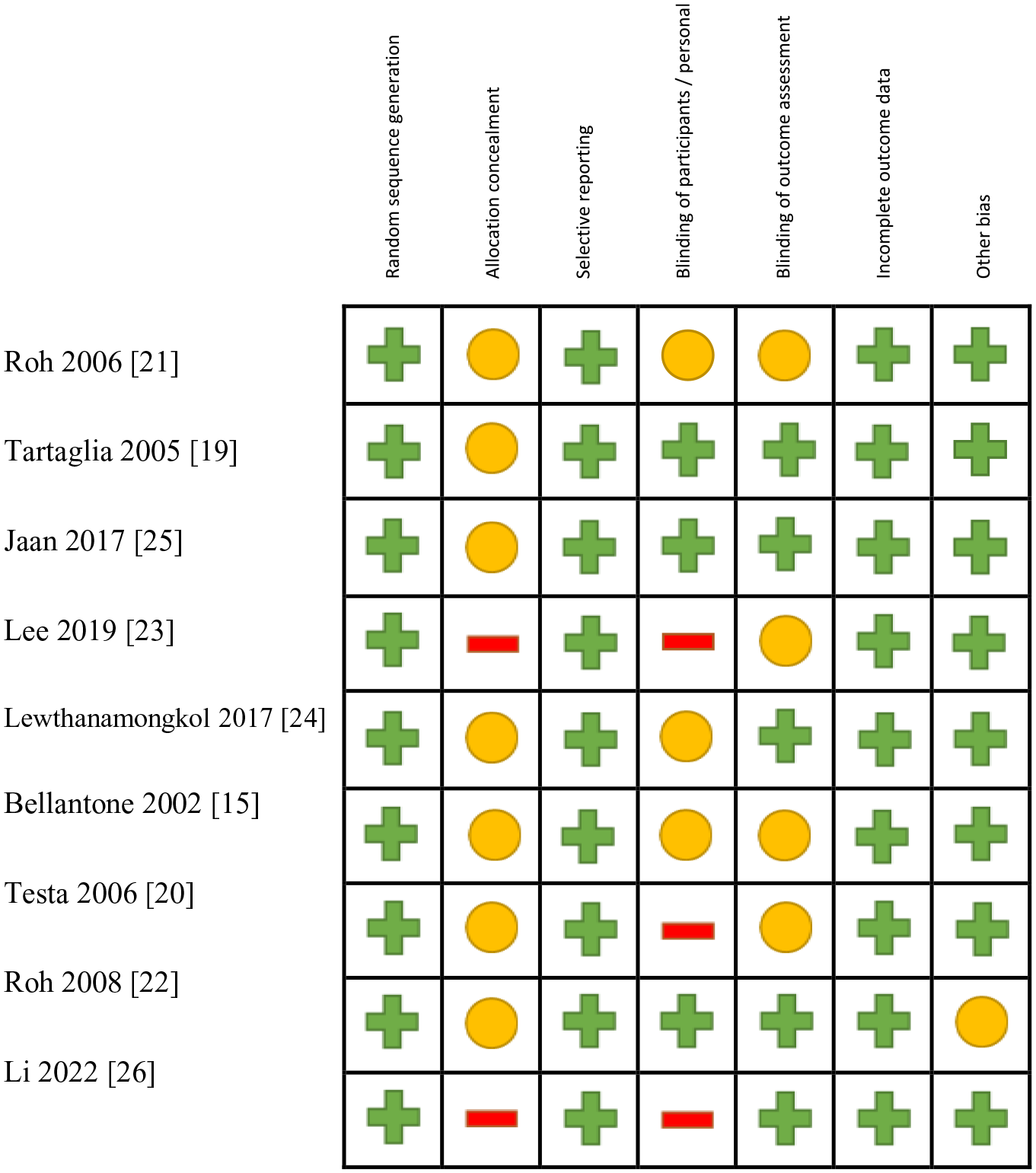
Risk of bias of the included studies.
The nine RCTs included were published from 2002 to 2022. Three from Italy,15,19,20 three from the Republic of Korea,21–23 one from Thailand,24 one from India,25 and one from China.26 There was a total of 1259 patients evaluated in these nine trials. The mean age was 36.43 ± 11.54 to 60.6 ± 8.6 among intervention and control groups, respectively. The majority of included patients were female (66% to 87%). The mean follow-up varied from 7 days to 36 months. Data related to calcium levels were included in eight of the included studies15,19 –23,25,26 and data for PTH levels from six studies15,20,22 –24,26 (Table 1).
Characteristics of included studies.
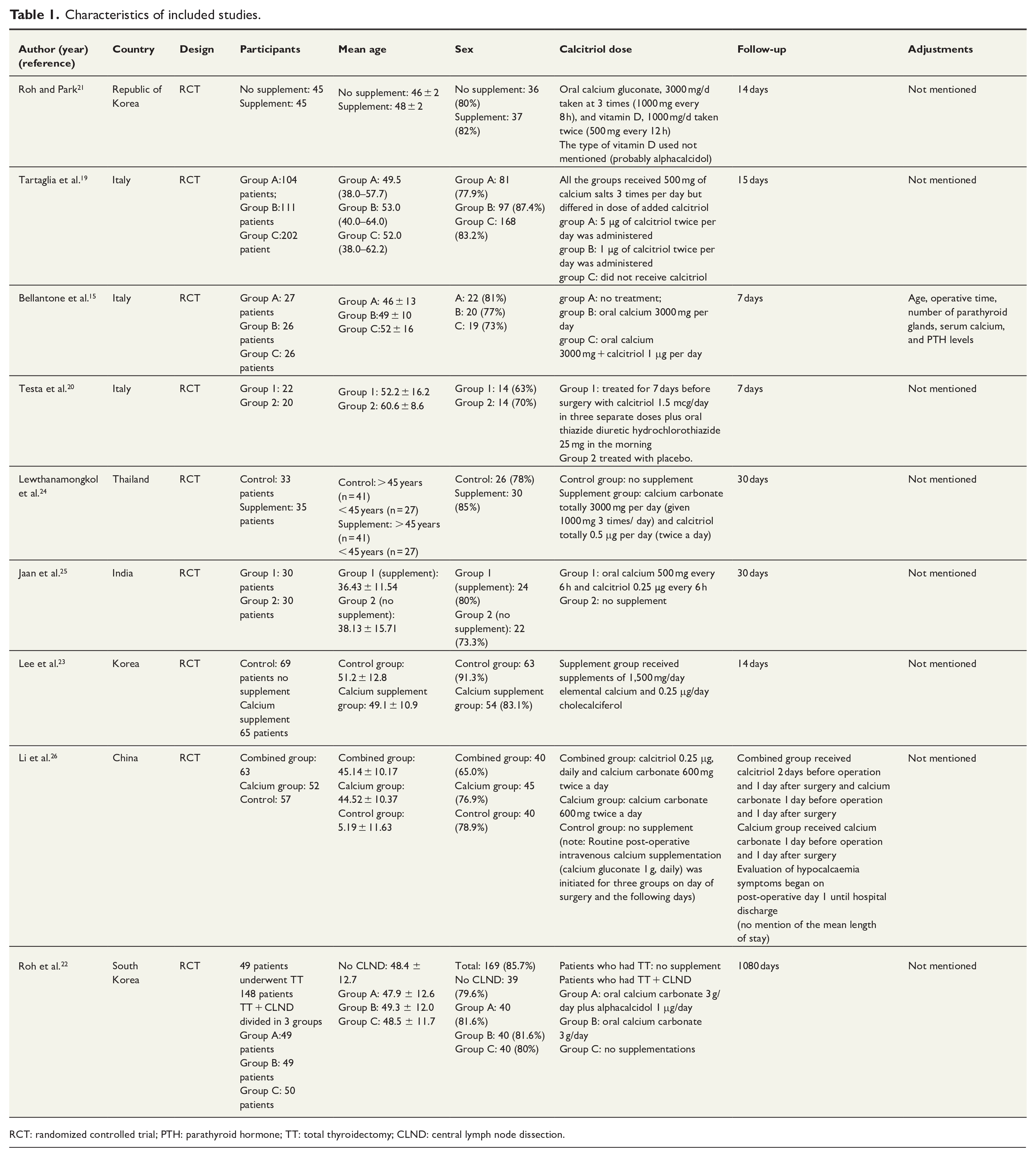
RCT: randomized controlled trial; PTH: parathyroid hormone; TT: total thyroidectomy; CLND: central lymph node dissection.
The group that received calcitriol had significantly higher levels of serum calcium with a WMD (95% CI): 0.18 (−0.00, 0.37), I2 = 98.7% (Fig. 3). In terms of PTH, no significant relation between calcitriol administration and pre- and post-operatively PTH levels was observed (WMD (95% CI): 0.49 (–1.91, 0.94), I2 = 79.1% (Fig. 4)). The funnel plot indicates lack of symmetry for both Ca and PTH levels, thus indicating a high level of publication bias among the included studies (Fig. 5).
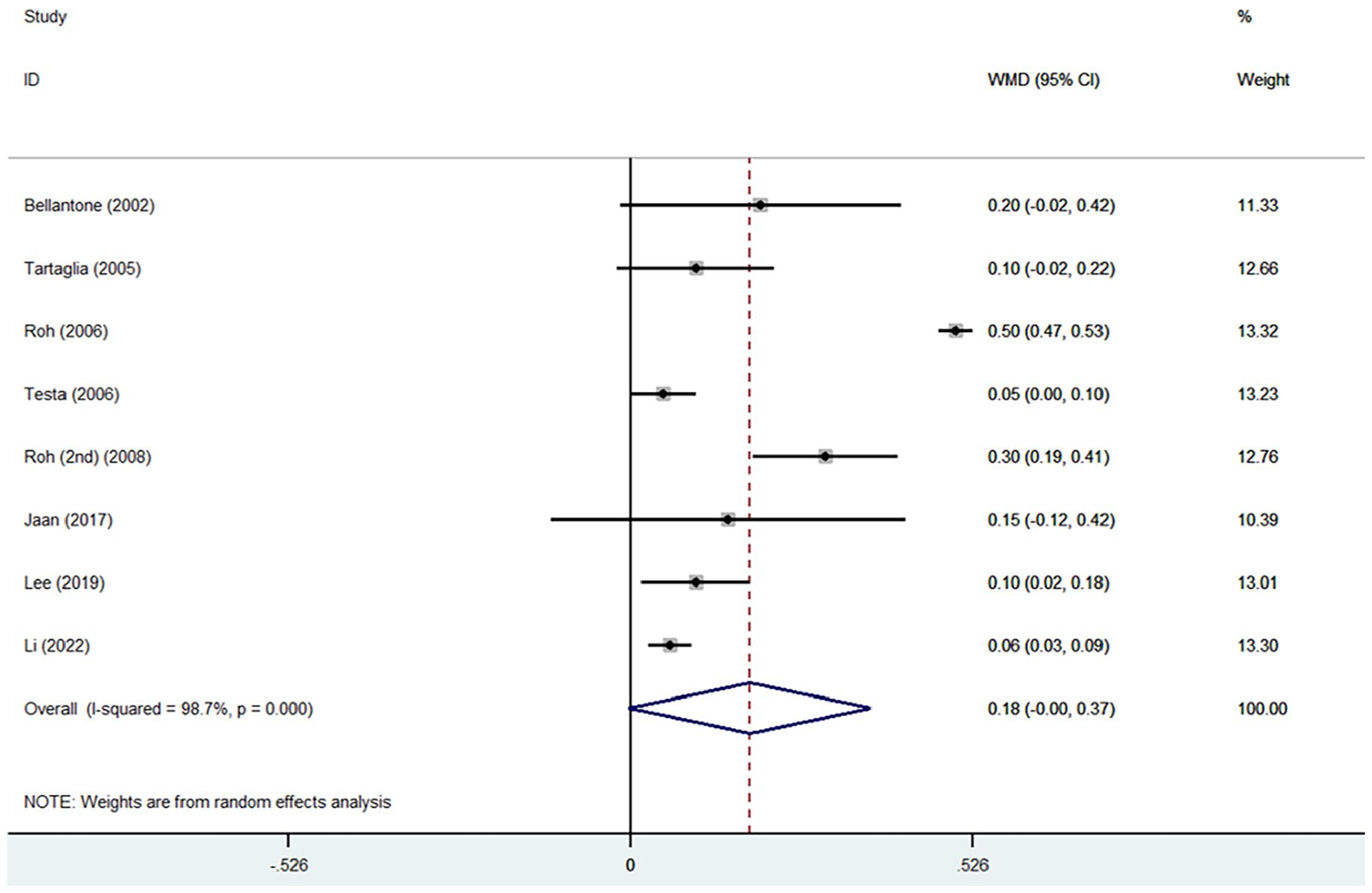
Forest plot of the studies on the impact of the calcitriol on calcium levels pre- and post-operatively.
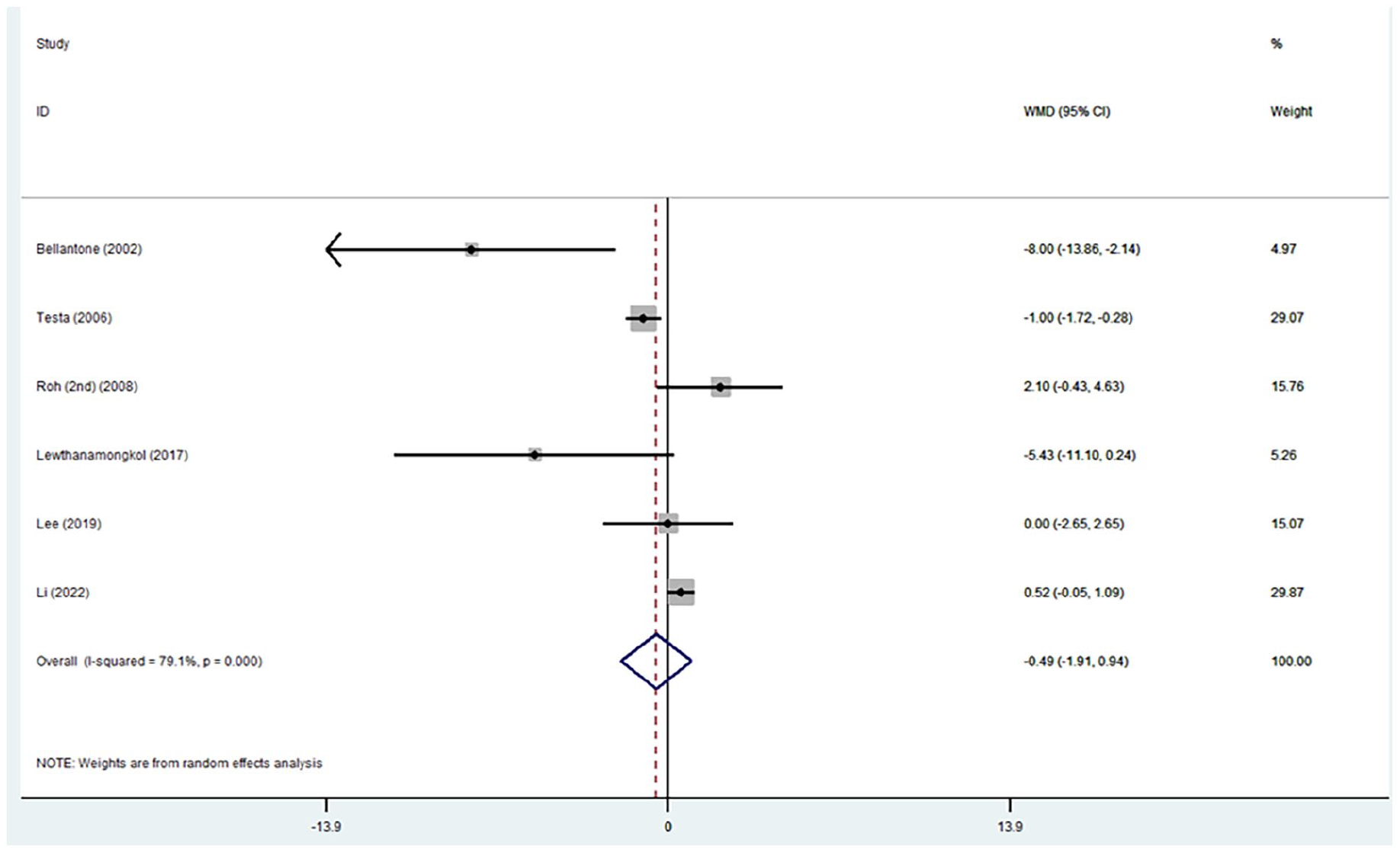
Forest plot of the studies on the impact of the calcitriol on PTH levels pre- and post-operatively.
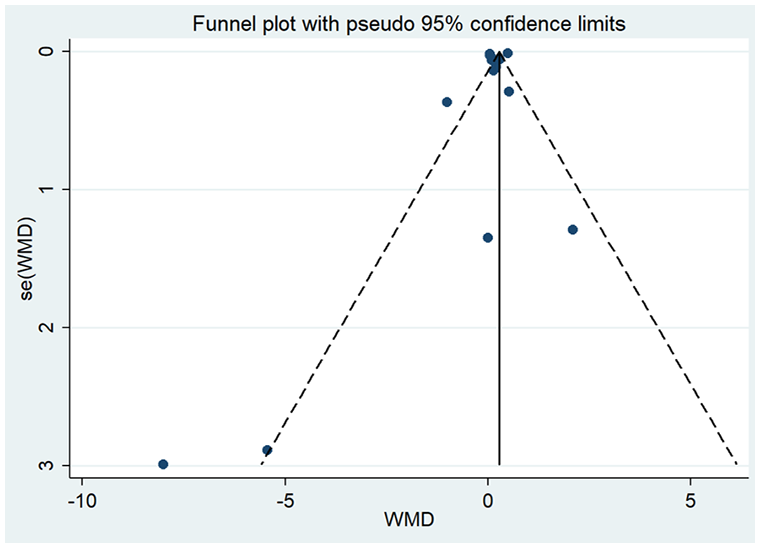
Funnel plot of the studies analyzed in the study for both Ca and PTH level after calcitriol supplementation.
A sensitivity analysis was performed and none of the studies were excluded, and no irrelevant data were found in the studied articles. A meta-regression analysis was conducted, and there was not a significant linear association between dose and response (p-value Ca: 0.000 and p-value PTH: 0.000).
Discussion
Total thyroidectomy is performed for both benign and malignant thyroid disease. This procedure has evolved from a hazardous to a safe one during the last century.27 Several complications, however, remain common, even when performed by experienced surgeons.28,29 Post-operative hypocalcaemia is a frequent complication occurring among 6.9% to 49% of patients within the first days after surgery.30 The exact pathogenesis is not clearly defined and is assumed to be multifactorial, although mostly attributed to transient hypoparathyroidism.31 One post-operative recommendation is to start oral calcium supplementation. This should begin with an initial dose adjustment and ultimately be tapered off based on the presence of hypocalcemic symptoms or serum Ca levels. In addition, calcitriol treatment can be started pre-operatively if serum PTH levels are low and finally, tapered off after surgery based on the PTH and/or Ca levels.15 Since the effect of calcitriol administration on the calcium and PTH levels has not been systematically investigated, this systematic review and meta-analysis was conducted to clarify its impact.
Overall, a total of nine RCTs (including 1259 patients) were included. Out of these eight studies (including 911 patients) were used to establish the correlation between calcitriol prescription and pre- and post-operative Ca level changes. The analysis showed a significant relation between calcitriol supplementation and Ca level changes. Moreover, six (encompassing 516 patients) were assessed to establish the association between calcitriol supplementation and pre- and post-operative PTH level changes. Calcitriol administration did not affect the serum PTH level significantly. By performing meta-regression test, no linear association found between the dose of calcitriol and clinical response.
Biochemical predictors of hypocalcaemia after thyroidectomy as defined in previous studies were perioperative PTH level, pre-operative vitamin D, and post-operative changes in Ca levels. In addition, female sex, Graves’ thyrotoxicosis, parathyroid gland auto-transplantation, and excision of parathyroid glands are also related to post-operative hypocalcaemia.32–34 Deficiency in the serum level of vitamin D has been introduced as an independent factor in predicting post-operative transient hypocalcaemia.35,36 A review with greater focus on pre-operative vitamin D deficiency, marked out the serum level of 25-hydroxyvitamin D equal or less than 20 ng/mL as a predictor of transient hypoparathyroidism, and the level equal or lower than 10 ng/mL as a predictor of permanent hypoparathyroidism.37 Moreover, certain group of patients are more likely to have vitamin D deficiency, including the patients with darker skin, higher age (possibly due to weakened skin 7-dihydrocholesterol, reduced renal 1α-hydroxylase function, and decreased calcium absorption38,39), obesity, and malabsorption diseases.38 It is also demonstrated that female sex is associated with lower vitamin D levels.40,41
This study is consistent with meta-analysis performed by Xing et al. that compared the effect of calcium tablets alone or with vitamin D in reducing post-operative hypocalcaemia. They found that a regimen containing vitamin D was more potent in lowering the need for intravenous supplementation, but both of regimens were effective in preventing post-operative hypocalcaemia. They also advised to reserve combination therapy with vitamin D for patients more inclined to show hypocalcaemia, in order to lower the rate of hypocalcemic incidents and readmissions.42
Multiple studies have shown the impact of post-thyroidectomy PTH level as a predictor of hypocalcaemia with a high sensitivity and specificity.43–45 In a pooled analysis by Noordzij et al.,46 a higher than 65% decrease in the serum PTH 6 h post-operatively was established to predict hypocalcaemia with a sensitivity >96% and specificity of 91%. It is, however, not cost beneficial to measure serial serum PTH levels after surgery; thus, many surgeons prefer to prescribe routine calcium and vitamin D after operations. It is assumed that these supplementations have impact on PTH levels.47 There is, however, no confirmed role of calcium and vitamin D supplementation. Previous studies have demonstrated that administration of calcitriol in contrast to calcium supplementations could actually decrease intact parathyroid hormone (iPTH) levels.40,48,49 Surprisingly, the present analysis showed that calcitriol administration did not change serum PTH levels significantly.
While the outcomes of this article may be anticipated, it conveys a vital message that, in general, it is advisable to integrate calcium and vitamin D supplements into daily practice at appropriate doses to prevent hypocalcaemia after thyroidectomy. The treating physician can determine the most suitable and effective medication strategy by considering the patient medical history, clinical symptoms of hypocalcaemia, and serum levels of calcium, vitamin D, and PTH before and after surgery.
There were several limitations to this study. First, due to the high rate of publication bias and the great heterogeneity in both Ca and PTH analysis in the included studies, a random-effect model was used to determine the overall effect. Second, the overall number of patients was rather small. Consequently, the power of this study to confirm previous results is limited. Third, due to variety of regimens used in each study, an exact dose and duration of treatment with calcitriol could not be established. Further research is recommended to explain the source of heterogeneity and to increase the population size. Also, more studies should be conducted to compare the impact of the different calcitriol regimens in duration, dosage, and whether it is better to prescribe pre-operatively and/or post-operatively. The calcitriol doses in the included articles varied from 0.25 µg per day to 5 µg twice per day. The optimal dose, timing, and duration of calcitriol administration before and after thyroidectomy to prevent hypocalcaemia and maintain normal PTH levels thus remain a matter of debate. Calcitriol resistance mechanisms may furthermore impair the suppressive effect of calcitriol on PTH levels, and other vitamin D analogues, such as paricalcitol, may have a superior PTH-lowering effect. The long-term effects of calcitriol administration on bone health, kidney function, and cardiovascular risk in thyroidectomy patients furthermore need to be evaluated in future studies.
Supplemental Material
sj-docx-1-sjs-10.1177_14574969241251899 – Supplemental material for Impact of oral administration of calcitriol to prevent symptomatic hypocalcemia after total thyroidectomy: Systematic review and meta-analysis
Supplemental material, sj-docx-1-sjs-10.1177_14574969241251899 for Impact of oral administration of calcitriol to prevent symptomatic hypocalcemia after total thyroidectomy: Systematic review and meta-analysis by Parisa A. Dadkhah, Amirali Farshid, Reza Khademi, Shayan Yaghoubi, Ailin Asadzadeh, Nima Moharamnejad, Faezeh Jadidian, Seyed A. Ziaei, Sepehr Haghshoar, Omid Salimi, Seyyed-Ghavam Shafagh, Negin Rabiei, Saleh Shahsavari, Samin Shokravi, Zahra Maroufi, Niloofar Deravi, Gisou Erabi and Mahdyieh Naziri in Scandinavian Journal of Surgery
Footnotes
Acknowledgements
The authors thank all the researchers whose their published articles were included in our study.
Author contributions
P.A.D., A.F., and R.K. contributed equally to the article. P.A.D. contributed to validation, data curation, writing, and editing. A.F. and R.K. contributed to investigation, validation, writing review, and editing. S.Y. contributed to investigation, writing, and data extraction. A.A. contributed to data extraction, validation, and writing. N.M. contributed to data extraction, validation, and screening. F.J. contributed to screening, data extraction, and writing. S.A.Z. contributed to writing. S.H. contributed to screening and data extraction. O.S. contributed to data extraction and writing. S.-G.S. contributed to screening and data extraction. N.R. contributed to data extraction. S.S. contributed to screening and data extraction. S.S. and Z.M. contributed to screening. N.D.: contributed to conceptualization and supervision. G.E.: contributed to formal analysis. M.N.: contributed to supervision.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
