Abstract
Background and objective:
Most of the studies on metastasectomy in renal cell cancer are based on metachronous, often oligometastatic disease. Prior data on the impact of metastasectomy in synchronous metastatic renal cell cancer (mRCC) is, however, very scarce. We aimed to investigate the role of complete and incomplete metastasectomy in a large, nationwide patient population.
Methods:
We analyzed nationwide data, including all synchronous mRCC cases in Finland diagnosed during a 6-year period identified from the Finnish Cancer Registry, and complemented with patient records from the treating hospitals. We only included the patients who underwent removal of the primary tumor by nephrectomy. We performed univariate and multivariable adjusted analysis to identify the effect of metastasectomy on overall survival (OS) and cancer-specific survival (CSS).
Results:
We included 483 patients with synchronous mRCC. Overall, 57 patients underwent complete and 96 incomplete metastasectomy, while 330 patients had no metastasectomy. The median OS was 17.9 and CSS 17.2 months for all patients. The median OS and the median CSS were 59.3 and 60.8 months for the complete, 21.9 and 25.1 for the incomplete, and 14.5 and 14.8 months for the no metastasectomy groups (p < 0.001 for differences). In both applied multivariable statistical models, the OS and CSS benefit from complete metastasectomy remained significant (hazard ratios (HRs) varied between 0.42 and 0.54, p < 0.001) compared with the no metastasectomy group. However, there was no improvement in survival estimates in the incomplete metastasectomy group compared with the no metastasectomy group (HRs varied between 1.04 and 1.10, p > 0.40).
Conclusions:
Complete metastasectomy, when possible, can be considered as a treatment option for selected patients with synchronous mRCC who are fit for surgery. By contrast, we found no survival benefit from an incomplete metastasectomy suggesting that such procedures should not be performed for these patients.
X summary: Nationwide data from Finland showed that patients with synchronous metastatic renal cell cancer benefit from complete metastasectomy. Read on!
Context and Relevance
Complete metastasectomy has been shown to improve the survival of selected patients with metachronous metastatic renal cell cancer (mRCC), whereas there is minimal previous data on the impact of metastasectomy in synchronous mRCC, which tends to have a worse prognosis in general. We investigated a large nationwide cohort of patients with synchronous mRCC to explore the possible benefits of complete and incomplete metastasectomy in this scarcely studied group. According to our findings, complete metastasectomy, when possible, should be considered as a treatment option for patients with synchronous mRCC who are fit for surgery.
Introduction
A gradual increase in the incidence of renal cell cancer (RCC) has been observed worldwide and in Europe during the past decades.1,2 At the time of diagnosis of RCC, approximately 16%–30% of patients present with synchronous distant metastases.3,4 Synchronous compared with metachronous mRCC has a poorer prognosis and tends to progress more rapidly.5 –7 Despite the advances in medical therapy after the introduction of vascular endothelial growth factor (VEGF)-targeted tyrosine kinase inhibitors (TKIs) and novel immuno-oncologic (IO) medications, surgical resection of selected metastases remains an attractive option, particularly in patients with a good performance status and oligometastatic disease.7 –13 To date, there is no randomized evidence on the effect of metastasectomies on the survival of patients with mRCC, but there is an increasing amount of retrospective data suggesting that metastasectomy could be beneficial in selected clinical scenarios. However, most of the studies on metastasectomy are focused on patients with mainly solitary metachronous metastatic lesions, usually lung metastases diagnosed in the follow-up of primarily surgically resected non-metastatic RCC.14,15 A long interval from the initial RCC diagnosis to the development of metastases is considered a favorable prognostic feature, and it may encourage clinicians to perform a complete metastasectomy in case of a late-onset metachronous oligometastatic disease.16,17 On the contrary, there is a relative lack of evidence on the results of metastasectomies in synchronous mRCC. A single-center retrospective study published in 2019, which included 37 patients with synchronous mRCC and 46 patients with metachronous mRCC who underwent metastasectomies, reported poorer overall survival (OS) and progression-free survival for patients with synchronous metastases regardless of the metastasectomy status. 6 Similarly, another recent single-institution retrospective study showed a worse cancer-specific survival (CSS) for synchronous mRCC in the first-line complete metastasectomy and medical therapy groups, but the statistical power regarding synchronous mRCC was limited, as only 19 such patients underwent first-line metastasectomies. 7
Aiming to evaluate the impact of metastasectomy on synchronous mRCC survival outcomes in a real-life setting, we present a nationwide observational study on all patients with synchronous mRCC, who underwent a nephrectomy, diagnosed over a 6-year period. To our knowledge, our study presents the largest published population-based data on metastasectomies in synchronous mRCC. In this cohort, we investigated the survival of patients with different metastasectomy statuses regarding various confounding factors. Our goal was to evaluate the effect of metastasectomy on survival and to evaluate the patient characteristics and clinical factors that should be considered when selecting patients for metastasectomies.
Methods
All patients who were diagnosed with RCC and synchronous distant metastases during the selected 6-year period (2005–2010) were identified from the database of Finnish Cancer Registry. Patients with missing treatment or follow-up details were excluded, as well as patients under 18 years of age, and with other advanced-stage cancer. Posthumously diagnosed cases were also excluded. The patient records were obtained from the treating hospitals, including information on diagnostics, surgical and medical oncologic treatment, and follow-up. Patients who did not undergo resection of the primary tumor via radical or partial nephrectomy were excluded. The medical reports were reviewed, and the following clinicopathological data were collected: sex, age at the time of diagnosis, T stage, Fuhrman grade and histology, the location of metastases and the number of metastatic sites, the Eastern Cooperative Oncology Group (ECOG) performance status, serum hemoglobin (Hb) and C-reactive protein (CRP), and cause of death. The T stage was reassigned according to the 2017 TNM classification, and the ECOG performance status was, in some cases, evaluated retrospectively if it was not clearly specified in the patient records. Due to the retrospective nature of this study, serum calcium, neutrophil, and lactate dehydrogenase levels were not available for analysis for all patients; therefore, the International Metastatic RCC Database Consortium (IMDC) or Memorial Sloan Kettering Cancer Center (MSKCC) risk criteria were not evaluated.
Treatment protocols, such as type of surgery and systemic treatment, as well as follow-up intervals and imaging modalities, were at the discretion of the treating physicians at the institutional level.
According to the metastasectomy status, the cohort was divided into the no metastasectomy, incomplete metastasectomy, and complete metastasectomy groups. The OS and CSS estimates and the time to systematic treatment were analyzed.
Data analysis
The primary outcome was OS. The secondary outcomes were CSS and time from diagnosis to initiation of medical oncologic therapy. Analysis of variance of baseline characteristics and risk factors between the treatment groups was performed using the chi-square test or Fisher’s exact test for categorical variables, and the independent-samples median test or the Kruskal–Wallis test adjusted with the Bonferroni correction for continuous and ordinal variables. Two-sided significance was taken at p < 0.05. Kaplan–Meier analysis was performed to estimate the median survival times and survival distributions. Univariate and multivariable Cox regression analyses were performed to estimate the hazard ratio (HR) for the association of risk factors for OS. Time-dependent multivariable adjusted Cox regression models were used to avert the immortal time bias considering metastasectomy status and TKI/mammalian target of rapamycin inhibitor (mTORi) therapy.18,19 Two different multivariable adjusted Cox regression models were created. Model 1 included all factors one by one that had a p-value < 0.3 according to OS univariate analysis. A variable was left in the final model if it had a p-value < 0.05 or if it had a significant impact on the Akaike information criterion compared with the previous model. In model 2, a directed acyclic graph (DAG) was constructed to identify the causal relationships between confounding factors and to only include variables needed to get an unbiased adjusted estimate of treatment effect (HR). The follow-up time was limited to 10 years in the survival analyses due to the decrease in the number of patients over time. Because there was missing data for some of the risk factors, a multiple imputation technique with 50 imputed datasets was applied to increase the precision and to reduce bias in the analyses. Statistical analysis was performed using SPSS version 28 (IBM Corp., Armonk, NY, USA) or SAS version 9.4 (SAS Institute, Inc., Cary, NC, USA).
Results
Overall, 483 patients were included in the study population. The flow chart for the selection of eligible cases for the study is presented in Fig. 1.
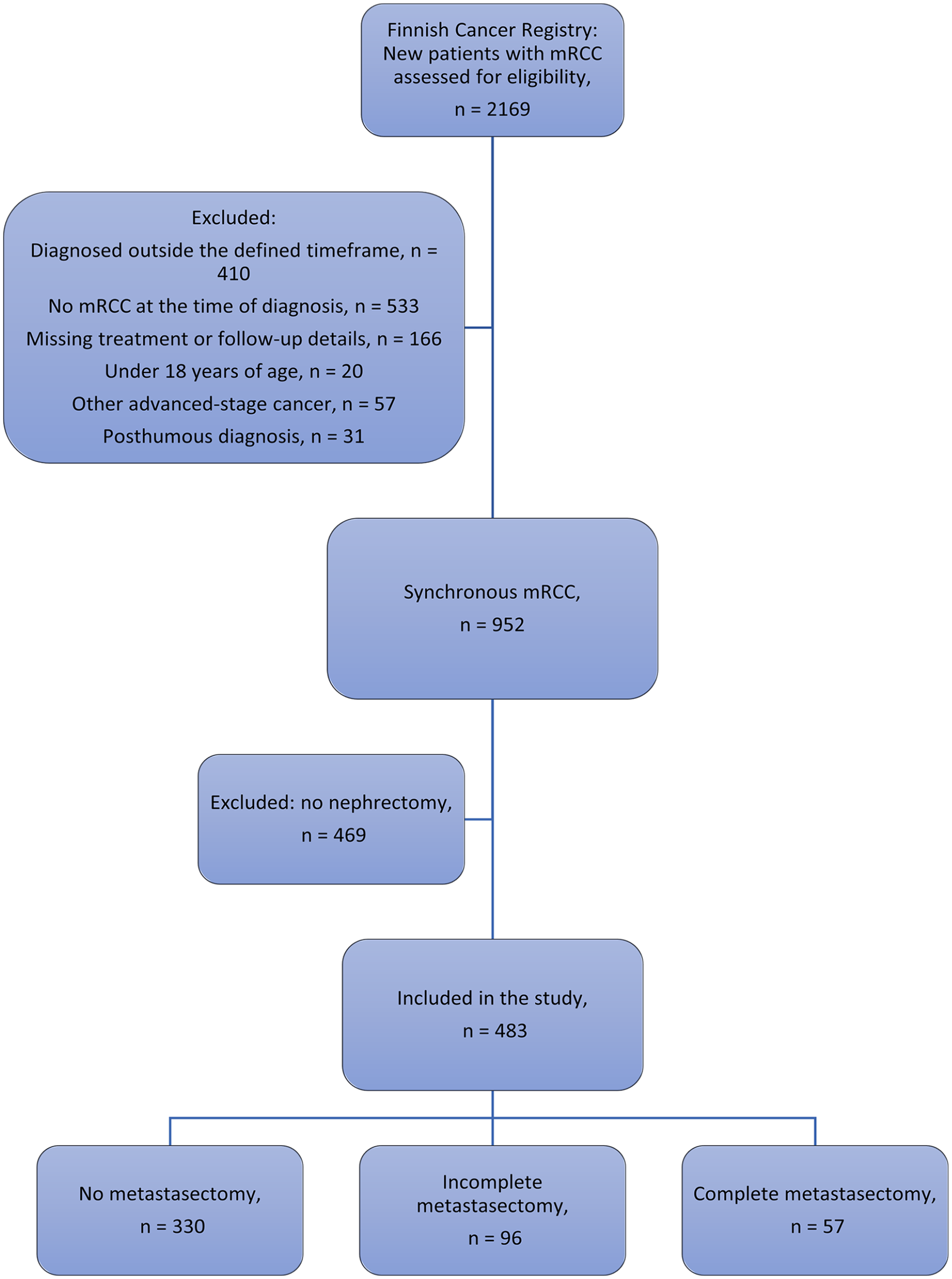
Flow chart of the patient exclusion and selection process. mRCC: metastatic renal cell cancer.
The mean follow-up for all patients was 34.0 (range, 0.4–120.0) months. The median OS, defined as the time interval from the first date of diagnosis of mRCC to death, was 17.2 (95% confidence interval (CI), 14.8–19.5) months. The median CSS was 17.9 (95% CI, 15.5–20.3) months. The median time from diagnosis to initiation of medical treatment was 3.0 months. The median age at diagnosis was higher in the no metastasectomy group compared with the complete and incomplete metastasectomy groups. The number of metastases and metastatic sites at disease onset was lower among the patients in the complete metastasectomy group versus the incomplete metastasectomy and no metastasectomy groups. In addition, there were some differences in the distribution of metastatic sites: Lung and bone metastases were least frequent among the complete metastasectomy group, whereas adrenal gland metastases were least common in the no metastasectomy group. The detailed clinicopathologic features are presented in Table 1.
Clinicopathologic features in the different metastasectomy status groups.
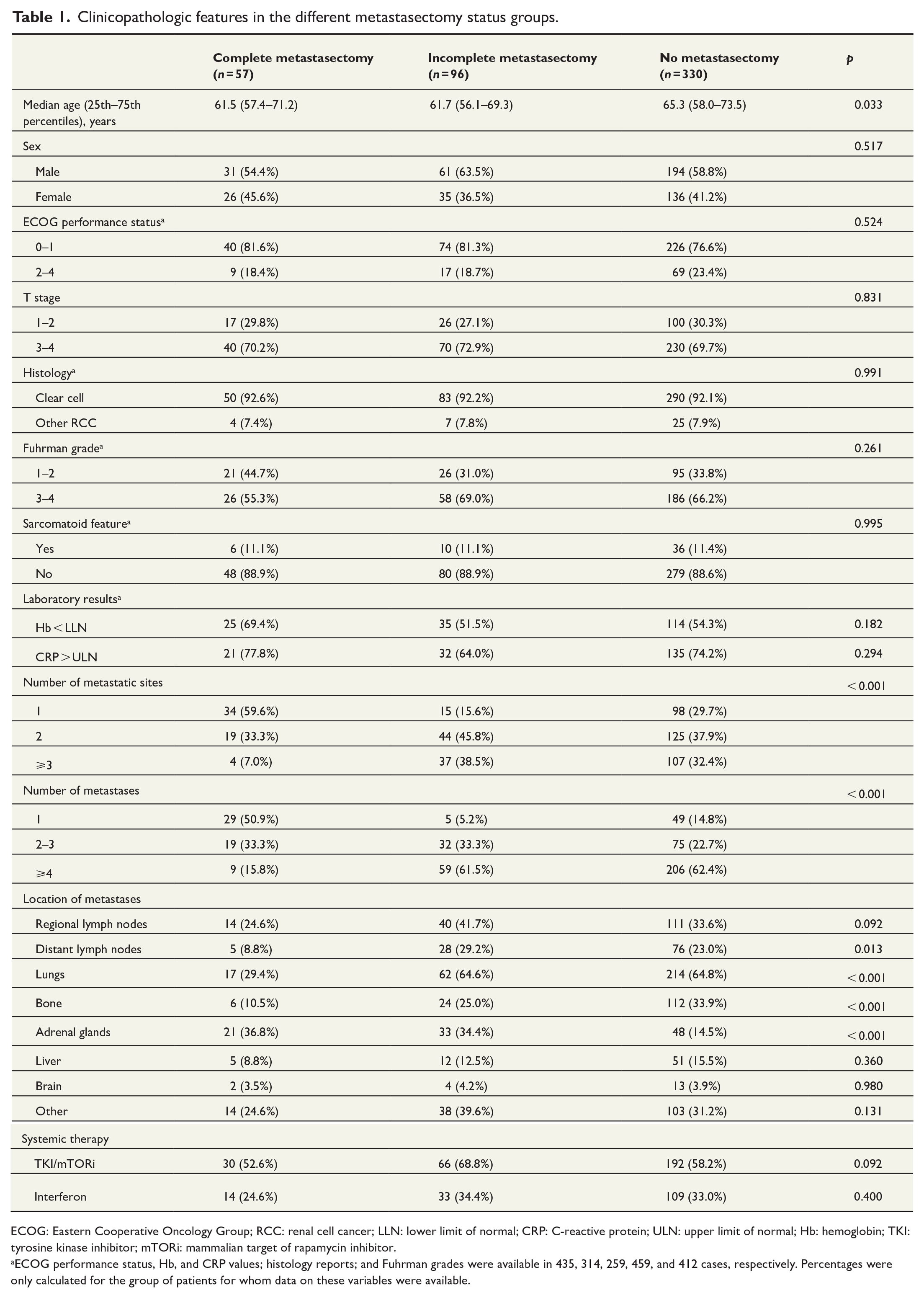
ECOG: Eastern Cooperative Oncology Group; RCC: renal cell cancer; LLN: lower limit of normal; CRP: C-reactive protein; ULN: upper limit of normal; Hb: hemoglobin; TKI: tyrosine kinase inhibitor; mTORi: mammalian target of rapamycin inhibitor.
ECOG performance status, Hb, and CRP values; histology reports; and Fuhrman grades were available in 435, 314, 259, 459, and 412 cases, respectively. Percentages were only calculated for the group of patients for whom data on these variables were available.
Risk factors for overall survival
We performed a univariate Cox regression proportional hazard analysis to assess the effect of various clinicopathologic features on OS. Age ⩾ 70 years, ECOG performance status 2–4, non-clear cell histology, sarcomatoid feature, serum Hb < the lower limit of normal (LLN), multiple metastatic sites, multiple metastases, regional lymph node metastases, bone metastases, liver metastases, and brain metastases increased the HR for OS, whereas TKI/mTORi therapy and complete metastasectomy decreased the HR for OS. The results of univariate analysis are shown in Table 2.
Univariate Cox regression analysis of risk factors for overall survival.
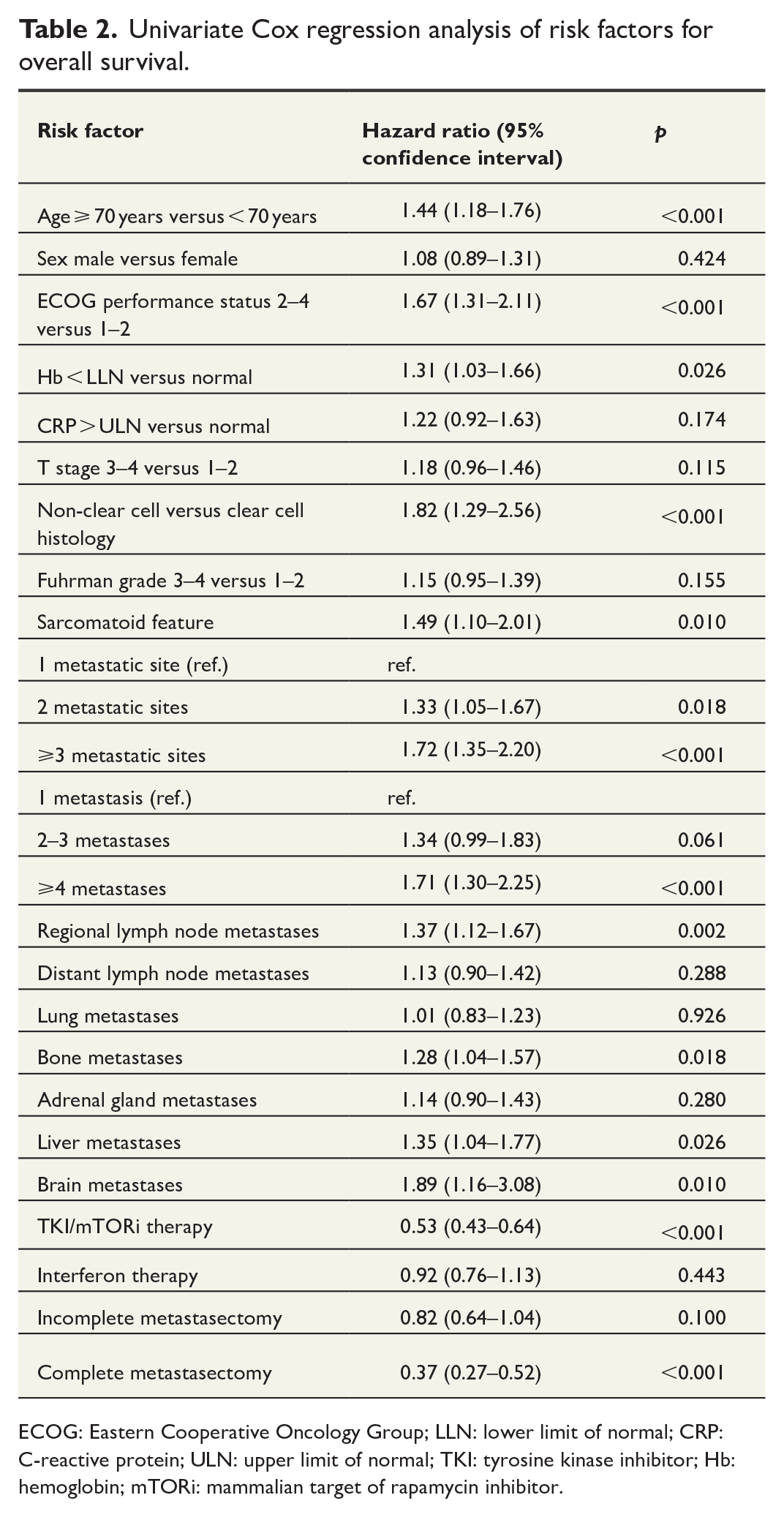
ECOG: Eastern Cooperative Oncology Group; LLN: lower limit of normal; CRP: C-reactive protein; ULN: upper limit of normal; TKI: tyrosine kinase inhibitor; Hb: hemoglobin; mTORi: mammalian target of rapamycin inhibitor.
Metastasectomy sites
Regional lymph nodes were the most frequent site of metastasectomy (54 patients), followed by adrenal glands, lungs, bones, and brain. As all patients had distant metastases, no patients in the complete metastasectomy group had metastasectomy of the regional lymph nodes only. The distribution of metastasectomy sites in the different metastasectomy status groups is shown in Table 3.
Distribution of metastasectomy sites.
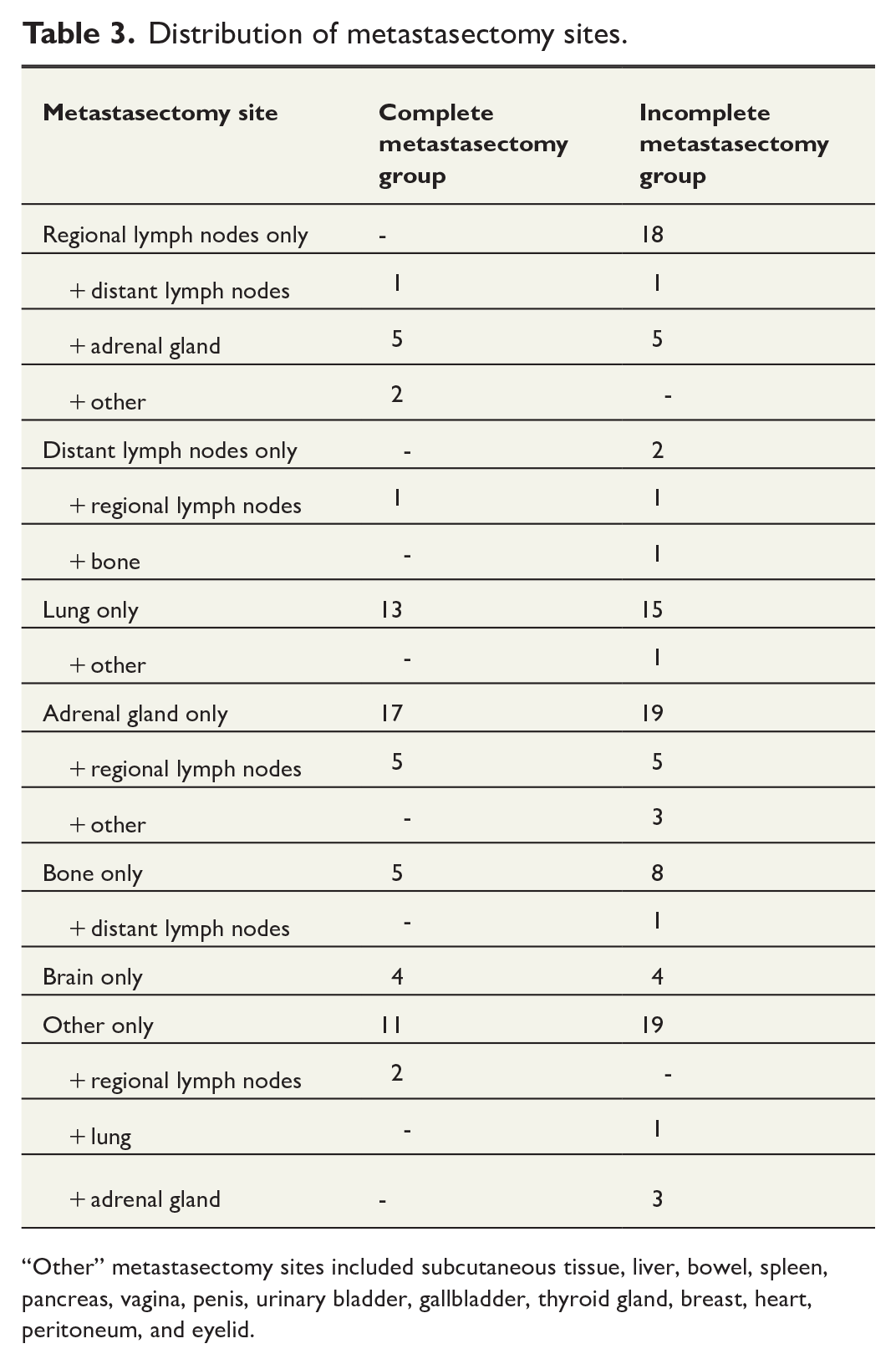
“Other” metastasectomy sites included subcutaneous tissue, liver, bowel, spleen, pancreas, vagina, penis, urinary bladder, gallbladder, thyroid gland, breast, heart, peritoneum, and eyelid.
Impact of metastasectomy on survival
The complete metastasectomy group had significantly better OS (59.3 versus 14.5 months) and CSS (60.8 versus 14.8 months) estimates compared with the no metastasectomy group (p < 0.001 for both). The survival outcomes according to metastasectomy status are shown in Table 4, and the Kaplan–Meier survival curves are presented in Fig. 2.
Overall survival (OS) and cancer-specific survival (CSS) outcomes in the different metastasectomy status groups.
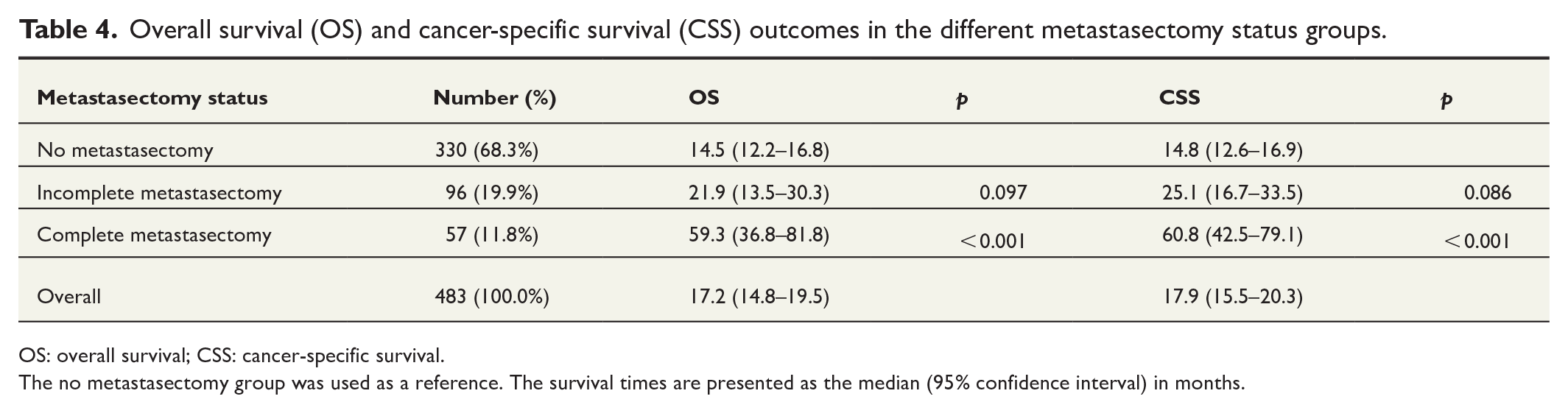
OS: overall survival; CSS: cancer-specific survival.
The no metastasectomy group was used as a reference. The survival times are presented as the median (95% confidence interval) in months.
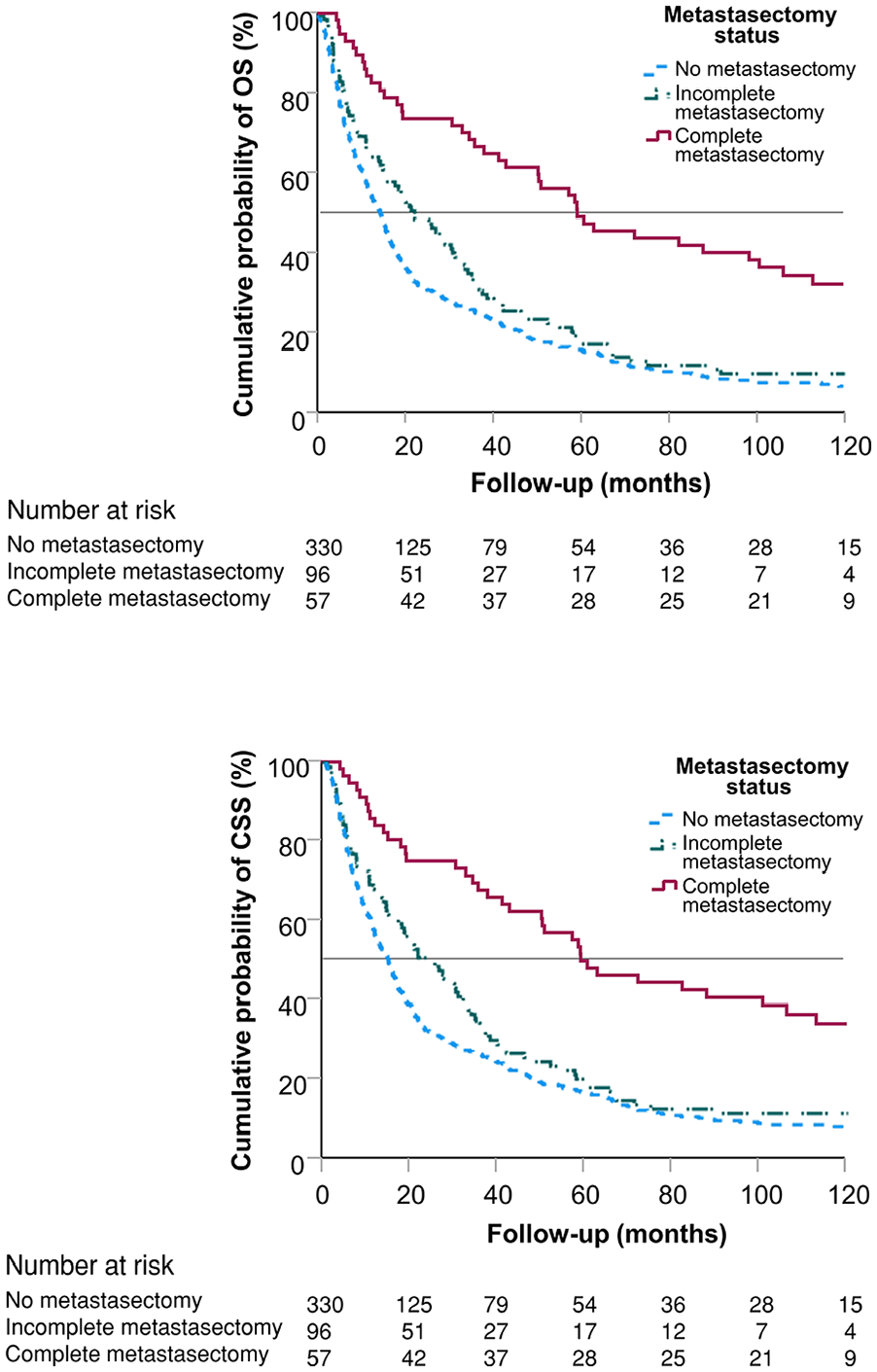
The Kaplan–Meier overall survival (OS) and cancer-specific survival (CSS) curves according to the metastasectomy status.
Multivariable adjusted analyses
In model 1, we used the following OS risk factors as adjusting covariates: age ⩾ 70 years, ECOG performance status 2–4, T stage ⩾ 3, non-clear cell histology, sarcomatoid histology, the number of metastatic sites, the number of metastases, Hb < LLN, CRP > the upper limit of normal (ULN), and TKI/mTORi therapy. In model 2, the DAG causal effect identification indicated that age, ECOG performance status 2–4, the number of metastases, and the number of metastatic sites are the minimally sufficient adjusting factors to estimate the total effect of metastasectomy on survival. In both models, due to some missing data in the adjusting covariates, we used a multiple imputation technique to increase the accuracy of the models.
In both multivariable adjusted models, complete metastasectomy was an independent significant predictor of improved HR for both OS and CSS (p < 0.001). However, incomplete metastasectomy, compared with no metastasectomy, did not have a significant effect on the HR values in either model. The detailed results for both models are presented in Table 5.
Multivariable (Model 1) and directed acyclic graph (DAG)–adjusted (Model 2) time-dependent Cox regression analyses on the effect of metastasectomy on survival. The results are presented as HR (95% CI). The No metastasectomy group was used as a reference.

MV: multivariable; OS: overall survival; CSS: cancer-specific survival.
The complete metastasectomy group: patient characteristics, treatments, and results
Fifty-seven patients underwent complete metastasectomy. The baseline characteristics and metastasectomy sites are shown in Tables 1 and 3, respectively. The most frequent sites of metastasectomy were adrenal gland (23 patients) and lung (13 patients). Fifty (87.7%) patients underwent a single metastasectomy session, while 7 (12.3%) patients underwent more than one metastasectomy session.
The median OS estimate was 59.3 (95% CI, 36.8–81.8) months and the median CSS estimate was 60.8 (95% CI, 42.5–79.1) months. The median time from diagnosis to initiation of medical treatment was 5.62 months, compared with 3.25 months (p = 0.040) and 2.83 months (p < 0.001) in the incomplete metastasectomy and no metastasectomy groups, respectively. Thirty-seven (64.9%) patients had no evidence of disease at the first follow-up imaging after metastasectomy, while the remaining 20 (35.1%) patients had recurrent disease at the first follow-up contact. At the end of the follow-up, 13 (22.8%) of the 57 patients who underwent complete metastasectomy had experienced no evidence of recurrence.
For the complete metastasectomy group, we performed univariate Cox regression analysis for risk factors for OS. We identified male gender (p = 0.043) and Hb < LLN (p = 0.017) as significant risk factors for OS. The OS HR results for all risk factors are presented in Supplementary Table S1.
Nine (15.7%) patients from the complete metastasectomy group died within 12 months from the time of diagnosis. For these nine patients, the median age at diagnosis was 72.9 years. Six patients had one metastatic site, and three had two metastatic sites. Seven patients had serum Hb < LLN and six had CRP > ULN. Seven patients had regional stage T3-4 tumor, and two had a Fuhrman grade 3–4 tumor. Three patients had medical treatment for RCC. Renal cancer was the cause of death for all except one patient in this group. Six patients had recurrent disease at the first follow-up imaging after metastasectomy. None of the patients experienced Clavien–Dindo grade 3–5 surgical complications requiring invasive treatment or prolonged hospitalization, but one patient did not recover sufficiently from pulmonary metastasectomy to receive medical oncologic treatment.
Patients with no evidence of recurrence during follow-up
Of all patients, 30 (6.2%) had no evidence of recurrent disease at the end of follow-up. The median follow-up for these patients was 119 (range, 82–120) months. Two (6.7%) patients died of other causes during the follow-up period, while the remaining 28 (93.3%) patients were alive at the last follow-up contact. At the time of diagnosis, 17 (56.7%) patients had only one metastatic site, 9 (30.0%) had two metastatic sites, and 4 (13.4%) had more than two metastatic sites. Sixteen (53.3%) patients underwent metastasectomies: 13 (43.3%) had complete metastasectomy and 3 (10.0%) had incomplete metastasectomy. Twelve (40.0%) patients received TKI/mTORi therapy and 7 (23.3%) received interferon. Of the no metastasectomy group, 13 patients had a permanent complete response by medical therapy and cytoreductive nephrectomy, and 1 patient experienced spontaneous and complete regression of pulmonary metastases after cytoreductive nephrectomy only.
Discussion
In the current era of novel IO- and VEGF-targeted combination therapy in mRCC, the proportion of patients with a complete response for medical therapy remains at 3%–16% according to phase III randomized studies.20 –22 Considering this, surgical resection for both the primary tumor and visible metastases remains a relevant component in a multidisciplinary approach to treat selected patients with mRCC, as there is an increasing interest toward metastasis-directed therapy via the use of surgical metastasectomy, thermal ablation, radiotherapy, and other minimally invasive options. 23 We investigated the impact of metastasectomy on patients with synchronous mRCC in a real-life population-based multi-center setting. As all the patients had the primary tumor surgically removed, the subjects were evaluated to be fit enough to have surgery; however, a proportion of the patients had a significantly decreased performance status (ECOG 2–4) at the time of diagnosis. Thus, we present important long-term results regarding the effect of metastasectomy on patients with synchronous mRCC in a nationwide dataset of heterogeneous patient population.
In the complete metastasectomy group, there was a significant improvement in median OS (59.3 (95% CI, 36.8–81.8) versus 14.5 (95% CI, 12.2–16.8) months, p < 0.001) and CSS (60.8 (95% CI, 42.5–79.1) versus 14.8 (95% CI, 12.6–16.9) months, p < 0.001) estimates compared with the no metastasectomy group. There were no differences in the median OS or CSS estimates between the no metastasectomy and incomplete metastasectomy groups. The survival benefit following complete metastasectomy also remained significant in the multivariable adjusted analysis, in terms of both OS and CSS. Although the evidence for metastasectomy in patients with synchronous mRCC remains scarce, our findings are well in line with the results of previous publications on general mRCC populations. A recent large Canadian population-based study reported a significant survival benefit for patients who underwent complete metastasectomy compared with the propensity score–matched group of patients who had not undergone metastasectomy (81 versus 61 months, p = 0.0001). 24 There were similar results in a recent Austrian retrospective study of 106 patients with metachronous mRCC. The authors reported an improved OS in the complete metastasectomy group compared with the no metastasectomy group, even after propensity score weighing and correction of immortal time bias; on the other hand, there was no survival benefit in the incomplete metastasectomy group. 25 Similarly, our findings suggest that incomplete metastasectomy has no effect on OS or CSS, and therefore cannot be recommended. This finding supports published retrospective data based on patient cohorts mostly consisting of patients with metachronous mRCC.26,27 It is to be noted that those studies report higher OS and CSS estimates than presented in our study, apparently mostly due to the small proportions of synchronous metastatic cancers among the patient cohorts. Indeed, Kim et al. 6 reported an even poorer median OS estimate of 11.61 (95% CI, 9.67–16.70) months for their synchronous mRCC cohort.
Nevertheless, it is noteworthy that 35.1% of the patients who underwent complete metastasectomy had a recurrence already at the first follow-up imaging. Moreover, 15.7% of the patients in the complete metastasectomy group died within a year, which underlines the severity of synchronous metastatic disease even in the cases where surgery with a curative aim is plausible.
In univariate analysis, the extent of metastatic burden, defined by number of metastases and the number of metastatic sites, seemed to be a significant prognostic factor in this patient cohort. This finding is consistent with earlier retrospective studies that observed an increased risk of death for patients with more than one tumor location24,27 –29 or multiple metastases. 25 We identified low serum Hb, non-clear cell histology, and liver metastases as clinically significant baseline prognostic factors in univariate analysis, which correlate with the MSKCC and IMDC models and other published studies, although mixed results on the significance of clear cell histology and liver metastases have been reported.6,16,17,24 In addition, VEGF–TKI targeted medical therapy improved median OS and CSS in univariate analysis, which emphasizes the importance of meticulous multidisciplinary consideration of treatment decisions, aiming for the best possible treatment of patients with synchronous mRCC.
The strength of our study is that it is based on complete national data from the Finnish Cancer Registry, which is notified of all suspected cancer cases by the treating hospitals. 30 To supplement this information, we examined the original patient records from the hospitals to make our insights as detailed as possible. As a result, this study provides reliable, essential, and new knowledge on the impact of metastasectomy on the scarcely investigated group of patients with synchronous mRCC, given that the results of earlier metastasectomy studies cannot necessarily be generalized to this sub-population. 25 In addition, using time-dependent DAG-adjusted multivariable analyses with a multiple imputation technique strengthens the methodology of our study. However, some limitations should be addressed. Most importantly, in a retrospective study, treatment selection bias should always be considered when interpreting the results because there is an inevitable tendency for the patients of the better overall prognosis and lower complication risk to be selected into the more active (i.e. surgical) line of treatment. Moreover, the medical treatments in the study population were based mostly on TKI drugs such as sunitinib, and to some extent on interferon, which deviates from the current practices and guidelines recommending IO-based or IO–TKI combination treatments.31,32 In the future, studies on the impact of metastasectomy on patients with mRCC who receive contemporary medical treatments are highly anticipated.
Supplemental Material
sj-docx-1-sjs-10.1177_14574969241234485 – Supplemental material for The impact of metastasectomy on survival of patients with synchronous metastatic renal cell cancer in Finland: A nationwide study
Supplemental material, sj-docx-1-sjs-10.1177_14574969241234485 for The impact of metastasectomy on survival of patients with synchronous metastatic renal cell cancer in Finland: A nationwide study by Lauri Laru, Hanna Ronkainen, Pasi Ohtonen and Markku H. Vaarala in Scandinavian Journal of Surgery
Footnotes
Acknowledgements
We would like to thank Kaisa Makkonen, MD, for the assistance on screening and analyzing the patient records. In addition, we would like to thank Ms Tuula Lähtevänoja, Ms Katja Vaihoja, and Ms Leena Heikkilä (Oulu University Hospital, Oulu, Finland) for the acquisition of the patient records.
Author contributions
All authors contributed to the study conception and design. L.L. prepared material and collected data. L.L. and P.O. analyzed the data. L.L. wrote the first draft of the manuscript. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: L.L. declares reimbursements from Orion Corp. for attending a scientific meeting, outside the submitted work. M.H.V., P.O., and H.R. declare that they have no competing interests.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Finnish government research funding.
Ethical approval
The local ethics council of Oulu University Hospital (Oulu, Finland) approved the present study, which was conducted according to the Declaration of Helsinki.
Clinical trial registration
Not applicable.
Informed consent
Informed written consent from included study patients was not obtained, for the National Institute for Health and Welfare (Helsinki, Finland) approved the present study and the use of registry data following local ethical approval, according to Finnish law. Patient data was anonymized and de-identified prior to statistical analysis.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
