Abstract
Background:
International guidelines recommend open surgery for atypical ductal hyperplasia (ADH) in the breast due to risk of underestimating malignant disease. Considering the ongoing randomized trials of active surveillance of low-risk ductal carcinoma in situ (DCIS), it seems reasonable to define a low-risk group of women with ADH where a conservative approach is appropriate. The aim here was to evaluate the management and risk for upgrade of lesions diagnosed as ADH in percutaneous breast biopsies in two Swedish hospitals.
Methods:
All women with a screen-detected or symptomatic breast lesion breast imaging-reporting and data system (BI-RADS) 2–4 and a percutaneous biopsy showing ADH between 2013 and 2022 at Sundsvall Hospital and Umeå University Hospital were included. Information regarding imaging, histopathology, clinical features, and management was retrieved from medical records. Odds ratio (OR) and 95% confidence intervals (CI) for upgrade to malignant diagnosis after surgery were calculated by logistic regression analysis.
Results:
Altogether, 101 women were included with a mean age 56.1 (range 36–93) years. Most women were selected from the national mammography screening program due to microcalcifications. Biopsies were performed with vacuum-assisted biopsy (60.4%) or core-needle biopsy (39.6%). Forty-eight women (47.5%) underwent surgery, of which 11 were upgraded to DCIS, and 7 to invasive breast cancer (upgrade rate 37.5%). Among the 53 women managed conservatively (median follow-up 74 months), one woman (1.9%) developed subsequent ipsilateral DCIS. The combined upgrade rate was 18.8%. No clinical variable statistically significantly correlating to risk of upgrade was identified.
Conclusions:
The upgrade rate of 37.5% in women undergoing surgery compared to an estimated 5-year risk of ipsilateral malignancy at 1.9% in women managed conservatively indicate that non-surgical management of select women with ADH is feasible. Research should focus on defining reproducible criteria differentiating high-risk from low-risk ADH.
Context and relevance
International guidelines recommend open surgery for atypical ductal hyperplasia (ADH) in the breast due to risk of underestimating malignant disease. In this study, surgery was performed in 48 out of 101 women with ADH on percutaneous breast biopsy in two Swedish hospitals. The upgrade rate to carcinoma was 37.5% in women undergoing surgery but only 1.9% in women managed conservatively. The decision to proceed to surgery was based on a multidisciplinary conference discussion considering clinical, hereditary, radiological, and pathological findings. Future guidelines of ADH management should include standardized and reproducible histopathological criteria to differentiate high-risk from low-risk ADH.
Introduction
Since the wide implementation of mammography screening, an increasing number of unclear lesions in the breast are being diagnosed. Atypical ductal hyperplasia (ADH) is a non-malignant proliferative lesion that shares a number of features with ductal carcinoma in situ (DCIS) and is recognized as a risk marker for developing breast cancer (BC), even in the contralateral breast. 1
The distinction between ADH and DCIS, and low-grade DCIS in particular, presents a challenge and is often made exclusively by the size of the lesion.2,3 Consequently, ADH found in a core needle biopsy (CNB) may underestimate the presence of DCIS or even invasive cancer in the breast. The reported percentage of upgrade to DCIS and/or invasive BC after surgical excision is extremely variable (range: 4%–54%), with rates of approximately 25%–30% generally reported.4–8 Variables associated with risk of upgrade include age, personal or family history of BC, lesion size, multifocality (several ADH lesions), breast imaging-reporting and data system (BI-RADS) classification, presence of a palpable mass, size and numbers of biopsy needle/tissue samples.9–14 In addition, cytological features such as presence of necrosis have been shown to influence the upgrade rate. 15 Pawloski et al. 16 and Deshaies et al. 11 found a significantly higher upgrade rate in “severe ADH” bordering on DCIS compared to conventional ADH.
Although management of ADH is still a matter of debate, surgery is generally recommended. Two international consensus conferences on B3 lesions have been held in 2016 and 2019.4,7 A panel of experts reached an agreement that underestimation rates should be below 5% for invasive BC and below 10% for DCIS. Given a reported underestimation rate of up to 14% even with complete removal of all microcalcifications by vacuum-assisted biopsy (VAB), the consensus panel recommend open surgery for ADH. This is also in accordance with US guidelines. 17 More conservative approaches have however been suggested for subsets of women.5,13,15,18 Nguyen et al. defined a low-risk group with ADH limited to two terminal duct lobular units (TDLUs), >95% removal of targeted calcifications, and no significant cytologic atypia and/or necrosis. This group could be managed conservatively with mammographic follow-up. 15 Pena et al. 5 have presented a similar definition of a low-risk subgroup of women with ADH by the absence of cell necrosis, and either one focus with ⩾50% of lesion removed, or 2–3 foci with ⩾90% of lesion removed. Prediction models based on clinical criteria without considering the type of cellular atypia have also been proposed,12,19 but these models have failed to be validated in independent cohorts.20,21 In the United Kingdom, current guidelines were published in 2018 in which vacuum-assisted excision (VAE) rather than open surgery is recommended in cases of ADH. 22 The authors emphasize the importance of collecting representative samples and if a lesion cannot be excised entirely, specimen should be of comparable volume to a diagnostic surgical excision with a minimum of 4 g. 22 Considering the ongoing randomized trials of active surveillance of low-risk DCIS, it seems reasonable to define a low-risk group of women with ADH where a conservative approach is appropriate. In Sweden, national documented guidelines for management of ADH and other B3 lesions are currently lacking.
The aim of this study was to evaluate the management of lesions diagnosed as ADH in percutaneous breast biopsies in two Swedish hospitals. The proportion upgraded to DCIS or invasive BC after surgical excision and factors influencing underestimation of malignancy is also presented.
Methods
All women with a screen-detected or symptomatic lesion with ADH found on percutaneous breast biopsy between 2013 and 2022 from two Swedish hospitals were identified. Imaging was classified according to BI-RADS as 2 (benign), 3 (indeterminate), or 4 (suspicious). Women with BI-RADS 5 (highly suspicious) or 6 (confirmed breast malignancy) in the ipsilateral breast were excluded. Biopsies were performed under standard technique using either 14G core biopsy needle or 9-12G VAB under local anesthesia by a specialized breast radiologist. Biopsy and surgical excision specimen were evaluated by in-house pathologists, not necessarily specialized in breast pathology.
Information regarding imaging, histopathology, and clinical features was retrieved from medical records. Collection of data included patient age, personal or family history of BC, mode of detection (screening or clinical), mammographic findings, lesion size, number of lesions, and biopsy technique (CNB or VAB). Family history was defined as at least one first-degree relative with a history of BC. Recommendations of excision versus observation were not made based on any formal criteria or guidelines, rather the decisions were made either by the breast radiologist alone after receiving the biopsy specimen histopathology report or after a multidisciplinary team (MDT) conference discussion. For the present analysis, the histopathology reports were retrospectively reviewed, and the features stated in the biopsy specimen formed the basis for categorizing the lesions into four groups: (1) simple ADH, (2) ADH present in fibroadenoma or papilloma, (3) ADH bordering on atypical columnar cell hyperplasia (CCH)/flat epithelial atypia (FEA), and (4) ADH bordering on DCIS.
Statistics
Chi-square test was used for comparisons between categorical variables. Significance was set at p < 0.05. Binary logistic regression analysis was used to estimate odds ratio (OR) and 95% confidence intervals (CIs) for upgrade to malignant diagnosis in the surgical specimen. SPSS version 28 was used for the statistical analyses.
Ethical considerations
Ethical approval was granted by the Swedish Ethical Review Board (Dnr 2021-05068 and 2022-03755-02). Informed consent was given by all women included in the study.
Results
Study population
From the Departments of Pathology at Sundsvall Hospital and Umeå University Hospital, a total of 63 and 38 percutaneous breast biopsies of ADH without concurrent malignancy in the ipsilateral breast were identified, respectively (n = 101). All patients were female. Mean age was 56.1 (range: 36–93) years. Patient characteristics are shown in Table 1. Most women were selected from the national mammography screening program due to microcalcifications. Biopsies were performed with either a 9G VAB (48.5%), 10-12G VAB (11.9%), or 14G CNB (39.6%).
Baseline characteristics of women with ADH on biopsy undergoing surgery or observation only.
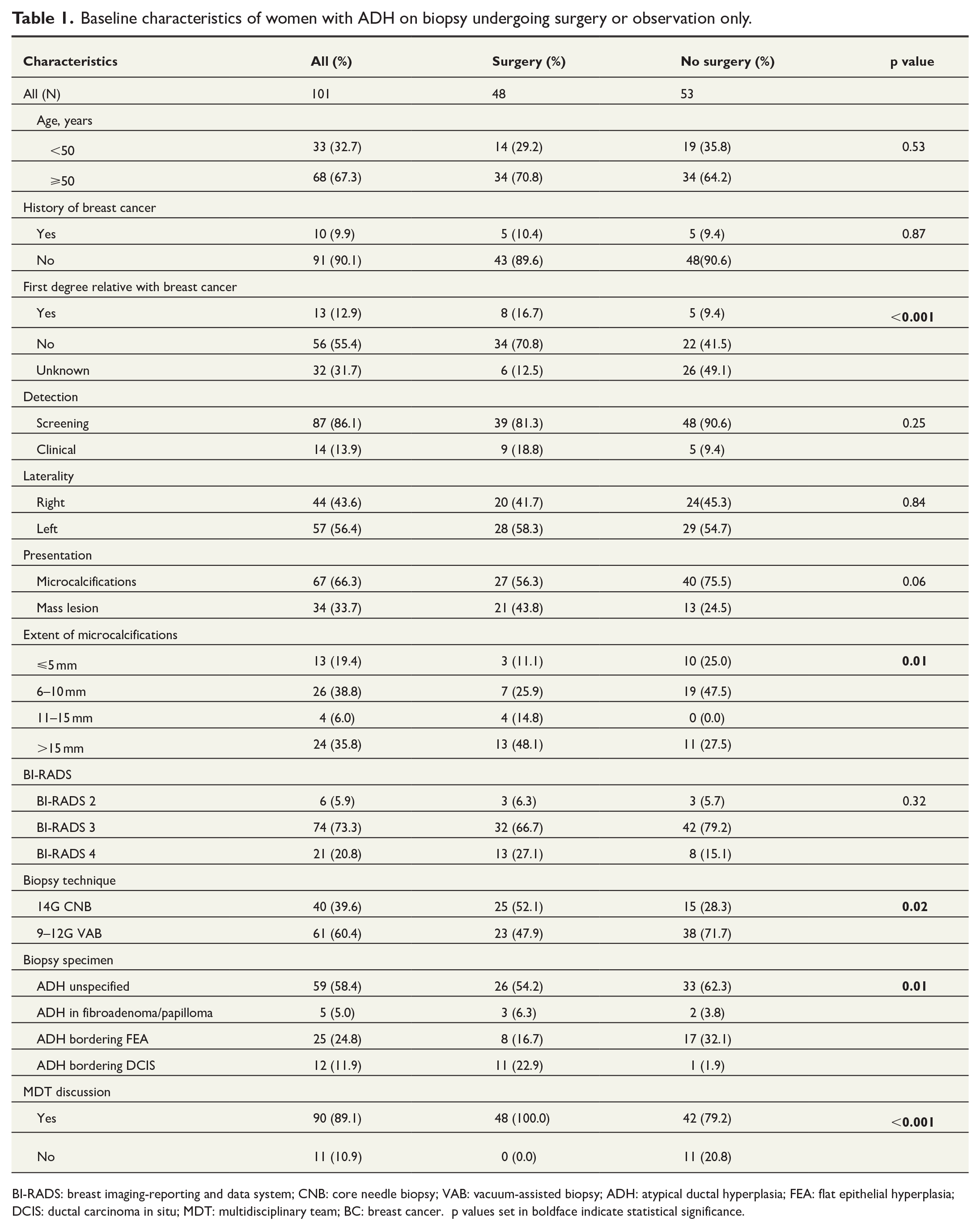
BI-RADS: breast imaging-reporting and data system; CNB: core needle biopsy; VAB: vacuum-assisted biopsy; ADH: atypical ductal hyperplasia; FEA: flat epithelial hyperplasia; DCIS: ductal carcinoma in situ; MDT: multidisciplinary team; BC: breast cancer. p values set in boldface indicate statistical significance.
Of the 101 women, 48 (47.5%) underwent subsequent surgical excision. In all of these, surgery was preceded by an MDT discussion. Women with a family history of BC were more likely to undergo surgery, as were women who underwent CNB as opposed to VAB, those with more extensive microcalcifications, and women in whom the histopathology report indicated suspicion of, or borderline DCIS in the biopsy specimen (Table 1). Forty-two out of 53 (79.2%) women who were managed conservatively were discussed at an MDT meeting, while the decision to forego surgery was made by the radiologist alone in 11 women.
Upgrade rates and predictors of upgrade
Overall, 19 out of 101 were upgraded to malignancy (upgrade rate 18.8%, 95% CI: 11.7%–27.8%). Among the 48 women who underwent surgery, 11 were upgraded to DCIS and 7 to invasive BC (upgrade rate 37.5%, 95% CI: 24.0%–52.6%) (Table 2). In 13 of these, there were residual microcalcifications left in the breast at the site of the biopsy, whereas in three women, all or close to all microcalcifications were removed by the biopsy. In two women, the biopsies were from mass lesions not completely removed. A review of the post-surgical specimen confirmed that the biopsy site was within the upgraded lesion in all patients. None of the variables investigated correlated with risk of upgrade, neither for the women undergoing surgery nor for the whole cohort (Figs 1 and 2). After a median follow-up time of 55 (interquartile range (IQR): 21–83) months, there were no subsequent ipsilateral recurrent or contralateral events in the women undergoing surgery.
Summary of histopathology results after surgery for ADH.
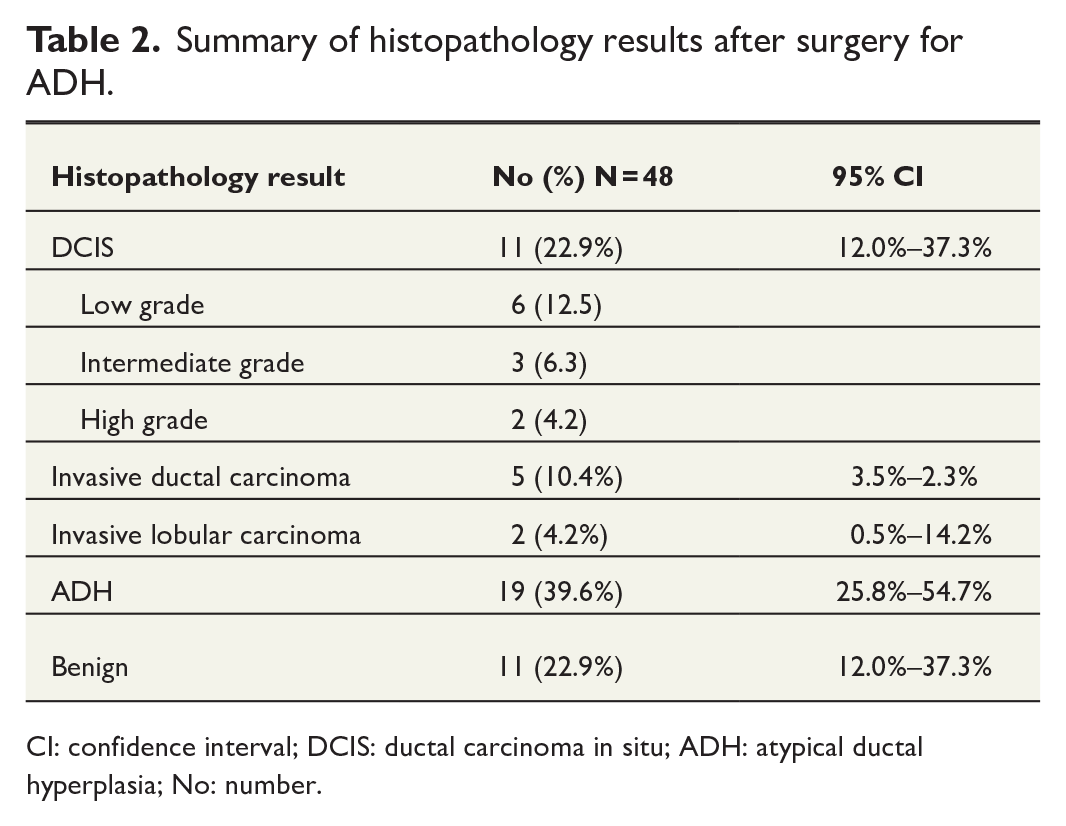
CI: confidence interval; DCIS: ductal carcinoma in situ; ADH: atypical ductal hyperplasia; No: number.
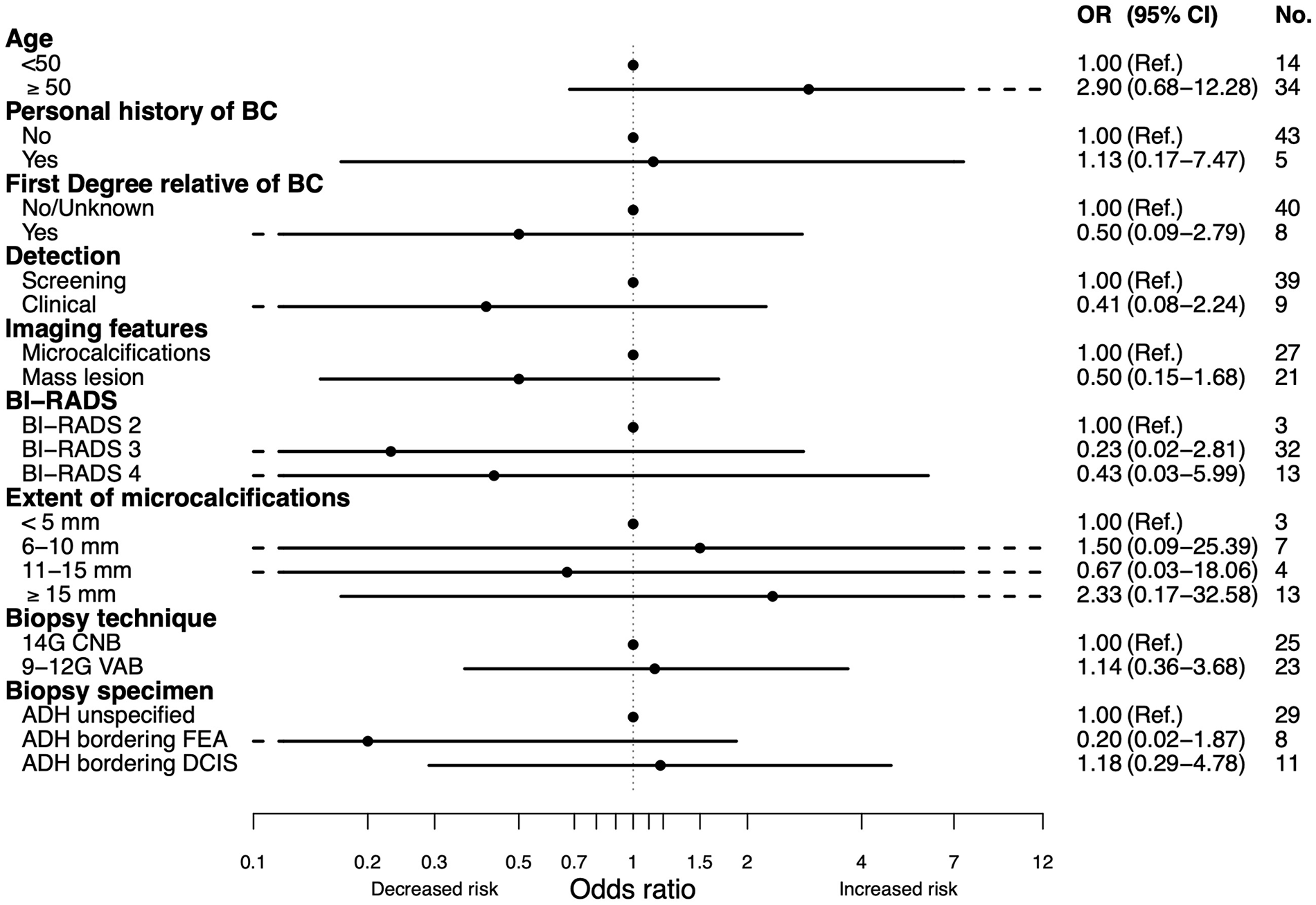
Correlation of clinicopathologic variables with risk of upgrade to carcinoma after surgical excision of ADH.
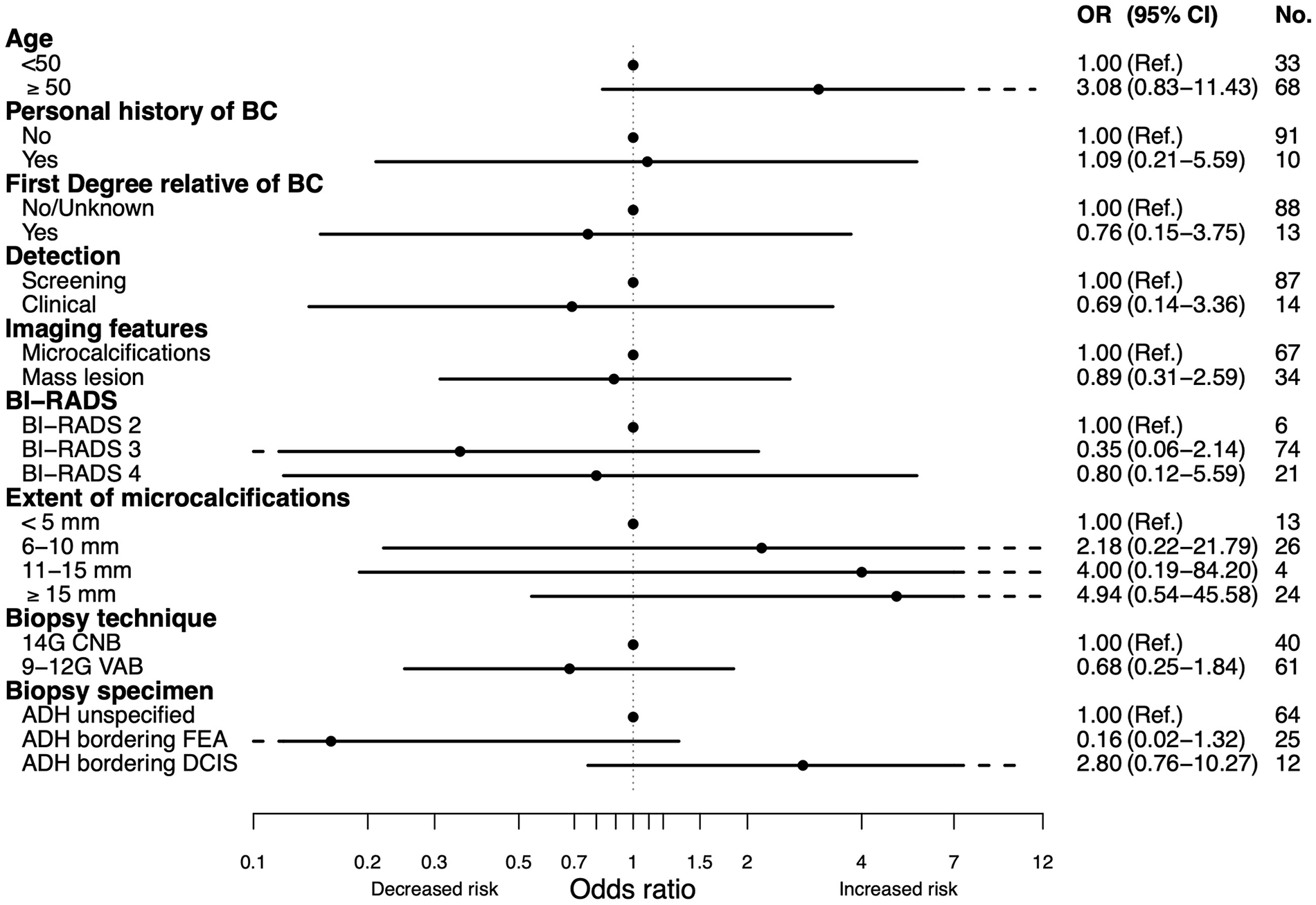
Correlation of clinicopathologic variables with risk of upgrade to carcinoma in women with ADH diagnosed on percutaneous biopsy.
In the group of 53 women managed conservatively, median follow-up was 74 (IQR: 49–92) months, with 47 women having a follow-up time of at least 24 months. One woman out of 53 underwent surgery 2 years after the biopsy due to the development of a new group of microcalcifications. The surgical specimen revealed DCIS grade I, thus providing an estimated 5-year ipsilateral upgrade risk among the women managed conservatively at 1.9% (95% CI: 0.01%–10.1%). The DCIS lesion was located at the site of the new group of microcalcifications that were close to, but not exactly at the index ADH site. At the index site, only ADH was found. In the group managed conservatively, there were three subsequent contralateral events, two women developed DCIS in the contralateral breast after 3 and 7 years, respectively, and one woman developed contralateral invasive BC after 4 years.
Discussion
The aim of this study was to assess management of ADH in daily practice in two Swedish Hospitals. It is shown here that only half of the cases with ADH diagnosed between 2013 and 2022 underwent subsequent surgery contrary to international guidelines that generally recommend surgical excision for all cases to rule out breast malignancy. In all women undergoing surgery, the decision had been preceded by an MDT discussion. After median 6 years, only one woman in the group without surgery developed DCIS in the ipsilateral breast, while 18 women in the surgical group were upgraded to DCIS or invasive BC. These results indicate that the selection of non-surgical treatment for subsets of women with ADH was appropriate.
Age above 50 years has by some been reported to increase the risk of upgrade10,12,14,19,23 but not by others,5,6,11,13,15,24,25 which is in line with this study. There was a trend toward increased risk of upgrade for women aged 50 years or older although not statistically significant. Similarly, several studies have shown a strong association between the extent of microcalcifications and risk of upgrade, but this has been contradicted by others.6,23 Here, women with microcalcifications of more than 10 mm were much more likely to undergo surgery compared to those with up to 10 mm, but the correlation between the extent of microcalcifications and risk of upgrade did not reach statistical significance. Farshid et al. performed a histology review of core biopsies in 110 women undergoing surgery for ADH with an overall upgrade rate of 40%. The biopsies were subclassified into classic ADH, ADH bordering hyperplasia, ADH with micropapillary architecture, and ADH bordering DCIS, with this sub-classification being the only variable significantly associated with malignancy. 25 Here, the biopsies were categorized in a similar way and, not surprisingly, the classification of ADH bordering on normal hyperplasia versus ADH bordering toward DCIS influenced the decision. The association between the different categories and risk of upgrade did however not reach statistical significance, neither among those who underwent surgery, nor for the whole cohort.
The upgrade rate after surgical excision of ADH is extremely variable in the literature. 6 There are a number of explanations for this. Inclusion criteria differ between studies. In this study, the aim was to evaluate ADH as a lesion of uncertain malignant potential. For this reason, BI-RADS 5 lesions were excluded, as these per definition have an estimated likelihood of malignancy greater than or equal to 95% based on imaging findings. 26 A biopsy showing ADH in this context would inevitably be eligible for a new biopsy or proceed to definitive surgery. This exclusion criterium has however not been used or has not been specified in many studies. Another reason for the wide range of upgrade rates is low reproducibility between pathologists for classifying ADH.12,27 The distinction between ADH and hyperplastic breast lesions without atypia and, at the other end between ADH and DCIS may be challenging. A review by Walia et al. 28 showed that the concordance for breast borderline lesions between pathologists was between 58% and 92%. Several attempts have been made to define ADH at increased risk of upgrade, or maybe even more important, lesions at low enough risk of upgrade making surgery avoidable. Among the women undergoing surgery in this study, none of the variables analyzed was statistically significantly associated with risk of malignancy and the upgrade rate of 37.5% exceeds the acceptable underestimation rate stated by the international consensus conferences on B3 lesions. 7 Only one out of the 53 women managed conservatively however developed DCIS in the ipsilateral breast. Definitions of low-risk ADH that could be managed conservatively have been suggested.5,12,15,19 In this study, reliable application of such definitions to this cohort was not possible due to the lack of standardized documentation of the suggested low-risk criteria such as the absence of pronounced atypia and absence of cell necrosis. Nevertheless, most women were discussed at an MDT, and it seems that differences presented in the histopathology reports together with a multidisciplinary management strategy made it possible to distinguish more benign atypia from ADH bordering on DCIS. The 1.9% estimated upgrade rate at 5 years is in line with the reported upgrade rates in the literature. Han et al. 24 reported the results of active surveillance in 35 patients with ADH where one patient underwent surgical excision after 11 months with upgrade to invasive BC. Together with seven other studies of active surveillance of ADH, the combined risk of upgrade was 2.2% at the biopsy site and 4.4% at any site after median 49 months of follow-up. The authors suggest that active surveillance of ADH on needle biopsy is a safe option with the possibility of delayed surgery if the lesion progresses radiographically.
This study has some limitations. One is that neither the biopsy specimen nor the specimen from final surgery has undergone central pathological review. The evaluations have been made by pathologists not necessarily specialized in breast pathology, which reflects management of ADH in daily practice. Another limitation is that data on the proportion of microcalcifications removed by biopsy were not systematically documented and could thus not be included. According to Pena et al. and Nguyen et al., one of the prerequisites for refraining from surgery is that at least 50%–95% of all microcalcifications are removed in the biopsy. Furthermore, the mean time of follow-up is rather short, with some women having been followed for less than 1 year. This time is too short to preclude underestimation of carcinoma in women managed conservatively.
Conclusion
International guidelines recommend open surgery for ADH in the breast due to underestimation rates of malignancy of up to 14%. In this study, 101 biopsies of ADH were identified over a period of 10 years in two Swedish hospitals. Surgery was performed in less than half of the cases based on histopathological features in the biopsy specimen, hereditary factors, extent of microcalcifications, and after an MDT discussion. The upgrade rate to carcinoma was 37.5% in women undergoing surgery, but only 1.9% in women managed conservatively. Longer follow-up of non-surgical management of women with ADH may be needed to confirm these results. For future guidelines of ADH management, the suggested low-risk criteria and their reproducibility in different independent cohorts should be validated.
Footnotes
Author contributions
Charlotta Wadsten: Conceptualization, data collection, formal analyses, writing—original draft, review and editing. Gunilla Rask: Data collection, writing—review and editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a regional agreement between Umeå University and Region Västerbotten (ALF) and Visare Norr (grant nos. 931408 and 968146).
Data availability
Data from medical records are not publicly available as they contain sensitive information. Researchers may have access to data through the corresponding author under standard rules of protecting data integrity and existing ethics permissions.
