Abstract
Background and aims:
Numerous studies have reported superior outcome for patients with hepatocellular carcinoma (HCC) in non-cirrhotic compared to cirrhotic livers. This cohort study aims to describe the clinical presentation, disease course, treatment approaches, and survival differences in a population-based setting.
Methods:
Data on patients diagnosed with HCC in Sweden between 2008 and 2018 were identified and extracted from the Swedish Liver registry (SweLiv). Descriptive and survival statistics were applied.
Results:
Among the 4259 identified patients, 34% had HCC in a non-cirrhotic liver. Cirrhotic patients presented at a younger age (median = 64 vs 74 years, p < 0.001) and with a poorer performance status (Eastern Cooperative Oncology Group (ECOG) = 0–1: 64% vs 69%, p = 0.024). Underlying liver disease was more prevalent among cirrhotic patients (81% vs 19%, p < 0.001). Tumors in non-cirrhotic livers were diagnosed at a more advanced stage (T3–T4: 46% vs 31%) and more frequently with metastatic disease at diagnosis (22% vs 10%, p < 0.001). Tumors were significantly larger in non-cirrhotic livers (median size of largest tumor 7.5 cm) compared to cirrhotic livers (3.5 cm) (p < 0.001). Curative interventions were more commonly intended (45% vs 37%, p < 0.001) and performed (40% vs 31%, p < 0.001) in the cirrhotic vs non-cirrhotic patients. Median survival was 19 months (95% confidence interval (CI) = 18–21 months), in patients with cirrhosis as compared to 13 months in non-cirrhotic patients (95% CI = 11–15) (p < 0.001). In the multivariable Cox regression model, cirrhosis was not an independent predictor of survival, neither among curatively nor palliatively treated patients.
Conclusion:
These population-based data show that patients with HCC in a cirrhotic liver receive curative treatment to a greater extent and benefit from superior survival compared to those with HCC in a non-cirrhotic liver. The differences in survival are more attributable to patient and tumor characteristics rather than the cirrhotic status itself.
Clinical trial registration:
not applicable. Patient confidentially: not applicable.
Context and Relevance
Superior outcome is assumed for patients with HCC in a non-cirrhotic compared to cirrhotic livers. In this national registry–based study, we did not find any independent associations between underlying liver cirrhosis and survival. However, patients with HCC in a cirrhotic liver had more favorable tumor and patient characteristics compared to those without cirrhosis, leading to a greater likelihood of receiving treatment with a curative intent and resulting in superior survival outcomes. Therefore, we conclude that the differences in survival between cirrhotic and non-cirrhotic HCC patients depend on patient and tumor characteristics rather than the cirrhotic status itself.
Introduction
The incidence of hepatocellular carcinoma (HCC) is increasing, and currently, it is the sixth most common cancer worldwide and ranks third in terms of mortality. 1 While liver cirrhosis is the primary risk factor for HCC, a proportion of patients develop HCC in a non-cirrhotic liver. 2 The percentage of non-cirrhotic HCC varies considerably, ranging from 7% to 54%, depending on the geographical area and the underlying liver disease’s etiology. 3
Patients with non-cirrhotic HCC often present at a more advanced stage, potentially due to fewer symptoms in early stages of HCC, lack of surveillance in non-cirrhotic patients (except for those with chronic hepatitis B), and a higher hepatic reserve in this population. 4 Although non-cirrhotic HCC patients tend to differ from those with cirrhosis regarding age, tumor size, and treatment allocation, non-cirrhotic HCC has generally been associated with better survival.5–8 However, contradictory findings have been reported regarding the survival outcomes of non-cirrhotic compared to cirrhotic HCC. Some studies have found similar survival rates between the two groups despite differences in patient and tumor characteristics, as well as treatments.9–13 Conversely, a recently published population-based study, which included patients not receiving curative treatment, found that non-cirrhotic HCCs were associated with worse survival. 14
The majority of studies on HCC in the absence of cirrhosis have only included patients undergoing curative interventions, leading to inconsistent data regarding whether cirrhosis itself is an independent prognostic factor for survival.5,9 In addition, many studies reporting patient data and survival outcomes originate from tertiary referral centers, which may not be representative of the general population. 6
Therefore, this study aims to address these knowledge gaps by utilizing a population-based national cohort to compare the clinical presentation, treatment modalities, and survival differences between patients with HCC in cirrhotic versus non-cirrhotic livers.
Methods
Study population and definition of study cohort
The Swedish National Registry for Cancer in the Biliary Tract and Liver (SweLiv) was introduced in 2008 and includes all primary and secondary tumors of the liver as well as all liver resections and ablations for primary cancers and/or metastatic disease. 14 The registry has a 95.6% nationwide coverage for inclusion compared with the Swedish Cancer Register. 15 Data in SweLiv are registered prospectively in four modules, the first of which includes data on diagnosis, staging, and treatment recommendations. The second module includes treatment-related information of curative treatments, that is, transplantation, hepatic resection, or local ablative therapy, and the third consists of data on complications and pathology following these treatments. The fourth module, launched in 2014, includes data on recurrence.
All patients registered with a diagnosis of HCC (ICD-10 code C22.0) between 1 January 2008 and 31 December 2018 were extracted from SweLiv. The variable “cirrhosis or non-cirrhosis” (or unknown cirrhotic status) is accounted for in a click box in the first module. In the second module, which includes data on patients undergoing a curative intervention, an additional variable “cirrhotic liver at clinical evaluation” was used to re-categorize cirrhotic status based on a clinical evaluation at the intervention. Patients with cirrhotic status not registered in the first module were excluded from further analysis if clinical re-evaluation did not re-categorize the patients as having either cirrhotic or non-cirrhotic liver. Non-cirrhotic patients undergoing more than one intervention, and who were categorized as cirrhotic in the subsequent intervention, were re-classified from non-cirrhosis to cirrhosis.
Collected variables
Patient characteristics such as sex, age, performance status (Eastern Cooperative Oncology Group (ECOG) score), presence of diabetes, and the underlying liver disease were extracted from the registry. Tumor size (the longest diameter of the largest tumor), number of tumors (based on radiologically detected tumors at diagnosis), and Tumor-Node-Metastases (TNM) stage were also extracted from the registry. Barcelona Clinic Liver Cancer (BCLC) was determined based on registry data, that is, number and size of tumors, Child–Pugh score, ECOG, and extrahepatic involvement.
Treatment allocation
Treatment recommendations at the multidisciplinary therapy conference are documented in the first module of SweLiv and include the curative modalities transplantation, hepatic resection, or local ablative therapy, as well as the palliative treatments transarterial chemoembolization (TACE) and systemic treatment (sorafenib or lenvatinib), or best supportive care (BSC). The provided curative treatments are described in detail in the registry, whereas for the palliative options, only the allocated treatment was registered, without data on treatment details or results. The same treatment categorization used in previous publications on HCC from SweLiv was applied on this study cohort.14,15 The patients were categorized to the following treatments: transplantation if the patient as any intervention had a liver transplant; resection if the patient underwent resection before local ablative therapy; local ablative therapy if the patient underwent ablation before resection; TACE if the patient was planned for TACE and did not undergo any curative interventions; systemic treatment if the patient was planned for systemic treatment and did not undergo any curative intervention; or BSC if the patient was not planned for any active treatment and not registered as undergoing any intervention during the study period. If patients underwent both curative and palliative treatments, it was the curative treatment that was registered.
In Sweden, a comprehensive national treatment program is in place for HCC patients, featuring a tailored treatment algorithm closely aligned with the BCLC algorithm. 16 It diverges in three key facets: first, the consideration of resection extends to patients with non-solitary lesions; second, assessment of ECOG is based on current performance status, including baseline symptoms, not only tumor-related symptoms and ECOG 2 can be accepted for curative treatment after rigorous assessment at the multidisciplinary therapy conference; third, the selection criteria for transplantation is based on the University of California San Francisco criteria. Throughout the study duration, the resection criteria have demonstrated relatively consistent constancy.
Study endpoints
The primary endpoint was overall survival (OS) calculated from the date of diagnosis of HCC for palliatively treated patients and from date of treatment in curatively treated to the date of death or last date for survival status update (1 October 2019). Secondary endpoints included comparisons of patient and tumor characteristics, and treatments that were provided to patients with or without cirrhosis, respectively. Ethical approval was obtained from the regional ethical review board, approval no. 2017/2267-31/2, who deemed informed consent could be waived due to the registry-based nature of the study.
Statistical analysis
Categorical patient and tumor characteristics were summarized as proportions and frequencies and compared using Pearson’s chi-square test or Fisher’s exact test. Non-normally distributed continuous data were presented as median values (min, max) and compared using the Wilcoxon rank-sum test.
Univariable and multivariable Cox proportional hazard regression was used to assess factors associated with survival and presented as hazard ratios (HRs) with 95% CIs. Patient characteristics not missing at random were excluded as potential variables in the regression model (Supplemental Table 1) as was variables with missing data of >25%. Variables were included in the multivariable model if p < 0.20 in the univariable and univariable survival estimates were illustrated with Kaplan–Meier curves and compared using log-rank test. p < 0.050 was considered statistically significant. STATA version 15.0 (StataCorp, Collage Station, TX, USA) was used for all data analyses.
Results
Patient and tumor characteristics in cirrhotic versus non-cirrhotic liver
After re-categorization of cirrhotic status, as illustrated in Fig. 1, the final study cohort consisted of 4259 patients, 2828 patients (66%) with HCC in a cirrhotic liver and 1431 patients (34%) having HCC in a non-cirrhotic liver. The diagnosis of HCC was based on imaging findings in 80% (n = 2270) of the cirrhotic patients (EASL/AASLD criteria) and in 54% (n = 776) of the non-cirrhotic patients if clinical signs demonstrated chronic liver disease or alpha fetoprotein (AFP) levels were elevated. Among those without cirrhosis, HCC was confirmed by a liver biopsy and/or cytology in 44% (n = 635) of cases.
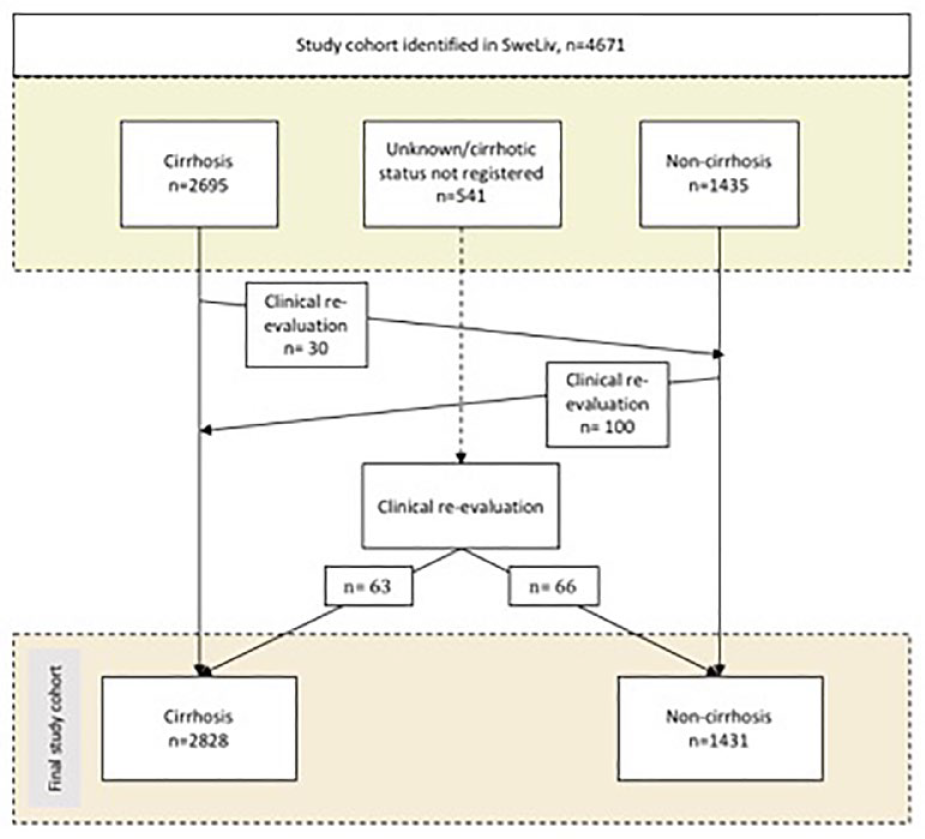
Flowchart illustrating the identification of patients from the Swedish Liver registry according to cirrhosis or non-cirrhosis status, re-categorization, and the final study cohort.
The baseline characteristics of all patients are summarized in Table 1. A higher proportion of females were diagnosed with HCC in a non-cirrhotic liver compared to a cirrhotic liver (30% vs 22%, p < 0.001). Underlying liver disease was more frequently present in those with cirrhosis, with 81% (n = 2291) having an underlying liver disease compared to 19% (n = 272) in the non-cirrhotic group. Hepatitis C was the most common underlying liver disease in both non-cirrhotic and cirrhotic patients. Non-cirrhotic patients were more likely to be positive for hepatitis B virus (HBV), while patients with cirrhosis were more likely to have alcohol-related liver disease.
Patient and tumor characteristics in 4259 patients with hepatocellular carcinoma.
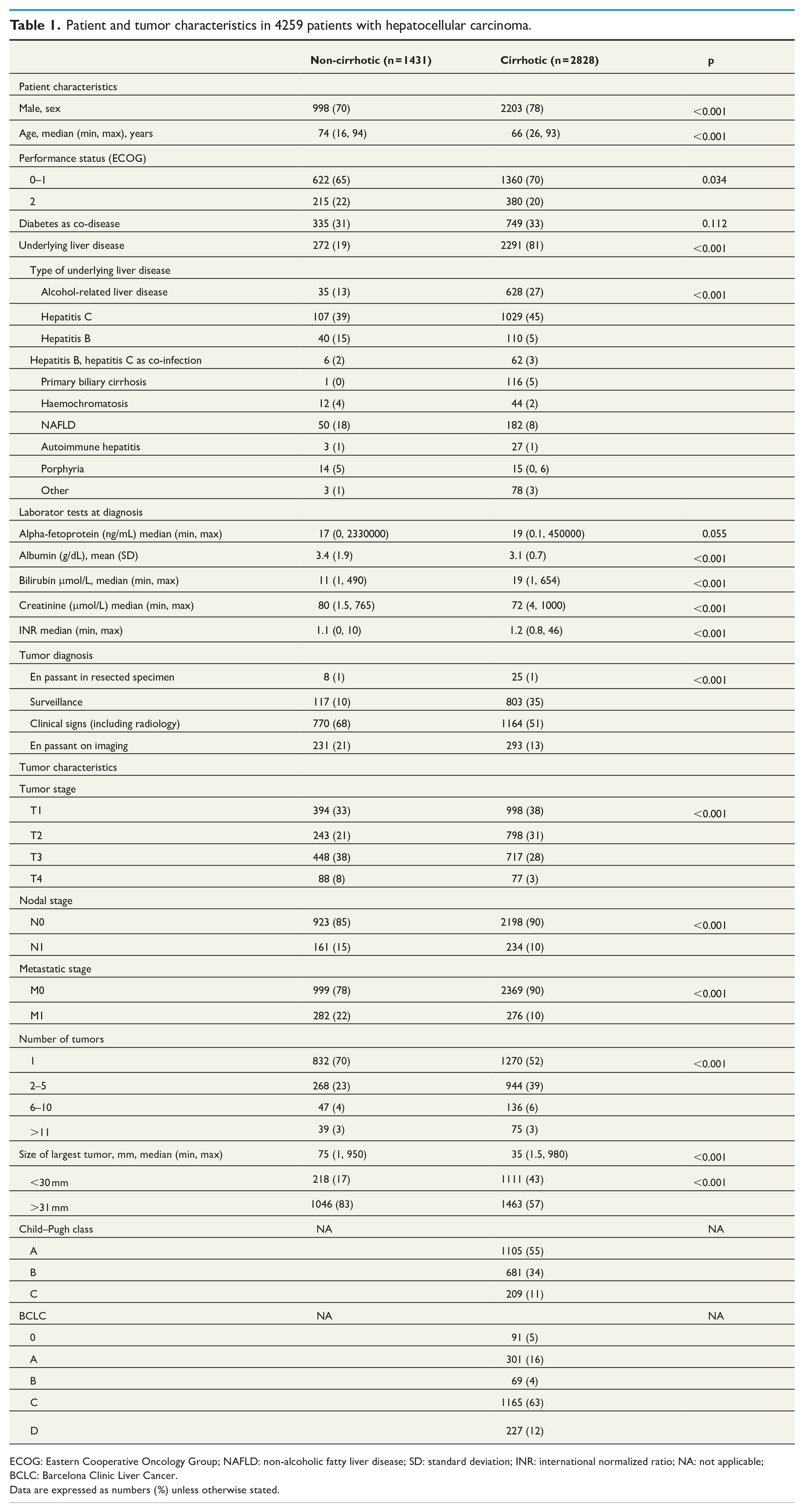
ECOG: Eastern Cooperative Oncology Group; NAFLD: non-alcoholic fatty liver disease; SD: standard deviation; INR: international normalized ratio; NA: not applicable; BCLC: Barcelona Clinic Liver Cancer.
Data are expressed as numbers (%) unless otherwise stated.
Non-cirrhotic patients presented with a lower number of tumors (a single tumor in 70% vs 52% in cirrhotic patients, p < 0.001), larger tumors (median size of largest tumor 7.5 cm vs 3.5 cm, p < 0.001), and at a more advanced TNM stage, with 22% having metastatic disease at diagnosis compared to 10% in those with cirrhosis.
Recommended treatment, actual treatment outcome, and re-interventions
A significantly greater proportion of cirrhotic patients, compared to those without cirrhosis, were recommended (45% vs 37%, p < 0.001) and subsequently underwent (40% vs 31%, p < 0.001) curative therapy, as outlined in Table 2. If curatively treated, cirrhotic patients underwent transplantation and ablation in 29% and 44%, respectively, while 85% of non-cirrhotic patients had a hepatic resection (Table 2). Cirrhotic patients more often underwent two or more interventions compared to those without cirrhosis (29% vs 12%, p < 0.001).
Recommended treatment at multidisciplinary team conference and actual curative treatments of HCC in non-cirrhotic versus cirrhotic patients.
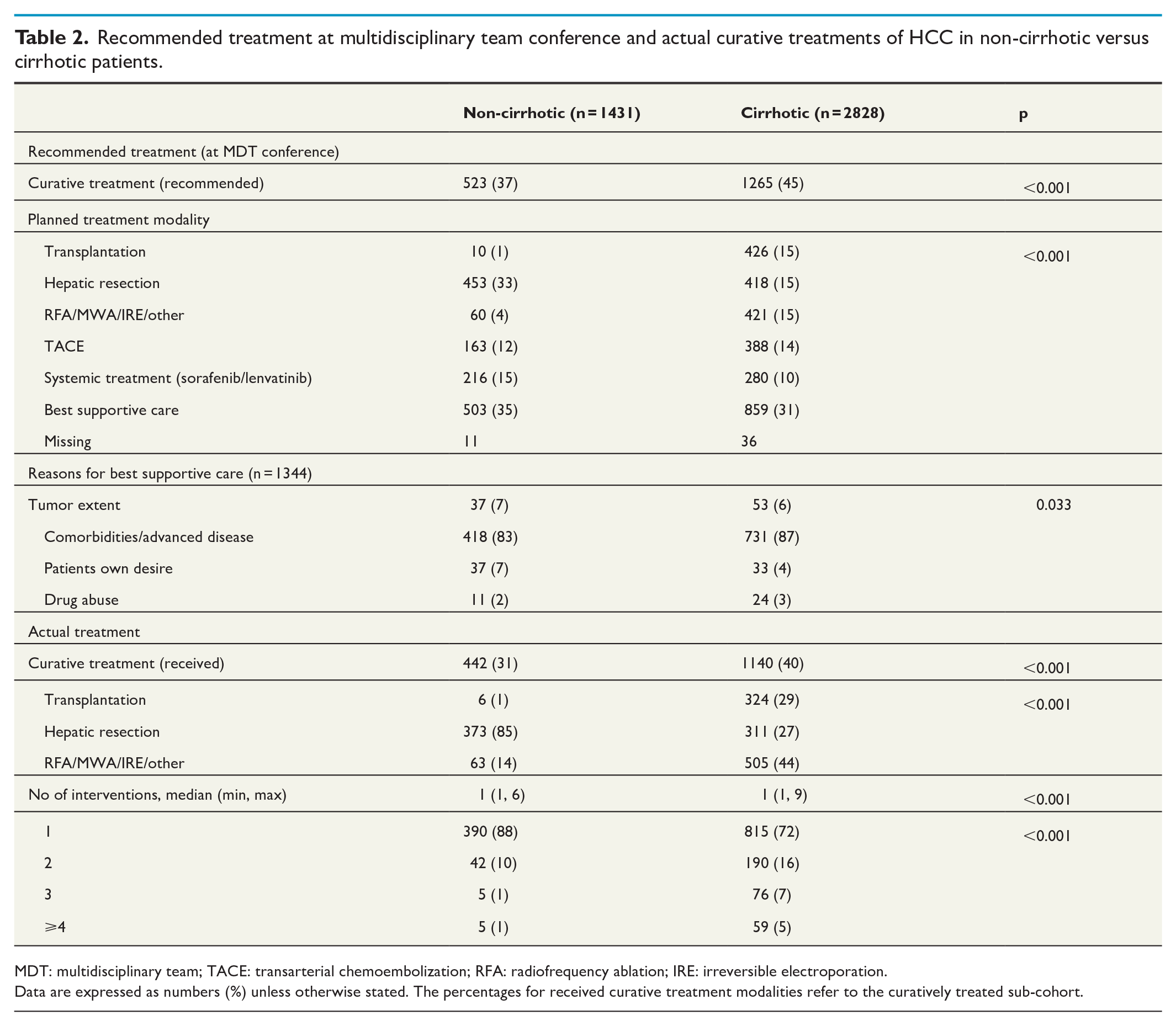
MDT: multidisciplinary team; TACE: transarterial chemoembolization; RFA: radiofrequency ablation; IRE: irreversible electroporation.
Data are expressed as numbers (%) unless otherwise stated. The percentages for received curative treatment modalities refer to the curatively treated sub-cohort.
Survival outcome
After a median follow-up time of 14.6 months from diagnosis, the estimated median survival was 13.1 months (95% CI = 11.5–14.6 months) and 19.2 months (95% CI = 17.8–20.8 months) for non-cirrhotic and cirrhotic patients, respectively, p < 0.001, irrespective of treatment strategy (Fig. 2).
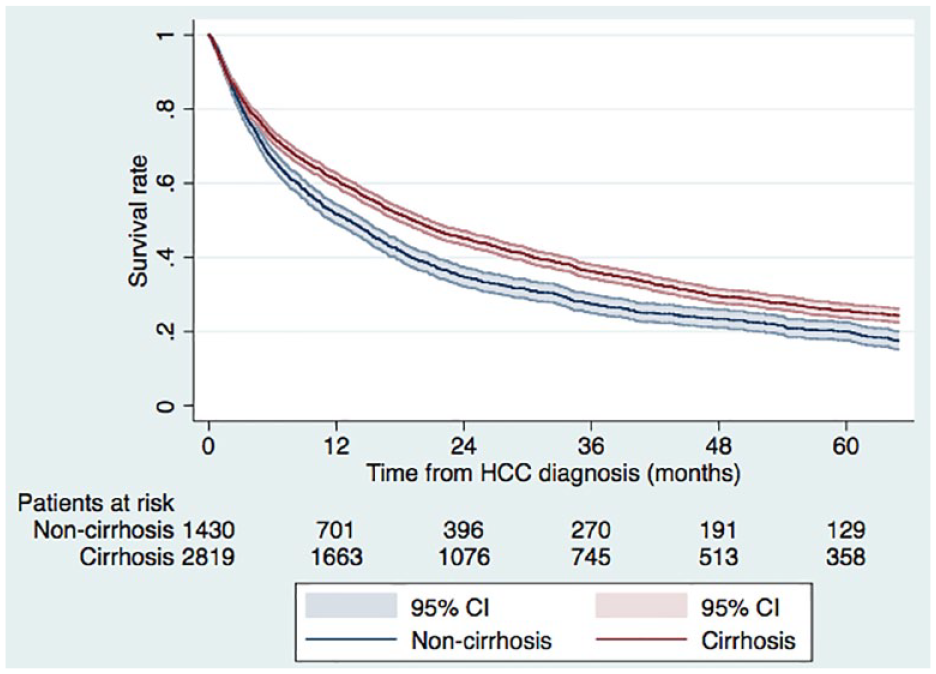
Survival among patients with hepatocellular carcinoma depending on cirrhotic status. The estimated 1- and 5-year overall survival, irrespective of a curative or non-curative treatment approach, was 50.7% (95% CI = 49.1%–54.3%) and 20.0% (95% CI = 17.7%–22.5%), respectively, among non-cirrhotic patients and 60.1% (95% CI = 59.1%–62.7%) and 25.6% (95% CI = 23.8%–27.5%), respectively, in cirrhotic patients, log-rank test p < 0.001. Survival status was missing in 10 patients, 1 in the non-cirrhotic group and 9 in the cirrhotic group.
In patients who received curative treatment (transplantation at any time-point, hepatic resection or ablation), the estimated median survival among cirrhotic patients was 66.2 months (95% CI = 58.0–78.7 months) compared to 57.1 months (9% CI = 48.0–65.9 months) in non-cirrhotic patients, p = 0.081 (Fig. 3A). Factors associated with OS in the subgroup of curatively treated patients are depicted in Table 3. Cirrhotic status was not significantly associated with survival (HR = 1.23; 95% CI = 0.89–1.70) while age, performance status (ECOG), tumor size, number of tumors, and type of curative treatment were (Table 3).
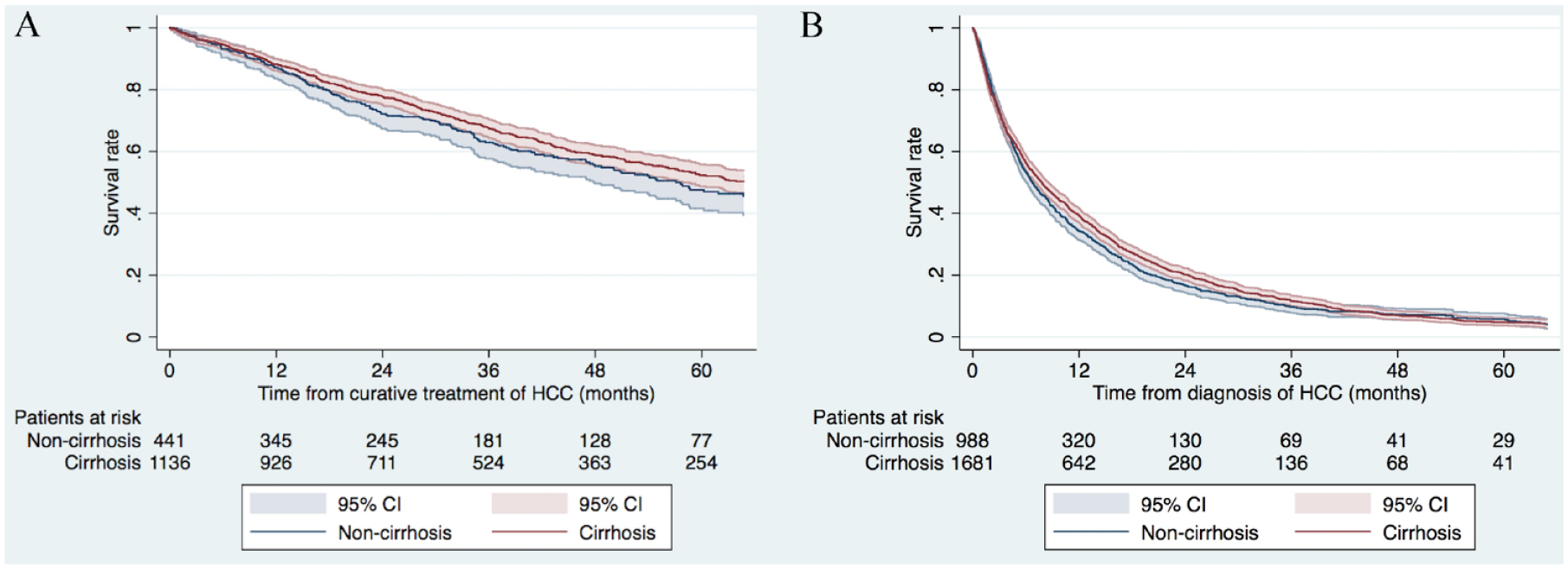
Survival among patients with hepatocellular carcinoma depending on treatment intention and cirrhotic status. (A) The estimated 1- and 5-year overall survival from date of intervention in curatively treated patients was 87.2% (95% CI = 83.6%–90.0%) and 47.6% (95% CI = 41.6%–53.4%), respectively, in the non-cirrhosis group and 88.2% (95% CI = 86.2%–90.0%) and 52.3% (95% CI = 48.7%–55.8%), respectively, in patients with cirrhosis, log-rank test p = 0.081. (B) Correspondingly, the estimated 1- and 5-year overall survival in non-curatively treated patients was 34.4% (95% CI = 31.4%–37.4%) and 5.7% (95% CI = 4.2%–7.6%) in the non-cirrhosis group and 39.2 (95% CI = 36.9%–41.6%) and 4.8% (95% CI = 3.6%–6.2%), respectively, in patients with cirrhosis, log-rank test p = 0.108.
Cox proportional hazard regression analysis on factors associated with survival in patients with hepatocellular carcinoma treated with a curative intent.
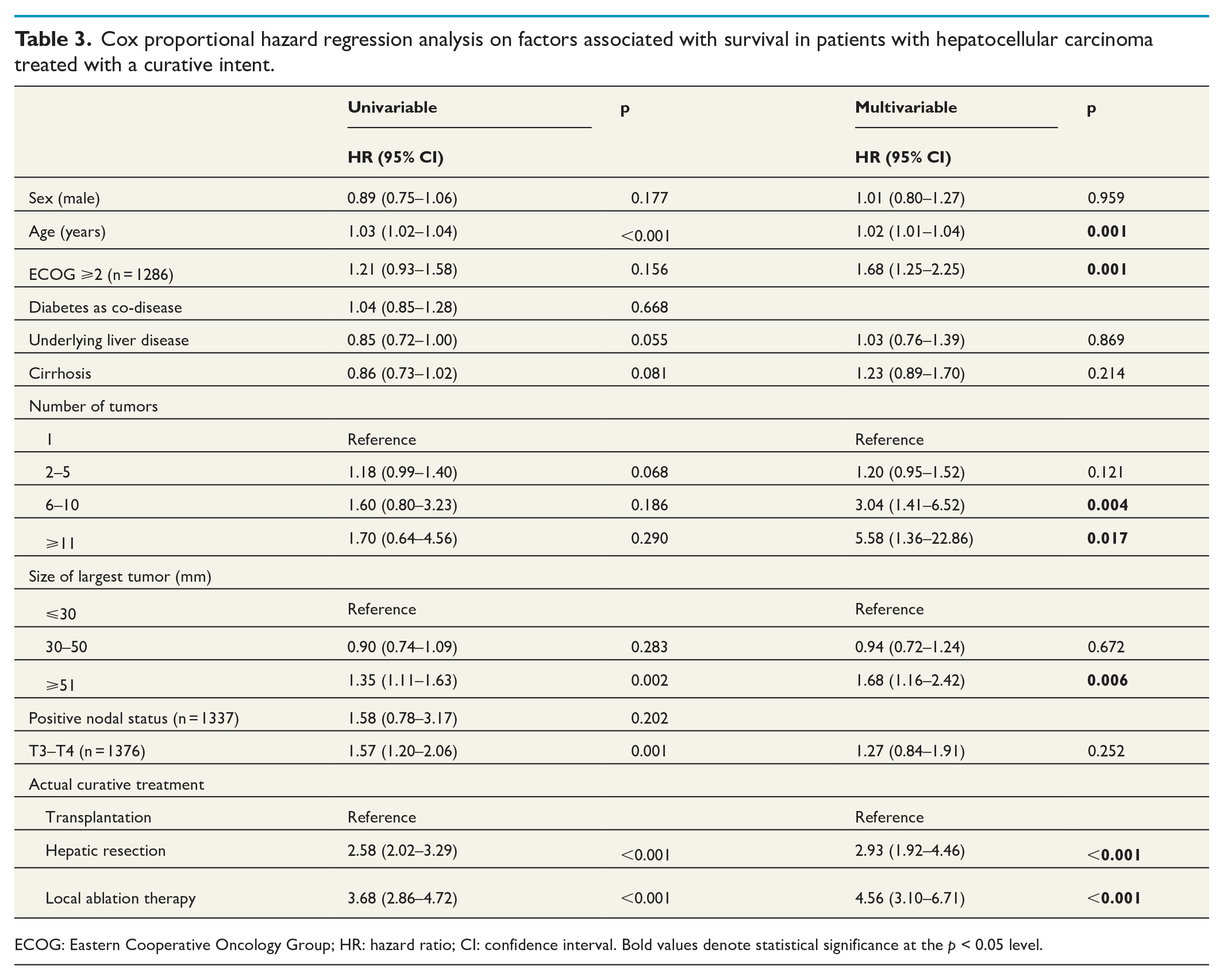
ECOG: Eastern Cooperative Oncology Group; HR: hazard ratio; CI: confidence interval. Bold values denote statistical significance at the p < 0.05 level.
In patients receiving palliative treatment (TACE or systemic therapy) or BSC, the estimated median survival was similar in those without cirrhosis (6.8 months, 95% CI = 6.0–7.5 months) compared to those with cirrhosis (7.8 months, 95% CI = 7.1–8.6 months), p = 0.108 (Fig. 3B).
The Cox regression analysis of the palliatively treated cohort was limited to a few variables since values from the registry partly depended on cirrhotic status and whether curative treatment was applied or not (Supplemental Table 1). Given these limitations, cirrhotic status remained a non-significant factor for survival (HR = 1.08, 95% CI = 0.95–1.22) while a high number of tumors and tumor size larger than 5 cm were significant predictors of survival (Table 4).
Cox proportional hazard regression analysis on factors associated with survival in palliatively treated patients with hepatocellular carcinoma.
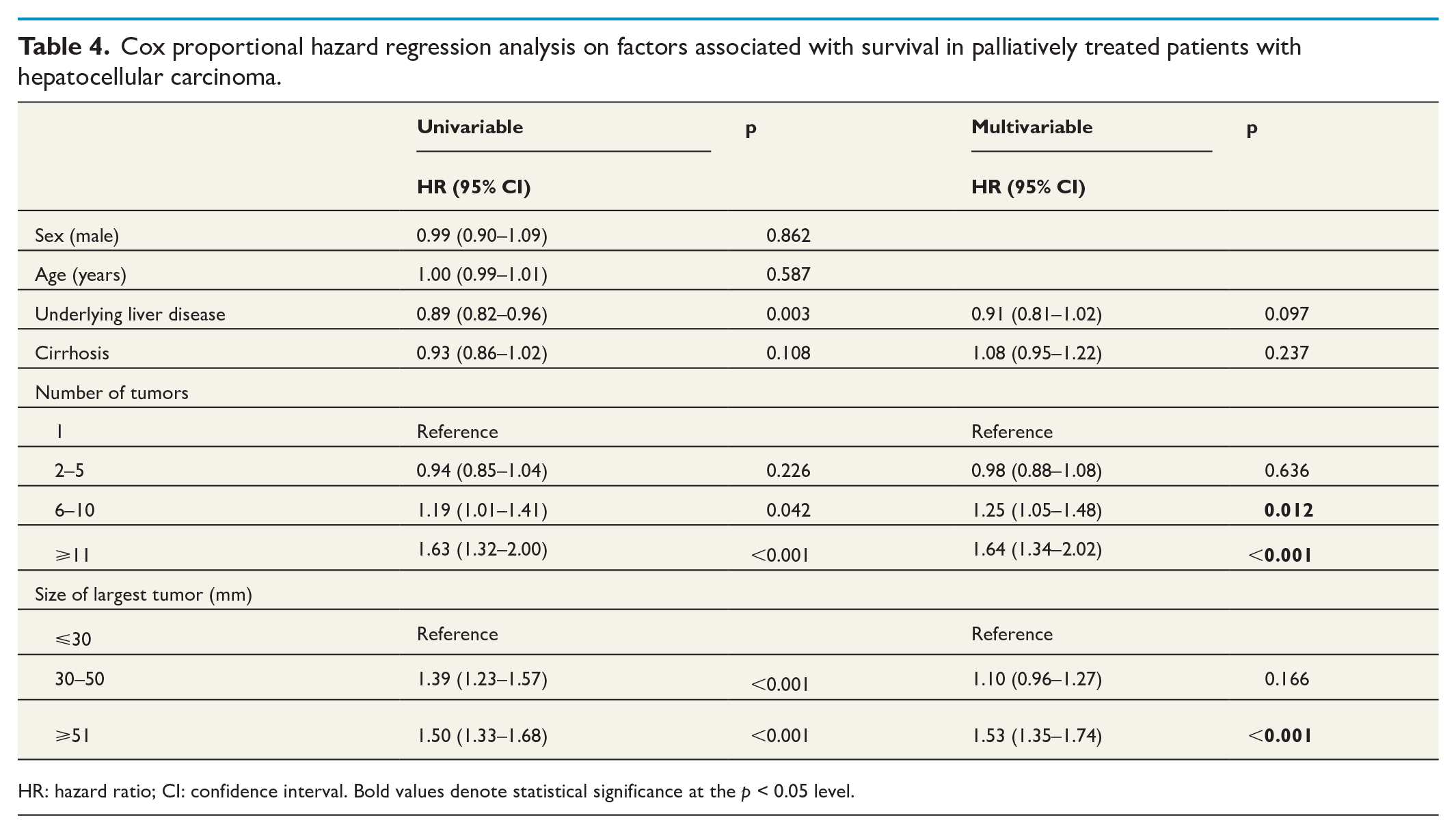
HR: hazard ratio; CI: confidence interval. Bold values denote statistical significance at the p < 0.05 level.
OS of cirrhotic and non-cirrhotic patients in the subgroup that underwent liver resection
Patient characteristics of the subgroup of patients who underwent resection are depicted in Supplemental Table 2. The patients with cirrhosis were younger, more often diagnosed with an underlying liver disease, had a less advanced tumor stage, smaller tumor size at diagnosis, and more often multiple tumors compared to the non-cirrhotic patients. Despite these differences, the Cox regression analysis showed no association between survival and cirrhotic status, number and size of tumors, while ECOG was associated with OS in this sub-cohort (Supplemental Table 3). The estimated median survival among resected patients was 58.6 months (95% CI = 48.0–79.7 months) and 59.8 months (95% CI = 45.3–83.4 months) for non-cirrhotic and cirrhotic patients, respectively, p = 0.896 (Supplemental Fig. 1).
Discussion
This nationwide population-based study examined an unselected cohort of patients diagnosed with HCC and revealed a higher proportion of non-cirrhotic HCC cases (33.6%) than anticipated. Furthermore, patients with HCC in a cirrhotic liver were more likely to receive curative treatment and experienced better survival compared to those with HCC in a non-cirrhotic liver. Regression analysis demonstrated that these differences were primarily due to variations in patient and tumor characteristics rather than the cirrhotic status itself. In fact, cirrhotic status did not emerge as an independent prognostic factor for survival among curatively treated patients nor among palliatively treated patients, possibly due to the higher number of curative treatments in the cirrhotic as compared to the non-cirrhotic group.
The proportion of patients with HCC in a non-cirrhotic liver varies across different geographic regions worldwide. A comprehensive review concluded that 7%–54% of HCCs develop without underlying cirrhosis. 3 Most studies report the proportion of HCC in non-cirrhotic livers to be below 30%,6–8,17 which contrasts with the 33.6% observed in this study. Among the US veterans, who are predominantly male, 13% of the HCC cases developed in patients without evidence of cirrhosis. 17 In a Western surgical cohort, 27% had HCC in non-cirrhotic liver, 9 while non-cirrhotic HCC was observed in 37.1% in a hospital cohort from South Korea. 12 A higher proportion of HCC in non-cirrhotic patients (37%) was reported in a subpopulation of patients with non-alcoholic fatty liver disease (NAFLD) 10 and in a single-center study from the Netherlands. 13 It is important to note that most studies on HCC in the absence of cirrhosis have included patients treated with a curative intent, either resection or liver transplantation, or have been conducted at tertiary referral centers, which may introduce selection bias.9,18
In this study, the slightly higher proportion of patients with HCCs in non-cirrhotic livers compared with some previous studies may be partly attributed to the population-based nature of this cohort, which minimizes selection bias. However, it is important to acknowledge the possibility that occasional patients with cirrhosis or advanced fibrosis may have been misclassified as non-cirrhotic.
An explanatory model for the high frequency of cirrhosis in HCC patients from Western countries has been chronic hepatitis C virus (HCV) and alcohol-related liver disease as underlying etiologies, whereas chronic HBV predominates in Asian countries. 19 However, with the advent of effective antiviral treatments against hepatitis C, together with the obesity epidemic, the panorama of underlying liver diseases in Western HCC is shifting toward an increased incidence of NAFLD. 20 In a recent study, cirrhosis was present in only 58.3% of patients with non-alcoholic steatohepatitis–related HCC. 21 This may have implications on screening guidelines and highlights the importance of obtaining a better understanding of the carcinogenic pathogenesis in fatty liver disease without cirrhosis.22–24
The treatment algorithm for HCC includes thermal ablation, liver resection, and transplantation as curative treatment options.25,26 The choice of surgical therapy depends on the extent of liver dysfunction and tumor burden (size and number of tumors), and presuming the absence of extrahepatic disease, and the appropriate treatment is decided at a multidisciplinary therapy conference. Our study revealed that the presence of liver cirrhosis strongly influenced treatment choices, with resection more commonly applied in non-cirrhotic patients, while transplantation or local ablative therapy was more frequently used in cirrhotic patients. In our cohort, a significantly higher proportion of cirrhotic compared to non-cirrhotic patients were recommended (45% vs 37%) and subsequently underwent curative therapy (40% vs 31%). Thus, future efforts should attempt to identifying patients at risk of HCC development in non-cirrhotic livers to diagnose the tumors at a treatable stage, targeting individuals with, for example, NAFLD and advanced fibrosis, or additional risk factors such as diabetes, obesity, tobacco smoking, or alcohol overconsumption. Non-cirrhotic patients in our study cohort were more likely to present with a larger overall tumor burden, consistent with findings from other studies. 9 In addition, one-fifth of our non-cirrhotic patients had metastatic disease at time of diagnosis, which aligns with the results from Schütte et al. 11 but is higher than previously reported. 27 The higher proportion of patients with metastatic disease was expected due to the population-based nature of our study, as patients with metastatic disease are less likely to be referred to tertiary centers for evaluation and appropriate treatment, and this may not be included in previously published surgical cohorts.
From other studies, we know that non-cirrhotic HCC patients tend to have more recurrences after resection compared to cirrhotic patients, likely due to factors such as the size of the resected tumor and the higher rates of transplantation as treatment option among cirrhotic patients.5,9 In our cohort, cirrhotic patients had more repeat interventions, indicating that recurrences in non-cirrhotic patients were less likely to be treated with a curative intent, which may be an effect of the larger tumor burden in this group. However, the limited data on recurrences from the registry prevented us from conducting a more detailed analysis on this topic.
Although one of the main findings of this study was that cirrhotic patients had a superior survival in this population-based cohort, cirrhosis itself was not an independent prognostic factor of survival. The observed survival benefit was attributed to differences in patient and tumor characteristics rather than cirrhotic status alone. This was true also in the subgroup of resected patients, where cirrhosis did not independently predict survival, probably reflecting the selection process that preceded the decision of resection in a cirrhotic liver. Several other studies have reported opposite results, showing a survival benefit in non-cirrhotic HCC patients.5–8 However, few other studies that compared differences in survival between cirrhotic and non-cirrhotic HCC patients included cirrhotic status as a variable in a regression model. In one of these studies, the presence of cirrhosis in NAFLD-HCC was not independently associated with mortality, 10 which aligns with our findings. In contrast, the absence of cirrhosis was associated with lower mortality in the multivariable analysis in the curatively treated cohort in the study by Van Meer et al., 6 in which only 19% had non-cirrhotic HCC.
Like all other studies on this topic, this study was retrospective in nature. One limitation is the uncertainty of non-cirrhotic status obtained from the registry. Some patients categorized as non-cirrhotic may have had advanced fibrosis (stage 3) or well-compensated cirrhosis. To minimize this bias, measures such as re-classification after the surgical procedure were taken, although the absence of misclassifications cannot be guaranteed. Another limitation is the restricted information on details on each individual patient in the non-curatively treated group and regarding recurrence. In addition, we recognize the presence of immortality bias in our survival estimates derived from the time of diagnosis of HCC (Fig. 2), given that patients undergoing curative treatments inherently survive for a duration sufficient to receive the intended intervention and potential subsequent re-interventions. To mitigate the immortality bias that would arise from estimating survival based on the date of diagnosis across all patients, separate regression analyses were conducted on the curatively and palliatively treated patients. Due to SweLiv’s focus on curative treatment of liver tumors, variable completeness relies on whether curative treatment was performed or not. As non-cirrhotic patients underwent curative treatments less frequently, essential tumor characteristics like T- and N-stage were more often missing among non-cirrhotic patients. Consequently, the regression analysis was constrained by this reduced set of variables.
The major strength of the study is the large number of patients and the population-based setting, which includes both curatively and palliatively treated patients, making the results generalizable to a Western cohort with similar risk factors. In this population-based study on patients with HCC, we did not find any independent associations between underlying liver cirrhosis and survival. However, patients with HCC in a cirrhotic liver had more favorable tumor and patient characteristics compared to those without cirrhosis, leading to a greater likelihood of receiving treatment with a curative intent and resulting in superior survival outcomes. Therefore, the differences in survival between cirrhotic and non-cirrhotic HCC patients depend on patient and tumor characteristics rather than the cirrhotic status itself.
Supplemental Material
sj-docx-1-sjs-10.1177_14574969231220179 – Supplemental material for Hepatocellular carcinoma in cirrhotic versus non-cirrhotic liver: Treatment and survival differences in a nationwide cohort
Supplemental material, sj-docx-1-sjs-10.1177_14574969231220179 for Hepatocellular carcinoma in cirrhotic versus non-cirrhotic liver: Treatment and survival differences in a nationwide cohort by Jennie Engstrand, Per Stål, Stefan Gilg, Anders Jansson and Cecilia Strömberg in Scandinavian Journal of Surgery
Footnotes
Author contributions
A.J. and C.S. contributed to conceptualization. J.E., A.J., and C.S. contributed to data curation. J.E. contributed to formal analysis. J.E., P.S., S.G., A.J., and C.S. contributed to methodology. P.S. contributed to supervision. J.E. and P.S. contributed to writing—original draft. J.E., P.S., S.G., A.J., and C.S. contributed to writing—review and editing. All authors have read and agreed to the submitted manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: J.E. was supported by Region Stockholm (Clinical Postdoctoral Appointment) and the Bengt Ihre Research Fellowship and P.S. by the Swedish Cancer Society (20 1355 PjF) and Region Stockholm (ALF FoUI-961954). None of the funding sources had any role in the design and conduct of the study and in the collection, management, and analysis of the data or in the preparation, review, and approval of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
