Abstract
Background and objective:
Barrett’s esophagus (BE) is associated with an increased risk of esophageal adenocarcinoma. The use of radio frequency ablation (RFA) for complete eradication of BE with confirmed low-grade dysplasia (LGD) or high-grade dysplasia (HGD) has been promising in multicenter trials. Our aim was to evaluate the safety and efficacy outcomes associated with RFA for patients with BE and LGD/HGD in a single-center setting.
Methods:
This was a retrospective single-center study conducted at Department of Surgery and Transplantation, Rigshospitalet, Denmark. Data were collected from all patients who had undergone RFA for LGD or HGD from January 2014 to December 2018. Effectiveness outcomes were based on histology: complete eradication of dysplasia (CE-D), defined as all esophageal biopsies being negative for dysplasia at the last biopsy session, and complete eradication of intestinal metaplasia (CE-IM) defined as esophageal biopsies being without intestinal metaplasia. Safety outcomes were based on the proportion of complications to the RFA treatment.
Results:
A total of 107 patients were identified during the follow-up period (75% men, median age = 65 years); 83% had LGD and 17% had HGD. The median follow-up was 25 months. After the last RFA treatment, CE-D was achieved in 89%. CE-D and CE-IM were achieved in 60%. Complications occurred in 6.5% of the patients.
Conclusions:
In patients with BE and confirmed LGD or HGD, RFA was associated with a high rate of CE-D and a low risk of complications. The observed safety and efficacy outcomes were comparable with those previously reported in multicenter trials, showing that the Danish treatment of BE with LGD and HGD is comparable with those of larger European expert centers.
Keywords
Context and relevance
The use of radio frequency ablation (RFA) for complete eradication of Barrett’s esophagus (BE) with confirmed low-grade dysplasia (LGD) or high-grade dysplasia (HGD) has been promising in multicenter trials. In this retrospective single-center study, patients with BE and confirmed LGD or HGD, RFA was associated with a high rate of complete eradication of dysplasia and a low risk of complications. The observed safety and efficacy outcomes were comparable with those previously reported in multicenter trials, showing that the Danish treatment of BE with LGD and HGD is comparable with those of larger European expert centers.
Introduction
Barrett’s esophagus (BE) is defined as a metaplastic change in the esophageal epithelium, ⩾1 cm proximal to the gastroesophageal junction (GEJ). The normal squamous epithelium is replaced by intestinal metaplasia (IM), a columnar epithelium with the presence of goblet cells.1–4 The prevalence of BE in the general adult population is estimated to be 1.6%. For patients with gastroesophageal reflux disease (GERD), the prevalence of BE is 2.3%. 2 BE is diagnosed by endoscopic biopsy and categorized histologically according to the absence or presence and severity of dysplasia as either non-dysplastic IM, indefinite for dysplasia, low-grade dysplasia (LGD), high-grade dysplasia (HGD), or adenocarcinoma.3–5
BE is strongly associated with esophageal adenocarcinoma (EAC). BE patients with verified LGD have a significantly higher risk of progression to HGD or EAC if not regressing spontaneously. The risk of EAC in BE patients with HGD is 7% per year.1,3,6–8
Available treatment modalities of BE include medical, endoscopic, and surgical techniques. Medical therapy with proton-pump inhibitors and endoscopic surveillance are recommended for patients with non-dysplastic BE as the risk of progression is very low.
In recent years, the treatment of choice for patients with BE and LGD or HGD has become endoscopic resection of any nodular lesions (either by endoscopic mucosal resection (EMR) or endoscopic submucosal dissection (ESD)), followed by radio frequency ablation (RFA). The aim is to eliminate the dysplastic Barrett’s epithelium, replacing it with a normal squamous epithelium. 5
Clinical trials have demonstrated a significantly reduced risk for malignant progression using RFA compared with endoscopic surveillance for patients with BE and LGD/HGD.9–13 In multicenter trials, RFA appears to result in a complete eradication rate >90% and the rate of complications related to RFA appears to be low.
The aim of this study is to evaluate the efficacy and safety outcomes associated with RFA for patients with BE with LGD/HGD in a single-center setting. Furthermore, it is to compare the findings to previous studies and guidelines for the treatment of BE with LGD/HGD.
Methods
Study design
This is a retrospective single-center study conducted at Department of Surgery and Transplantation, Rigshospitalet, Denmark. Data were collected from all patients who had undergone RFA treatment of BE with LGD or HGD from January 2014 to December 2018.
Patients
Study participants gave informed consent prior to study inclusion. All patients underwent confirmatory endoscopy with esophageal biopsies. Biopsies were evaluated by two independent pathologists using internationally accepted standards for BE. We included patients with histologically confirmed IM and either LGD or HGD who had received treatment of RFA. Patients with progression to early esophageal cancer during treatment were counted as failures. All patients received a daily double dose proton-pump inhibitor. Patient characteristics are shown in Table 1.
Patient characteristics.
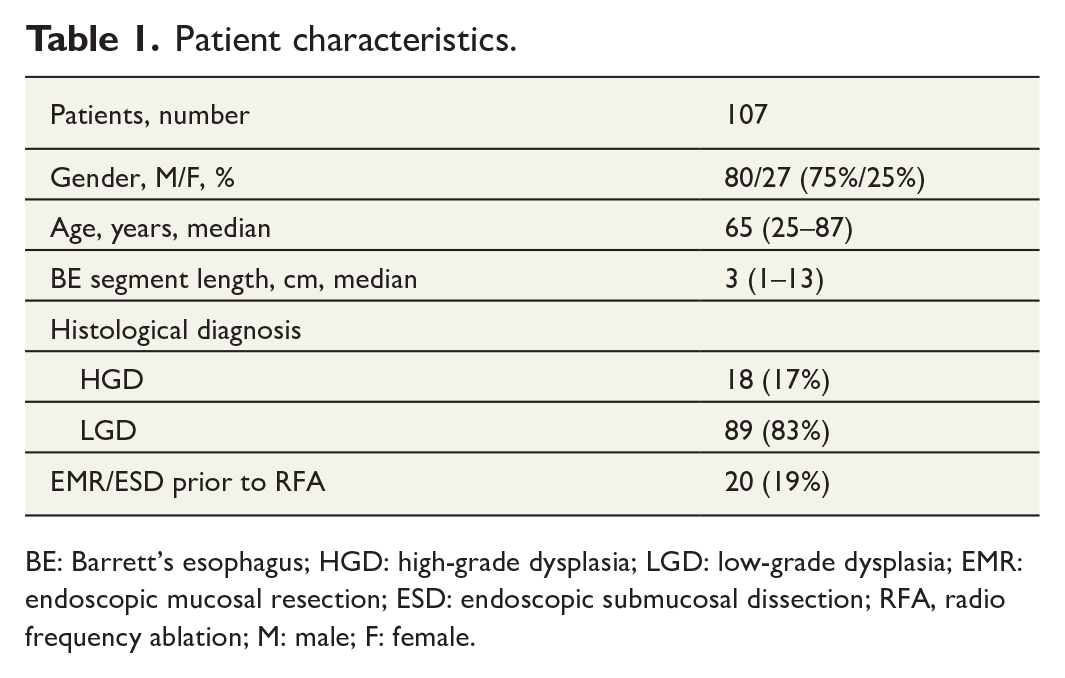
BE: Barrett’s esophagus; HGD: high-grade dysplasia; LGD: low-grade dysplasia; EMR: endoscopic mucosal resection; ESD: endoscopic submucosal dissection; RFA, radio frequency ablation; M: male; F: female.
Endoscopic treatment
Patients with elevated nodules underwent EMR prior to RFA treatment. An additional endoscopy with biopsies was performed for these patients to confirm remaining dysplasia before starting RFA.
Endoscopic circumferential RFA was performed using the Medtronic Barrx™ 360 express RFA balloon catheter (Medtronic, Minneapolis, MN, USA). Patients treated before 2016 were treated with the prior HALO device. The balloon catheter is self-adjusting, and the energy mode was placed at standard setting. The BE segment was cleaned with an acetylcysteine solution prior to treatment. The balloon catheter was introduced over a guidewire, and the BE segment was ablated. The ablation zone was cleaned with a cap, and a second ablation was performed. If necessary, circumferential lesions were treated with repeated circumferential ablation.
For minor lesions, the Medtronic Barrx™ channel endoscopic catheter was used through the working channel of the endoscope. The Barrx™ 90 focal catheter was occasionally used to treat larger areas. The patients underwent 2 × 2 ablations per session. BE segments were prepared with acetylcysteine solution and ablated twice using a standard setting. Again, the ablated area was cleaned and treated twice more. The patients underwent repeated endoscopy and treatment every third month until eradication of visible BE was achieved. When there was no sign of BE, new biopsies were performed from four quadrants. Cases not achieving CE-D following EMR/ESD and repeated RFA were surveilled by annual endoscopy.
Follow-up RFA and biopsy sessions
After the final RFA, patients underwent follow-up endoscopy every 6 months for 2 years and thereafter yearly. The degree of eradication of visible BE was assessed, and biopsies were taken. The results were decisive for any further treatment. In the case of recurrence, patients underwent a second round of RFA until the histology proved non-dysplasia.
Data collection and data analysis
Effectiveness outcomes were based on histology from biopsies taken after the last treatment of RFA. The primary study outcomes were the proportion of patients with complete eradication of dysplasia (CE-D), defined as all esophageal biopsies being negative for dysplasia at the last biopsy session, and complete eradication of intestinal metaplasia (CE-IM) defined as the proportion of patients with esophageal biopsies being negative for IM. Safety outcomes were based on the proportion of patients who experienced complications to the RFA treatment.
Data were collected from electronic journals and registered in an encrypted database. Statistical Package for Social Sciences software version 17 (SPSS Inc., Chicago, IL, USA) was used for the descriptive data. The study participants provided informed consent, and the study was approved by the board of data inspection and the regional Committee for the Capital Region of Denmark as a quality assessment.
Results
The cohort included 108 patients. One patient was excluded due to the development of dementia. A total of 107 patients remained in the study for the follow-up period, 75% men, with a median age of 65 years (25–87 years). All patients had dysplasia at entry, 89% had LGD, and 17% had HGD. Twenty (19%) of the patients passed EMR/ESD prior to RFA. The median baseline endoscopic BE segment length was 3.0 cm (1.0–13.0 cm) by the first RFA session and 1.0 cm by the second RFA session. The median number of RFA ablations was 3. The follow-up period had a median of 25 months (6–73 months).
After the last RFA treatment, CE-D and CE-IM were achieved in 60% of the patients (n = 64). CE-D was achieved in 89% of the patients (n = 95). Efficacy outcomes are shown in Table 2. By the cut-off date, 9 patients had LGD and 31 patients had IM. During the follow-up period, recurrence of dysplasia was seen in 14 patients which makes the recurrence rate 14.7%. Eleven of these patients were treated to the state of IM and three patients are in current treatment.
Efficacy outcomes.
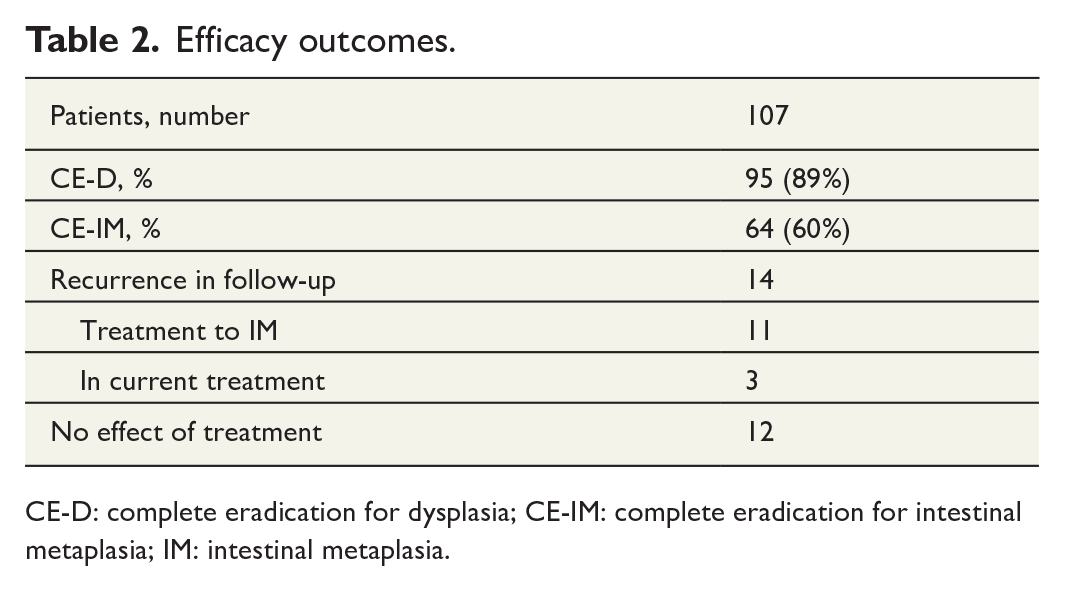
CE-D: complete eradication for dysplasia; CE-IM: complete eradication for intestinal metaplasia; IM: intestinal metaplasia.
HGD progressed to >T1a cancer for three patients during ongoing treatment. Two cases had recurrence of cancer after removal by ESD; they underwent surgery (Ivor Lewis esophagectomy). The third case was most likely cancer prevalent at the time of enrollment; this cancer was removed by ESD.
Complications occurred in 6.5% of the patients (n = 7). One patient developed a stricture after treatment with both a balloon and a focal catheter. The stricture was resolved with dilatations. Four patients developed pain and fever (temperature >38.5 °C). They had normal examinations and were managed with analgetic and were discharged within 48 h. One patient was admitted with upper gastrointestinal (GI) bleeding 8 days after RFA treatment. The patient had an implantable cardioverter-defibrillator (ICD) and was treated with warfarin. The international normalized ratio (INR) was 3.9. Warfarin was paused, and the bleeding stopped. The last patient had a small aspiration pneumonia following general anesthesia. The patient was treated with antibiotics for 3 days and discharged.
Discussion
In this retrospective study of a single-center cohort, we evaluated RFA as a treatment modality for patients with BE and confirmed LGD or HGD. The efficacy and safety of RFA are compared with other treatment modalities in previous studies, including endoscopic surveillance. Several studies such as Pouw et al., 10 Small et al., 14 and Kahn et al. 15 found that the progression of LGD to either HGD or EAC was significantly lower in patients treated with RFA compared with endoscopic surveillance, with an absolute risk reduction of 6%–32% and a relative risk reduction of 40%–96%. 16
We found efficacy outcomes of RFA to be promising in the treatment of patients with BE and LGD or HGD. The proportion of CE-D was achieved in 89% of the patients. As a result, the risk of progression of dysplasia to EAC in these patients is markedly reduced. CE-IM was observed in 60% of the patients. In similar studies using RFA as a treatment for BE patients with LGD, the efficacy results are comparable with the outcomes in this study. Specifically, a multicenter study by Lyday et al. 9 evaluated RFA as a treatment for BE with dysplasia in 429 patients, CE-D was achieved in 89%–100%, and CE-IM was achieved in 72%–77%. In a randomized controlled trial by Shaheen et al., 11 RFA was evaluated in 127 patients with BE and dysplasia, CE-D was achieved in 81%–91%, and CE-IM was achieved in 77%. In a retrospective cohort study by Pouw et al., 10 CE-D was achieved in 90% of the patients in the RFA group. Overall, efficacy outcomes for the treatment of RFA for patients with BE in this study are compared favorably with the outcomes reported in similar studies.9–11,14 Yet, the CE-IM of 60% in this study is slightly lower compared with other studies. A reason for this difference can most likely be found in the methods of treatments including the RFA procedure itself and the less aggressive use of EMR/ESD.
Current guidelines for the treatment of BE with LGD and HGD are endoscopic resection/ablation. Historically, a surveillance strategy designed to detect disease progression at the earliest possible stage was utilized. In the last decade, clinicians have utilized a prophylactic strategy by treating patients with BE and LGD with endoscopic resection and RFA.3–5 By achieving CE-D in 89% of the patients and CE-IM in 60% of the patients, the findings in this study support these current guidelines.
Safety outcomes in the study were evaluated as well. Overall, the proportion of patients with complications to the treatment was 6.5%. The severity of these complications can be measured as extended hospitalization stays and possibly permanent injuries. Due to the sample size in this study, we may not conclude any risk rates for specific adverse events to be representative of the background population. Nevertheless, the study provides information on possible complications of the treatment of RFA, including stricture, pain, fever, upper bleeding, and aspiration pneumonia following general anesthesia. Four patients developed pain and fever, which resulted in 48 h of extended hospitalization. One patient had a small aspiration pneumonia following anesthesia which was treated with antibiotics and resulted in 3 days of extended hospitalization. A few serious adverse events were related to the procedure, including upper GI bleeding and aspiration pneumonia. None of the above complications resulted in permanent injuries. These results are in accordance with safety outcomes reported in other similar studies using RFA. The most common adverse event is an esophageal stricture with a 0%–6% rate per patient, 2.1% per patient for Lyday et al., 9 and 6.0% per patient for Shaheen et al. 11 Upper GI bleeding was seen in the study by Shaheen et al. 11 In several studies, RFA was associated with pain and fever.9–11
We observed recurrence in 14 patients (14.7%). Several systematic reviews and meta-analyses have estimated recurrence rates for IM and dysplasia after achieving CE-IM following the treatment of BE with RFA. Orman et al. 17 observed a recurrence rate of about 13%, including 18 studies. 17 Leclercq et al. 18 found the recurrence to range between 8% and 32%. Fujii-Lau et al. 19 included 25 studies and demonstrated an incidence of recurrence of 8.6 per 100 patient-years.
This study is a retrospective study of a single-center cohort. The study has some limitations. In the study, we observed only one group of patients, who all underwent RFA. The lack of a control group of patients complicates the interpretation of the results. We cannot measure an absolute risk reduction of the progression of dysplasia to HGD or EAC caused by the treatment of RFA compared with endoscopic surveillance. Despite these limitations, the results are useful by supporting already existing evidence for the treatment of RFA.9–11,14 The strengths of the study are the long follow-up period and the low loss of follow-up. The biopsy sampling protocol was standardized, and the fact that histology was assessed by two expert pathologists is a strength.
In conclusion, in this retrospective single-center study, the use of RFA to patients with BE with LGD and HGD is associated with a high complete eradication rate and a low rate of complications. The results of this study resemble the results from previous similar studies, thereby ascertaining that the Danish treatment of BE with LGD and HGD is up to the highest quality standards and comparable with those of larger European expert centers. Furthermore, the results support current guidelines for the treatment of BE with LGD and HGD.
Footnotes
Author contributions
J.H. and M.T. contributed to analysis and design. M.T. collected data. M.T. and J.H. performed analysis. M.M., J.H. and M.T. contributed to drafting the article. M.M., J.H., and M.T. contributed to reviewing and final approval.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
