Abstract
Background and objective:
Minimally invasive liver surgery is evolving worldwide, and robot-assisted liver surgery (RLS) can deliver obvious benefits for patients. However, so far no large case series have documented the learning curve for RLS.
Methods:
We conducted a retrospective study for robotic liver surgery (RLS) from June 2019 to June 2022 where 100 patients underwent RLS by the same surgical team. Patients’ variables, short-term follow-up, and the learning curve were analyzed. A review of the literature describing the learning curve in RLS was also conducted.
Results:
Mean patient age was 63.1 years. The median operating time was 246 min and median estimated blood loss was 100 mL. Thirty-two patients underwent subsegmentectomy, 18 monosegmentectomies, 25 bisegmentectomies, and 25 major hepatectomies. One patient (1.0%) required conversion to open surgery. Five patients (5%) experienced postoperative major complications, and no mortalities occurred. Median length of hospital stay was 3 days. R0 resection was achieved in 93.4% of the malignant cases. The learning curve consisted of three stages; there were no significant differences in operative time, transfusion rate, or complication rate among the three groups. Postoperative complications were similar in each group despite an increase in surgical difficulty scores. The learning effect was highlighted by significantly shorter hospital stays in cohorts I, II, and III, respectively. The included systematic review suggested that the learning curve for RLS is similar to, or shorter, than that of laparoscopic liver surgery.
Conclusions:
In our experience, RLS has achieved good clinical results, albeit in the short term. Standardization of training leads to increasing proficiency in RLS with reduced blood loss and low complication rates even in more advanced liver resections. Our study suggests that a minimum of 30 low-to-moderate difficulty robotic procedures should be performed before proceeding to more difficult resections.
Keywords
Context and Relevance
Currently, very little data regarding robotic liver surgery (RLS) have been reported, but results appear to be similar to conventional laparoscopic liver surgery (LLS), at least in terms of blood loss, morbidity, mortality, and length of hospital stay (LOS). In order to safely disseminate RLS in the future, it is essential to examine the learning curve. However, the learning curve in RLS has not yet been fully evaluated. Due to the limited data regarding RLS, a learning curve model in RLS should be based on the experience with conventional LLS. The purpose of this study was to compare our results with several LLR learning curve reports and to identify the number of cases required to create the RLS learning curve.
Introduction
Hepatectomy once used to be associated with a high mortality rate. However, recent advances in surgical procedures, preoperative evaluation of liver function reserves, and pre- and post-operative patient management make hepatectomy now a relatively safe procedure. 1 Since the first case of laparoscopic liver surgery (LLS) was documented in 1991, 2 a multitude of techniques with favorable outcomes have been reported. Compared to open liver surgery (OLS), LLS has been associated with less blood loss and shorter hospital stays.3–10 However, a laparoscopic approach to the liver has its drawbacks, with restrictions imposed on the instruments and the indications for liver resection. 11 The da Vinci Surgical System offers many advantages compared to laparoscopic surgery; these include articulating instruments that re-create the 7 degrees of freedom of the human wrist and a three-dimensional view of the operative field in high-definition on the console, and its indications have expanded to include hepato-pancreato-biliary (HPB) cases that were once considered difficult to treat with laparoscopic surgery.
Robot-assisted liver surgery (RLS) was first introduced in 2003 by Giulinotti et al. 12 ; by 2011, 70 cases of RLS had already been reported. 13 With the growing body of evidence, a consensus has emerged around the safety of RLS. Therefore, it is likely that more surgeons will take advantage of da Vinci when performing RLS in the future.13–15 Experience on the da Vinci platform is growing among HPB surgeons starting from the pioneering work of Giulianotti and colleagues,12–16 but the learning curve for proficiency is yet to be defined. Therefore, it is crucial to define learning curves to ensure patients’ safety while developing RLS. This article draws on our experience from the 100 cases of RLS conducted at the Copenhagen University Hospital, Denmark and discusses the following points: adaptation of short-term surgical results and the learning curve in RLS.
Methods
Clinical factors studied included demographics, IWATE criteria, surgical and postoperative outcomes. Estimated blood loss (EBL) was obtained from anesthesia records. Operative time (OT) was defined as the time from skin incision to wound closure. No adjustment for adhesion release was made. Postoperative complications were evaluated based on the Clavien-Dindo classification (CD). Major complications were defined as events requiring surgical, endoscopic, or radiological intervention (CD grade ⩾ 3).
Developing a RLS program and training in RLS
Our hospital performs approximately 400 liver operations per year, of which 10%–15% have been performed as LLS since 2009. Robotic systems have several advantages over conventional laparoscopic surgery, including 7 degrees of freedom with Endowrist technology, the ability to reach posterior superior lesions, improved suturing capabilities, elimination of physiological tremors and superior surgeon ergonomics. Despite the downside is added cost and longer operative times compared to a laparoscopic approach,17 we have decided to introduce RLS from 2019 due to the aforementioned advantages and the strong belief in this technology which is meant to stay and will further develop. Our team members in RLS are required to attend a series of courses: (1) Copenhagen Academy for Medical Education and Simulation (CAMES) course; (2) Intuitive Surgical’s basic robotic training course (Orsi Academy, Melle, Belgium). In addition to the basic courses, robotic console surgeons are required to complete all Intuitive simulation exercises (scoring 90% or higher) with a minimum of 80 h of independent training. Robotic console surgeons are then trained in dry and wet-lab models. In addition, surgeons, anesthetists, OP nurses, and anesthetic assistants responsible for RLS are required to be trained to deal with emergency situations before clinical introduction (CAMES training model 6). With regards to the clinical introduction of RLS, there was no on-site sponsored proctoring. The team, however, has an extensive experience in OLS and LLS. Regarding the increased in surgical complexity of RLS surgery, we have incorporated the Morioka Conference (2014) recommendations for LLS into our RLS program, starting with minor liver resection. As our experience and proficiency increased, we gradually increased the difficulty of the operation and attempted major liver resection according to the IWATE criteria.
Difficulty scoring
As one of the measures to evaluate the learning curve and its effects, we used the IWATE criteria introduced by Wakabayashi 11 in 2016 (Fig. 1).
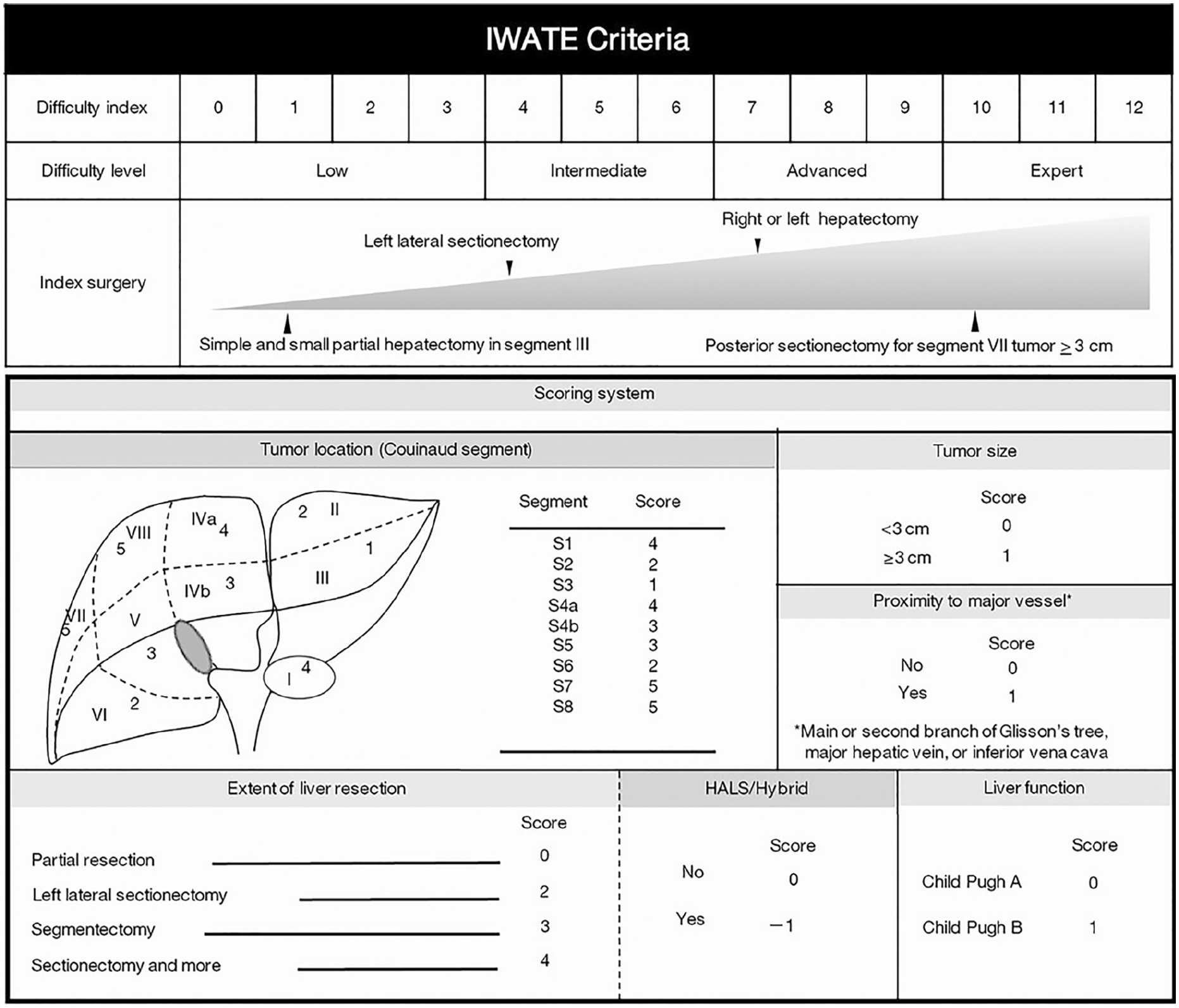
IWATE criteria.
Definitions and resection type
Minor liver resection was defined as a Couinaud’s resection of two or fewer segments. 18 A major liver resection was defined as a Couinaud’s trisection or greater 18 and a technically difficult liver resection, that is either right posterior sectionectomy (S6 + S7), extended right posterior sectionectomy (S6 + S7 + right hepatic vein), or Right anterior sectionectomy (S5 + S8).
Exclusion criteria
Exclusion criteria included general contraindications to pneumoperitoneum, cardiac or respiratory insufficiency, and/orASA (American Society of Anesthesiologists) physical status higher than III. Any patients with a history of OLS were excluded.
Surgical procedures of RLS
The robot system used is the da Vinci® Si Surgical System. All operations were performed by the senior surgeon.
Transection method
Since our introduction to LLS, we have used only the Robotic Harmonic scalpel® (Ethicon Endo-Surgery) for liver parenchymal dissection (LPD), not in conjunction with the Cavitron Ultrasonic Surgical Aspirator (CUSA). Due to the EndoWrist function of the Maryland bipolar forceps/fenestrated bipolar forceps, they are effective in both controlling of minor bleeding from a cut surface and for dissecting and isolating major vessels in the deep LPD. As in LLS,19,20 in our opinion, that LPD in RLS is best performed with a caudo-dorsal approach. Surgeons proceeded with the transection layer by layer using the Harmonic scalpel from the cortical aspect of the liver toward the core of the parenchyma. The left side of the transection line was held by the third robotic arm in order to provide exposure of the hepatic section line. Blood vessels or bile ducts with a diameter of >3 mm were ligated using Weck® Hem-o-lok® clips. Both the Glissonean pedicle and the major hepatic vein >10 mm was transected intrahepatically using a laparoscopic Endo-GIA stapler introduced by the bedside surgeon. Pringle’s maneuver was used for anatomical liver resection of more than one segment. During transection of the liver parenchyma, the central venous pressure was maintained below 5 mm Hg to prevent venous hemorrhage. Pneumoperitoneal pressure was established at 8–12 mm Hg and maintained stable using an Airseal® System. After confirming that there was no bile leakage or bleeding from the resection margin, the operation was terminated.
Postoperative care
All our liver resected patients followed the ERAS program as previously published.21,22
Evaluation of the learning curve
The learning curve effect was investigated by separately analyzing three cohorts over three periods with the goal to identify differences in patient outcomes that reflect the surgeon’s proficiency: the first stage (Cohort 1: initial experience 30 cases), the second stage (Cohort II: pushing the limits 40 cases), and the third stage (Cohort III: the steady state 30 cases); the following points were considered: IWATE criteria, open conversions rate, OT, EBL, LOS, postoperative complications, and major morbidity. A literature review (PUBMED/MEDLINE and EMBASE) of publications on learning curves in both RLS and LLS from 2006 to 2022 was performed based on the following terms: “Robot-Assisted liver surgery,” “Robot-Assisted liver resection,” “Laparoscopic liver resection,” “Laparoscopic liver surgery,” and “ Learning curve.” After screening the manuscripts, we selected six series that included more than 10 cases on RLS and LLS. Data were extracted and compared with our results. All included articles reported intraoperative and postoperative outcomes (Table 6).
Statistical analyses were performed using IBM SPSS v.27. We applied Fisher’s exact test for categorical data and Student’s T test for continuous data to detect statistically significant differences. Differences in outcomes were considered statistically significant at p < 0.05.
Results
Initial experience with RLS
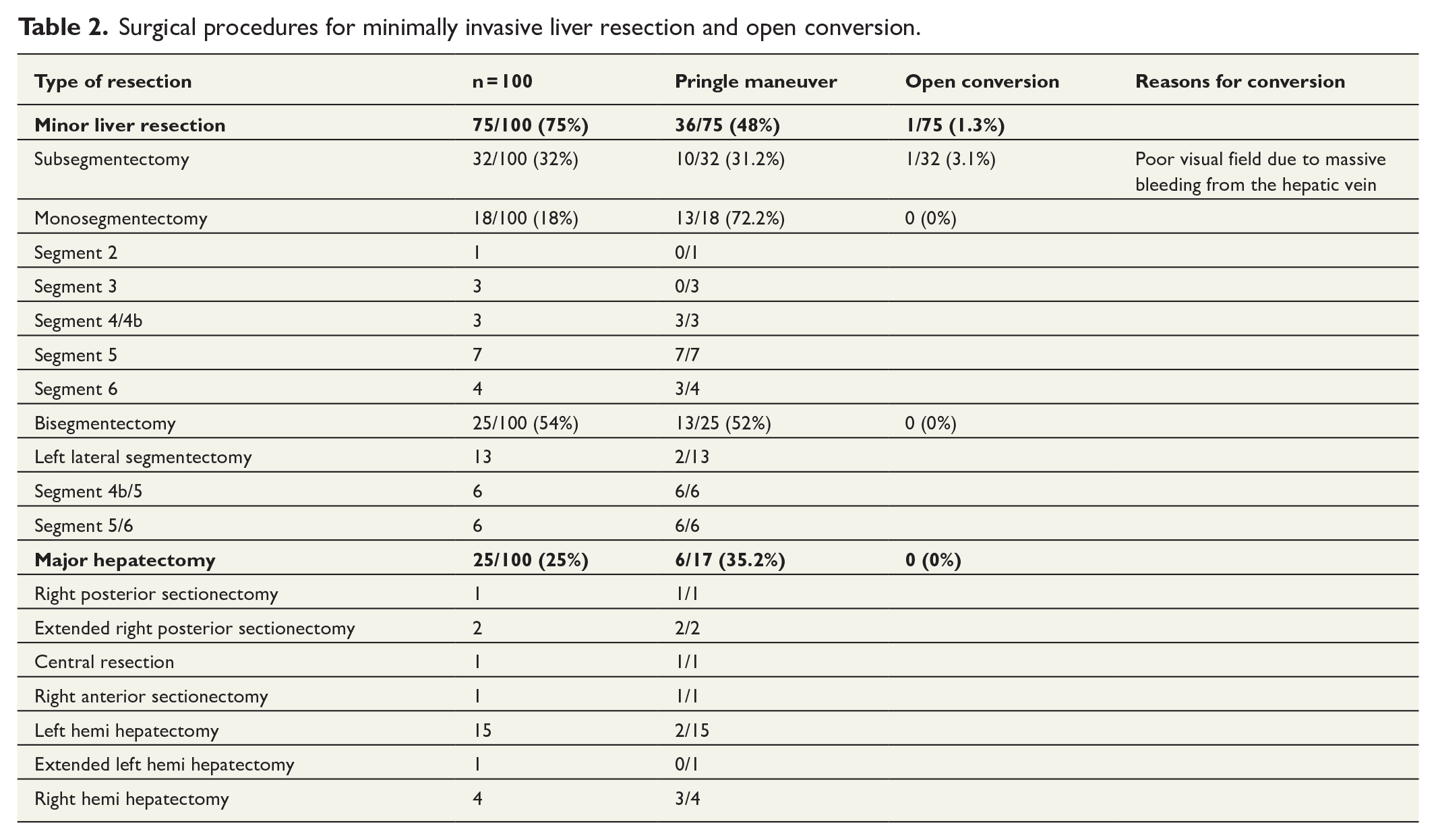
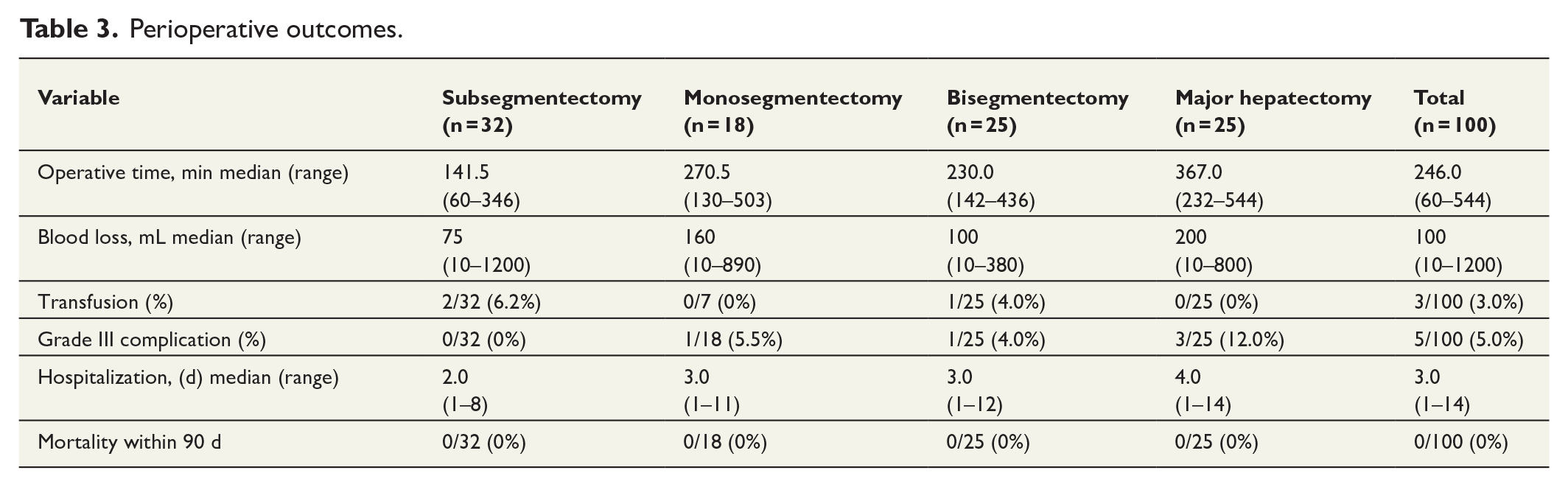
Clinicopathologic characteristics are summarized in Table 1. Mean patient age was 63.1 years (range, 25–92 years). Body mass index (BMI) was 26.5 kg/m2 (range 16.7–38.9). There were 70 malignant liver tumors and 30 benign liver tumors. The mean surgical margin for malignant lesions was 7.3 mm (range 0–100). In four patients (5.2%), surgical margin was positive (R1). We performed 32 subsegmentectomy, 18 monosegmentectomies, 25 bi-segmentectomies, and 25 major hepatectomies (Table 2). The median OT was 246.0 min (range 60–544 min), and median EBL during surgery was 100 mL (range 10–1200 mL). Three patients (3.0%) needed perioperative blood transfusion. One patient (1.0%) who underwent subsegmentectomy on the surface of S6 was converted to open surgery because of poor visual field due to massive bleeding from the hepatic vein (Table 3). No conversions occurred in any of the 93 most recent resections.
Clinicopathologic characteristics.
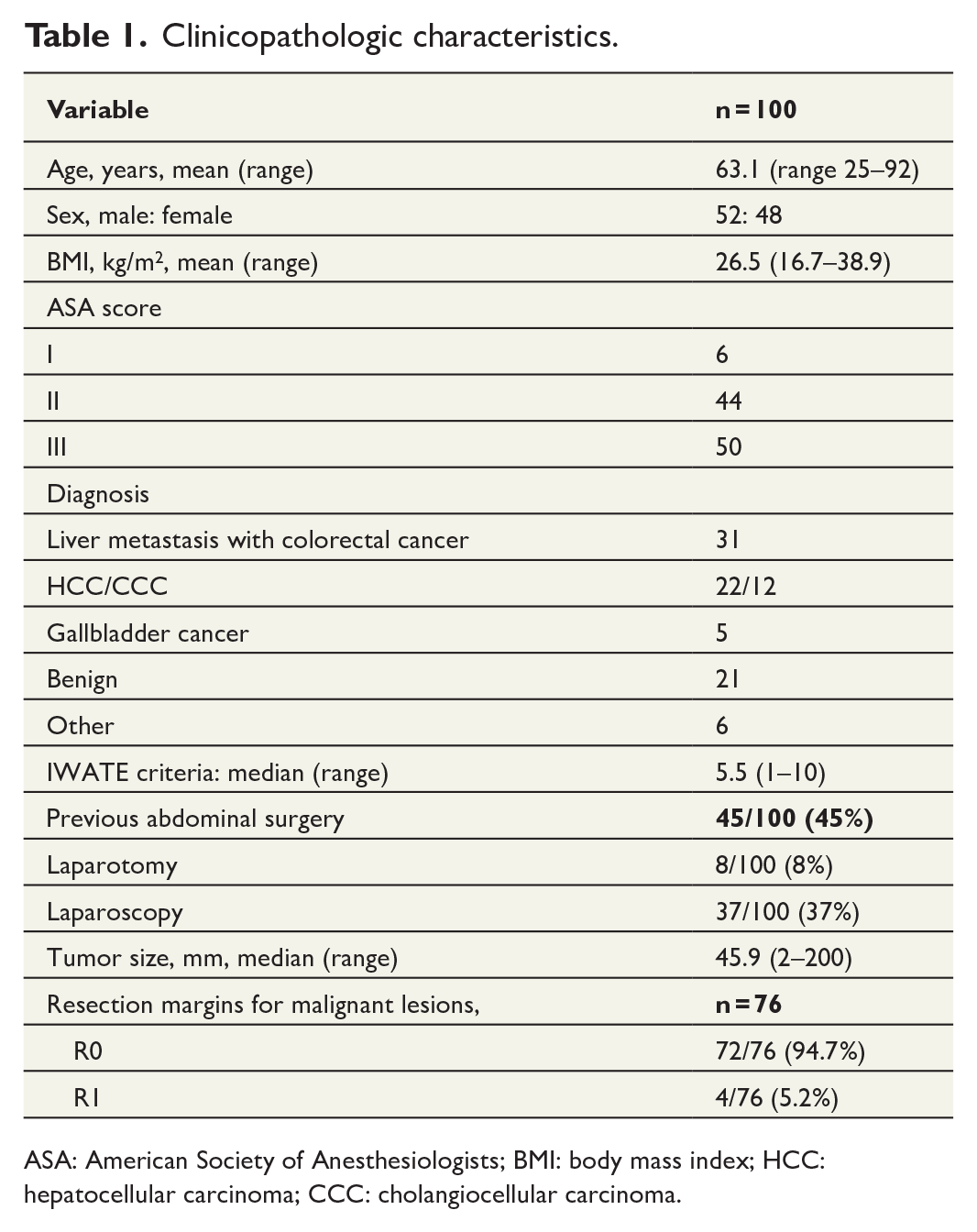
ASA: American Society of Anesthesiologists; BMI: body mass index; HCC: hepatocellular carcinoma; CCC: cholangiocellular carcinoma.
Surgical procedures for minimally invasive liver resection and open conversion.
Perioperative outcomes.
Morbidity and mortality
The overall morbidity rate (⩽ 30 postoperative day (POD)) was 8.0% (8/100), which is more precisely described as follows: n = 3 for CD grade I (3.0%), n = 5 for CD grade III/IV (5.0%). The CD grade III complications included the following: intra-abdominal fluid collection in two cases (2.0%) necessitating UL-guided percutaneous drainage, and biliary leakage in two (2.0%) cases necessitating UL-guided percutaneous drainage and Endoscopic retrograde cholangiopancreatography. The remaining one case had postoperative acute renal injury. We did not experience CO2 gas embolization with severe intraoperative hypotension as previously reported. Other complications were a CD grade I complication with pulmonary atelectasis and wound infection. The median length of stay in the hospital was 3 days (range 1–14 d). No mortality occurred (Table 3). Perioperative outcomes were compared between major and minor RLS, as shown in Table 4, with no significant differences between the two groups in operative time, estimated blood loss, rate of conversion to laparotomy, or complications.
Perioperative outcomes: comparison between minor and major resections with RLS.
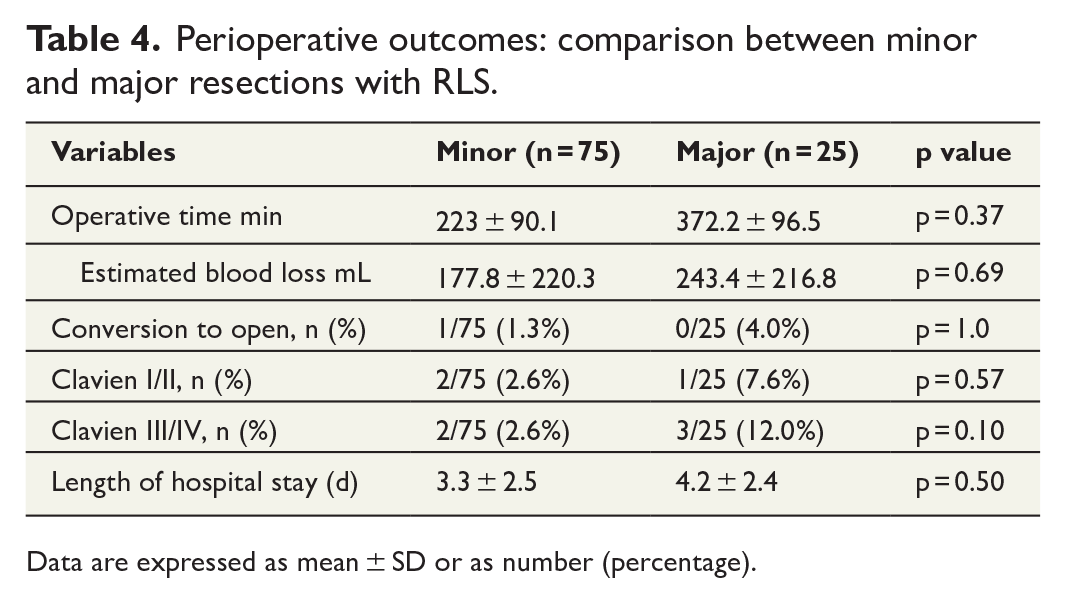
Data are expressed as mean ± SD or as number (percentage).
Learning curves
The learning curve was evaluated by comparing the results of the first 30 cases (Cohort I), the middle 40 cases (Cohort II), and the latest 30 cases (Cohort III; Table 5). According to stepwise difficulty, the average IWATE score was assessed as 4.7 ± 2.1 for Cohort I, 6.0 ± 2.3 for Cohort II, and 5.8 ± 2.8 for Cohort III. There was a trend toward increasing difficulty scoring from Cohort I to II and III, without any statistically significant difference between the three groups. With regards to minor resection, there was a decreasing trend from Cohort I to II as well as II to III, with a significant difference between Cohorts I and III (26 versus 18, p = .004). For major liver resection, these showed an increasing trend from I to II and II to III, with significant differences between cohorts I and III (4 versus 12, p = .004); one patient (1%) had the procedure changed to OLS. The overall mean OT was 261.0 ± 112.5 min, although there was a trend toward increased OT without any statistically significant difference between the three groups. The overall median EBL was 194.0 ± 222.5 mL, showing a decreasing trend from Cohort I to II, but no significant difference. The predominant increase of major and more complex liver resections from Cohort II to III has possibly resulted in a significant increase in blood loss in Cohort III compared to Cohort II (151.1 versus 251.8 mL, p = .004). The median length of hospital stay was 3 d (3.5 ± 2.5), with significantly shorter lengths of stay in Cohorts II and III compared to cohort I. There were no significant differences among the three groups in the comparison of postoperative complications.
Learning curve: Comparison of perioperative outcomes of RLS in subgroups of early, intermediate, and late experience.
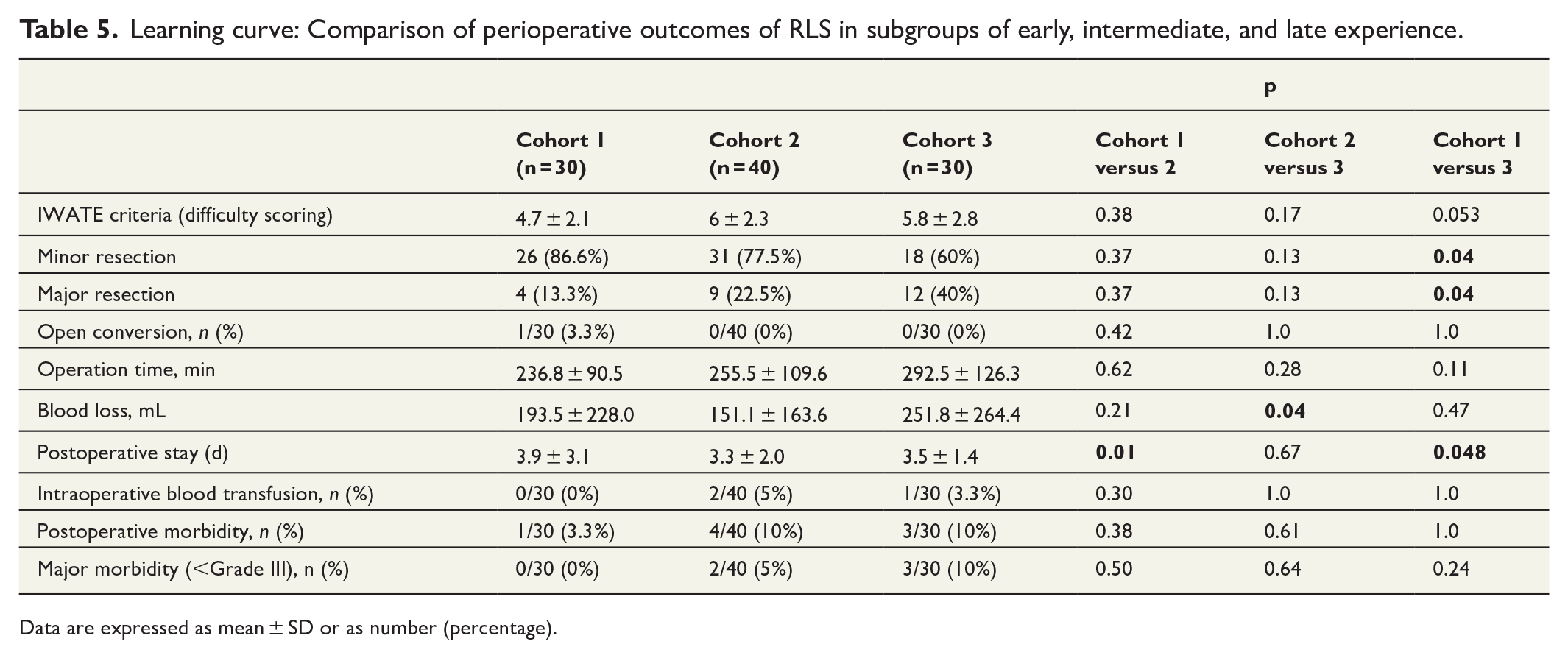
Data are expressed as mean ± SD or as number (percentage).
A literature search (PubMed/MEDLINE, EMBASE) identified six series describing LLS and RLS learning curves. These results were extracted and compared with our results (Table 6). The learning curve in LLS is currently reported to require 20 cases for minor LLS and 50 cases for major LLS; the learning curve in RLS is reported to require 13–33 cases.
Studies evaluating learning curves for laparoscopic and robotic liver surgery.
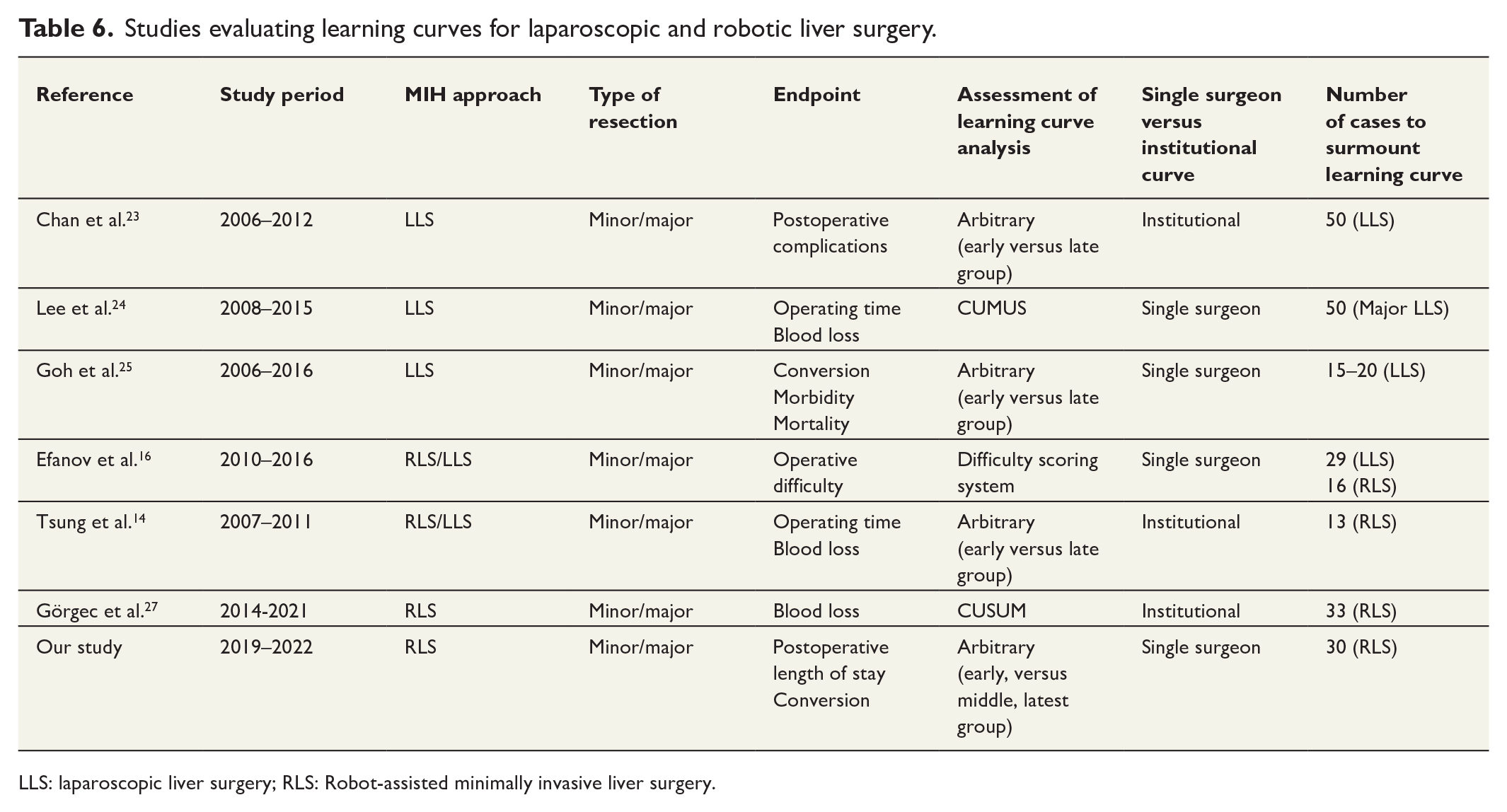
LLS: laparoscopic liver surgery; RLS: Robot-assisted minimally invasive liver surgery.
Discussion
Although RLS has been reported to produce similar surgical results to LLS,14,15 its reproducibility and feasibility are still questionable, and it has not yet been widely adopted. In our study, after careful introduction of RLS with adequate pre-robotic training. The overall results of RLS were good, indicating that RLS can be performed safely. Our results compare favorably with other published series of LLS, and RLS.4–10,13–16,23,24
Review of literature
Learning curve
In the past, operative time, blood loss, conversion rate, morbidity, mortality, transfusion rate, intensive care unit (ICU) days, and LOS have served as factors to determine the learning curve in LLS. Unfortunately, a wide variety of learning curves have been reported by each center, however, a comprehensive modality to describe the learning curve has still not been identified.23–25 Based on our review of literature, the learning curve for LLS is currently reported to require 20–40 cases for minor LLS and 40–60 cases for major LLS. 26
There are still only few reports published regarding the RLS learning curve. RLS is the most recent evolution in the field of minimally invasive liver surgery and has been proposed as a valuable alternative to LLS. Therefore, the learning curve model in RLS should be analyzed based on reports of conventional LLS, which is similar in surgical technique and outcomes. Although statistical methods and endpoints for learning curves have not yet been standardized, we cited three references reporting learning curves in LLS as well as three for RLS and compared them with our study (Table 6). Chan et al. 23 describe their learning curve experience with results of an early and late study with 100 patients. Using the postoperative complication rate as the primary endpoint, they conclude that LLS requires a learning curve of about 50 cases or more. Lee et al.24 analyzed perioperative data from the medical records of 170 consecutive cases in which a surgeon performed and reported on LLR. The necessary number of cases to achieve a stable learning curve for LLR differs according to the extent of liver resection. This variance suggests that a surgeon needs to perform LLS in 50, 25, and 35 cases for major LLS, minor LLS, and tumorectomy, respectively. On the other hand, Goh et al. 25 described their learning curve experience of 324 consecutive LLR from 2006 to 2016 performed by a single surgeon. Analysis of LLR for tumors in the posterosuperior segments demonstrated that there was a significant decrease in conversion rates after a surgeon had experienced 20 LLR. Similar results have been obtained with major liver resection. They suggest that improvements in postoperative complications and conversion rates could be achieved in as few as 20 cases.
Efanov et al. 16 retrospectively analyzed the results of 131 consecutive liver resections (40 RLS, 91 LLS); for all approaches, groups with beginner and intermediate levels of experience were compared. The difficulty index for robotic surgery increased significantly from 5.0 (3.0–7.7) to 7.3 (4.3–10.2), and 16 procedures were required for RLS. They concluded that the learning curve for RLS was shorter than that for LLS. Tsung et al.14 analyzed RLS cases occurring before January 2010 (n = 13) and compared these cases with those that had occurred during or after January 2010 (n = 44). Significant differences were observed in EBL (300 versus 100 mL), overall operating room time (466 versus 314.5 min), operation time (381 versus 232 min), and LOS (5 versus 4 d), in favor of RLS performed at a later date. They suggested that improvements in the postoperative complications and conversion rate could be achieved after only 13 RLS cases. Görgec et al.27 reported a multicenter retrospective cohort study including consecutive patients who underwent RLS for all indications in nine Dutch centers between August 2014 and March 2021. Outcomes were stratified for minor, technically major, and anatomically major RLS. CUSUM analysis for blood loss found a learning curve of at least 33 major RLS procedures that needed to be surpassed.
In our study, the learning curve comprised three characteristic phases (Table 5). The clinical outcome in increasingly complex procedures depends on improved and refined surgical skills of the primary surgeon. Therefore, liver resection with a higher difficulty index is inevitably likely to result in poorer surgical outcomes, mainly in terms of morbidity in an early stage and period of the learning curve. Interestingly, the predominant increase in the proportion of major and more complex liver resections from Cohort II to III has possibly resulted in a significant increase in blood loss in Cohort III compared to Cohort II. In terms of length of stay, a significant difference between Cohorts I and II/III was observed. In addition, compared to Cohort I, no cases in Cohorts II and III were converted to open surgery. Thus, when the learning curve was examined in terms of surgical difficulty, LOS, and rate of conversion to OLS, no significant differences in OT or morbidity were observed over time despite the expansion of surgical indications and complexity; this confirmed that surgical outcomes remained stable even as resection techniques became more complex. Based on these results, we believe that a learning curve for RLS can be achieved with 30 patients, including both minor and major RLS.
By performing a systematic review on learning curves in RLS, our study and the reports of Tsung et al., 14 Efanov et al., 16 and Görgec et al. 27 suggest that 13–33 cases are required, including both minor and major hepatectomies (Table 6). The learning curve in RLS suggests that it could be equivalent to or even shorter than that of LLS despite having dissimilar endpoints. In gynecology, urology, and thoracic surgery, it has been reported that robotic manipulation is rather similar to open and that its learning curve allows for much faster progress than in laparoscopic surgery.28–30 In fact, the procedural strategy for RLS is very similar to that of LLS, despite the limitations of the instruments used for LPD.11,17–20 Therefore, if RLS is performed by expert HPB surgeons having an extensive experience in both LLS and OLS and understanding the characteristics of the robotic equipment, the learning curve in RLS may be shorter. Although it is not clear whether the learning curve in RLS reported in the above-mentioned and our study can be directly applied to other institutions, these results may be a good reference for liver surgeons introducing RLS in their department.
The present series has several limitations. This study addresses skilled HPB surgeons with years of experience in both OLS and LLS.14,16 Ideally, however, different surgeons should have been included in this study. It is, therefore, very difficult to directly apply the results of these learning curves in RLS to both HPB fellows and liver surgeons in their early career. Based on the above, we believe that the results of the learning curves shown in our study may be applied as a base for further estimates of surgeon volume load in future learning programs for the implementation of RLS.
Conclusion
In our experience, RLS has achieved good clinical results, albeit in the short term. Standardization of training leads to increasing proficiency in RLS with reduced blood loss and low complication rates even in more advanced liver resections. Our study suggests that a minimum of 30 low-to-moderate difficulty robotic procedures should be performed before proceeding to more difficult resections.
Footnotes
Author contributions
The authors confirm their contributions to the paper as follows: study conception and design: Daisuke Fukumori, Christoph Tschuor, Jens Hillingsø, Lars Bo Svendsen, and Peter Nørgaard Larsen; data collection, Daisuke Fukumori; analysis and interpretation of results: Daisuke Fukumori, Christoph Tschuor, and Luit Penninga; draft manuscript preparation: Daisuke Fukumori. All authors reviewed the results and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Due to the fact that this study is retrospective quality control-based study no ethical committee approval was required.
