Abstract
Background and objective:
An association between body mass index (BMI) and pancreatic cancer is suggested in observational studies. However, further studies are required to substantiate available evidence. The aim of this study was to explore the association between BMI and pancreatic ductal adenocarcinoma (PDAC) risk, treatment, and mortality.
Methods:
A registry-based cohort study was performed by combining data from four registries in Norway. Baseline data were collected between 1963 and 1975 with follow-up data collected until 2018. Kaplan–Meier curves and multivariable Cox regressions were estimated. Chi-square tests were used to analyze differences between groups.
Results:
The study cohort consisted of 1,723,692 individuals. A total of 8973 PDAC cases were identified during 55,744,749 person-years of follow-up. A 5 kg/m2 increase in BMI was associated with an increased risk of PDAC if high BMI at young age (16–29 years) (hazard ratio (HR): 1.21, 95% confidence interval (CI): 1.13–1.31), both for men (HR: 1.30, 95% CI: 1.15–1.46) and women (HR: 1.16, 95% CI: 1.05–1.28). In men, there was a 52% increase in risk of early-onset PDAC (<age 50) (HR: 1.52, 95% CI: 1.13-2.03) with 5 kg/m2 increase in BMI. A total of 2645 individuals were diagnosed with stage 1–3 disease, of whom 1131 underwent curative surgery. In all, 49% of normal weight, 38% overweight, and 30% obese individuals with stage 1–3 PDAC underwent surgery with curative intent (p < 0.001). Cancer survival was lower in overweight and obese individuals for both sexes. Analysis adjusted for sex, age, and period of diagnosis confirmed increased risk of cancer death in obese individuals.
Conclusion:
This study suggests that increased BMI in young adults may increase the risk of PDAC, and higher BMI in men is associated with an increased risk of early onset PDAC (<age 50). Overweight and obese individuals were less likely to undergo curative surgery and obese individuals had a higher risk of cancer-related death.
Context and relevance
Several studies suggest an association between body mass index (BMI) and pancreatic cancer; however, further studies are required to elaborate on the present evidence. We have performed a large registry-based cohort study to explore the association between BMI and pancreatic adenocarcinoma in detail, including risk, treatment, and mortality.
Background
Obesity is a major health concern worldwide, with the prevalence tripled the last four decades. 1 Obesity is related to several health care challanges 2 and excess weight is estimated to cause up to 20% of all cancers 3 including cancers of the gastrointestinal tract.4–6
Pancreatic cancer is the 12th most common cancer worldwide, with approximately a half million new cases in 2020. 7 The disease is often diagnosed late rendering a high mortality. Although use of chemotherapy and centralization of pancreatic surgery has improved prognosis,8–10 1-year overall survival and 5-year relative survival in Norway are still reported to be 30% and 15%, respectively.8,11
Several observational studies and meta-analyses indicate a positive relationship between excess weight and pancreatic cancer.5,12–16 In a British study, 12.8% of all pancreatic cancers was attributed to either overweight or obesity. 17 Others have reported a higher risk of developing pancreatic cancer with excess body fat in early adulthood.16,18,19 Cancer-specific mortality is also observed to increase with excess body weight.18,20–22 However, most studies have limited sample sizes, with the largest meta-analysis including 9504 patients aggregated from 23 studies. 13 Furthermore, histological subtypes of pancreatic cancer are typically not reported, and studies on the association between weight and risk of early-onset cancer (before the age of 50) are lacking.
The main objective of this study was to explore the association between body mass index (BMI) and risk of pancreatic ductal adenocarcinoma (PDAC), based on a large Norwegian cohort with up to 50 years of follow-up. Secondary objectives were to investigate whether high BMI in early adulthood is associated with early-onset PDAC, to explore associations between BMI and cancer survival after PDAC, and to assess rates of curative surgery across weight categories.
Methods
Study design
An observational registry-based cohort study was performed with baseline data from the Norwegian Tuberculosis Screening Program (NTSP) cross-linked with follow-up data from Norwegian public health registries. The study is conducted in adherence to the STROBE guidelines for reporting of observational studies. 23
The NTSP
The NTSP was a screening program conducted between 1943 and 1999 in Norway. Between 1963 and 1975, a nationwide unselected mass survey was performed, where height and weight were measured and registered by health professionals for all participants. 24 A total of 1,911,598 individuals (aged 7–99 years) out of a total population of 4 million were included. NTSP data are owned by the Norwegian Institute of Public Health.
Registries used for follow-up data
All cancer cases diagnosed in Norway are reported by law to the Cancer Registry of Norway (CRN), ensuring high quality and close to complete acquisition of data. 25 Data are registered using both International Classification of Diseases (ICD), 10th edition and ICD for Oncology, 3rd edition (Topography and Morphology). Data on tumor location, histological subtypes, cancer stage, and the use of curative surgery were retrieved for all patients with verified pancreatic cancers diagnosed between 1963 and 2018.
Date and cause of death were retrieved from the Norwegian Cause of Death Registry, while information on vital status and emigration, with dates, was obtained from the National Population Register. Data from the different registries were linked using a unique personal identification number allocated to all Norwegian residents.
Study cohort and outcomes
Persons aged <16 and >75 years at NTSP survey, with missing data, BMI (weight in kg/height in m2) <15 or >50, short stature (women <150 cm and men <161 cm), cancer prior to or 1 year after the survey, or without follow-up time were all excluded from further analyses (Fig. 1). Only 0.02% of the cohort had extreme BMI values <15 or >50 and were excluded to reduce statistical uncertainty and to avoid model extrapolation. Two BMI measurements were available in a subgroup of patients; the first at NTSP screening and the second at time of surgery for a given cancer, registered in the CRN.
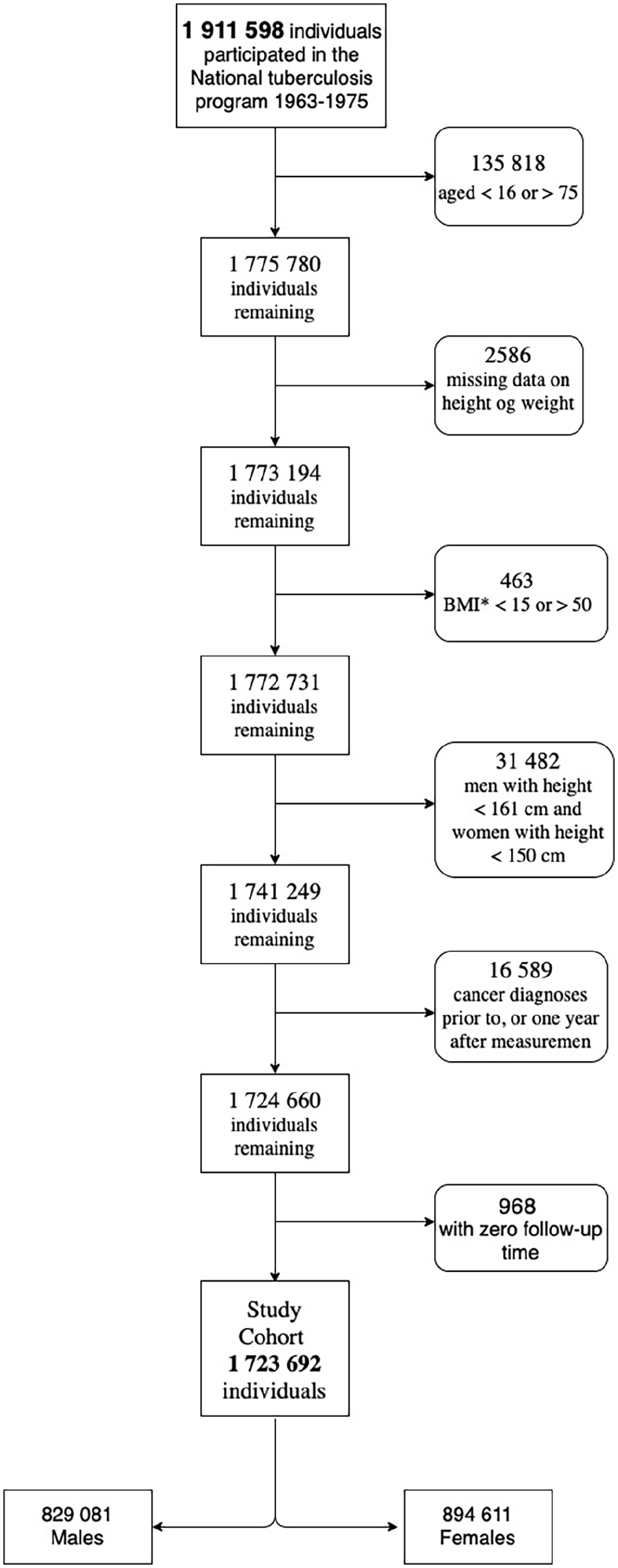
Flowchart of the study cohort from the Norwegian Tuberculosis Screening Program 1963–1975.
Pancreatic cancer was classified using the ICD-10 lexicon. Only adenocarcinomas were included using ICD-O-3 morphology lexicon (Supplementary Methods S1). Based on clinical and pathological information on metastasis, the CRN classifies stage as local disease (no metastasis); regional metastasis (regional lymph nodes, satellites, and in-transit metastasis); distant metastasis (non-regional lymph node and organ metastasis); or unspecified (no verified information on metastasis). These data were utilised to classify stage in accordance with the Union for International Cancer Control (UICC). 26
To account for potential BMI changes at population level over time, the study cohort was stratified into three time periods according to year of screening: 1963–1967, 1968–1971, and 1972–1975 (Supplementary Table S1). Age at screening was categorized as age group 1 (16–29 years) and age group 2 (30–75 years).
Statistical analysis
Means and standard deviations were used for continuous variables, while absolute and relative frequencies were used for presenting categorical variables.
The association between BMI and PDAC incidence was analyzed using multivariable Cox proportional hazard regression models, adjusted for age groups at screening and gender.
In the main analysis, BMI was handled as a continuous variable (assuming log-linearity) and presented as estimated hazard ratios (HRs) with 95% confidence intervals (CIs) per 5 kg/m2 increase in BMI. To explore potential non-linear effects of BMI and PDAC incidence, BMI was modeled using restricted cubic splines with 5 degrees of freedom. Individuals were followed from the date of NTSP screening until either PDAC diagnosis, 75 years of age, emigration or end of follow-up (31 December 2018). Attained age was used as timescale. Separate analyses were done to assess the risk of early-onset PDAC, censoring individuals at the age of 50.
Standard chi-square test was used for comparing proportions of patients who underwent surgery.
Survival curves were estimated using the Kaplan–Meier method and presented by BMI categories as defined by the World Health Organization (WHO). 27 The p values were calculated using log-rank tests. Univariate and multivariable Cox regression models were used to estimate the relative risk of cancer-related death, with normal weight used as reference BMI category. In the multivariable models, we adjusted for age at screening, year of diagnosis, and sex. Adjustment for stage was not performed as excess body weight may affect stage and thus is mediating the association of BMI on risk of cancer death, rather than confounding. Individuals were followed from PDAC diagnosis to death, or end of follow-up, whichever occurred first. As the outcome of interest was death from PDAC, individuals that died from other causes were censored. Time since diagnosis of PDAC was used as timescale.
All statistical analyses were performed using Stata version 16.1. 28 The significance level was set to 5%.
Ethics and approvals
The study was approved by the Regional Committee for Medical and Health Research in South-Eastern Norway (REC#: 2018/670) and the Oslo University Hospital data protection officer (SD0759843). Approvals from the Norwegian Institute of Public Health (NTSP data), the CRN, and the Norwegian Tax Administration (the National Population Register data) were also obtained. Individual consent for using register data was waived by the Regional Committee for Medical and Health Research.
Results
A total of 1,911,598 individuals participated in the NTSP mass survey. After exclusions, the study cohort consisted of 1,723,692 individuals, of which 829,081 were men (48.1%) with a mean follow-up of 32.4 (SD 14.8) years (Fig. 1). The study cohort characteristics are given in Supplementary Table S1.
A total of 8973 PDAC cases were observed during 55,744,749 person-years of follow-up, 4511 (50.3%) men and 4462 (49.7%) women (Table 1). Mean time from screening to PDAC diagnosis was 27.1 years (SD 12.7), ranging from 29.4 years (SD 12.5) in normal weight individuals to 21.4 years (SD 11.6) for obese individuals. Mean age at BMI measurement in PDAC patients was 43.0 years (SD 14.2) and mean age at diagnosis of PDAC was 69.7 years (SD 9.5).
Characteristics of patients developing PDAC in the cohort.
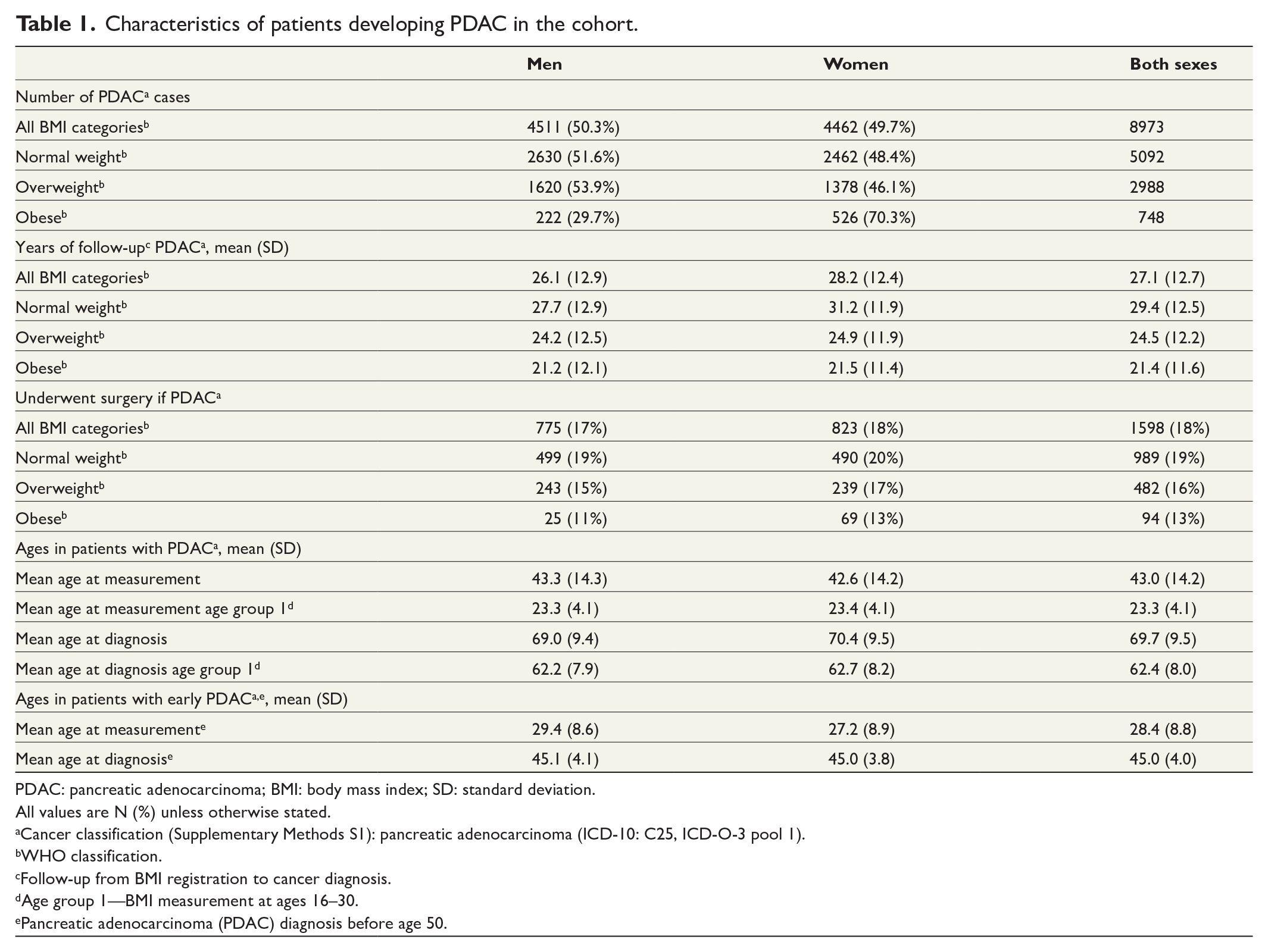
PDAC: pancreatic adenocarcinoma; BMI: body mass index; SD: standard deviation.
All values are N (%) unless otherwise stated.
Cancer classification (Supplementary Methods S1): pancreatic adenocarcinoma (ICD-10: C25, ICD-O-3 pool 1).
WHO classification.
Follow-up from BMI registration to cancer diagnosis.
Age group 1—BMI measurement at ages 16–30.
Pancreatic adenocarcinoma (PDAC) diagnosis before age 50.
Risk of PDAC
There was no association between BMI (per 5 kg/m2 increase in BMI) and risk of developing PDAC for the cohort as a whole (HR: 0.99, 95% CI: 0.96–1.02), neither for men (HR: 1.02, 95% CI: 0.97–1.07) nor women (HR: 0.98, 95% CI: 0.95–1.02) (Fig. 2A). For individuals in age group 1 (16–29 years at screening), an increased risk of PDAC per 5 kg/m2 increase in BMI was observed (HR: 1.21, 95% CI: 1.13–1.31) for both men (HR: 1.30, 95% CI: 1.15–1.46) and women (HR: 1.16, 95% CI: 1.05–1.28).
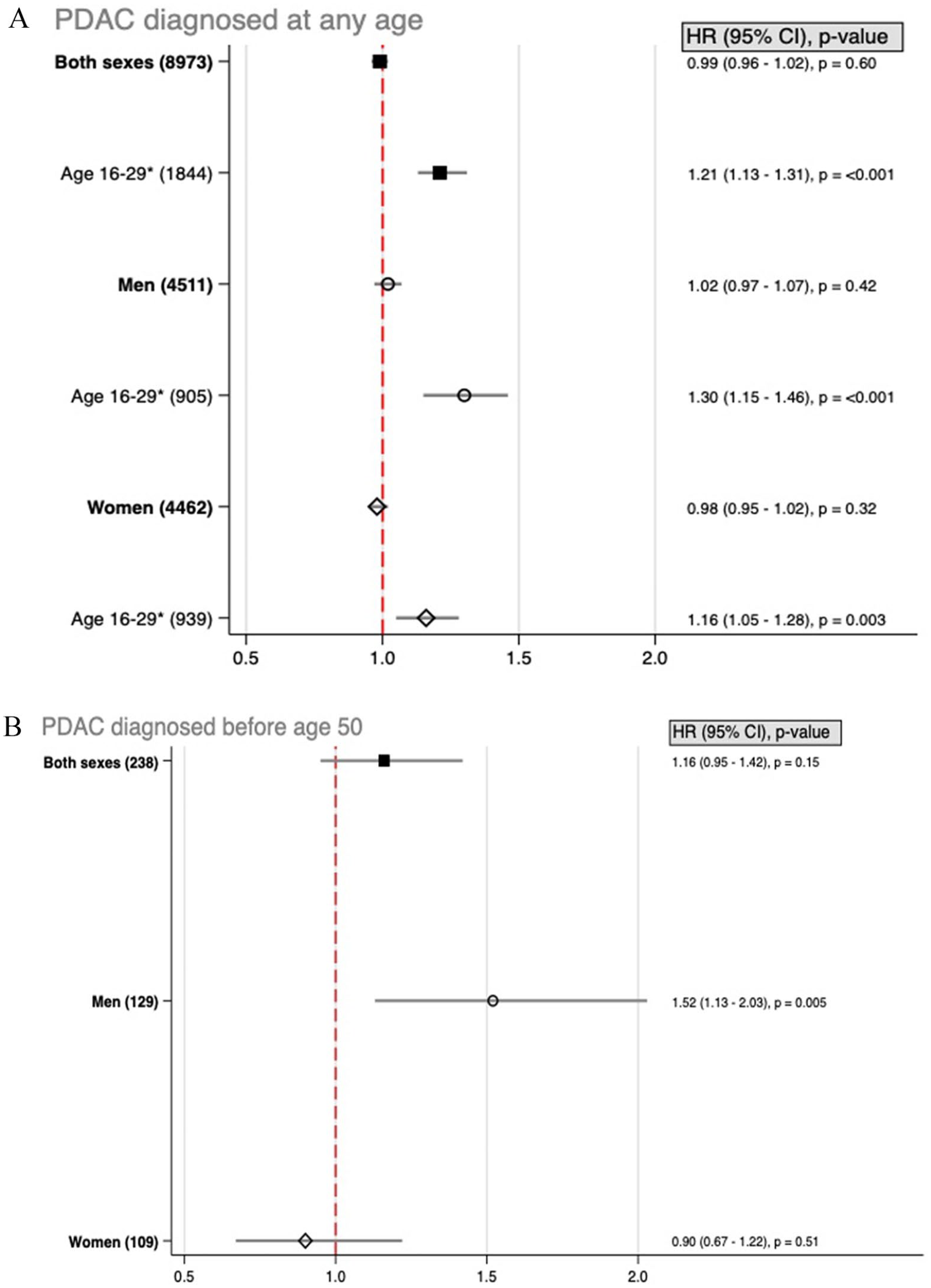
Risk of pancreatic adenocarcinoma (PDAC) incidence with increasing body mass index (BMI). (A) HR with 95% CI for risk of developing PDAC at any age, stratified by BMI measurement at any age or at age 16–30. (B) HR with 95% CI for risk of PDAC before age 50.
Analyses of non-linear associations supported these findings with no increased risk of PDAC across BMI for the cohort at large, but with an increased risk of developing PDAC with increasing BMI for age group 1 (Fig. 3).
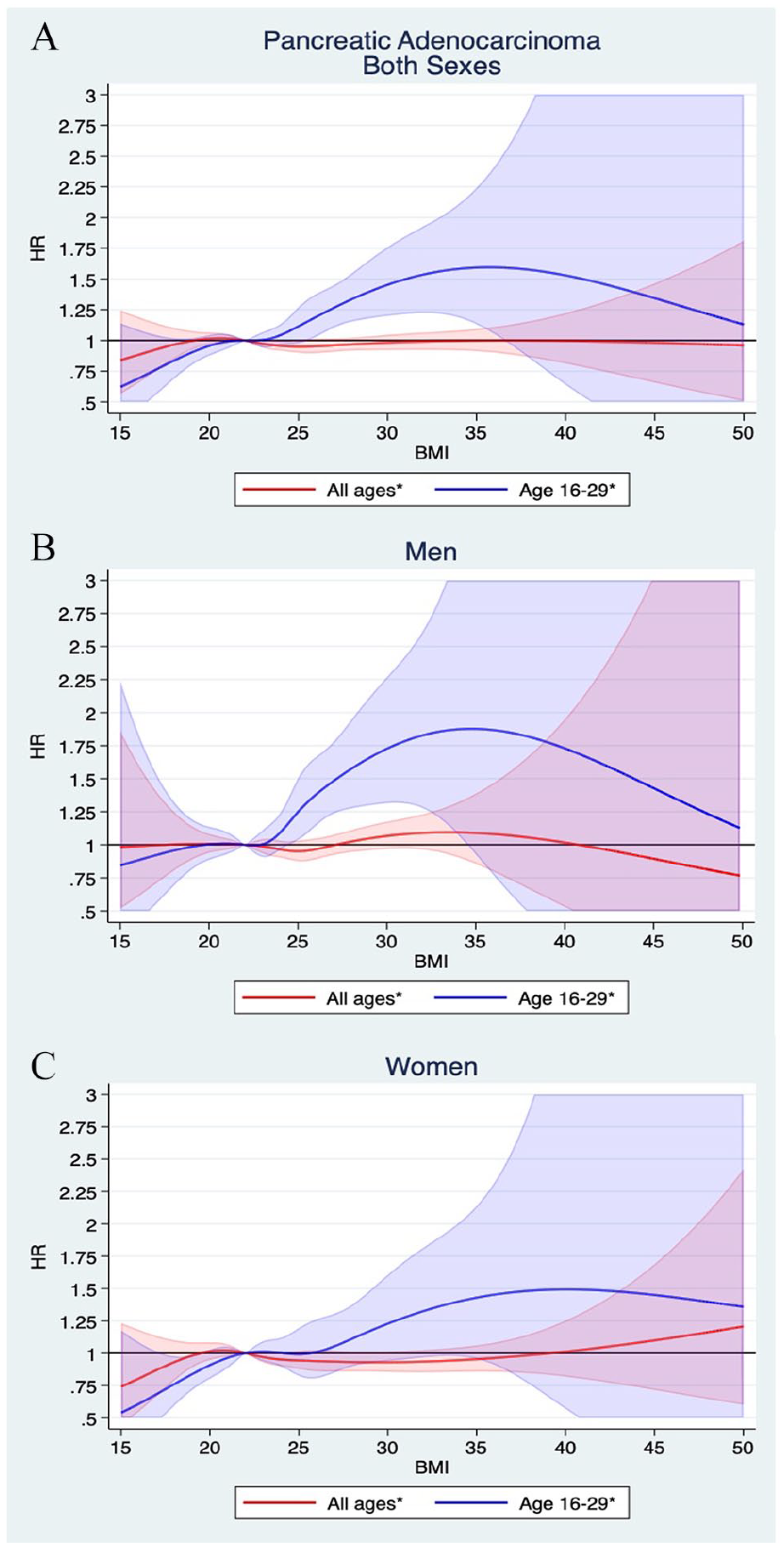
Non-linear relationship between PDAC incidence and BMI. Curves are plotted for (A) both sexes (B) men, and (C) women with 95% confidence intervals, allowing for non-linear effects modeled using restricted cubic splines.
Risk of early-onset PDAC
A tendency of increased risk of early-onset PDAC (diagnosis before age 50) per 5 kg/m2 increase in BMI was found (HR: 1.16, 95% CI: 0.95–1.42) although not statistically significant. When stratified by sex, a 52% increased PDAC risk (<age 50) per 5 kg/m2 increase in BMI was observed in men (HR: 1.52, 95% CI: 1.13–2.03), but not for women (HR: 0.90, 95% CI: 0.67–1.22).
Survival and mortality after PDAC
Kaplan–Meier survival curves, stratified by BMI categories, showed reduced cancer survival in overweight and obese individuals compared to normal weight individuals (p=<0,001) (Fig. 4). Univariate Cox regression analyses showed an increased risk of PDAC-related death in both overweight and obese individuals, for both genders. However, in multivariate analyses adjusted for sex, age, and time period of diagnosis, the increased risk of PDAC-related death was significantly increased only in individuals with obesity (both genders combined and when analyzing women only) (Fig. 5).
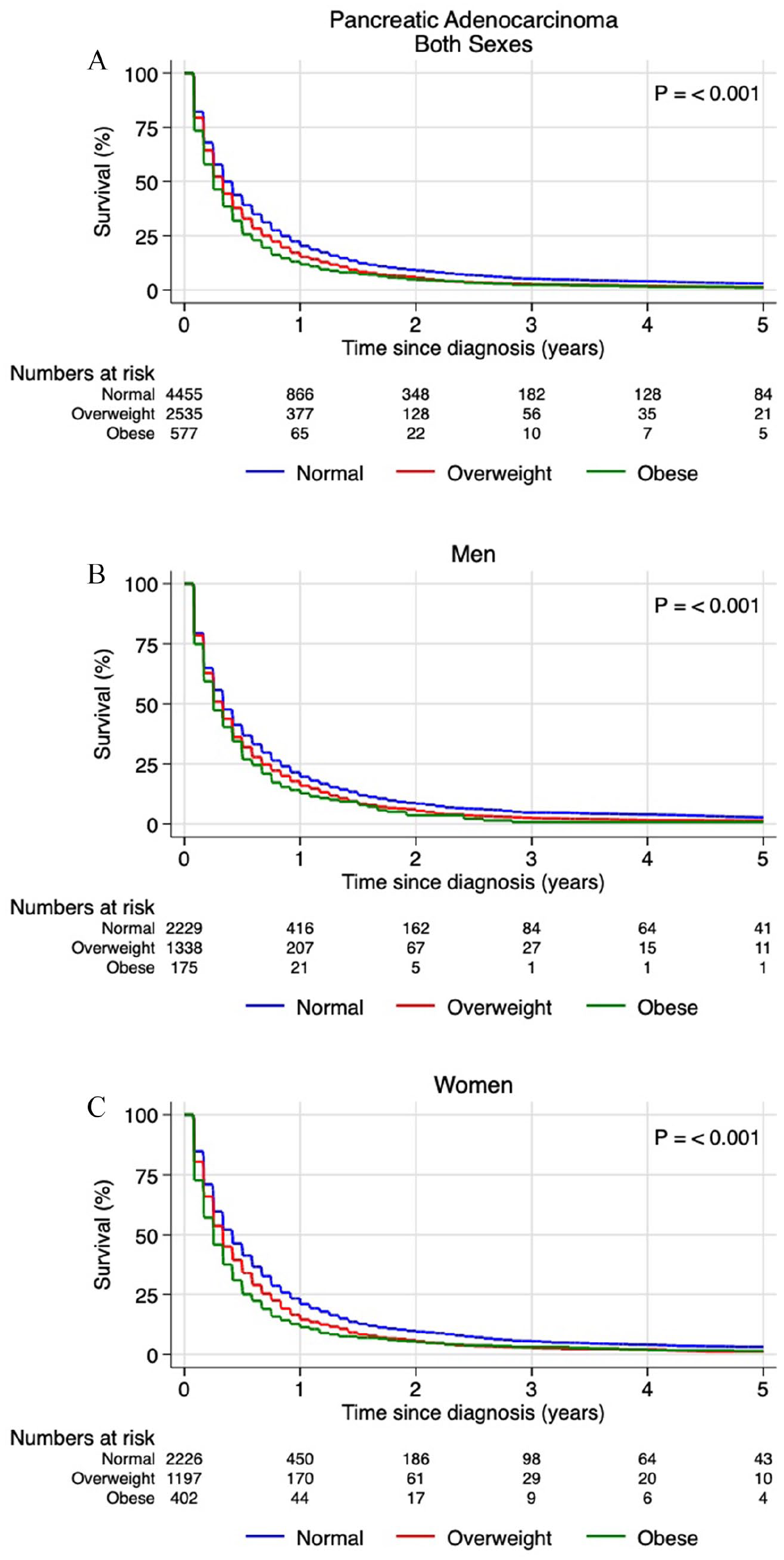
Kaplan–Meier survival curves for patients with pancreatic adenocarcinoma (PDAC). Curves are plotted for (A) both sexes, (B) men, and (C) women.
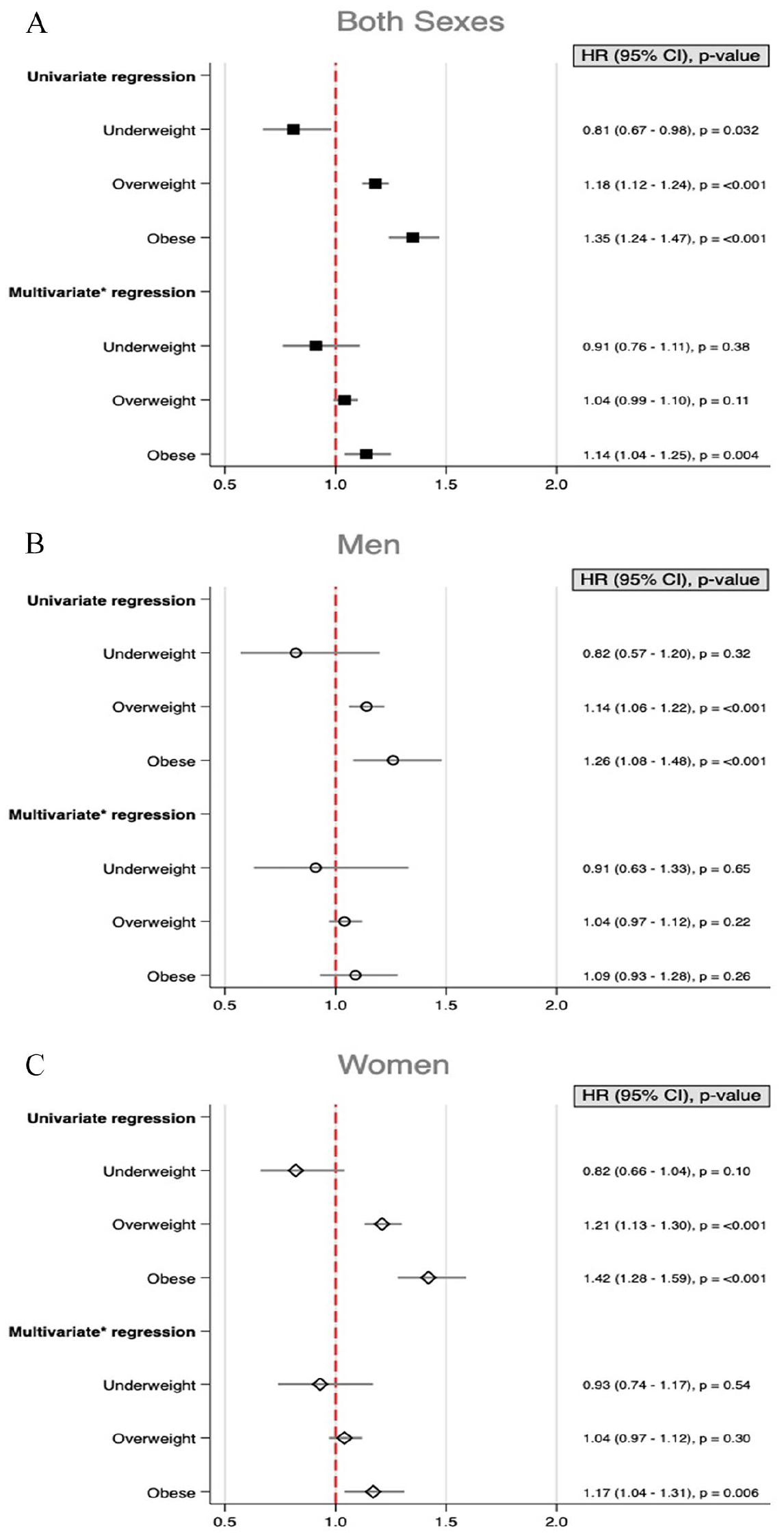
Risk of cancer-related death for patients diagnosed with pancreatic adenocarcinoma, stratified by body mass index (BMI) categories. Separate figures for (A) both sexes, (B) men, and (C) women.
Surgery for PDAC
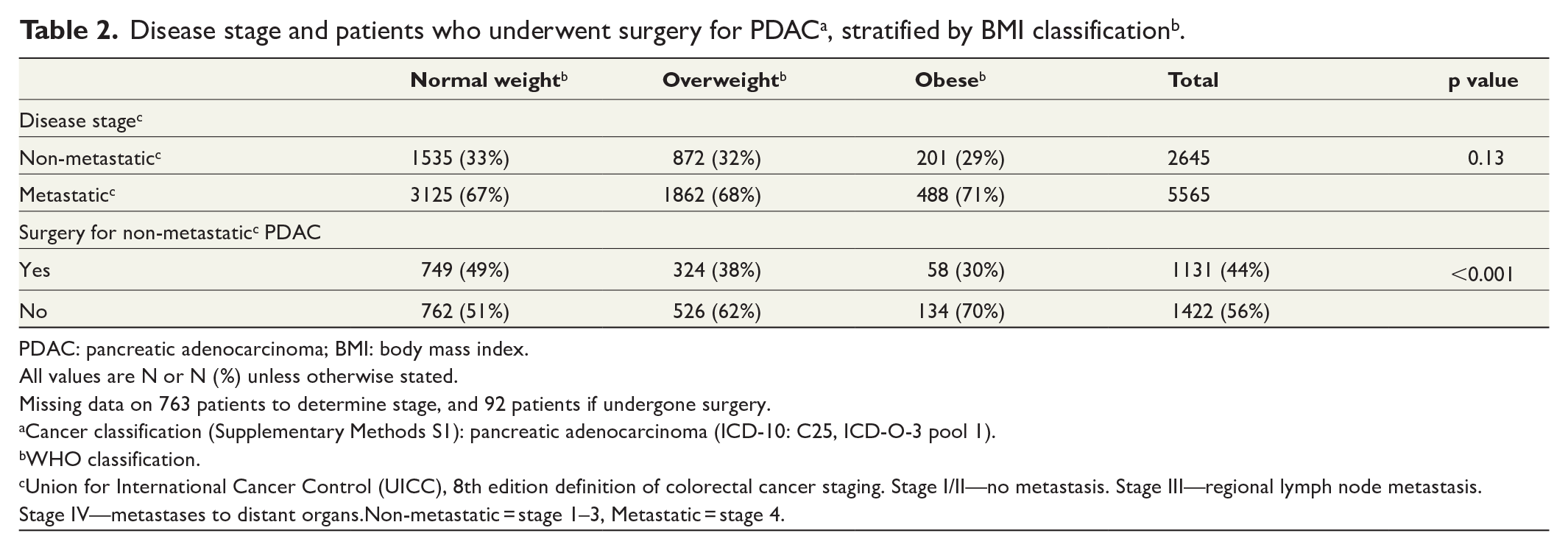
Of all patients diagnosed with PDAC, 2645 (32%) presented with UICC stage 1–3 (non-metastatic) disease (Table 2), of whom 1131 (44%) underwent curative surgery. When stratified by BMI categories, a higher proportion (49%) of normal weight non-metastatic PDAC patients underwent curative surgery than patients with overweight (38%) or obesity (30%) (p < 0.001).
PDAC: pancreatic adenocarcinoma; BMI: body mass index.
All values are N or N (%) unless otherwise stated.
Missing data on 763 patients to determine stage, and 92 patients if undergone surgery.
Cancer classification (Supplementary Methods S1): pancreatic adenocarcinoma (ICD-10: C25, ICD-O-3 pool 1).
WHO classification.
Union for International Cancer Control (UICC), 8th edition definition of colorectal cancer staging. Stage I/II—no metastasis. Stage III—regional lymph node metastasis. Stage IV—metastases to distant organs.Non-metastatic = stage 1–3, Metastatic = stage 4.
Discussion
To our knowledge, this is the largest cohort study of cancer cases investigating the association between pancreatic cancer and BMI that only includes adenocarcinomas. Our results extend and elaborate available evidence on the association between BMI and PDAC risk and survival/mortality. A main finding is no increased risk of developing PDAC, for both sexes with increasing BMI except for persons with high BMI at young age. A novel finding is the increased risk of PDAC at ages <50 years with increasing BMI in men.
The association of BMI and pancreatic cancer has been shown in several epidemiological studies, including meta-analyses, but with some discepencies.6,13,15 Most publications describe the association as strong, 6 but some also report no or weak associations. 5 However, as far as we know, no publications discriminate between histological subtypes. Our findings of no increased risk of developing PDAC for all age groups combined, and for both sexes, support the notion that increasing BMI may not be associated with an increased risk of PDAC.
However, our findings support an increased PDAC risk with increasing BMI in young adulthood (16–29 years), for both sexes, in accordance with previous studies.16,18,19
The biological etiology fueling the risk of PDAC with increasing BMI is not fully understood. Suggested mechanisms include expansion of adipose tissue causing desmoplasia, promoting transformation to malignant cells, or hyperinsulinemia and rise in insulin-like growth factor, which activates several cancer pathways. 29 A plausible explanation for the increased risk of PDAC with high BMI in early life could be sustained exposure to cancer-promoting factors from a young age, also supporting a hypothesis that obesity in early life may cause early-onset (<age 50) pancreatic cancer. There are reports of 2–6 years earlier onset of pancreatic cancer in patients with overweight and obesity; however, the median age at diagnosis was approximately 60 years. 18 As early-onset PDAC is uncommon, 30 a cohort with large number of cancer cases is needed to explore a potential association between BMI and early-onset PDAC. Among men, a 52% increased risk of early-onset PDAC (<age 50) was found in our study per 5 kg/m2 increase in BMI. This association was not seen in women. The reason for this gender difference is unclear. Female sex hormones have been suggested to protect against pancreatic cancer thus potentially postponing the increase in incidence for females to the postmenopausal period. 31 Although early onset PDAC is uncommon, an increased incidence has been observed with time that appears not related to earlier detection of the disease. 30 Increasing BMI in younger individuals could be a potential explanation.
Our findings in young individuals could be relevant for public health care strategies. Potential preventive measures for high-risk groups could include individualized lifestyle modification strategies or the use of bariatric surgery for selected obese younger individuals. A reduced risk of pancreatic cancer following weight loss induced by bariatric surgery has been suggested. 32 Future studies are needed to strengthen the evidence for this finding.
The stigma of obesity is reported present in various aspects of daily life but also within health care. 33 However, a large-scale study did not find any evidence of inferior care given to individuals with overweight or obesity. 34 We found that patients with overweight and obesity were less likely to undergo curative surgery for PDAC compared with normal weight patients. Challenging surgery with potential high risk of complications and morbidity related to obesity may contribute to these findings. 35 However, a recent report described the same postoperative morbidity after pancreatic surgery across BMI categories, concluding that pancreatic surgery should be provided to eligible patients regardless of BMI. 36
Our findings of reduced cancer survival in PDAC for individuals with high BMI are consistent with previous studies.18,21,22 However, the multivariable analysis shows an increased risk of PDAC-related death in individuals with obesity when analyzing both genders combined and women alone.
A challenge of using BMI as a measure of overweight or obesity is that it does not distinguish fat from muscle or bone, nor indicate visceral fat or muscle fat distribution. However, at a population level the impact of this potential source of misclassification is likely to be small as individuals with BMI of 30 or more who are not obese represent a small proportion of the total population. 37
In our data set, BMI was measured once and thus acted as a proxy for lifetime BMI. However, several studies have shown increase in weight and BMI at a population level over time and by increasing age. 38 Since the time of NTSP screening, the rate of obesity in the Norwegian population increased from 10% to some 20%–25%. 37 For 3190 individuals (1690 women), we had a second BMI measurement, obtained at time of cancer diagnosis. The mean interval between the measurements was 47.3 years (SD 3.0). As expected, we found an increase in BMI, mean 2.2 kg/m2 (SD 4.6) for both genders combined, 2.4 kg/m2 (SD 4.3) for men, and 2.0 kg/m2 (SD 4.8) for women. The BMI at the second measurement could be affected by the malignancy with a possible even larger increase prior to cancer diagnosis.
Strength/limitation
This study presents an unprecedented number of PDAC patients, and to the best of our knowledge, the largest series in a single cohort study. Long follow-up, high-quality, and complete data are strengths of this study which enables accurate precision and statistical power with in-depth sub-analysis. Only adenocarcinomas were included reducing tumor heterogeneity. At the time of NTSP screening, the Norwegian population was homogeneous Caucasian, making our results applicable to Caucasian populations. There was no major selection bias in the NTSP, and the chance of reverse causality is eliminated with BMI measurements long time before cancer diagnosis.
Limitations include lack of data on potential confounders such as smoking, alcohol, diet, physical activity, medications, and medical conditions including diabetes. There are studies that report a weak association between BMI and pancreatic cancer in general, but an increased risk in individuals who have never smoked. 5 An association between high BMI, smoking, and pancreatic cancer has been previously shown. 39 As smoking is inversely associated with obesity, 40 the lack of information on smoking might underestimate our results.
Conclusion
Our findings suggest that high BMI in early adulthood increases the risk of pancreatic cancer and that men with high BMI have increased risk of early-onset PDAC (age <50 years). Individuals with overweight and obesity were less likely to have curative surgery for PDAC, and individuals with obesity had a higher risk of PDAC-related death.
Supplemental Material
sj-docx-1-sjs-10.1177_14574969221127530 – Supplemental material for Body mass index and pancreatic adenocarcinoma: A nationwide registry-based cohort study
Supplemental material, sj-docx-1-sjs-10.1177_14574969221127530 for Body mass index and pancreatic adenocarcinoma: A nationwide registry-based cohort study by Usman Saeed, Tor Å. Myklebust, Trude E. Robsahm, Bjørn Møller, Tom Mala, Bjørn S. SkÅlhegg and Sheraz Yaqub in Scandinavian Journal of Surgery
Supplemental Material
sj-docx-2-sjs-10.1177_14574969221127530 – Supplemental material for Body mass index and pancreatic adenocarcinoma: A nationwide registry-based cohort study
Supplemental material, sj-docx-2-sjs-10.1177_14574969221127530 for Body mass index and pancreatic adenocarcinoma: A nationwide registry-based cohort study by Usman Saeed, Tor Å. Myklebust, Trude E. Robsahm, Bjørn Møller, Tom Mala, Bjørn S. SkÅlhegg and Sheraz Yaqub in Scandinavian Journal of Surgery
Supplemental Material
sj-jpg-3-sjs-10.1177_14574969221127530 – Supplemental material for Body mass index and pancreatic adenocarcinoma: A nationwide registry-based cohort study
Supplemental material, sj-jpg-3-sjs-10.1177_14574969221127530 for Body mass index and pancreatic adenocarcinoma: A nationwide registry-based cohort study by Usman Saeed, Tor Å. Myklebust, Trude E. Robsahm, Bjørn Møller, Tom Mala, Bjørn S. SkÅlhegg and Sheraz Yaqub in Scandinavian Journal of Surgery
Footnotes
Correction (February 2024):
Article updated online to include the affiliation for the first author Usman Saeed to the following: “Institute of Clinical Medicine, University of Oslo, Norway”.
Author contribution
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Tweet
Data from a large cohort study show that men with high BMI have an increased risk of early-onset pancreatic cancer and individuals with obesity have a higher risk of pancreatic cancer-related death.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.