Abstract
Background and objective:
Exposures of gallstones and treatments thereof in relation to development of cancer have not been explored before in long-term follow-up studies. Our objective was to determine whether symptomatic gallstones, cholecystectomy, or sphincterotomy were associated with development of upper gastrointestinal cancers.
Methods:
This is a nationwide cohort study of persons born in Denmark 1930–1984 included from age 30 years with long-term follow-up (1977–2014). Exposures were hospital admissions with gallstones, cholecystectomy, and sphincterotomy. Time-varying covariates were included in analyses to allow the impact of exposures to change with time. Follow-up periods were 2–5 and > 5 years. Hazard ratios (HR) with 95% confidence intervals (CI) were reported.
Results:
A total of 4,465,962 persons were followed. We found positive associations between sphincterotomy and biliary (>5 years HR 4.34, CI [2.17–8.70]), gallbladder (2–5 years HR 20.7, CI [8.55–50.1]), and pancreatic cancer (2–5 years HR 3.68, CI [2.09–6.49]). Cholecystectomy was positively associated with duodenal (2–5 years HR 2.94, CI [1.31–6.58]) and small bowel cancer (2–5 years HR 2.75, CI [1.56–4.87]). Inverse associations were seen for cholecystectomy and biliary (>5 years HR 0.60, CI [0.41–0.87]), pancreatic (>5 years HR 0.45 CI [0.35–0.57]), esophageal (>5 years HR 0.57, CI [0.43–0.74]), and gastric cancer (>5 years HR 0.68, CI [0.55–0.86]) and for gallstones and pancreatic cancer (>5 years HR 0.66, CI [0.47–0.93]). Gallstones were positively associated with gallbladder (>5 years HR 3.51, CI [2.02–6.10]) and small bowel cancer (2–5 years HR 3.21, CI [1.60–6.45]).
Conclusions:
A positive association between sphincterotomy and biliary cancer was identified. Cholecystectomy seems to be inversely associated with biliary, pancreatic, esophageal, and gastric cancer. Associations should be explored in similar large cohorts.
Keywords
Context and Relevance
Specific exposures of gallstones, cholecystectomy, and sphincterotomy in relation to development of upper gastrointestinal cancer have not been explored before in long-term follow-up studies. In this nationwide cohort study including persons born in Denmark 1930–1984, we find a positive association between sphincterotomy and biliary cancer after more than 5 years of follow-up. Cholecystectomy seems to be inversely associated with biliary, pancreatic, esophageal, and gastric cancer.
Introduction
Gallstones and possible derived treatments have shown to be associated with occurrence of several gastrointestinal cancers.1,2 Previous smaller clinical studies have suggested associations for cancer and sphincterotomy3–5 and larger cohorts also for cholecystectomy, 2 but have been unable to explore whether it is the presence of gallstones or their treatments that cause development of gastrointestinal cancers. This may be due to unavailability of lifelong follow-up data, not exploring the impact of sphincterotomy, and for not including the changing exposures for gallstones and treatments in time-to-event analyses with long-term follow-up. There is a need for exploring potential harmful effects of specific gallstone treatments.
Gallstones are highly prevalent in general populations and their associated clinical conditions lead them to be among the most frequent gastrointestinal diagnoses for hospital admissions causing high health care costs and burden.6,7 Standard treatment for symptomatic gallstones is cholecystectomy. 8 Laparoscopic cholecystectomy is the most frequent surgical procedure of the gastrointestinal system in Scandinavian countries, and frequencies are on the rise in other European countries.9,10 Endoscopic retrograde cholangiography (ERC) is performed when common bile duct stones are suspected with the aim of performing therapeutic sphincterotomy and stone extraction. 8 ERC rates have been rapidly increasing within the recent decade in North America.11,12
The sequence of clinical events in persons with gallstones may include a period from diagnosis of symptomatic gallstones to the performance of surgical treatments, which may be years apart. This causes a long period with changing exposures of gallstones, sphincterotomy, and cholecystectomy. All hospital contacts for Danish residents are registered centrally with diagnose and intervention codes, enabling long-term and complete follow-up. 13 Thereby, the full impact of gallstones, sphincterotomy, cholecystectomy, and the changing exposures throughout a lifetime on incidence of gastrointestinal cancers may be explored.
The objective was to determine whether hospital admissions with gallstones, cholecystectomy, or sphincterotomy were associated with development of site-specific upper gastrointestinal cancer divided into biliary, gallbladder, hepatic, pancreatic, esophageal, gastric, duodenal, and small bowel cancer in a nationwide population cohort with long-term follow-up.
Methods
This is a nationwide cohort study including all persons born in Denmark in the years 1930–1984. Persons were included when turning 30 years of age as this was considered relevant for gallstone formation and for established socioeconomic conditions. Persons were followed until occurrence of upper gastrointestinal cancer, death, hepatic metastasis, or were censored if moving out of the country, whichever came first. Hepatic metastasis may be the debuting sign of disseminated gastrointestinal cancer without a site-specific cancer of origin ever being identified, and therefore these cases were censored if occurring before a site-specific gastrointestinal cancer. Persons were excluded if the explored cancers or liver metastasis had occurred before the age of 30 years. A varying number of persons may therefore have been included at baseline in analyses of site-specific cancer outcomes. When exploring gallbladder cancer, we censored patients that had cholecystectomy performed.
Persons were followed-up through Danish national registers. The Danish Central Person Registry contains daily updated information on vital status and migration of the entire nation. A unique personal registration number enables linkage to other registers. 13 The National Patient Registry contains data on all hospital admissions from the year 1977 and emergency room visits from 1995. It includes diagnose codes according to the International Classification of Disease (ICD 8 or ICD 10—ICD 9 was never used in Denmark) and surgical intervention codes according to the Nordic Classification of Surgical Procedures. Codes are registered at the time of discharge from the hospital. 14 The Danish Register of Causes of Death contains one underlying cause of death with up to three contributory causes according to the ICD since 1970. 15 The National Patient Registry and The Danish Register of Causes of Death were used for identification of cancer outcome diagnoses, and The National Patient Registry was used to assess gallstone and treatment exposures. Statistics Denmark was used to obtain data on socioeconomic conditions. The National Patient Registry was updated at the end of 2014, The Danish Register of Causes of Death at the end of 2015, and The Danish Central Person Registry and Statistics Denmark in 2016. All registers have been used frequently in epidemiological research.13–15
The outcome of interest was site-specific upper gastrointestinal cancer divided into biliary, gallbladder, hepatic, pancreatic, esophageal, gastric, duodenal, and small bowel cancer. Exposure variables included cholecystectomy, sphincterotomy, presence of both interventions performed, or gallstones without interventions performed. For detailed definitions of exposures and outcomes see Table 1.
Intervention and diagnosis codes.
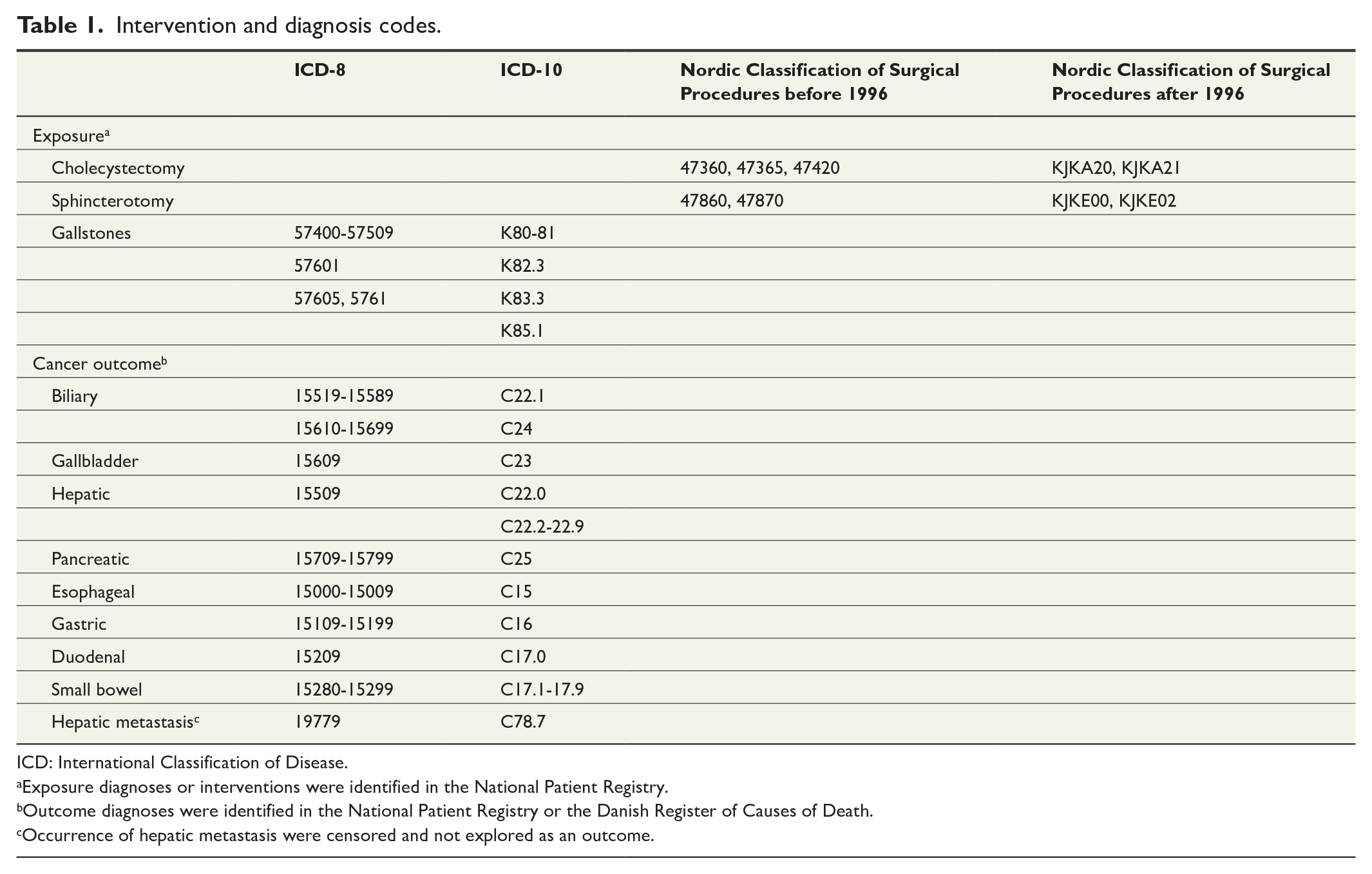
ICD: International Classification of Disease.
Exposure diagnoses or interventions were identified in the National Patient Registry.
Outcome diagnoses were identified in the National Patient Registry or the Danish Register of Causes of Death.
Occurrence of hepatic metastasis were censored and not explored as an outcome.
Socioeconomic variables were included as covariates in multivariable models and served as proxy for lifestyle and anthropometric data, as both gallstones and gastrointestinal cancer may be associated with these. Socioeconomic variables were assessed once when the person was 30 years of age where possible. Covariates included sex (female/male), socioeconomic status (employee, self-employed/manager, retired, student, temporary transfer income/on leave, transfer income/unemployed), civil status (married/registered partner, unmarried, widowed, divorced), level of education (high school/elementary school, vocational training, short/medium-term education, higher university education), and personal annual income (DKK). Primary sclerosing cholangitis is highly associated with biliary cancer when compared with the risk in the general population. 16 Sensitivity analyses were, therefore, performed to explore the possible confounding pathway for primary sclerosing cholangitis, sphincterotomy, and biliary cancer. The study was reported according to the STROBE statement.
Ethical approval for performing the study was given through the Danish Data Protection Agency and the Capital Region of Denmark (journal number CSU-FCFS-2017-007, I-Suite number 05575). As no new human data were collected, no participants provided informed consent was required.
Statistical analyses
Competing risk analyses 17 were used with age as the underlying timescale. Death was used as the competing cause when analyzing the occurrences of the site-specific cancer outcomes. We modeled the cause-specific hazard functions using the Cox proportional hazards model 18 with time-varying covariates to allow the impact of the different exposures to change with time. Time periods of 0–2 years, 2–5 years, and more than 5 years since exposure was used. Unexposed persons served as the controls. Based on the cause-specific hazard models, we computed cumulative incidence functions to quantify exposure effects on the probability scale. We did not report results from the period of 0–2 years since exposure to avoid prevalent cancers. We report number of outcome cases, total persons in exposure category, person years, incidence rate as cases per 100,000 person years, crude cumulative incidence curves, and hazard ratios (HR) with 95% confidence intervals (CI) both unadjusted and adjusted for baseline covariates. Significance level is set as a 95% CI not including one. Statistical software R with the “survival” package was used for analyses.
Results
The nationwide cohort included 4,465,962 persons in the period 1977–2014. For baseline characteristics, see Table 2. During follow-up, the most frequently occurring cancers were pancreatic, gastric, and esophageal. The rarest occurring cancers were duodenal, gallbladder, and small bowel. Hepatic and biliary cancers occurred with frequencies in between (Table 3).
Baseline characteristics of the study population born 1930–1984 (n = 4,465,962 a ).
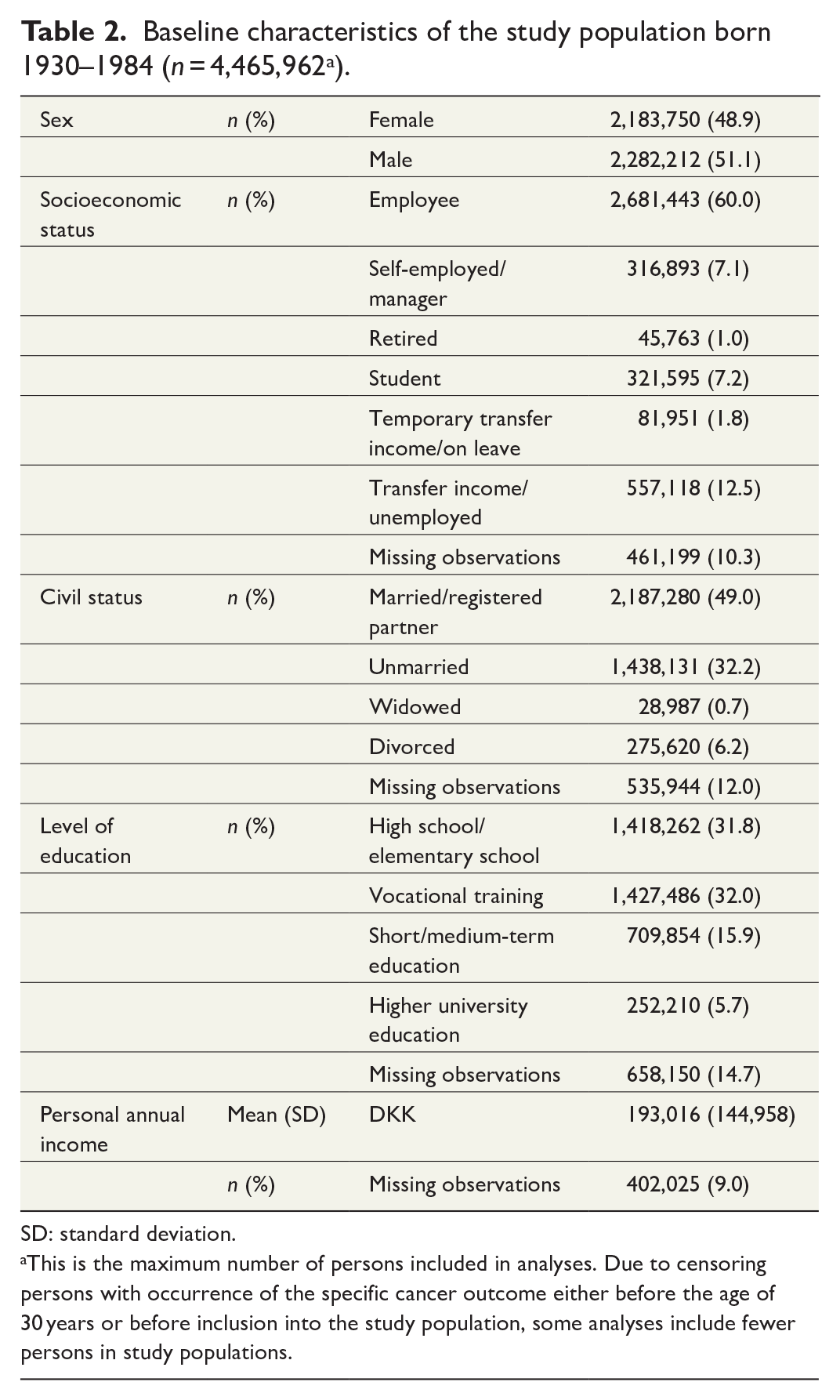
SD: standard deviation.
This is the maximum number of persons included in analyses. Due to censoring persons with occurrence of the specific cancer outcome either before the age of 30 years or before inclusion into the study population, some analyses include fewer persons in study populations.
Gallstones and treatments thereof in relation to development of upper gastrointestinal cancers.
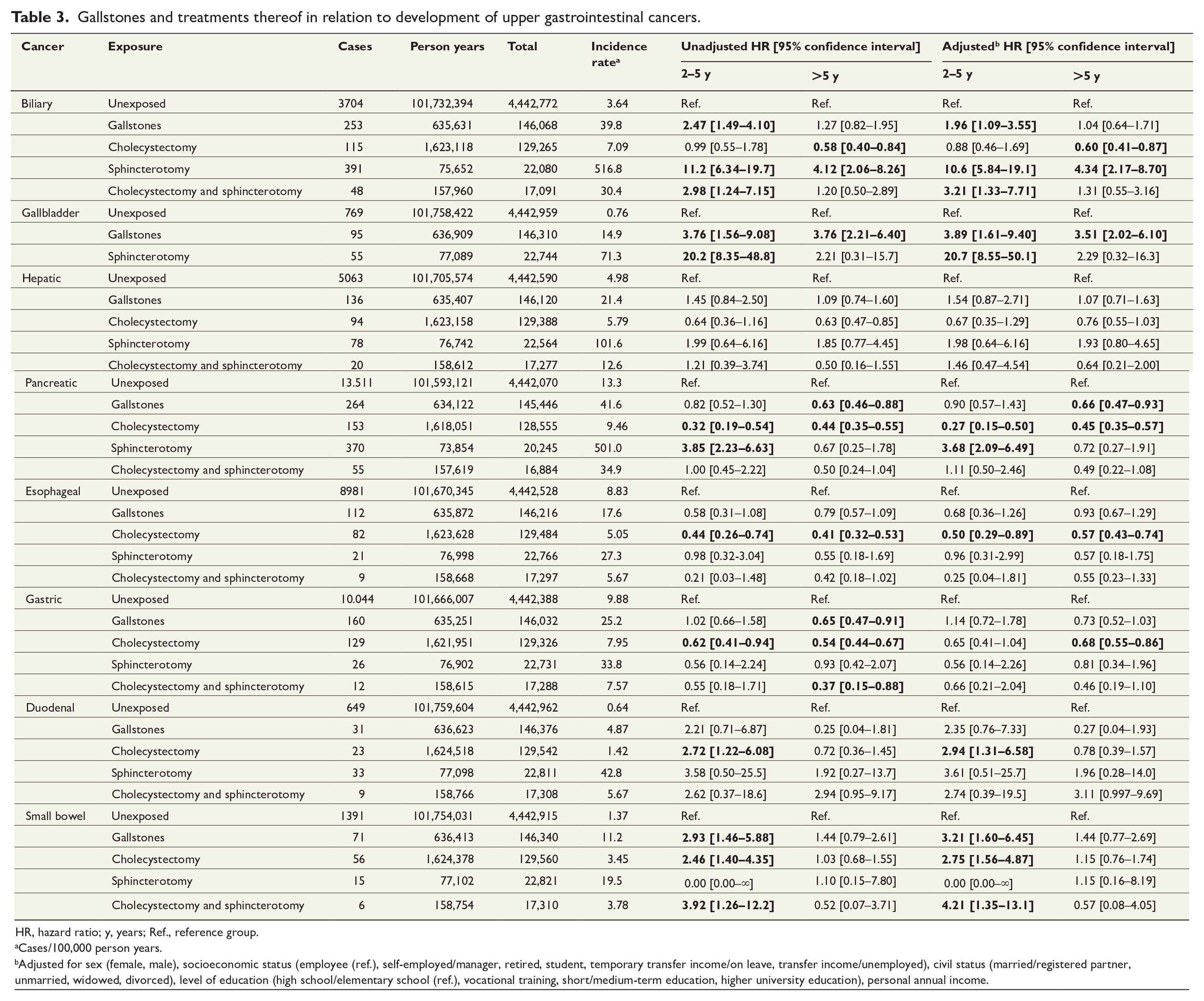
HR, hazard ratio; y, years; Ref., reference group.
Cases/100,000 person years.
Adjusted for sex (female, male), socioeconomic status (employee (ref.), self-employed/manager, retired, student, temporary transfer income/on leave, transfer income/unemployed), civil status (married/registered partner, unmarried, widowed, divorced), level of education (high school/elementary school (ref.), vocational training, short/medium-term education, higher university education), personal annual income.
When inspecting the crude cumulative incidence curves, patients with gallstones have higher incidence of biliary and gallbladder cancer than controls, whereas patients after sphincterotomy have much higher incidence of biliary, gallbladder, hepatic, and pancreatic cancer. Patients with both cholecystectomy and sphincterotomy have higher incidence of biliary and pancreatic cancers. Patients with cholecystectomy have lower incidence of pancreatic and gastric cancer than controls (Fig. 1).
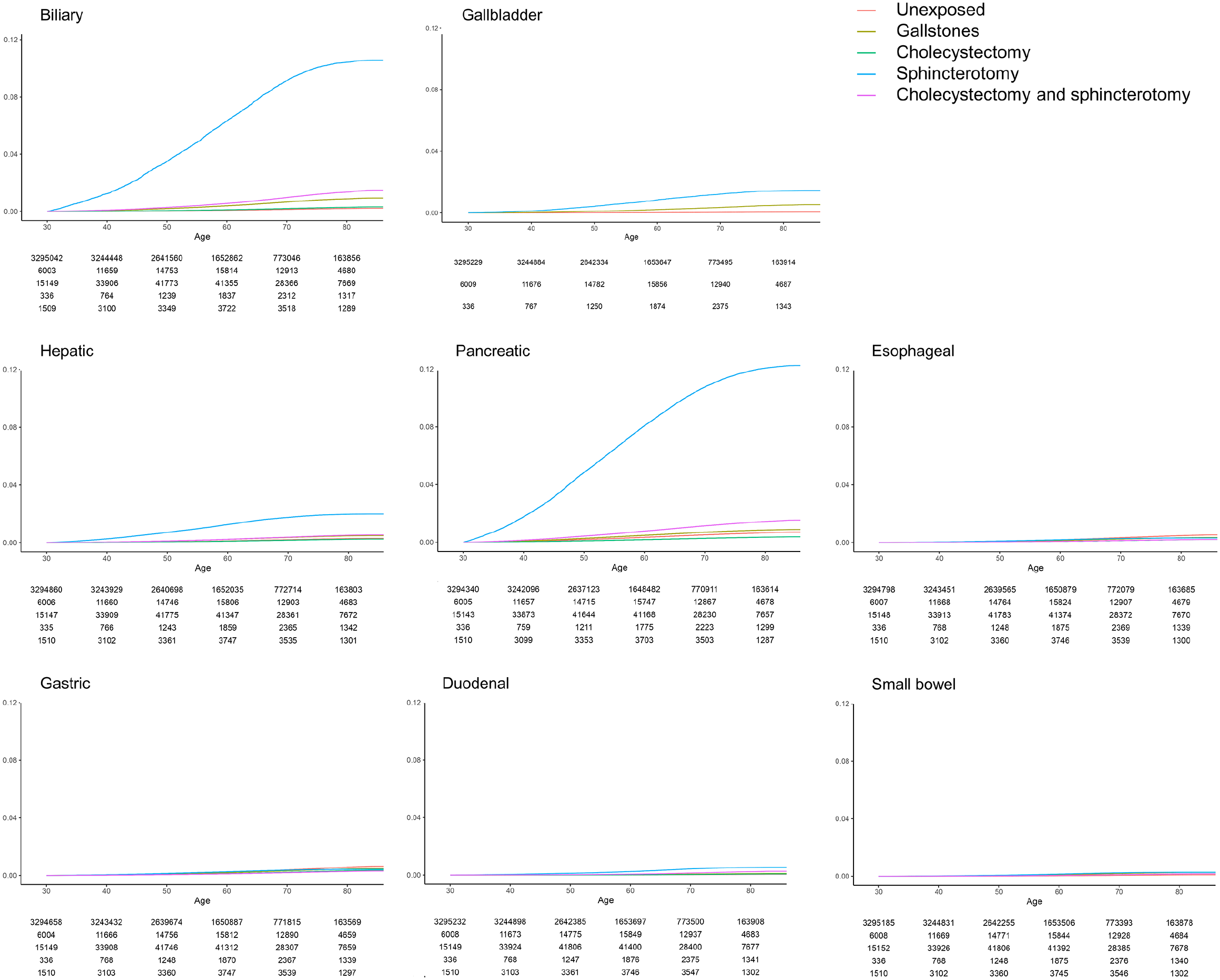
Cumulative incidence curves for cancer outcomes.
Estimates for unadjusted and adjusted analyses were very similar regarding significance level in both exposure periods, except for gastric cancer (Table 3).
Patients with gallstones significantly more often developed biliary and small bowel cancer at 2–5 years of follow-up and gallbladder cancer at above 5 years compared with controls. An inverse association for gallstones and pancreatic cancer at above 5 years follow-up was seen. Those with cholecystectomy significantly more often developed duodenal and small bowel cancer at 2–5 years. Sphincterotomy caused both gallbladder and pancreatic cancer development significantly more often at 2–5 years. Sphincterotomy caused biliary cancer development significantly more often at above 5 years. Those with both cholecystectomy and sphincterotomy had significant associations for biliary and small bowel cancer at 2–5 years. Inverse associations for cholecystectomy and biliary, pancreatic, esophageal, and gastric cancer at above 5 years were found (Table 3).
Sensitivity analysis for biliary cancer with addition of primary sclerosing cholangitis to the multivariable adjusted model did not change estimates substantially (Supplemental Material).
Discussion
Through long-term follow-up of a nationwide population sample and with time-varying exposures for symptomatic gallstones and treatments thereof, we found associations for sphincterotomy and biliary, gallbladder, and pancreatic cancer. Cholecystectomy was associated with duodenal and small bowel cancer. Gallstones were associated with gallbladder and small bowel cancer. Inverse associations were found for cholecystectomy and biliary, pancreatic, esophageal, and gastric cancer and for gallstones and pancreatic cancer.
We define strong and precise associations as statistically significant estimates with narrow confidence intervals and as being present after 5 years of follow-up. Such associations were identified for sphincterotomy and biliary cancer, for gallstones and gallbladder cancer, and the inverse association for cholecystectomy and pancreatic cancer. The association for sphincterotomy and biliary cancer was not altered with the inclusion of primary sclerosing cholangitis in the multivariable model indicating no issues with confounding. Both gallstones and cholecystectomy were associated with small bowel cancer. Associations did not persist beyond 5 years of exposure and it may therefore be difficult to interpret whether gallstones or cholecystectomy caused cancer development.
Another study based on the National Health Insurance Research Database in Taiwan suggested associations for sphincterotomy or sphincter of Oddi balloon dilatation and biliary cancer and also the inverse association for cholecystectomy and biliary cancer when compared with controls with gallstones. 19 The study is not consistent with our study as no general population controls without gallstones or treatments were used, adjusted time-to-event analysis were not performed, the study population was much smaller, and changing exposures were not accounted for. A population-based cohort study from Sweden identified gallstones to be associated with development of biliary cancer 20 consistent with our study. The study also found that cholecystectomy reduced the risk of biliary cancer back to the level of the background population with time after cholecystectomy based on similar standardized incidence ratios. The authors suggested mechanisms of chronic biliary inflammation in presence of gallstones which may be altered by cholecystectomy. 20 Another study finds gallstones associated with pancreatic cancer when compared with population controls, but finds no significant associations following cholecystectomy. 21 These results are not consistent with our findings of an inverse association for cholecystectomy and biliary or pancreatic cancer when compared with the general population, but they do suggest a protective mechanism of cholecystectomy in patients with gallstones. The association for gallstones and gallbladder cancer has been reported several times before. 2 To our knowledge, this is the first study to report an inverse association for cholecystectomy and pancreatic cancer as well as the other inverse associations for cholecystectomy in patients with gallstone treatments when compared with the background population.
Cholecystectomy causes a continuous bile flow into the bowel22,23 and an altered microbiota with increased bacterial dehydroxylation of primary bile acids into secondary bile acids23,24 with the latter being carcinogenic.25,26 Secondary bile acids and the bacteria producing them are also found in higher amounts in persons with gallstones and gallbladders in situ compared with persons without gallstones,27–30 suggesting similar pathophysiological mechanisms in the non-cholecystectomy state. Sphincterotomy eliminates the function of the sphincter of Oddi and removes the common bile duct to duodenal pressure gradient which is believed to cause reflux of duodenal content into the common bile duct and pancreatic duct. 31 Following sphincterotomy, a number of changes in bile and common bile duct are found, including colonization with bowel bacteria, 32 higher amounts of secondary bile acids and cytotoxic agents, histological chronic inflammation, 33 and atypical cells indicating cell proliferation. 34 These experimental observations indicate a plausible and coherent cancer transformation mechanism seen with gallstones and sphincterotomy for gallbladder and biliary cancer. Reflux with chronic biliary inflammation has also been suggested as mechanism for biliary cancer transformation in smaller clinical series following patients with sphincterotomy.3,5 The suggested mechanisms may also explain the possible mechanisms for duodenal and small bowel cancer development following cholecystectomy. No apparent mechanisms may be suggested to explain the inverse associations for cholecystectomy and cancer of the more oral sites as found in this study. Further studies are awaited.
The strength of this study is that it is the first cohort study to explore associations for specific and changing exposures of gallstones, treatments thereof, and cancer outcome in the general population. The comprehensiveness of the databases used in this study enabled all exposure treatments and occurrences of cancer to be registered nationwide without loss. The study population was also the largest reported in cohort studies exploring gallstones, treatments thereof, and cancer at the time of publication. Symptoms from a cancer may be clinically misinterpreted as gallstones as suggested earlier. 35 To avoid such diagnostic mimicry and risk of detection bias and to secure temporal associations, we did not include estimates during the first 2 years of follow-up. Limitations of this study include that only symptomatic gallstones causing hospital admission were identified. We know from a previous cohort study of screen-detected gallstones found in a population with systematic ultrasound screening, that clinically identified gallstones leading to hospital admission are only accountable for about one-fifth of persons with gallstones. Furthermore, specific characteristics determine symptomatic gallstones.36,37 The true gallstone prevalence is therefore underestimated in this study with risk of potential differential misclassification bias. This could cause both significant and nonsignificant estimates when the gallstone exposure was explored, and these results should therefore be interpreted with caution. Estimates for sphincterotomy and cholecystectomy do not have such risk. Anthropometric data were not available which could cause residual confounding, but we included socioeconomic variables as proxy in models to minimize the impact of such potential bias. Due to differences in initiation dates of the registries used in this study, persons may have been older than 30 years when included. This baseline difference was addressed through having age as the underlying timescale.
The strong and precise association for sphincterotomy and biliary cancer found in this large nationwide population-based cohort with long-term and advanced follow-up analyses suggest a causal association. The association’s consistency should be explored in future cohort studies including similarly large populations and design. ERC with sphincterotomy is presently the most utilized treatment for common bile duct stones. 38 Alternatives for therapeutic common bile duct stone extraction without the use of sphincterotomy are available and have comparable clinical outcomes. 39 For now, caution with performance of sphincterotomy is advised and especially in younger patients.
In conclusion, this large nationwide population-based cohort study identified a novel association for endoscopic sphincterotomy and development of biliary cancer after more than 5 years of follow-up. Caution with use of sphincterotomy in younger patients may be advised until further studies are performed. Cholecystectomy seems to be inversely associated with biliary, pancreatic, esophageal, and gastric cancer. These associations should be explored in larger cohort studies with similar designs in the future.
Supplemental Material
sj-docx-sjs-10.1177_14574969221116941 – Supplemental material for Development of upper gastrointestinal cancer in patients with symptomatic gallstones, cholecystectomy, and sphincterotomy: A nationwide cohort study
Supplemental material, sj-docx-sjs-10.1177_14574969221116941 for Development of upper gastrointestinal cancer in patients with symptomatic gallstones, cholecystectomy, and sphincterotomy: A nationwide cohort study by Daniel M. Shabanzadeh, Torben Martinussen and Lars T. Sørensen in Scandinavian Journal of Surgery
Footnotes
Acknowledgements
The authors give credit to Sofie Korn and Frederik Mølkjær Andersen for performing data management and analyses, to Karin Mønsted Shabanzadeh for linguistic edit, and to Torben Jørgensen for his insights and help with the manuscript.
Author contributions
D.M.S. contributed to the study design, performing analyses, interpretation of results, drafting, and final acceptance of manuscript. T.M. contributed to performing analyses, interpretation of results, critical review, and final acceptance of manuscript. L.T.S. contributed to study design, interpretation of results, critical review, and final acceptance of manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by The Faculty of Health and Medical Sciences at the University of Copenhagen. The University of Copenhagen had no role in the study design or in the collection, analysis, and interpretation of data.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
