Abstract
Background and objective:
Gallstones are highly prevalent, and more than 9000 cholecystectomies are performed annually in Denmark. The aim of this guideline was to improve the clinical course of patients with gallstone disease including a subgroup of high-risk patients. Outcomes included reduction of complications, readmissions, and need for additional interventions in patients with uncomplicated gallstone disease, acute cholecystitis, and common bile duct stones (CBDS).
Methods:
An interdisciplinary group of clinicians developed the guideline according to the GRADE methodology. Randomized controlled trials (RCTs) were primarily included. Non-RCTs were included if RCTs could not answer the clinical questions. Recommendations were strong or weak depending on effect estimates, quality of evidence, and patient preferences.
Results:
For patients with acute cholecystitis, acute laparoscopic cholecystectomy is recommended (16 RCTs, strong recommendation). Gallbladder drainage may be used as an interval procedure before a delayed laparoscopic cholecystectomy in patients with temporary contraindications to surgery and severe acute cholecystitis (1 RCT and 1 non-RCT, weak recommendation). High-risk patients are suggested to undergo acute laparoscopic cholecystectomy instead of drainage (1 RCT and 1 non-RCT, weak recommendation). For patients with CBDS, a one-step procedure with simultaneous laparoscopic cholecystectomy and CBDS removal by laparoscopy or endoscopy is recommended (22 RCTs, strong recommendation). In high-risk patients with CBDS, laparoscopic cholecystectomy is suggested to be included in the treatment (6 RCTs, weak recommendation). For diagnosis of CBDS, the use of magnetic resonance imaging or endoscopic ultrasound prior to surgical treatment is recommended (8 RCTs, strong recommendation). For patients with uncomplicated symptomatic gallstone disease, observation is suggested as an alternative to laparoscopic cholecystectomy (2 RCTs, weak recommendation).
Conclusions:
Seven recommendations, four weak and three strong, for treating patients with symptomatic gallstone disease were developed. Studies for treatment of high-risk patients are few and more are needed.
Endorsement:
The Danish Surgical Society.
Context and Relevance
More than 9000 cholecystectomies are performed annually in Denmark. The latest Danish clinical practice guideline for the treatment of symptomatic gallstone disease was published in 2006. Other published international guidelines may not be entirely suitable for a healthcare system such as the Danish. There is a need for an updated guideline to optimize the course of patients with gallstone disease and the hospital resources. An interdisciplinary group working under the Danish Surgical Society developed new guidelines according to the GRADE methodology. Seven recommendations were reported including three strong and four weak. The recommendations describe the treatment of patients with uncomplicated gallstone disease, acute cholecystitis, and common bile duct stones including treatments for the subgroup of high-risk patients. We suggest several areas of interest for future research.
Introduction
Laparoscopic cholecystectomy is one of the most frequently performed surgical procedures in Scandinavia. More than 9000 procedures are performed annually in Denmark. 1 Standard treatment for gallstone disease is laparoscopic cholecystectomy, but treatments may also include other endoscopic and radiological procedures. 2 Gallstone disease causes high costs and is a burden to health care providers due to admissions and treatments. 3 Danish clinical practice guidelines were published in 2006. 4 An updated guideline including novel treatments and evidence is needed to ensure optimal patient treatment and appropriate use of health care resources. Current clinical challenges involve treatment of uncomplicated gallstone disease, acute cholecystitis, common bile duct stones (CBDS) and of the subgroup of high-risk patients.
Gallstones cause a continuum from an asymptomatic state to symptomatic disease including uncomplicated pain attacks and complications such as acute cholecystitis, CBDS, pancreatitis, and cholangitis. 5 The prevalence of gallstones is about 10%–20% in the general Danish population depending on age and gender. 6 About 20% of gallstone carriers will develop symptomatic gallstone disease requiring hospital admission of which 12% will have uncomplicated disease and 8% complicated disease. 7
There is no consensus regarding the diagnosis of acute cholecystitis. The Tokyo guidelines from 2013 and 2018 define this pathology as: (1) local signs of inflammation like Murphy’s sign or right upper quadrant tenderness, (2) systemic signs of inflammation such as fever or elevated serum markers of inflammation, and (3) imaging findings of acute cholecystitis such as gallstones and ultrasonic wall thickening, edema, or transducer tenderness. Acute cholecystitis is suspected if local and systemic signs are present and the diagnosis is definitive with imaging. It is graded as mild, moderate, or severe with the latter defined by the presence of organ failure. 8 The World Society of Emergency Surgery (WSES) does not suggest a set of criteria but merely emphasizes that the diagnosis should be based on a combination of detailed history, complete clinical examination, laboratory tests, and imaging. 9
Abdominal ultrasound is usually sufficient to diagnose gallbladder stones in patients with symptomatic gallstone disease. 10 A diagnosis of CBDS often requires invasive or non-invasive examinations that are not readily available and unnecessary for most cases. Simple clinical risk estimation tools have been developed to estimate the need for further diagnostic workup in patients with gallbladder stones. The European Society of Gastrointestinal Endoscopy (ESGE) and the American Society for Gastrointestinal Endoscopy (ASGE) define a high, intermediate and low risk of CBDS.11,12 High risk may be characterized by clinical signs of acute cholangitis or detection of CBDS on ultrasound. Furthermore, the ASGE includes severely elevated serum bilirubin in the high-risk group. Patients with a high risk may be treated directly with endoscopic retrograde cholangiography (ERC). An intermediate risk may be defined as elevated hepatic function tests and/or bile duct dilation on abdominal ultrasound. ASGE also includes an age above 55 years or clinical presence of gallstone pancreatitis. Intermediate risk patients should undergo magnetic resonance cholangiopancreatography (MRCP) or endoscopic ultrasound (EUS) examination to rule out CBDS. No risk factors predict a low risk of CBDS, and no further diagnostic workup is needed.
The definition of the high-risk surgical patient often relies on both surgical and anesthesiologic aspects. Several anesthesiologic risk estimation tools have been developed to identify high-risk patients based on pre-, intra-, and post-operative factors. 13 Previous laparotomy and obesity often result in exclusion from clinical trials due to a suspected higher risk of surgical complications.14,15 The perioperative mortality has significantly decreased over the last decades, especially throughout the 1990–2000s probably due to persistent changes and improvements of anesthesiologic and surgical practice. 16 The definition of the surgical high-risk patient is ultimately ill-defined and may have changed over time. Possibilities for and willingness to perform surgery in an aging high-risk patient population have increased over time.
Several pending clinical challenges have promoted the need for updated guidelines. Following cholecystectomy in patients with uncomplicated symptomatic gallstone disease, up to one in three patients report persistent upper abdominal pain.17,18 This may suggest challenges in patient selection or that too many cholecystectomies are currently performed. For acute cholecystitis, delayed versus acute surgery is persistently debated. Several arguments support delayed surgery, such as the idea of “cooling off a hot gallbladder,” patient comorbidity, delayed patient presentation, surgeon preference, and resource constraints with limited access to the operating room. 19 Gallbladder drainage is used as an alternative to surgery in high-risk patients or as a bridge to surgery. In high-risk patients with acute cholecystitis, arguments for the use of gallbladder drainage are the same as for delayed surgery in non-high-risk patients. 20 Gallbladder drainage may be used to treat moderate or severe acute cholecystitis according to the Tokyo guidelines. 21 The most commonly used treatment of CBDS is ERC-guided removal of CBDS and laparoscopic cholecystectomy as two separate procedures. 22 Newer treatments including one-step procedures have gained popularity recently including intraoperative ERC-guided or laparoscopic removal of CBDS during laparoscopic cholecystectomy.23,24
There are a few updated published international guidelines for treatment of symptomatic gallstones. The Tokyo Guidelines from 2018 propose frequent use of gallbladder drainage treatment and/or reference to an expert center for treatment of acute cholecystitis. 21 Such algorithms are not suitable for smaller countries like Denmark where high-volume laparoscopic cholecystectomy surgical centers are readily available as compared to larger countries such as Japan. Treatments for CBDS and acute cholecystitis are described in ESGE guidelines from 2019 11 and in WSES guidelines from 2020. 9 These comprehensive guidelines emphasize the multiple lines of treatments that may be applied based on local expertise. A small country like Denmark with a publicly financed healthcare system may apply nationwide changes in treatments more readily according to the highest level of evidence enabling more uniform treatments and guidelines.
The overall aim of this clinical guideline was to improve the clinical course of patients with gallstone disease including the subgroup of high-risk patients. The objectives were to reduce the rates of complications, readmissions with gallstone disease, and the need for additional interventions in patients with uncomplicated gallstone disease, acute cholecystitis, and CBDS. This guideline does not include treatment of patients with acute pancreatitis, severe acute cholangitis, or acalculous cholecystitis.
This guideline primarily addresses clinicians involved in the treatment of patients with gallstone disease in hospital settings, but also general practitioners in primary care and policy makers. Practice guidelines are essential to optimize the clinical course and reduce costs in a publicly financed healthcare system. These guidelines are particularly relevant for Scandinavian countries and other high or middle-income countries with similar healthcare systems. Although we aimed at the shortest patient course with lowest costs, we summarize the highest level of evidence for treatment of gallstone disease rather than mere cost-effectiveness analyses.
Methods
The steering group included two of the authors (LTS and DMS) who explored the need for updated guidelines in gallstone disease, which was confirmed by the Danish Surgical Society. The steering group invited a broad group of Danish healthcare professionals to participate in the guideline working group. The final working group included 14 clinicians from the five Danish regions involved in treatment of patients with gallstone disease and included surgeons, advanced endoscopy specialists, a gastroenterologist, a radiologist, and a nurse. The guideline was developed from May 2020 to October 2021 with regular online meetings. The working group was split into six subgroups that each worked with one or two of the defined clinical questions (see below). A final 2-day seminar with the entire working group was held in Copenhagen June 2021, and the entire guideline was presented and discussed. Consensus regarding recommendations was reached according to the used methodology and was defined as all group members agreeing on the recommendations. To ensure process and quality according to Danish Health Authority standards, a methodology consultant was hired to supervise the guideline process and analyses. The final guideline was reviewed at the Danish Health Authorities, Danish Regions, Danish Patients, the board of the Danish Surgical Society, and all other Danish Medical Societies. An experienced hepato-pancreatic-biliary surgeon and a clinical professor in general surgery performed external peer review. The reviewer comments were considered, and the guidelines were modified accordingly in agreement with the entire working group. Reviewer comments and answers were published online together with the final revised guideline. 25 The full report in Danish is available as Supplemental Material (Supplemental Material 1).
Based on current challenges in the treatment of gallstone disease, seven clinical questions were developed. The PICOS (patient, intervention, comparator, outcome, and study design) format served as the strategy that facilitated a systematic review process. Search strategies were based on keywords and Medical Subject Heading (MeSH) terms (Table 1). An information specialist designed search strategies and performed the literature searches. Five search strategies were conducted in September 2020. Systematic searches were performed in the databases CENTRAL, MEDLINE, Embase, LILACS, Science Citation Index Expanded, and Cochrane Hepato-Biliary Group Controlled Trials Register. Ovid was used to search, and results were transferred to the online reference program Covidence. All group members had access to Covidence. At least two independent working group members performed study selection, data extraction, and study quality assessment in each PICOS. Final assessment was according to subgroup discussion and consensus. No language restrictions were applied at study selection. PRISMA study flow-charts were obtained in Covidence. Available preliminary results from relevant ongoing trials were sought identified through online sources such as a trial webpage. The RIGHT statement was used to report recommendations.
Specific clinical questions answered in this guideline.
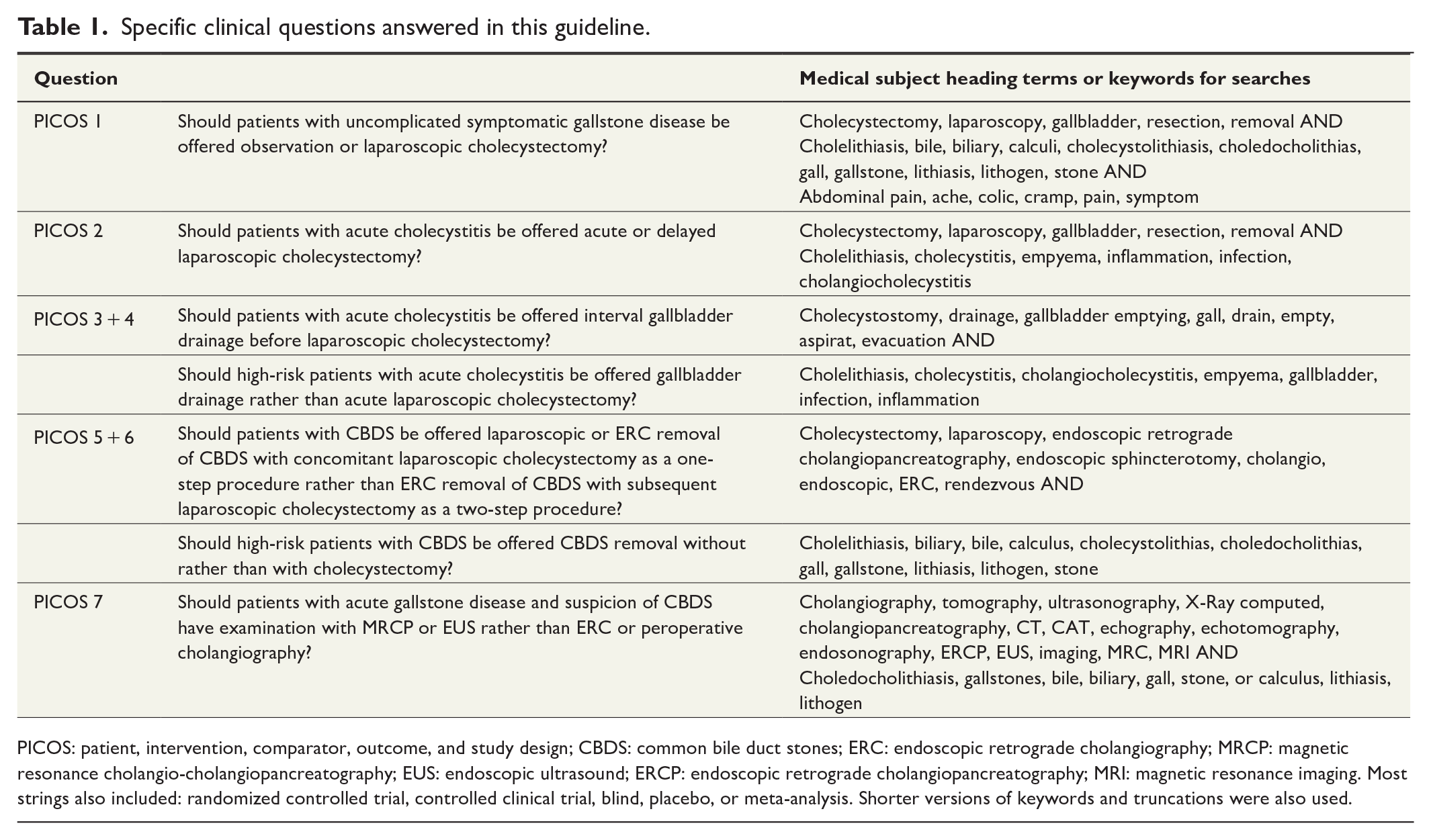
PICOS: patient, intervention, comparator, outcome, and study design; CBDS: common bile duct stones; ERC: endoscopic retrograde cholangiography; MRCP: magnetic resonance cholangio-cholangiopancreatography; EUS: endoscopic ultrasound; ERCP: endoscopic retrograde cholangiopancreatography; MRI: magnetic resonance imaging. Most strings also included: randomized controlled trial, controlled clinical trial, blind, placebo, or meta-analysis. Shorter versions of keywords and truncations were also used.
Odds ratio (OR) or mean difference were calculated with corresponding 95% confidence intervals (CIs). Risk in comparator and intervention groups were reported. Meta-analyses were performed where relevant with the random effects model. Review Manager version 5.4 was used for analyses. Analyses were performed by the first author (DMS) and the methodology consultant.
Evidence was assessed across studies on an outcome-by-outcome basis as suggested by the GRADE Working Group. GRADE methodology separates the assessment of the quality of evidence and of the strength of recommendations. Quality of evidence was assessed based on the risk of bias according to the Cochrane’s tool for randomized controlled trials (RCTs), imprecision, indirectness of evidence, inconsistency of results, and reporting bias. 26 Risk of bias in nonrandomized studies was assessed according to ROBINS-I. 27 Quality of evidence was defined as high, moderate, low, or very low. We defined quality of evidence as our confidence in the estimate of the effect to support a recommendation. The strength of the final recommendation was assessed as either strong or weak depending on the quality of evidence, desirable, and undesirable effects, and patient preferences. 26 All working group members either had experience with the GRADE methodology or participated in an online course held by the Danish Health Authorities.
Relevant clinical outcomes were chosen based on clinical experience and in respect of healthcare resources. Between two and three outcomes were considered critical for each PICOS and moderate to good quality of evidence was required to allow a strong recommendation. Post-operative complications and readmissions with gallstone disease were considered critical for most PICOS. Quality of life was included in most PICOS. Need for additional interventions and length of hospital stay were included when comparing specific procedures. When exploring high-risk patients, mortality was included. Assessing uncomplicated disease, absence or reduction of pain intensity, and the rate of cholecystectomies were included. Successful CBDS diagnosis or removal and conversion to open surgery were included if relevant. Patient-reported outcome measures (PROMs) were sought in the identified literature and in a separate literature search to explore patient perspectives. Outcome measures were defined prior to the results of the search strategies. All outcomes were defined in detail in Table 2.
GRADE summary of findings.
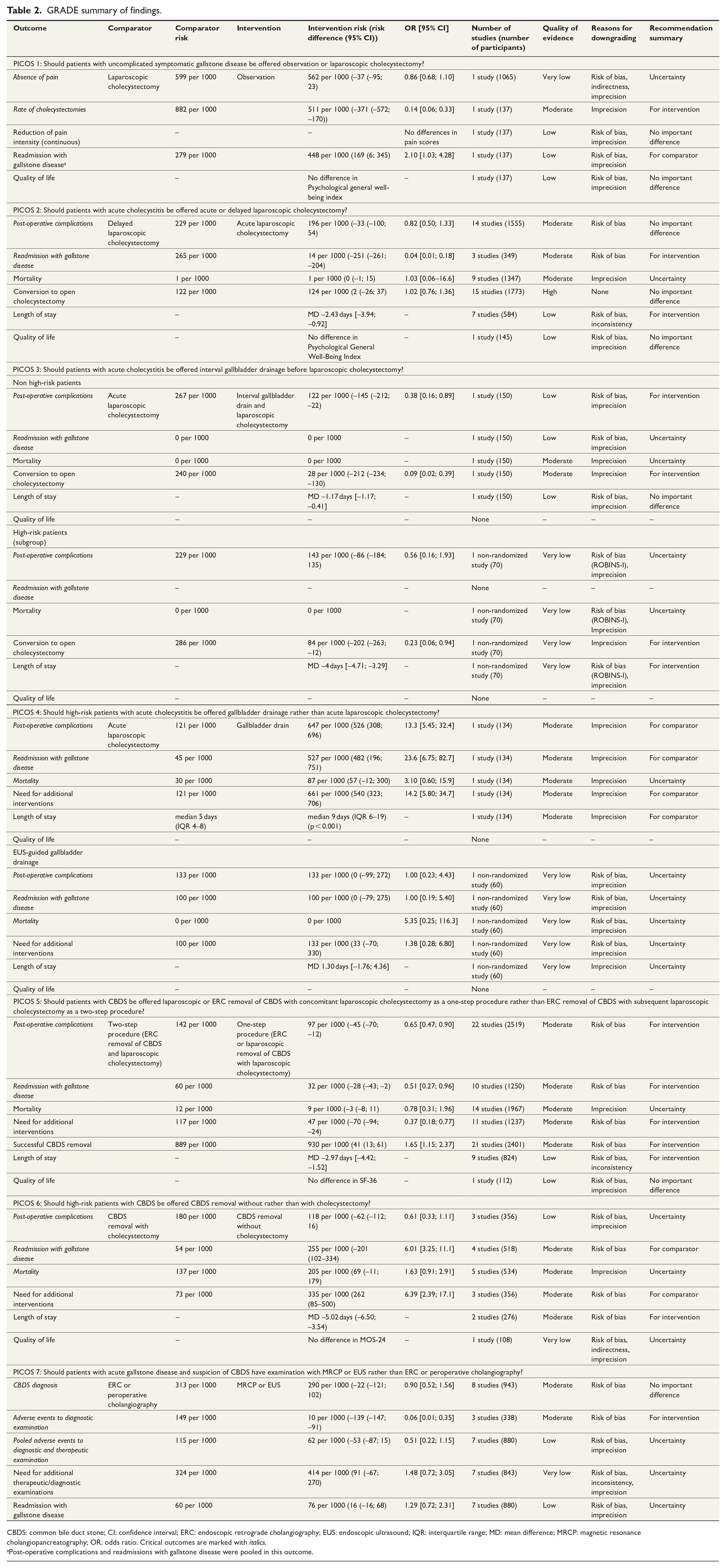
CBDS: common bile duct stone; CI: confidence interval; ERC: endoscopic retrograde cholangiography; EUS: endoscopic ultrasound; IQR: interquartile range; MD: mean difference; MRCP: magnetic resonance cholangiopancreatography; OR: odds ratio. Critical outcomes are marked with italics.
Post-operative complications and readmissions with gallstone disease were pooled in this outcome.
Results
A total of 58 original studies were identified and published as 61 papers including 56 RCTs and two non-randomized studies. Non-randomized studies were included to answer PICOS three and four. PRISMA study flow-charts for all seven PICOS are presented in the Supplemental Material (Supplemental Material 2). During the study selection, we decided not to include published systematic reviews and meta-analyses as they did not match our defined outcomes, were not up to date, or included non-randomized studies. Multiple RCTs with comparable outcomes were identified for PICOS 2, 5, 6, and 7 allowing meta-analyses to be conducted. The remaining PICOS were answered by referencing single studies qualitatively. Table 2 shows GRADE summary of findings for all PICOS, analyses, quality of evidence, and reasons for downgrading at the outcome level. A summarizing flowchart for the Danish clinical practice guidelines is presented in Fig. 1.
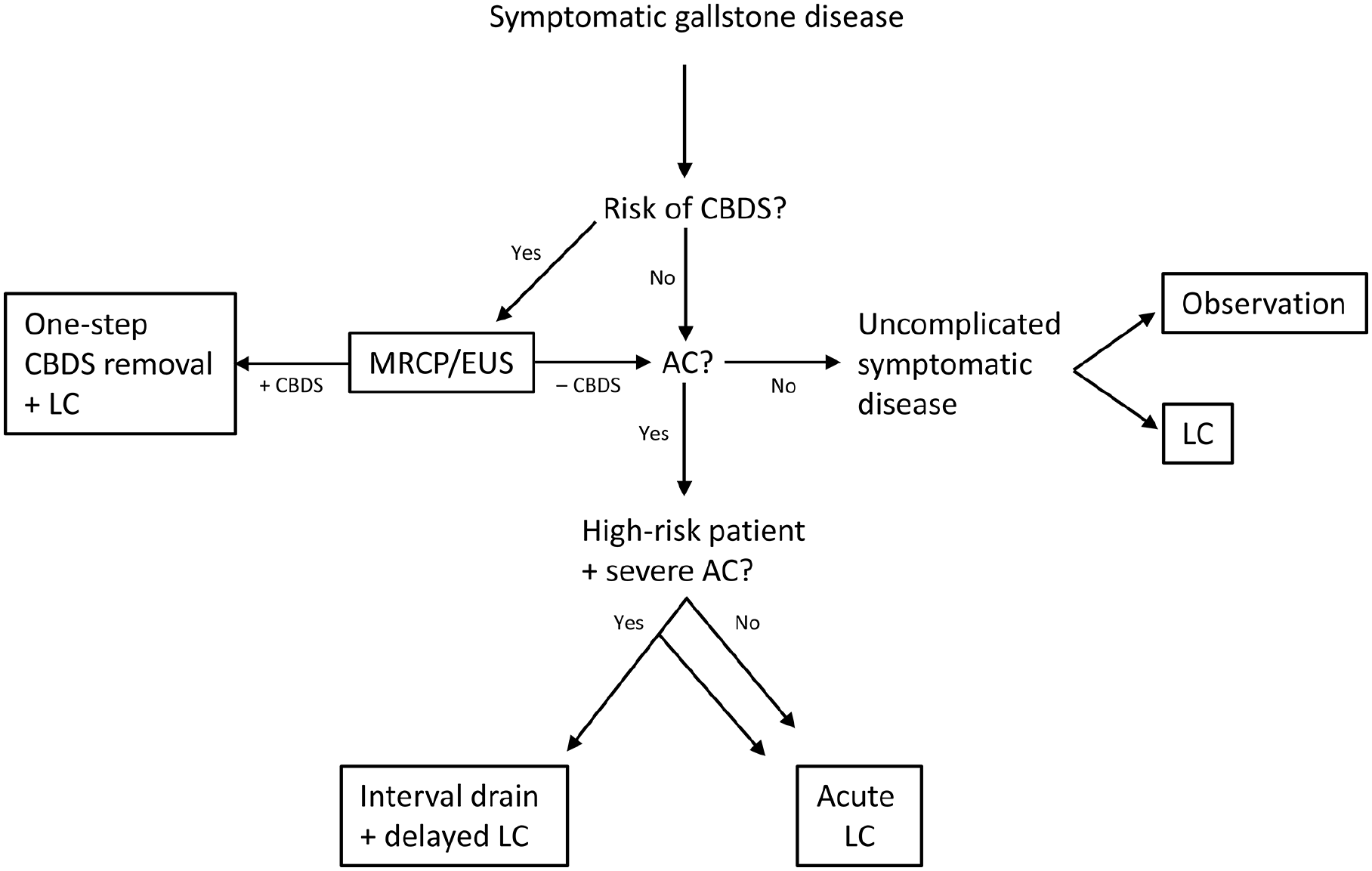
The Danish clinical practice guidelines for treatment of symptomatic gallstone disease flowchart.
Two studies were identified that specifically explored patient preferences with PROMs using questionnaires. One study included both emergency and elective surgery. Patient satisfaction was not associated with traditional clinical outcome measures, but with higher self-perceived health, less patient-reported wound pain, and return to normal leisure activities. 28 In another study patients scored long-term quality of life after emergency surgery as the most important factor. 29 PROMs for specific treatments are described in the individual recommendations below. Lack of evidence with respect to PROMs prompted us to try to anticipate patient preferences. Highest quality of life, shortest length of stay and no readmissions were appreciated.
We included two original studies that were published as four papers. The first study explored observation versus cholecystectomy30,31 and included a 14-year long-term follow-up study. 32 The other study explored a restrictive strategy versus usual care for cholecystectomy. The restrictive strategy required presence of a five symptoms complex before cholecystectomy was offered: (1) severe pain attacks, (2) pain lasting 15–30 min or longer, (3) epigastric or right upper quadrant pain, (4) pain radiating to the back, and (5) a positive pain response to simple analgesics. 33 Only 72% of patients remained in the restrictive strategy group in one study 33 and only 49% in the observation group in the other study 32 whereas most patients stayed in the usual care (98%) 33 and surgery groups (88%). 32 Due to large cross-over in both studies, both per-protocol and intention-to-treat analyses were performed.
One of the studies30,32 was downgraded due risk of bias as neither patients or outcome assessors were blinded and imprecision due to the presence of only one study. The other study was further downgraded due to indirectness since it did not explore observation directly but a restrictive strategy. 33
Absence of pain was examined in one study exploring restrictive strategy versus usual care. 33 No significant differences were identified. No differences in per-protocol and intention-to-treat analyses were identified (very low quality of evidence). Observation versus operation resulted in a significant and clinically relevant reduction in rate of cholecystectomies in the observation group at long-term follow-up 32 (moderate quality of evidence).
Readmissions and post-operative complications were pooled in the analyses of this PICOS, and they were more frequent in the observation group.30,32 The difference was significant in intention-to-treat analysis but not in per-protocol analysis. Readmissions were largely due to uncomplicated disease comprising 92% of readmissions in the observation group and 89% in the surgery group.
No significant differences were found in reduction of pain intensity or quality of life measured through the Psychological General Well-Being Index at 60 months of follow-up, 31 but with low quality of evidence due to the presence of one study only. No specific studies explored patient preferences, but we assume that patients prefer surgery due to the large cross-over from observation to surgery in the studies.
Findings suggest no change in quality of life at long-term follow-up of laparoscopic cholecystectomy compared to observation. In accordance with an increasing rate of laparoscopic cholecystectomies in Denmark and a significant rate of patients with persistent post-operative pain, we suggest an observational strategy to reduce the number of procedures in uncomplicated gallstone disease. Observation causes more readmissions, but these are largely due to uncomplicated gallstone disease.
We recommend observation in presence of non-severe symptoms, long interval between pain attacks, high age, or presence of multimorbidity. Low quality of evidence and discrepancy between presumed patient preferences and desirable and undesirable effects, result in a weak recommendation. Observation versus laparoscopic cholecystectomy in case of symptoms that can be ascribed to uncomplicated gallstone disease ultimately must involve shared decision holding.
We included 16 original studies published as 17 papers. The studies concerned either suspected or definitive acute cholecystitis when graded according to the current Tokyo guidelines. The preoperative maximum patient-reported symptom duration was 7,34–40 5,41,42 3 days,43–46 or was not reported.47–50 All studies intended laparoscopic cholecystectomy. The intervention constituted a conservative approach with delayed surgery after 6–8 weeks. Most studies excluded high-risk patients defined as those deemed unfit for surgery due to high age, comorbidities, an American Society of Anesthesiologists (ASA) score of more than 3, sepsis, or severe acute cholecystitis according to the Tokyo guidelines. Studies were published in 1998 to 2016 from Asia (India, Pakistan, and Hong Kong),34,35,37–39,41–44,50 Europe,36,40,46,47,49 and the Middle East.45,48
No studies reported blinding of outcome assessment and most studies reported insufficiently on follow-up measures or completeness. There is a risk of bias across studies for most outcomes, and we downgraded to moderate for critical outcomes. Meta-analyses were performed for all outcomes except for quality of life.
No significant differences in post-operative complications were identified (Fig. 2). Estimates were accurate and indicated no true clinical important difference. Readmissions were much less frequent in the acute surgery group and the difference was significant (moderate quality of evidence) and clinically relevant.
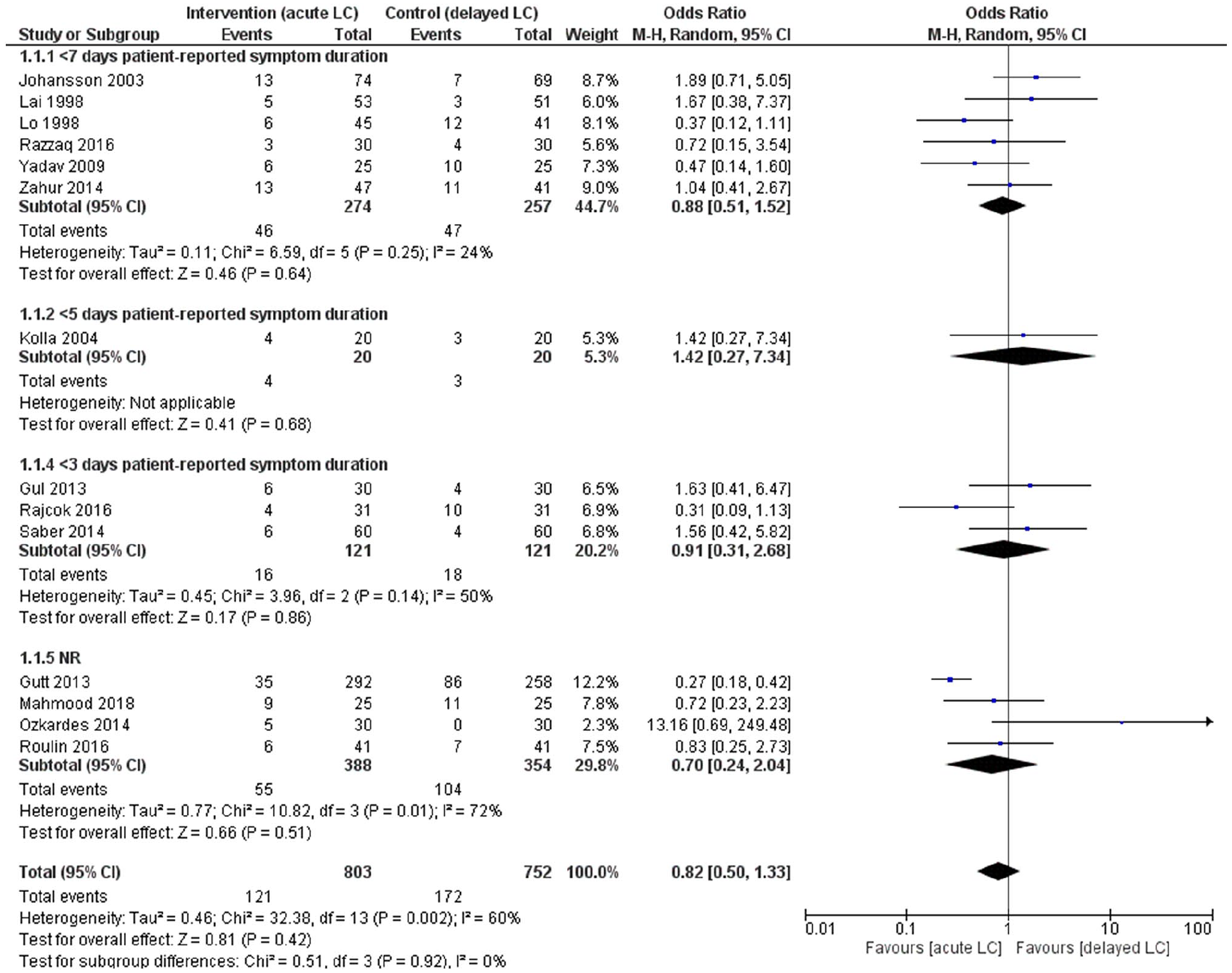
Subgroup analysis of preoperative maximum patient-reported symptom duration and post-operative complications following acute versus delayed laparoscopic cholecystectomy for patients with acute cholecystitis (PICOS 2)
Length of stay was shorter with acute surgery, and no significant or clinically relevant differences were found for conversion to open surgery. Eight studies reported no mortalities and one study reported one death in each group, 47 resulting in a mortality proportion of less than 0.1%. No significant differences in Psychological General Well-Being Index after 6 months were noted in one study assessing quality of life. 40 Estimates for mortality and quality of life were not significantly different between the treatment arms.
Subgroup analysis of preoperative maximum patient-reported symptom duration showed no differences in significance levels compared to the original meta-analysis for post-operative complications (Fig. 2), readmissions, and conversion to open surgery. Length of stay was significantly shorter for acute surgery in all subgroups, except for studies that did not report symptom duration (data not shown).
Patient perspectives were explored in one of the included studies through non-validated measures showing higher patient satisfaction for acute surgery. 45 Meta-analysis confirmed that delayed surgery causes readmissions, and we thus assume that patients prefer acute rather than delayed laparoscopic cholecystectomy.
The strong recommendation in favor of acute laparoscopic cholecystectomy is largely based on fewer readmissions and non-inferiority regarding post-operative complications. Preoperative maximum patient-reported symptom duration does not seem to have an impact on post-operative complications and should not influence the choice of treatment. In case of acute laparoscopic cholecystectomy, we recommend surgery as soon as possible, but only when a competent surgeon is present and preferably during daytime.
We included one RCT and one non-randomized retrospective study. The RCT included high-risk and non-high-risk patients with moderate acute cholecystitis according to the Tokyo guidelines and a preoperative patient-reported symptom duration of more than 72 hours. 51 The non-randomized retrospective study included high-risk patients defined as age 65 years or more, morbidities, severe acute cholecystitis according to the Tokyo guidelines, assessed as unfit for surgery, and without a sufficient response to an initial conservative approach. The interval drainage group was matched with a similar acute laparoscopic cholecystectomy group. 52 The study was included to represent high-risk patients, as the option for drainage often is considered in this subgroup. Both studies included ultrasound-guided percutaneous transhepatic gallbladder drainage and delayed laparoscopic cholecystectomy after 6–10 weeks compared to acute laparoscopic cholecystectomy. No meta-analyses were performed due to differences in study designs.
Quality of evidence of the RCT was low, due to risk of bias because of no reporting of allocation sequence, no blinding of outcome assessment, and due to the presence of only one study, causing imprecision. Quality of the non-randomized study was downgraded due to risk of bias according to ROBINS-I for measurements of outcome. Further downgrading was for imprecision due to the presence of only one study with wide CIs for critical outcomes.
A significant and clinically relevant reduction in post-operative complications occurred in the interval drainage group. The estimate for readmissions was uncertain (low quality of evidence).
Conversion to open surgery was significantly decreased in the interval drainage group and assessed as clinically relevant. Estimate for mortality was uncertain and no important difference was found for length of stay.
In the subgroup of high-risk patients, estimates for post-operative complications were uncertain (very low quality of evidence). Conversion to open surgery and length of stay were significantly decreased in the drainage group and assessed as clinically relevant. The estimate for mortality was uncertain.
No studies reported quality of life or patient preferences. We assume that interval drainage may cause patient discomfort as the catheter is left in situ for several weeks while the patient is at home. Our clinical experience is that drainage treatment may cause several readmissions due to catheter-related dysfunction.
We generally recommend acute laparoscopic cholecystectomy for patients with acute cholecystitis (see recommendation 2) including high-risk patients (see recommendation 4). Currently available literature on gallbladder drainage as a bridge to surgery does not sufficiently address critical outcomes as post-operative complications and readmissions and especially not for high-risk patients. But studies do, however, suggest that interval drainage may result in fewer post-operative complications, fewer conversions to open surgery, and may reduce length of stay. The mechanism may be infection control by drainage, resulting in preoperative optimization of the frail high-risk patient. We suggest that clinicians may consider interval drainage as an option in the presence of severe acute cholecystitis in high-risk patients with advanced age, low performance score, or multi-morbidity. The clinical case may be represented by the frail patient with severe acute cholecystitis admitted at the intensive care unit or with the need for intensive care treatment but deemed unfit.
We included one RCT and one non-randomized retrospective cohort study. The RCT included high-risk patients defined as an APACHE II score of 7–14. APACHE II is a scoring system prediction of mortality based on 12 acute physiological parameters, age, and morbidities. Intervention was ultrasound-guided percutaneous gallbladder catheter for at least 3 weeks. 53 The non-randomized study included high-risk patients defined as age of 80 years or more, ASA score of 3 or above, a Charlson comorbidity index of 5 or above or a Karnofsky score of 50 or below. Propensity score matching was performed on these baseline variables. Intervention was an EUS-guided drain placed between the gallbladder and the duodenum (lumen-apposing metal stent). Most patients had moderate acute cholecystitis. 54 The comparator was acute laparoscopic cholecystectomy in both studies.53,54 The non-randomized study was included to explore the impact of a novel EUS-guided technique compared to conventional percutaneous drainage.
Quality of evidence of the RCT was high with no risk of bias but we downgraded to moderate due to the presence of only one study causing imprecision. Quality of the non-randomized study was downgraded due to risk of bias according to ROBINS-I for missing data and for difference in follow-up lengths in measurements of outcome. The study was further downgraded for imprecision due to the presence of only one study with wide CIs.
Percutaneous gallbladder drainage caused a significant and highly clinically relevant increase in post-operative complications and readmissions. Estimate for mortality was uncertain (moderate quality of evidence).
Both the need for additional interventions and length of stay were significantly higher in the drainage group.
Intervention with EUS-guided gallbladder drainage caused uncertain estimates for both critical and non-critical outcomes (very low quality of evidence).
No identified studies explored PROMs. Due to increased post-operative complications, readmissions, and need for additional interventions as well as increased length of stay, we believe that most high-risk patients with acute cholecystitis would decline percutaneous gallbladder drainage if they were well-informed.
The decision not to recommend percutaneous gallbladder drainage is based on the highly clinically relevant and significantly higher risk of post-operative complications and readmissions in frail high-risk patients.
We included 22 original studies. Fourteen studies included patients with CBDS confirmed by MRCP, EUS, or ERC15,55–67 and eight with merely clinical suspicion of CBDS.14,68–74 Most studies excluded high-risk patients defined as an ASA score of 3 or above, age of 70 years or more, cardiac or pulmonary morbidity or otherwise defined as unfit for surgery. Only one study exclusively included high-risk patients. 14 Control groups underwent laparoscopic cholecystectomy performed during the same admission or up to 8 weeks after ERC with CBDS removal. One-step interventions varied between studies
Laparoscopic common bile duct exploration with cholecystectomy (LCBDE) in 14 studies.14,15,55,56,58,60,62,66,68–71,73,74
Rendezvous technique where a guidewire is inserted through the cystic and common bile duct at laparoscopic cholecystectomy to facilitate simultaneous ERC and CBDS removal in five studies.59,63,65,67,72
Laparoscopic cholecystectomy and concomitant ERC and CBDS removal without a guidewire (non-rendezvous) in three studies.57,61,64
No report on how the one-step procedure with ERC was performed in one study. 69
Studies were published between 1999 and 2020 from Europe,14,59,63,67,70–72,74 Asia,15,55,56,60–62,65 the Middle East,57,58,64,66 South or Middle America,68,69 and North America. 73
No studies reported blinding of outcome assessment, and most studies did not report sufficiently on follow-up measures or completeness causing a risk of bias across studies for most outcomes. Quality of evidence was downgraded to moderate for critical outcomes. Meta-analyses were performed for all outcomes except for quality of life.
One-step procedures reduced post-operative complications and readmissions significantly and clinically relevant (moderate quality of evidence).
The need for additional interventions was significantly lower and the rate of successful CBDS removal higher for the one-step procedure. Mortality was reported in 14 studies and 20 deaths occurred in six studies resulting in a mortality proportion of approximately 1%. No significant differences were found for mortality. Length of stay was significantly less with the one-step procedure and assessed as clinically relevant. Only one study reported quality of life measured through the Short Form 36 for LCBDE versus the two-step procedure and no significant differences were found. 73
Subgroup analysis revealed that patients with CBDS imaging confirmation experienced significantly fewer post-operative complications (OR 0.57, CI 95% 0.40–0.83; Fig. 3), less need for additional interventions (OR 0.31, CI 95% 0.12–0.79), and higher rate of successful CBDS removal (OR 1.87, CI 95% 1.22–2.86) following the one-step procedure just like in the original meta-analysis. In the group without imaging confirmation, a substantial number of patients did not have CBDS at surgery and meta-analyses showed more non-significant estimates and substantial heterogeneity (data not shown). Estimates from the group without imaging confirmed CBDS were inconsistent and uncertain.
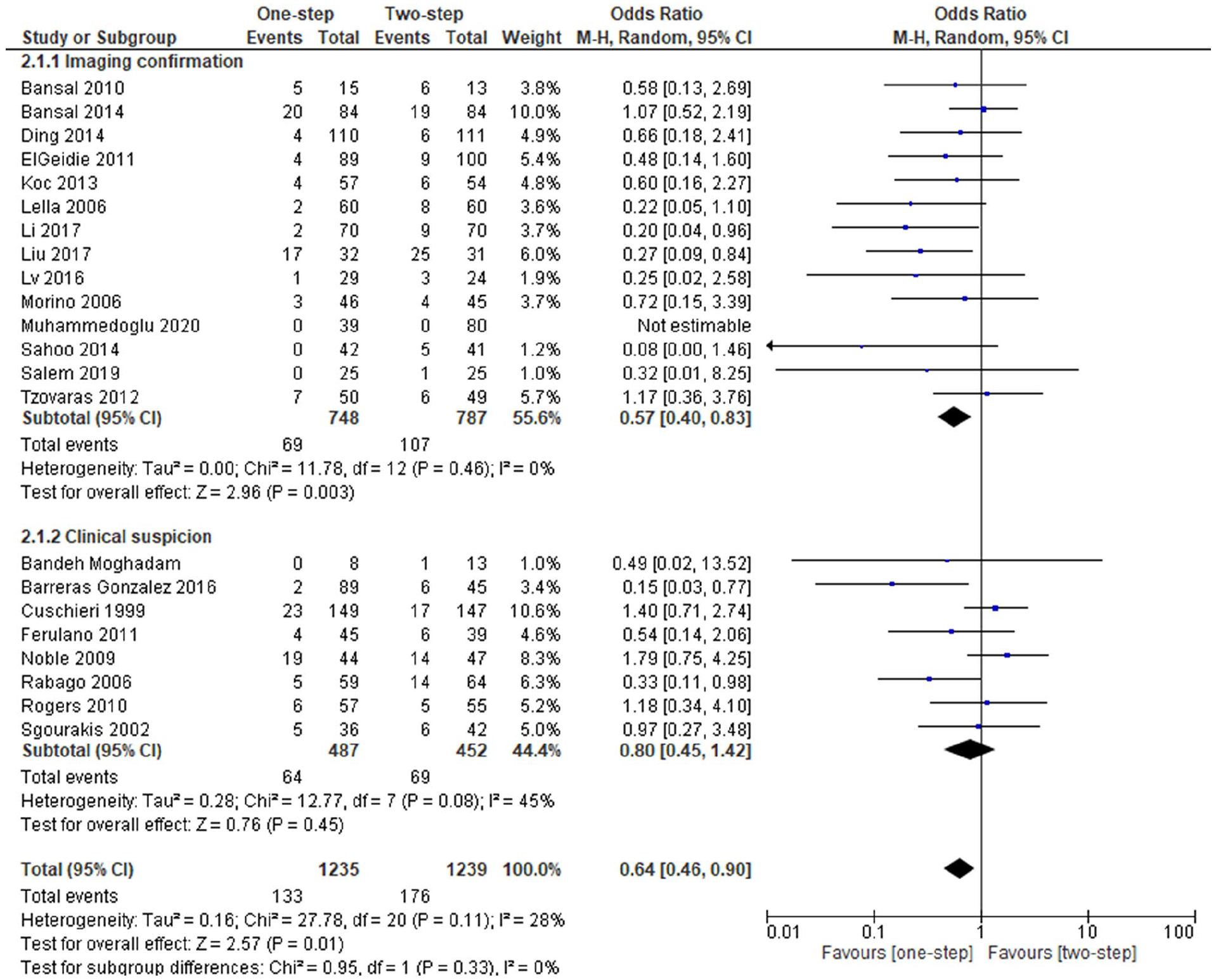
Subgroup analysis of imaging confirmed CBDS and post-operative complications following one-step versus two-step procedures for patients with CBDS (PICOS 5).
One-step interventions resulted in higher patient satisfaction in two studies using unvalidated measures.15,66 We assume that patient preferences are in favor of the one-step interventions.
We strongly recommend a one-step procedure based mainly on a lower risk of post-operative complications, readmissions, but also on the higher rate of CBDS clearance and lesser need for additional interventions. The one-step procedure is currently not standard treatment at all hospitals and thorough staff training should be pursued prior to implementation.
We included six original studies. High-risk patients were defined as age above 60 or 75 years,75,76 age above 70 years and morbidity, 77 age above 75 years and ASA score of 3 or above, 78 or presence of either body mass index above 30 kg/m2, age above 70 years, or a severe degree of morbidity. 79 ERC confirmation of CBDS prior to inclusion of patients was performed in three studies,75,76,79 one study included patients based only on clinical suspicion of CBDS, 78 and one study did not specify preoperative CBDS diagnosis. 77 All five studies reported interventions with ERC and sphincterotomy for CBDS removal. Control group treatments varied and included:
Open surgical common bile duct exploration with cholecystectomy in three studies.76,78,79
ERC and laparoscopic cholecystectomy after 11 weeks in one study. 75
No report of control group cholecystectomy method in one study. 77
One supplemental study reported PROMs, but included patients were not high-risk and therefore only results for quality of life were included. 80
No studies reported blinding of outcome assessment and most studies did not report sufficiently on follow-up measures or completeness causing a risk of bias across studies for most outcomes. Quality of evidence was downgraded to moderate for critical outcomes. Meta-analyses were performed for all outcomes except for quality of life.
No significant differences were found for post-operative complications (low quality of evidence) and mortality, but estimates were imprecise and uncertain and therefore downgraded. Mortality proportions were 20% in the group with no cholecystectomy and 14% in the group with cholecystectomy. Significantly higher rate of readmissions was found in the non-cholecystectomy group which was assessed as clinically relevant (moderate quality of evidence).
The need for additional interventions was significantly higher and length of stay was shorter in the no cholecystectomy group. Quality of life measured with MOS-24, which is a shorter version of the Short Form 36, showed no significant differences between groups, but estimates were uncertain due to imprecision, indirectness of evidence, and risk of bias causing a very low quality.
Subgroup analysis showed that the group with imaging confirmed CBDS had a significantly higher mortality of 24% with no cholecystectomy compared to 13% in the cholecystectomy group (Fig. 4). The other estimates were not scientifically different when compared to the original meta-analyses (data not shown).
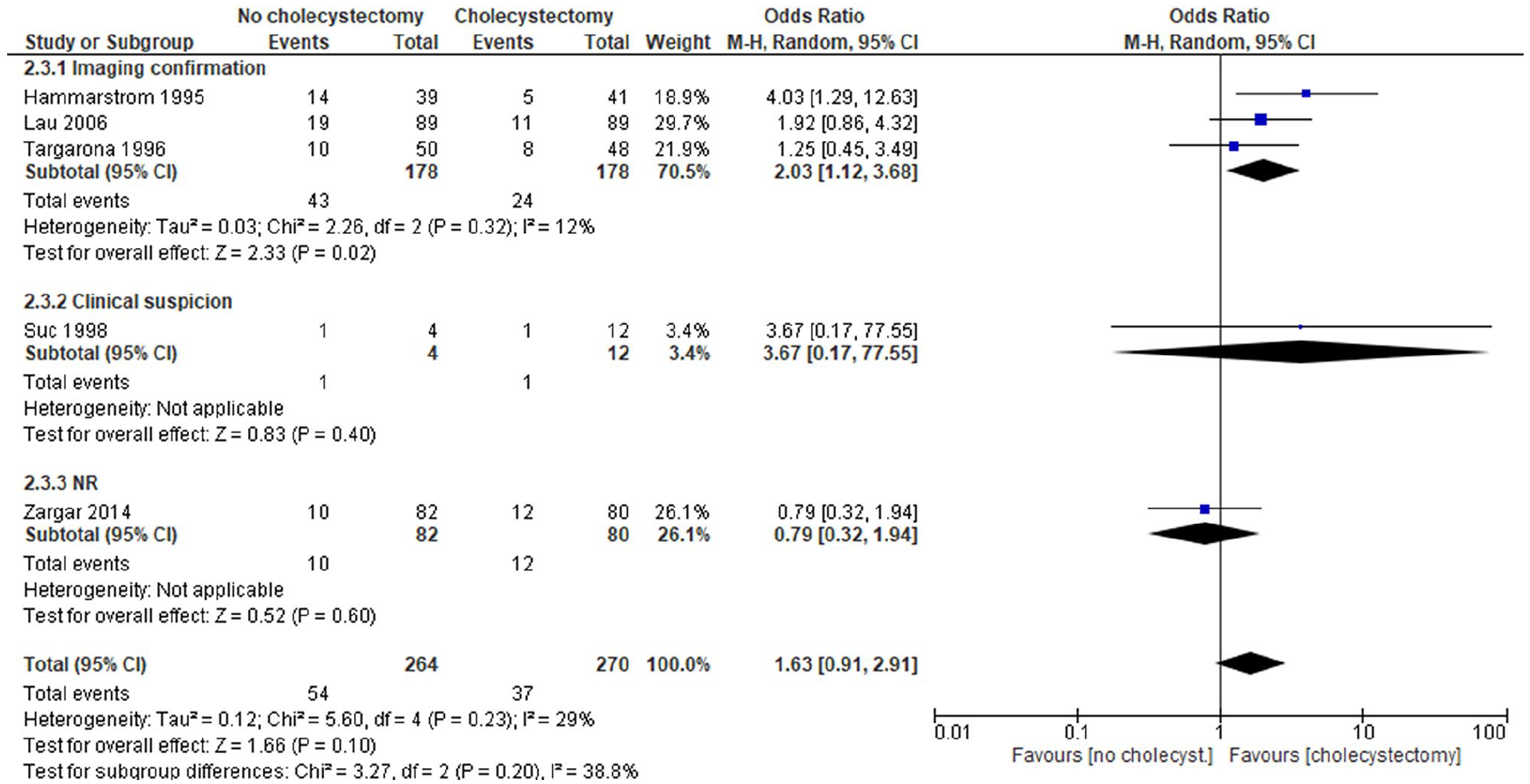
Subgroup analysis of imaging confirmed CBDS and mortality following CBDS clearance without versus with cholecystectomy for high-risk patients with CBDS (PICOS 6).
Quality of life could not be explored for high-risk patients. We assume that most high-risk patients prefer cholecystectomy in addition to CBDS removal, given a higher risk of readmission, additional interventions, and probably a higher risk of mortality without cholecystectomy. On the contrary, some patients may not want to run the immediate risk of surgery to avoid uncertain future complications and therefore abstain from cholecystectomy at first CBDS presentation. We expect variation in patient preferences.
The decision to suggest cholecystectomy to high-risk patients with CBDS is based on lower risk of readmissions and the possible lower risk of mortality. The recommendation is weak since two critical outcomes have uncertain estimates.
We included eight original studies concerning patients with gallbladder stones and intermediate risk of CBDS, defined as either elevated bilirubin and/or dilated bile duct on ultrasound,81–84 mild gallstone pancreatitis,85,86 elevated liver enzymes, 87 or presence of one of the mentioned risk factors. 88 All studies excluded patients with high risk of CBDS such as presence of cholangitis, sepsis, or imaging confirmed CBDS. EUS versus ERC was explored in five studies,81,83–85,88 EUS versus peroperative cholangiography in one study, 87 MRCP versus ERC in one study, 82 and MRCP versus peroperative cholangiography in one study. 86 In all studies, CBDS were removed by ERC. Pooled adverse events were reported for diagnostic examination, CBDS removal, and CBDS disease in most studies. Only three studies reported on adverse events for the diagnostic examination separately and all used EUS.84,85,88 PICO 7 was intended as a diagnostic question and analyses were therefore reported for both the pooled rates of adverse events and adverse events related to examination only.
Most studies did not report blinding of outcome assessment or report sufficiently on follow-up measures causing a risk of bias across studies for most outcomes. Quality of evidence was downgraded to moderate for critical outcomes. Meta-analyses were performed for all outcomes.
There were no important differences in CBDS diagnosis between MRCP/EUS and cholangiographic examinations (moderate quality of evidence). Rates of pooled adverse events for examination, treatment, and disease were uncertain for EUS and cholangiographic examinations (low quality of evidence). There were significantly lower rates of adverse events for EUS compared to cholangiographic examinations, and it was assessed as clinically relevant (moderate quality of evidence).
Estimates for additional examinations and readmissions were not significantly different between groups. Estimates for additional examinations had high heterogeneity and were therefore further downgraded for inconsistency. Estimates for both outcomes were uncertain.
Subgroup analyses of the intervention group examinations showed less frequent adverse events for EUS (OR 0.41, CI 95% 0.17–0.99) and no significant difference for MRCP (OR 1.37, CI 95% 0.42–4.42). Need for additional examinations was higher with MRCP when compared to cholangiography (OR 2.53, CI 95% 1.13–5.69), and no significant differences were found for EUS (OR 1.28, CI 95% 0.52–3.12). Subgroup analyses of the control groups with peroperative cholangiography and ERC showed lower rates of adverse events with MRCP/EUS when ERC was the control group (OR 0.40, CI 95% 0.18–0.89) and no significant differences when peroperative cholangiography was the control group (OR 1.87, CI 95% 0.51–6.85). No differences were seen for need for additional treatments.
No studies reported patient preferences for diagnosis of CBDS. We assume that patients prefer MRCP or EUS as the least-invasive examinations, with a minimum of discomfort and risk of complications.
The decision to recommend MRCP or EUS in favor of ERC to diagnose CBDS was based on a higher risk of adverse events for ERC and on a comparable diagnostic yield in case of intermediate CBDS risk. Hospitals may choose between MRCP or EUS depending on local availability. ERC should not be used for diagnostic purpose only. Peroperative cholangiography may be used for diagnosis at the discretion of the surgeon. However, we generally recommend MRCP or EUS for CBDS diagnosis prior to surgery for gallstone disease.
Discussion
We have reported seven recommendations for treatment of gallstone disease. Three recommendations are strong and four are weak due to a lack of published studies. We suggest several areas of interest for future research.
Treatment with gallbladder drainage for acute cholecystitis in high-risk patients was found to increase readmissions and complications. A large British non-randomized study of high-risk patients with acute cholecystitis confirmed that about half of patients initially treated with gallbladder drainage subsequently experienced readmission due to gallstone disease and that drainage or conservative treatment may result in higher mortality when compared to cholecystectomy. 89 A recent RCT suggested that EUS-guided gallbladder drainage may be superior to percutaneous drainage in high-risk patients. 90 EUS-guided drainages may serve as a treatment of high-risk patients with acute cholecystitis. Future RCTs should compare laparoscopic cholecystectomy and EUS-guided drainage and explore the feasibility of EUS-guided drainages in an emergency setting. There was a general paucity in published studies on gallbladder drainage for acute cholecystitis and more studies are needed to make stronger recommendations. Further research is also needed to determine if interval drainage as a bridge to surgery is a useful tool in high-risk patients.
Subgroup analyses of patients with acute cholecystitis showed that the preoperative maximum patient-reported symptom duration, does not predict the development of complications following acute laparoscopic cholecystectomy and should therefore not influence treatment choices. Large database studies have shown that increased length of preoperative admission is associated with post-operative complications, bile duct injuries, mortality, conversion to open surgery, reoperations, and length of stay.91,92 If laparoscopic cholecystectomy is indicated, we recommend surgery as soon as possible and, preferably, during daytime and when appropriate surgical competency is available.
The one-step compared to the two-step procedures for treatment of high-risk patients with CBDS have only been explored in one RCT included in this guideline, 14 and more studies are needed. We suggest that laparoscopic cholecystectomy should be added to the treatment of high-risk patients with CBDS to reduce readmission and possibly mortality. However, all except one of the included RCTs have outdated interventions such as open cholecystectomy with surgical exploration of the common bile duct. Non-randomized studies of high-risk patients treated with CBDS removal also suggest that laparoscopic cholecystectomy is performed to decrease complications, readmissions, and the need of additional interventions.93,94 Future RCTs should explore minimally invasive one-step procedures (recommendation 5) versus CBDS removal without laparoscopic cholecystectomy in high-risk patients.
Symptomatic uncomplicated gallstone disease comprises a large group of the patients undergoing laparoscopic cholecystectomy, but only two RCTs were identified exploring observation or a restrictive patient selection for surgery. More studies should explore observation or patient selection strategies for cholecystectomy. A large ongoing RCT from the United Kingdom exploring observation versus surgery is expected to be published soon. 95
The use of MRCP for diagnosis of CBDS was only explored in two studies. Since MRCP is the least-invasive examination for CBDS, we suggest that more studies are performed in the future. A large ongoing RCT from the United Kingdom exploring MRCP versus observation is expected to be published in some years. 96
Most studies were not published in Denmark or Scandinavia and one may therefore question if they are representative for treatments performed at Danish hospitals. For patients with acute cholecystitis and treated with laparoscopic cholecystectomy, conversion to open surgery was 12% and post-operative complications 20% in our meta-analyses. Estimates for mortality are generally low but were also uncertain in our included data (0.1%) due to overall low number of patients (Recommendation 2, Table 2). A Danish database study from the period 2006–2011 shows a conversion rate of 15% 97 and a mortality of 1.2%. 98 A Swedish database study from 2006 to 2014 shows post-operative complications in 12%. 91 The operative and post-operative courses of patients undergoing laparoscopic cholecystectomy for acute cholecystitis in our meta-analysis were thereby comparable to observational data from Denmark and Sweden. No Scandinavian observational data on one versus two-step procedures for treatment for CBDS were identified.
Generally, most studies reported no blinding of outcome assessment and many did not report sufficiently on follow-up measures or completeness causing a risk of bias. Future studies should be performed according to current recommendations for high-quality evidence. Quality of life or PROMs were rarely explored in any of the included studies. Patient preferences for high-risk populations are unexplored. We highly recommend the inclusion of PROMs in future RCTs of treatment for gallstone disease.
Strengths in this guideline development include the multidisciplinary working group which represent all specialties and all five Danish regions involved in the treatment of patients with gallstone disease. Limitations include sparsely reported patient preferences in identified studies, and we did not include relevant patient groups to explore patient preferences any further.
Supplemental Material
sj-docx-1-sjs-10.1177_14574969221111027 – Supplemental material for National clinical practice guidelines for the treatment of symptomatic gallstone disease: 2021 recommendations from the Danish Surgical Society
Supplemental material, sj-docx-1-sjs-10.1177_14574969221111027 for National clinical practice guidelines for the treatment of symptomatic gallstone disease: 2021 recommendations from the Danish Surgical Society by Daniel Mønsted Shabanzadeh, Dorthe Wiinholdt Christensen, Caroline Ewertsen, Hans Friis-Andersen, Frederik Helgstrand, Lars Nannestad Jørgensen, Anders Kirkegaard-Klitbo, Anders Christian Larsen, Jonas Sanberg Ljungdalh, Palle Nordblad Schmidt, Rikke Therkildsen, Peter Vilmann, Jes Sefland Vogt and Lars Tue Sørensen in Scandinavian Journal of Surgery
Supplemental Material
sj-docx-2-sjs-10.1177_14574969221111027 – Supplemental material for National clinical practice guidelines for the treatment of symptomatic gallstone disease: 2021 recommendations from the Danish Surgical Society
Supplemental material, sj-docx-2-sjs-10.1177_14574969221111027 for National clinical practice guidelines for the treatment of symptomatic gallstone disease: 2021 recommendations from the Danish Surgical Society by Daniel Mønsted Shabanzadeh, Dorthe Wiinholdt Christensen, Caroline Ewertsen, Hans Friis-Andersen, Frederik Helgstrand, Lars Nannestad Jørgensen, Anders Kirkegaard-Klitbo, Anders Christian Larsen, Jonas Sanberg Ljungdalh, Palle Nordblad Schmidt, Rikke Therkildsen, Peter Vilmann, Jes Sefland Vogt and Lars Tue Sørensen in Scandinavian Journal of Surgery
Footnotes
Acknowledgements
The authors thank Henning Keinke Andersen from the Danish Health Authorities for cooperation during the entire process, methodology consultant Jeanett Friis Rohde from The Parker Institute for method supervision and analyses, information specialist Sarah Louise Klingenberg at the Cochrane Hepato-Biliary Group for performing search strategies and literature searches, and Karin Mønsted Shabanzadeh for language editing.
Author contributions
DMS contributed to the design, acquisition of data, analyses, interpretation of results, and drafting of the manuscript. The remaining authors contributed to the design, acquisition of data, interpretation of results, and critical review of manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Consent
No new patients were involved in this study and therefore no consent required.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported financially by the Danish Health Authorities. The Danish Health Authorities accepted the PICOS that the work group had defined, but otherwise had no roles in guideline development, dissemination, or implementation of the recommendations.
Ethical approval
No ethical statement required since no new data were collected from individuals.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
