Abstract
Background and objective:
Laparoscopic resection for obstructive colon cancer (CC) after insertion of self-expanding metallic stents (SEMSs) is reportedly difficult. However, this has not yet been thoroughly investigated. Therefore, we investigated the influence of SEMS insertion on laparoscopic resection.
Methods:
This retrospective comparative study included 87 patients with obstructive left-sided CC (December 2011–December 2019). Patients were assigned to two groups based on elective laparoscopic surgery for (1) obstructive CC necessitating emergent decompression with SEMS insertion (SEMS group) and (2) obstructive CC without emergent decompression (control group).
Results:
The SEMS group had a longer operation time (283.3 ± 79.3 min vs 222.2 ± 79.4 min, P = 0.002) and greater blood loss (204.8 ± 417.6 mL vs 53.7 ± 166.1 mL, P = 0.029) on univariate analysis; however, in multiple linear regression analysis, SEMS was not an independent risk factor for both operation time (Δ operation time 25.5 min: P = 0.19) and blood loss (Δ blood loss 33.6 mL: P = 0.58). The complication rate based on Clavien−Dindo grade II did not differ significantly (17% vs 20%, P = 1.00), whereas the rates of conversion to laparotomy (17% vs 2%, P = 0.016) and stoma creation (26% vs 2%, P = 0.001) were higher in the SEMS group. In oncological outcomes, there was no intergroup difference in the 5-year disease-free survival (80.0% vs 72.2%, P = 0.76) and overall survival (100% vs 86.3%, P = 0.25).
Conclusions:
Laparoscopic surgery after SEMS for left-sided CC is demanding due to higher conversion rates to open surgery. However, this study also revealed that it is as safe as laparoscopic surgery for cases without SEMS because of comparable complication rate and long-term outcomes.
Introduction
Approximately 15%–20% of patients with colon cancer (CC) present with large-bowel obstruction, requiring emergent medical care.1,2 Previously, emergency surgery was the treatment option for acute obstructive CC and was often accompanied by stoma creation. Emergent colorectal surgery is associated with high morbidity and mortality. 3 - 5 The self-expanding metallic stent (SEMS) was first introduced by Dohmoto in 1991 to relieve colonic obstruction and was increasingly used as a bridge to surgery (BTS) for potentially resectable obstructive CC. 6 SEMS altered the treatment of obstructive CC from emergency surgery to elective surgery and resulted in lower mortality, morbidity, and stoma creation rate.7,8 Furthermore, decompression of the colon using SEMS facilitates the laparoscopic resection of obstructive CC.
Laparoscopic colectomy for obstructive CC is reportedly difficult after SEMS insertion; the rigidity of the colon containing the stent makes tumor and peritumoral dissection challenging. 9 Studies investigating the usefulness of laparoscopic surgery 10 - 12 have compared it with open surgery; they were thus unable to elucidate if the difficulty in laparoscopic colectomy is associated with SEMS insertion. Furthermore, the features and outcomes of laparoscopic resection after SEMS insertion for obstructive CC have been compared with those of laparoscopic resection for non-obstructive CC;12,13 however, as non-obstructive CC is far less advanced than obstructive CC, this comparison failed to identify the effect of SEMS insertion on laparoscopic colectomy in obstructive CC.
Therefore, this study aimed to elucidate the influence of SEMS insertion for obstructive CC on laparoscopic resection by comparing the clinicopathological features, as well as short- and long-term outcomes, of laparoscopic resection after SEMS insertion with those of laparoscopic resection without SEMS insertion.
Methods
Patients
This retrospective study included patients with obstructive left-sided CC who underwent elective laparoscopic surgery at our institution between December 2011 and December 2019. In this study, obstructive CC was defined as CC through which a small-diameter colonoscope could not be passed. We excluded double cancer cases and combined resection cases of distant metastasis, such as those involving the liver and ovary. Thus, 87 patients were included in the analysis and assigned to two groups based on whether emergent decompression by SEMS insertion was needed before laparoscopic resection (SEMS group) or not (control group). Twenty-three patients underwent laparoscopic surgery after SEMS insertion for obstructive CC, and the remaining 64 patients underwent laparoscopic resection for obstructive CC without SEMS insertion. For each group, we extracted information from the maintained database on age, sex, body mass index (BMI), tumor location, diameter, and depth, lymph node metastasis, distant metastasis, type of operation, operation time, blood loss, the presence/absence of a stoma, conversion to laparotomy, combined resection, and postoperative complications, categorized according to the Clavien–Dindo classification. The 5-year disease-free survival (DFS) and overall survival (OS) were calculated.
Stent insertion
All patients with symptoms of bowel obstruction, such as abdominal pain, abdominal distention, and failure to pass gas or feces, underwent a computed tomography scan. When CC-induced large-bowel obstruction was suspected, SEMS insertion as BTS was considered. SEMS was contraindicated in patients diagnosed with peritonitis, perforation, and lower rectal cancer, and they were excluded.
The SEMS was inserted by expert endoscopists. 14 When CC-induced large-bowel obstruction was suspected on flexible colonoscopy, the guidewire was placed over the stenosis, followed by a catheter through which a water-soluble contrast solution was injected to estimate the length of the stenosis. WallFlex (Boston Scientific), Jentlly (Japan Lifeline), Niti-S (Century Medical Inc.), or Naturfit (Boston Scientific) was used as the SEMS. Both the technical and clinical success of SEMS placement was defined as previously described. 14 After SEMS insertion, patients were discharged, and approximately 1 month later, they were readmitted for elective surgery. This study was approved by the ethics committee of the institution (no. 3252-(9)). All study participants provided written informed consent.
Statistical analyses
Statistical analyses were conducted using the BellCurve for Excel. Categorical variables were compared using Fisher’s exact test or the χ2 test, while continuous variables categorized in two groups were compared using Student’s t-test or Mann–Whitney U-test. To assess statistical dependence between two continuous variables within a group, Spearman’s correlation test was used. Variables with P < 0.1 on univariate analysis were included in a multiple linear regression analysis to identify the independence of such variables. Survival function was analyzed using the Kaplan–Meier curves with the log-rank test. A P-value of <0.05 was considered statistically significant.
Results
The clinicopathological features of the SEMS and control groups are summarized in Table 1. In the SEMS group, both technically and clinically successful SEMS insertion was obtained in all patients. There was no difference in tumor location; however, for rectosigmoid junction CC, the rate of high anterior resection was lower (n = 2 (9%) vs n = 16 (25%)) and that of low anterior resection (n = 4 (17%) vs n = 1 (2%)) was higher. For descending CC, the rate of left hemicolectomy (n = 3 (13%) vs 4 (6%)) was higher in the SEMS group than in the control group. The operation time was longer (283.3 ± 79.3 min vs 222.2 ± 79.4 min, P = 0.002), the amount of blood loss was larger (204.8 ± 417.6 mL vs 53.7 ± 166.1 mL, P = 0.029), and the postoperative hospital stay was longer in the SEMS group (18.6 ± 7.7 days vs 13.3 ± 4.7 days, P < 0.001) than in the control group. There was no difference in the rate of complications with Clavien–Dindo classification grade II (n = 4 (17%) vs n = 13 (20%), P = 1.00).
Patients’ characteristics and perioperative surgical outcomes.
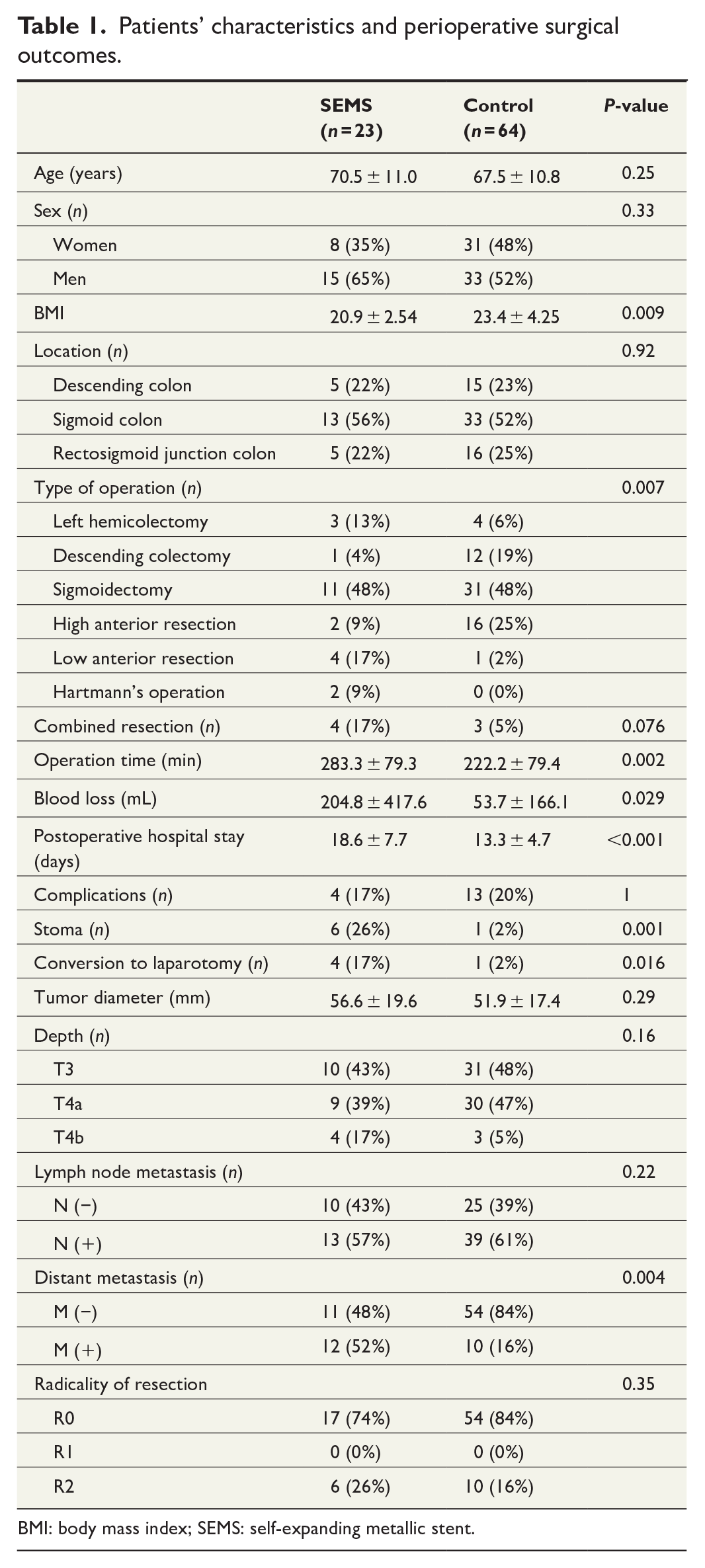
BMI: body mass index; SEMS: self-expanding metallic stent.
The rate of stoma creation was higher in the SEMS group (n = 6 (26%) vs n = 1 (2%), P = 0.001). Among six patients with stoma creation in the SEMS group, primary anastomosis was performed in four patients. In four of the six patients, the reasons for stoma creation were related to either patient or tumor factors such as severe comorbidities and highly advanced tumors. The other reason for stoma creation was the presence of obstructive colitis and peritumoral inflammation.
The rate of conversion to open surgery was higher in the SEMS group (n = 4 (17%) vs n = 1 (2%), P = 0.016). The reason for conversion to open surgery in the SEMS group was to evaluate the resectability of the disseminated lesion in one case, and in the other three cases, the reason was suspected invasion of the adjacent organs, such as the pelvic wall, ureter, and seminal duct. However, in these three cases, invasion of the adjacent organs was not confirmed pathologically. In the control group, adhesion after open sigmoidectomy was the reason for conversion.
Pathologically, the prevalence of distant metastasis (n = 12 (52%) vs n = 10 (16%), P = 0.004) was much higher in the SEMS group, which meant that the tumor was more advanced in the SEMS group.
A multiple linear regression analysis was performed to confirm whether SEMS insertion was an independent risk factor for the operation time or the amount of blood loss. As the pT4b was closely associated with combined resection, we did not evaluate the impact of combined resection on the operation time and amount of blood loss to avoid collinearity. The multiple linear regression analysis confirmed that the type of operation (Δ operation time, 57.2 min, P = 0.002) and conversion to open surgery (Δ operation time, 84.3 min, P = 0.017) were independent risk factors for a longer operation time; however, SEMS insertion was not an independent risk factor (Δ operation time, 25.5 min, P = 0.19; Table 2). As for blood loss, multiple linear regression analysis revealed that conversion to open surgery (Δ blood loss, 644.5 mL, P < 0.001) was an independent risk factor for blood loss; however, SEMS insertion was not an independent risk factor (Δ blood loss, 33.6 mL, P = 0.58; Table 3).
Univariate analysis and multiple linear regression analysis for associations with operation time.
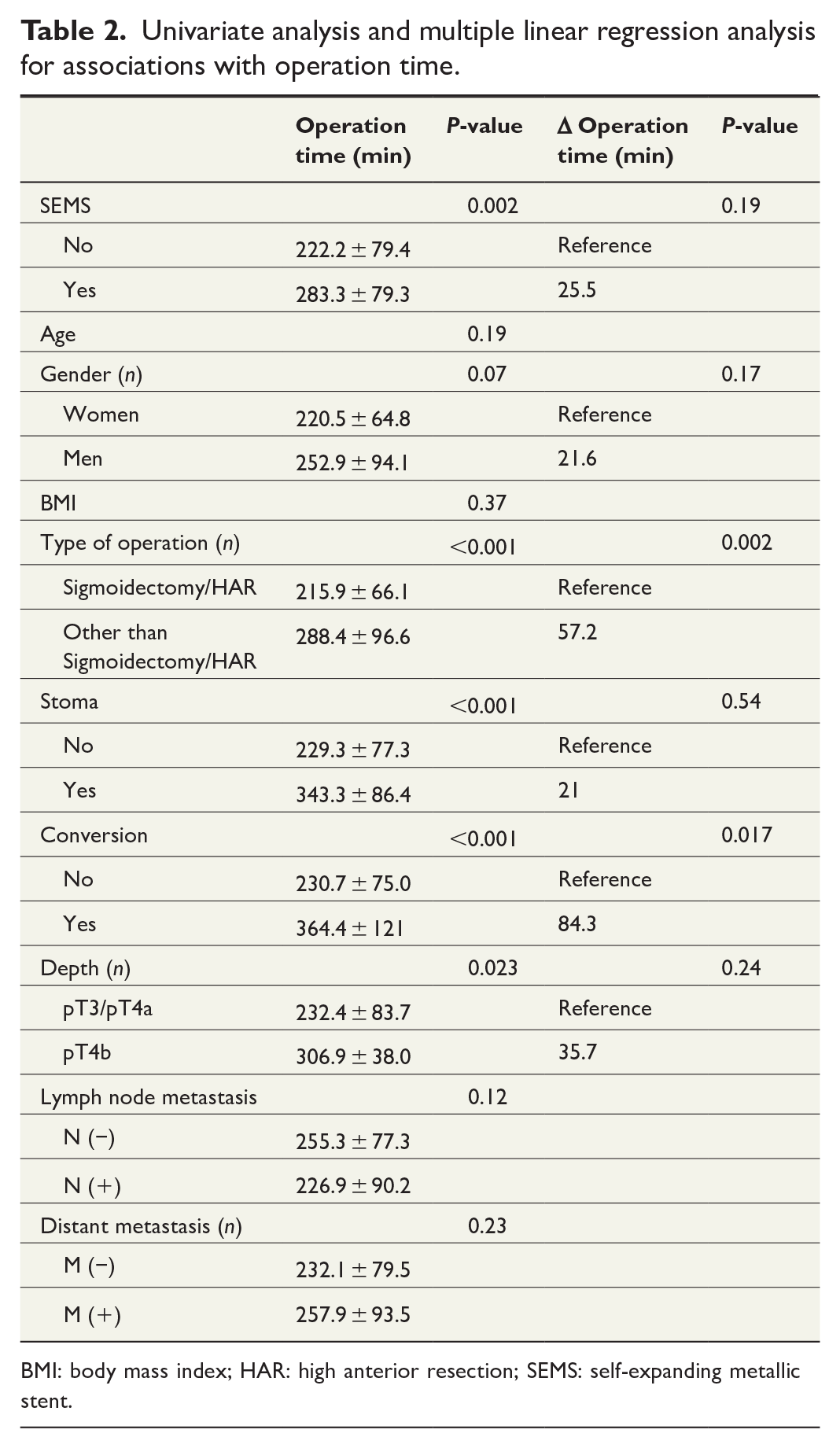
BMI: body mass index; HAR: high anterior resection; SEMS: self-expanding metallic stent.
Univariate analysis and multiple linear regression analysis for associations with blood loss.
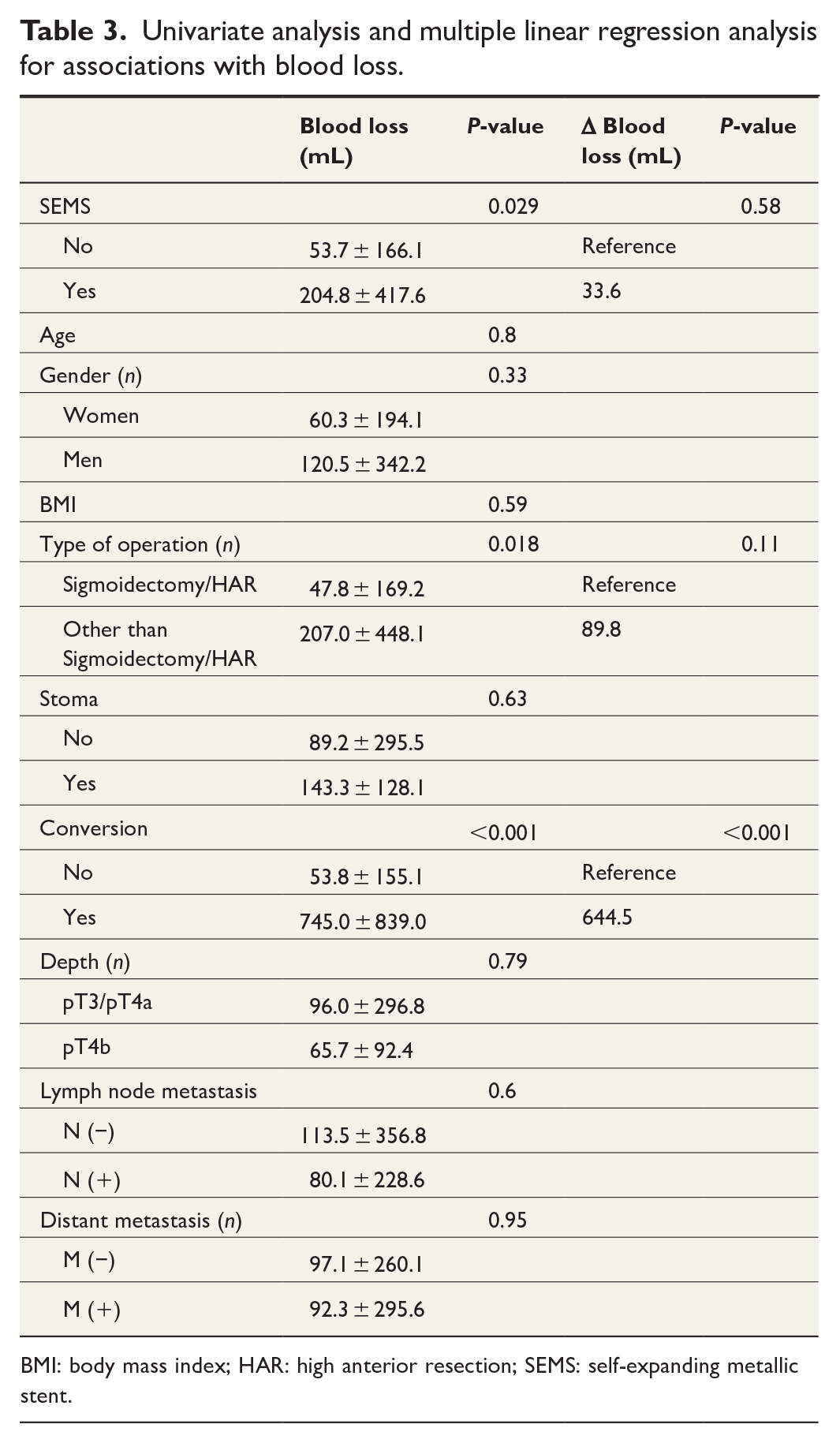
BMI: body mass index; HAR: high anterior resection; SEMS: self-expanding metallic stent.
Finally, to assess the long-term oncological outcome, the 5-year OS and DFS were analyzed. In this analysis, all patients with synchronous metastases were excluded. Both OS (100% vs 86.3%, P = 0.25) and DFS (80.0% vs 72.2%, P = 0.76) were compared between the SEMS and control groups and showed no significant differences (Fig. 1).
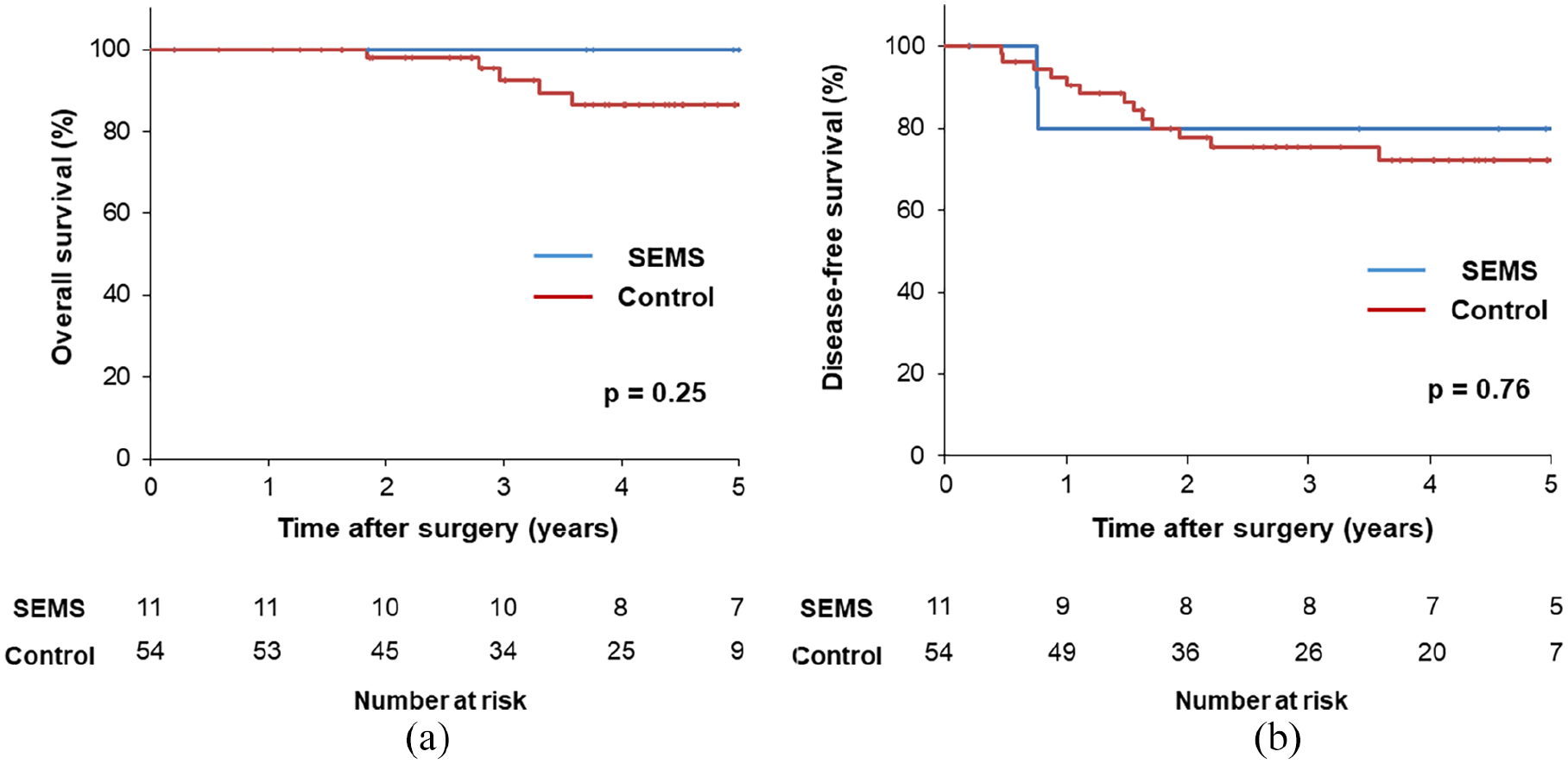
Overall survival (a) and disease-free survival (b) curve of the SEMS and control group. In this analysis, all patients with synchronous metastases were excluded.
Discussion
To identify the influence of SEMS on laparoscopic resection of left-sided obstructive CC, cases of SEMS insertion should be compared with those of emergent decompression by stoma creation or long-tube insertion. However, in our institution, for decompression of left-sided obstructive CC, SEMS was chosen initially; there was no case of laparoscopic resection after emergent decompression without SEMS. Therefore, we compared cases requiring laparoscopic resection for obstructive CC after emergent decompression with SEMS with obstructive CC cases that did not require emergent decompression. However, neither presentation would allow a small-diameter colonoscope to pass through. The major limitation of this study is that these two groups are not exactly comparable because of different tumor factors. Although the results should be interpreted with caution, we revealed that laparoscopic resection of obstructive CC with SEMS could be challenging owing to a highly advanced tumor and high conversion ratio to laparotomy in comparison with that laparoscopic resection of obstructive CC without SEMS. In contrast, the comparable rate of complications greater than the Clavien–Dindo classification grade II and long-term oncological outcomes indicated that laparoscopic resection with SEMS could be safely performed in patients with obstructive left-sided CC.
Previous studies compared laparoscopic resection after SEMS insertion with standard laparoscopic resection for non-obstructive CC and found no difference in the operation time, intraoperative blood loss, and postoperative hospital stay.12,13 In this study, regardless of the comparison of laparoscopic resection after SEMS with that for obstructive CC without SEMS, which is considered more difficult than laparoscopic resection for non-obstructive CC, the operation time was longer, the blood loss was greater, and the hospital stay was longer in the SEMS group than in the control group. However, in the multiple regression analysis, SEMS was not an independent risk factor for both operation time and blood loss. The independent risk factors were operation type, conversion to laparotomy for operation time, and conversion to laparotomy for blood loss. Shimizu et al. documented no difference in the distribution of operation types between laparoscopy after SEMS and standard laparoscopy, with no conversion case. 13 In this study, the percentages of left hemicolectomy for descending CC and low anterior resection for rectosigmoid junction CC were much higher in the SEMS group, and the rate of conversion to laparotomy was higher in the SEMS group, which possibly accounts for the longer operation time and the larger amount of blood loss in the SEMS group.
An important advantage of SEMS insertion for obstructive CC is one-stage resection and primary anastomosis without stoma creation. This study revealed that the stoma creation rate was 26% in the SEMS group, which was much higher than that in the control group (1%). In this study, the SEMS group was not really comparable to the control group, since the SEMS group was considered more advanced in terms of both necessity of emergent preoperative decompression and higher rate of distant metastasis than the control group. In fact, in most cases, the reason for stoma creation was not due to SEMS insertion itself but due to patient and tumor factors. It could not be concluded that laparoscopic surgery after SEMS insertion caused the high rates of stoma creation, although the inflammatory effect of the stents may have necessitated stoma creation in some cases.
This study showed that the conversion rate in the SEMS group was 17%, which was much higher than that in the control group (1%). The conversion rate of laparoscopic resection after SEMS insertion has been reported to range from 0% to 25%.8,12,13,15 In one case, peritoneal dissemination was the reason for conversion. In three other cases, despite no pathological evidence of invasion, the dissection between the tumor and normal organ was difficult, resulting in conversion to open surgery. In the control group, the reason for conversion was adhesion. Therefore, SEMS insertion could make separating the tumor from the normal organ difficult because of tumor rigidity or peritumoral inflammation. In addition, the highly advanced nature of tumors in the SEMS group is a possible reason for the high conversion rate.
This study showed no intergroup differences in the rate of complications over Clavien–Dindo classification grade II. This result was consistent with that of a previous report. In addition, there was no anastomotic leakage, and grade III or higher complications of the Clavien–Dindo classification occurred in only one case. These data suggest that even in highly advanced cases, to select stoma creation and conversion to open surgery correctly, laparoscopic surgery after SEMS insertion is as safe as laparoscopic surgery without SEMS in terms of short-term outcomes.
With respect to long-term outcomes, owing to the poor outcome and low success rate of SEMS insertion, elective surgery after SEMS insertion was not recommended in obstructive CC. 16 - 18 However, recent large-scale studies have reported comparable oncological outcomes between surgery after SEMS insertion and emergency surgery. 19 - 21 This suggests that elective surgery after SEMS insertion is acceptable for long-term survival. A few studies have compared the oncological outcomes of laparoscopic surgery after SEMS insertion with those of laparoscopic surgery without SEMS insertion in obstructive CC. This study revealed that in stage II/III CC patients, there was no intergroup difference in oncological outcomes.
As mentioned above, the major limitation of this study was the difference in the tumor factor of both groups; the control group did not require emergent decompression whereas the SEMS group needed it. Furthermore, the very small sample size and retrospective non-randomized design were other limitations because, apart from the tumor factor, the patient’s background was also not homologous between the two groups. The results should therefore be interpreted with caution. Further investigation is needed to elucidate the clinicopathological features and short- and long-term outcomes of laparoscopic surgery after SEMS insertion.
In conclusion, laparoscopic surgery after SEMS insertion for left-sided CC is demanding due to higher conversion rates to open surgery. However, this study also revealed that it could be performed as safely as laparoscopic surgery without SEMS insertion because of comparable complication rate and long-term outcomes.
Footnotes
Acknowledgements
Author Contributions
Y.Y. conceived the study, collected data, and drafted the manuscript. S.E. conceived the study, corrected, and revised the manuscript. H.N., Kazushige K., K.S., K.M., R.I., Kazuhiko K., and S.I. collected data, corrected, and revised the manuscript. All authors have read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Grants-in-Aid for Scientific Research (C: grant no. 18K07194; C: grant no. 19K09114; C: grant no. 19K09115; C: grant no. 20K09051; C: grant no. 20K09028; Challenging Research (Exploratory): grant no. 20K21626) from the Japan Society for the Promotion of Science. This research was supported by the Project for Cancer Research and Therapeutic Evolution (P-CREATE; grant no. JP 19cm0106502) endowed by the Japan Agency for Medical Research and Development.
