Abstract
Background and objective:
There are several forms of relevant epi-aortic branching anomalies, and perhaps that is why different views as to the best approach have been reported. To help resolve this dilemma, we examined the unforeseen arch anomalies found at thoracoscopic repair of esophageal atresia and the outcomes.
Methods:
In a retrospective cohort, all consecutive patients who were thoracoscopically approached for esophageal atresia over a 5-year period with unforeseen aortic/epi-aortic branching were identified and grouped. Thoracoscopic views, operative interventions, and outcomes were studied.
Results:
A total of 121 neonates were thoracoscopically approached for EA, of whom 18 cases with aberrant aortic architecture were selected. Four (3%) cases were diagnosed on a preoperative echocardiography as a right-sided aortic arch, whereas unforeseen anomalous anatomies were reported in 14 cases (11.6%): left aortic arch with an aberrant right subclavian artery (ARSA) (n = 10), right-sided aortic arch with an aberrant left subclavian artery (ALSA) (n = 3), and mirror-image right arch (n = 1). Single postoperative mortality was reported among the group with left arch and ARSA (10%), whereas all the cases with right arch and ALSA died.
Conclusions:
In all, 11.6% of the studied series exhibited unexpected aberrant aortic architecture, with higher complication rates in comparison to the typical thoracoscopic repairs. For EA with left aortic arch and ARSA, the primary esophageal surgery could safely be completed. Meanwhile, curtailing surgery—after ligating the TEF—to get advanced imaging is still advised for both groups with the right arch due to the significant existence of vascular rings.
Keywords
Introduction
Conventional medical thinking dictates that every case of esophageal atresia (EA) should undergo preoperative echocardiography. The purpose of such an investigation is to detect cardiac anomalies that help stratify patients into different prognostic categories and aortic arch anatomies (AAAs) that might influence the clinical approach if an anomalous position is suspected.1 –4 The problem with this protocol is that it converges all aortic arch anomalies into one—right-sided aortic arch—which, in fact, is over-simplified. There are several forms of relevant epi-aortic branching anomalies, and perhaps that is why different views as to the best approach have been reported. General pediatric surgeons, unaware of this, have been faced with cases of unfamiliar thoracic vascular anatomy and have documented their successes or failures in establishing esophageal continuity while ignoring the precise form of vascular anomaly.2,3
Being the standardized approach for management of EA at our facility, thoracoscopy offered us an opportunity to study the aortic/epi-aortic anatomy and to verify the suitable operative plan for each variant.
Materials and method
A retrospective cohort study of all thoracoscopically approached cases of EA at Cairo University Specialized Pediatric Hospital between 2016 and 2021 was carried out to identify cases with unforeseen aortic/epi-aortic anomalies. These refer to congenital abnormalities of the arch position, branching pattern, or both, not documented in routine preoperative echocardiography. Medical records of the sorted cases were checked by two authors individually to report the anomalous vascular anatomy, type of EA, gap length, and approach followed for establishing esophageal continuity. Postoperative outcomes and follow-up results for the enrolled groups were reviewed.
A specialized consultant radiologist (RZ) examined multi-slice computed tomography (MSCT) images of pertinent aortic and epi-aortic anatomies with the thoracoscopic profiles to help the authors confirm the delineation of the unanticipated vascular anomalies.
Patients with proven aberrant aortic/epi-aortic architectures were grouped according to its pattern. To avoid misinterpretation of the dextroposition of the left arch with aberrant right subclavian artery (ARSA) as a right arch, the authors of this article set the following scheme (Fig. 1). Detecting the aortic arch during right thoracoscopy should prompt evaluation of the following anatomic characteristics: first, the association of the aortic arch with the trachea, whether emerging dorsal to the trachea (i.e. left-sided aortic arch [LAA]) or arching ventral to the trachea (i.e. right-sided aortic arch [RAA]) and second, once arch laterality is established, the pattern of its great vessels should be set. Detection of an aberrant subclavian artery emerging low from an LAA, passing dorsolateral to the trachea toward the thoracic inlet, should confirm the diagnosis of LAA + ARSA. While the detection of an aberrant subclavian artery emerging low from an RAA and passing dorsally circum-around the trachea forming a vascular sling around it should narrow the diagnosis to an RAA with aberrant left subclavian artery (ALSA) ± Kommerell’s diverticulum (Edward type II) or double aortic arches. The Edward type I arch anomaly presents itself as a right-sided aortic arch—emerging ventral to the trachea—with no aberrant subclavian artery obscuring the required mediastinal dissection for tracheoesophageal fistula (TEF) or upper esophageal pouch mobilization. Supplementary Figure 1 illustrates the three patterns of anomalous arch anatomy in the studied group.
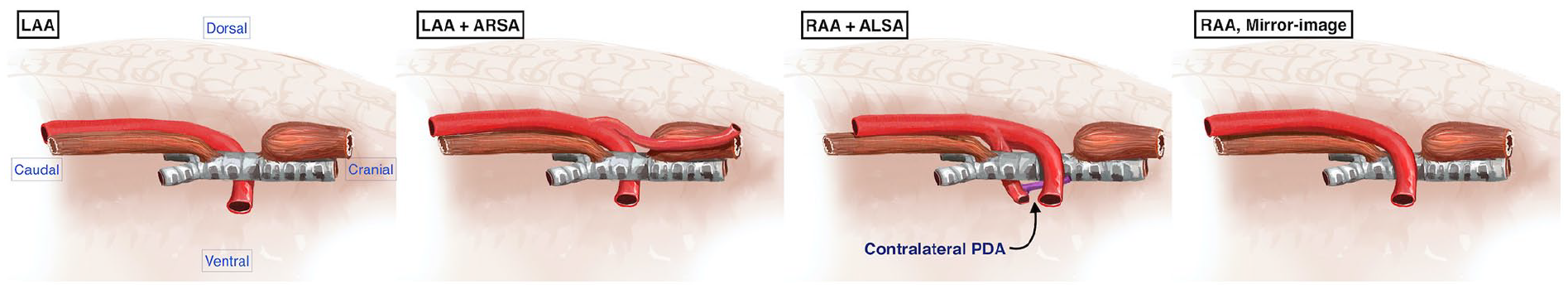
Labeled schematic representation of the thoracoscopic view of posterior mediastinum for common AA variants encountered with type-C-esophageal atresia.
Statistical analysis
Patient characteristics were analyzed using descriptive statistics presented as mean with standard deviation, median with range, and/or numbers with percentages. For comparing categorical data, chi-square test was performed. The Exact test was used instead when the expected frequency is less than 5. P values < .05 were considered statistically significant.
Results
Patients
During the inclusion period, 121 neonates with EA were approached thoracoscopically, in the fully prone position at the side edge of the table, with the surgeon facing the right side.5,6 Four (3%) cases with aberrant aortic architecture in the form of RAA were identified on preoperative echocardiography. Meanwhile, unforeseen AAAs were reported in 14 (11.6%) cases of EA; all were of Gross-type C with proximal blind pouch and distal tracheoesophageal fistula (EA/TEF).
The identified unforeseen vascular anatomies were grouped as follow: LAA with an aberrant right subclavian artery (LAA + ARSA) (n = 10), RAA with an aberrant left subclavian artery (RAA + ALSA) arising from the remnant of the left dorsal aortic root (Kommerell’s diverticulum) (n = 3), and a mirror-image right arch (n = 1). Supplementary Table 1 illustrates the perioperative data for the 14 cases with unforeseen AAA.
Operative dissection
In the cases with LAA + ARSA, dissection of the upper pouch was accomplished in a plane ventral to the ARSA close to the trachea, without the mobilization of the ARSA (Supplementary Fig. 2).
For the cases with RAA + ALSA, upper pouch dissection was accomplished in a plane dorsal to the right arch, and the dissected pouch was then delivered through the window between the RAA and the ALSA after being widened bluntly (Supplementary Fig. 3).
For the case with mirror-image right arch, TEF ligation and upper pouch dissection was performed in a plane dorsal to the right arch. The patient had an esophagostomy and gastrostomy with failure to achieve an otherwise applicable primary esophageal anastomosis after a lengthy struggle on operation. Supplementary video (1) shows the operative plan in the three atypical forms of aortic/epi-aortic architectures at thoracoscopic EA repair: https://drive.google.com/file/d/1jYXbyBgylJ8jaH0Jbb5G43QRs8o72oIA/view?usp = sharing
Outcomes
Table 1 demonstrates the peri-operative characteristics of the studied cohort in comparison to the typical thoracoscopic repairs of EA with LAA. There was one reported mortality among the cases with LAA + ARSA (10%), related to leak-associated mediastinal sepsis. The three patients with RAA + ALSA died. Despite the modest number of recruits, the failure to cease postoperative ventilatory support was the most serious issue in all. Retrospectively, it became clear that although the operative plan was successful in completing the task, tracheal impingement was inevitable because of incarcerating the upper esophageal pouch into the dissected window between the RAA and the ALSA. In such cases, the anatomical challenge posed by the existent vascular ring was not realized in due time.
Perioperative characteristics of the AAA cohort in comparison to the typical thoracoscopic repairs at authors’ facility.
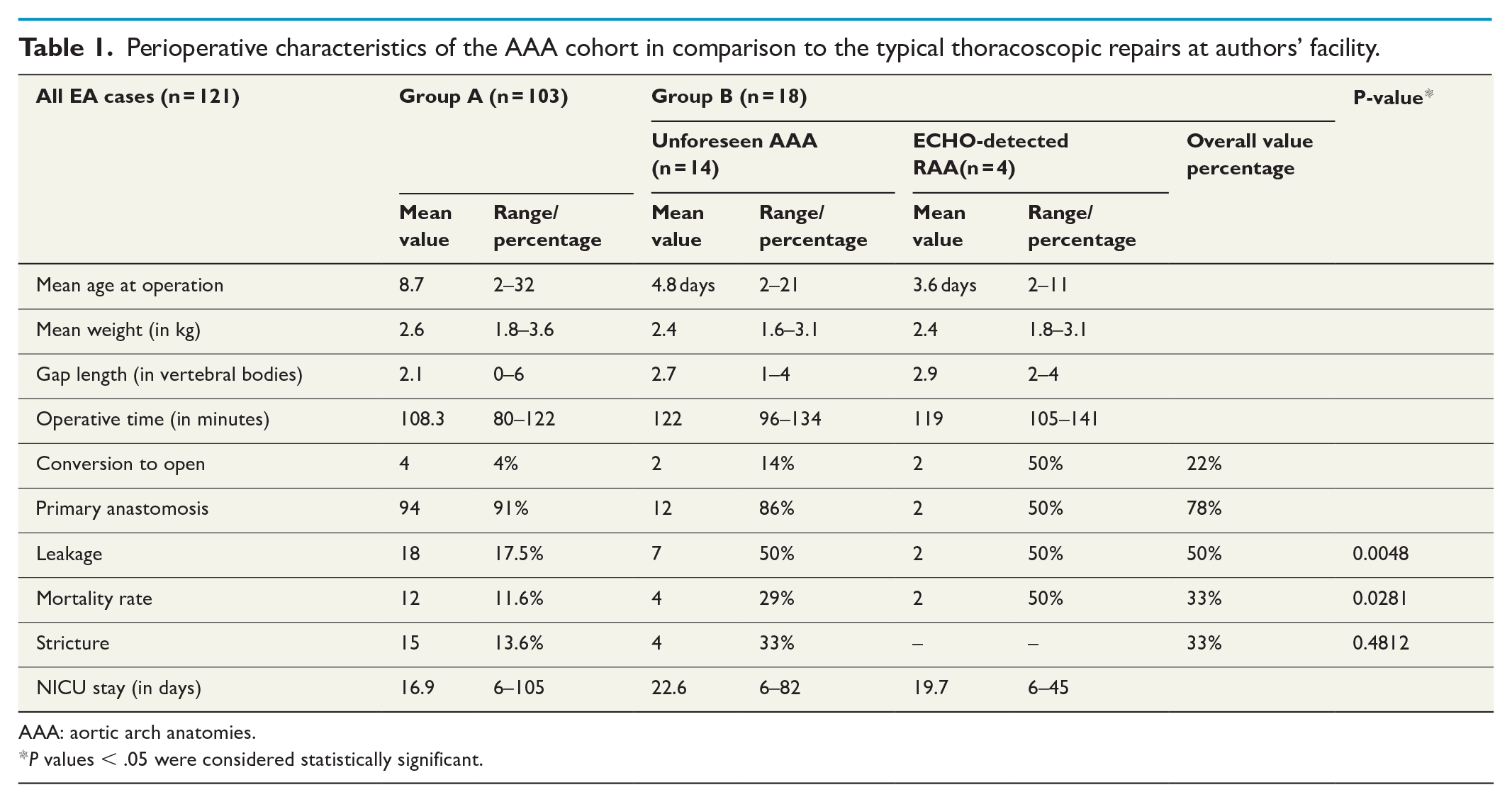
AAA: aortic arch anatomies.
P values < .05 were considered statistically significant.
Discussion
Although thoracoscopy offered a unique opportunity to view the mediastinal anatomy and plan the surgery with minimal disturbance to the chest wall,7,8 proper knowledge of aortic/epi-aortic anatomy is the clue to such planning, to quote Tempest-Tost by Robertson Davies “The eye sees only what the mind is prepared to comprehend.” 9
The routine preoperative echocardiography for EA is advised. However, it may provide false-negative findings, particularly in newborns with epi-aortic branching anomalies (i.e. ARSA or ALSA).10,11 MSCT with virtual 3D reconstruction can sharply delineate vascular anatomy corresponding with the targeted esophageal ends and trachea, hence it is recommended once arch anomalies are suspected in a preoperative investigation.12 –14
Visualization of a Kommerell’s diverticulum at the origin of an aberrant subclavian artery—namely, the 60% prevalence with RAA + ALSA and the occasional association with the more frequent LAA + ARSA combination (15–30%)—is another substantial issue that might have impact on the operative plan.15 –17 This diverticulum represents the remnant of the contralateral dorsal aortic root and is thought to be related to the presence of a ductus arteriosus (patent or obliterated) contralateral to the persistent AA, 14 thereby forming a complete vascular ring around a congenitally pliable trachea in the absence of a coexisting esophageal segment.
In the presented series, the combination of LAA + ARSA was the most common arch anomaly reported in cases of EA (8%). This is consistent with other published series17,18 as well as with the natural prevalence of such arch configuration (0.5–2%; 35% among those with Down syndrome).19,20 Table 2 reports the literature review for cases of EA with AAA.
Literature review for cases of EA with AAA.
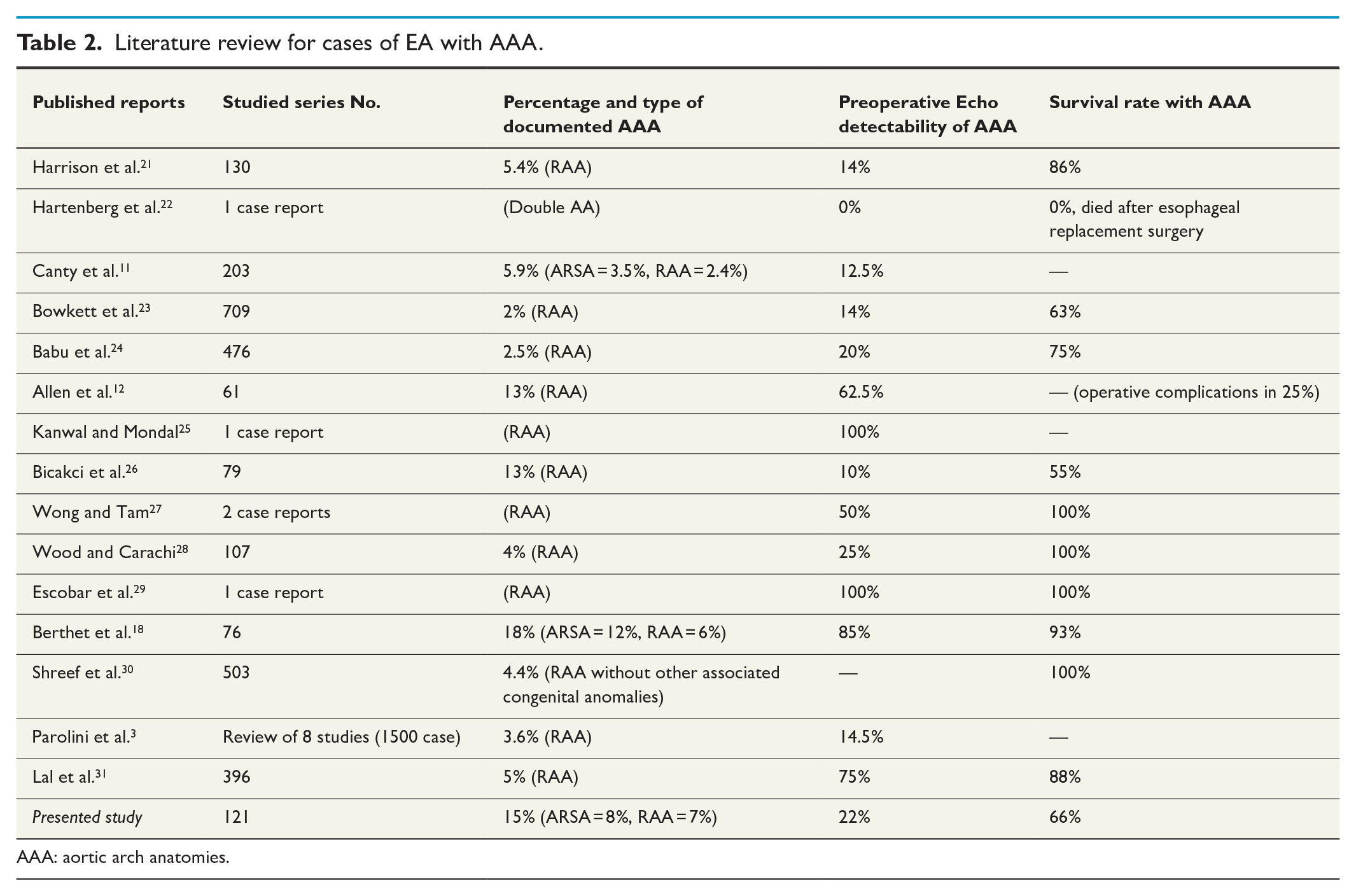
AAA: aortic arch anatomies.
Although echocardiography aids in the diagnosis of ARSA, it may still go unnoticed. Moreover, ARSA is difficult to identify during a conventional thoracotomy for EA, in contrast to RAA, and the postoperative esophagram has a sensitivity of 66%. 18 According to the presented series, thoracoscopy offered a unique opportunity to clearly visualize this aortic anatomy and meticulously plan the surgery and the post-operative care.
Although it typically has a benign natural history, 10% of otherwise normal infants with ARSA will develop a later presentation of dysphagia lusoria as identified in the literature, being caused by the oblique retro esophageal course of the ARSA (Supplementary Fig. 4). 32 Likewise, it is reported that this—not rare—abnormality is the source of EA-related respiratory and/or GI symptoms, where a ring partially—or occasionally completely—encircles the trachea and/or esophagus, creating an extrinsic compression. 18
Hence, it is critical to consider the likelihood of an aortic anomaly in EA patients, not only because these defects can be symptomatic, but also because they have been shown to be fatal. Lo et al described two cases of arterio-esophageal fistula development and significant upper gastrointestinal bleeding following post-EA repair stenting, each with an unidentified ARSA. 33 Accordingly, it has been advocated to use magnetic resonance imaging or angioscan to rule out such anomalies when stenting of the esophagus is needed, before esophageal replacement surgery, or when prolonged (> 2 weeks) use of a nasogastric tube is needed.34,35
The right-sided thoracic approach for primary repair in EA/TEF is not regarded contraindicated by RAA.26,28 However, an RAA with an ALSA frequently forms a complete vascular ring, and, unlike ARSA, this anomaly causes dysphagia and respiratory distress in more than half of otherwise normal newborns, with EA having a higher frequency.36 –38 As a result, early surgical repair of the vascular ring is indicated to provide complete symptom relief and enhance overall survival. 18
In EA cases with the RAA + ALSA or double AA, although dissection of the upper pouch and primary esophageal anastomosis to the left of the RAA could be accomplished, the authors of this report realized that it was not the esophagus that posed a challenge in such cases, but rather, keeping the fragile trachea open. This type of AA has a high propensity to form a vascular ring. A left-sided patent ductus arteriosus (PDA) running from the ALSA (at its junction with Kommerell’s diverticulum) to the pulmonary artery completes a vascular ring that develops at a cross-sectional congenital EA, creating a window just fitting a collapsible trachea (Supplementary Fig. 5). The presented study proved that this developmentally tight window cannot accommodate the pouched esophagus, regardless of how skillful the surgeon is. Division of PDA in such cases could release the constricting ring. Moreover, ALSA could be divided whenever it causes refractory tracheal/esophageal impingement.
It is traditionally recommended to ligate the fistula and defer EA until the unforeseen anomalous vascular anatomy is properly studied; a postoperative radiological assessment would help guide a different life-saving approach for such cases, preferably involving a cardiothoracic vascular surgeon. 21 However, many authors suggest alternate approaches, albeit perhaps with some controversy.21,39 Nevertheless, with cumulative experience, thoracoscopy offered a precise operative delineation of unforeseen vascular anomalies and prompt a tailored surgical dissection for each. Hence, consideration should be given to this modality of anatomic verification preferentially over curtailing surgery in all anatomic variants. However, curtailing surgery—after ligating the TEF—is still recommended in the case of right arch-sidedness ± Kommerell’s diverticulum to search for a constricting vascular ring, which has been demonstrated to take precedence over the primary esophageal anastomosis.
Limitations
The main limitations of this study were the small number of enrolled cases and the lack of long-term follow-up for possible aero-digestive manifestations of vascular rings or slings (incomplete rings).
Conclusion
According to the data given, 11.6% of the studied series exhibited unexpected aberrant aortic/epi-aortic architecture, with higher complication rates in comparison to the typical thoracoscopic repairs. Thoracoscopy provided a unique opportunity to accurately identify previously unknown vascular anatomy. For EA with left aortic arch and ARSA, the primary esophageal surgery could safely be completed. Meanwhile, curtailing surgery—after ligating the TEF—to get advanced imaging is still advised for both groups with the right arch; the mirror-image type and the right arch with ALSA due to the significant existence of vascular rings. It is not the esophagus that posed a challenge in such a situation, but rather, keeping a rickety trachea open.
Supplemental Material
sj-docx-1-sjs-10.1177_14574969221090487 – Supplemental material for The dilemma after an unforeseen aortic arch anomalies at thoracoscopic repair of esophageal atresia: Is curtailing surgery still a necessity?
Supplemental material, sj-docx-1-sjs-10.1177_14574969221090487 for The dilemma after an unforeseen aortic arch anomalies at thoracoscopic repair of esophageal atresia: Is curtailing surgery still a necessity? by Hamed M. Seleim, Ahmed M.K. Wishahy, Basma Magdy, Mohamed Elseoudi, Rania H. Zakaria, Sherif N. Kaddah and Mohamed M. Elbarbary in Scandinavian Journal of Surgery
Supplemental Material
sj-jpg-3-sjs-10.1177_14574969221090487 – Supplemental material for The dilemma after an unforeseen aortic arch anomalies at thoracoscopic repair of esophageal atresia: Is curtailing surgery still a necessity?
Supplemental material, sj-jpg-3-sjs-10.1177_14574969221090487 for The dilemma after an unforeseen aortic arch anomalies at thoracoscopic repair of esophageal atresia: Is curtailing surgery still a necessity? by Hamed M. Seleim, Ahmed M.K. Wishahy, Basma Magdy, Mohamed Elseoudi, Rania H. Zakaria, Sherif N. Kaddah and Mohamed M. Elbarbary in Scandinavian Journal of Surgery
Supplemental Material
sj-jpg-4-sjs-10.1177_14574969221090487 – Supplemental material for The dilemma after an unforeseen aortic arch anomalies at thoracoscopic repair of esophageal atresia: Is curtailing surgery still a necessity?
Supplemental material, sj-jpg-4-sjs-10.1177_14574969221090487 for The dilemma after an unforeseen aortic arch anomalies at thoracoscopic repair of esophageal atresia: Is curtailing surgery still a necessity? by Hamed M. Seleim, Ahmed M.K. Wishahy, Basma Magdy, Mohamed Elseoudi, Rania H. Zakaria, Sherif N. Kaddah and Mohamed M. Elbarbary in Scandinavian Journal of Surgery
Supplemental Material
sj-jpg-5-sjs-10.1177_14574969221090487 – Supplemental material for The dilemma after an unforeseen aortic arch anomalies at thoracoscopic repair of esophageal atresia: Is curtailing surgery still a necessity?
Supplemental material, sj-jpg-5-sjs-10.1177_14574969221090487 for The dilemma after an unforeseen aortic arch anomalies at thoracoscopic repair of esophageal atresia: Is curtailing surgery still a necessity? by Hamed M. Seleim, Ahmed M.K. Wishahy, Basma Magdy, Mohamed Elseoudi, Rania H. Zakaria, Sherif N. Kaddah and Mohamed M. Elbarbary in Scandinavian Journal of Surgery
Supplemental Material
sj-jpg-6-sjs-10.1177_14574969221090487 – Supplemental material for The dilemma after an unforeseen aortic arch anomalies at thoracoscopic repair of esophageal atresia: Is curtailing surgery still a necessity?
Supplemental material, sj-jpg-6-sjs-10.1177_14574969221090487 for The dilemma after an unforeseen aortic arch anomalies at thoracoscopic repair of esophageal atresia: Is curtailing surgery still a necessity? by Hamed M. Seleim, Ahmed M.K. Wishahy, Basma Magdy, Mohamed Elseoudi, Rania H. Zakaria, Sherif N. Kaddah and Mohamed M. Elbarbary in Scandinavian Journal of Surgery
Supplemental Material
sj-tiff-2-sjs-10.1177_14574969221090487 – Supplemental material for The dilemma after an unforeseen aortic arch anomalies at thoracoscopic repair of esophageal atresia: Is curtailing surgery still a necessity?
Supplemental material, sj-tiff-2-sjs-10.1177_14574969221090487 for The dilemma after an unforeseen aortic arch anomalies at thoracoscopic repair of esophageal atresia: Is curtailing surgery still a necessity? by Hamed M. Seleim, Ahmed M.K. Wishahy, Basma Magdy, Mohamed Elseoudi, Rania H. Zakaria, Sherif N. Kaddah and Mohamed M. Elbarbary in Scandinavian Journal of Surgery
Footnotes
Author contributions
All contributing authors made substantial contributions to conception and design, acquisition of data, analysis and interpretation of data; participated in drafting the article or revising it critically for important intellectual content; and approved the version to be published.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Consent was given for registration of medical data for anonymous research.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
